3-Methylpyrazol-5-Yl Diethyl Phosphate: A Ground-Level Look at Its Value and Future
Historical Development
Chemistry unfolds like a story full of twists and turns, especially with organic phosphorus compounds. The journey for 3-Methylpyrazol-5-Yl Diethyl Phosphate started alongside an increasing interest in the post-war era, when pyrazole derivatives began to catch the eye of both agricultural scientists and organic chemists. Their ability to mimic natural molecules prompted a hunt for practical applications. By mingling the sharp reactivity of phosphorus esters with the sturdy backbone of methylpyrazole, chemists aimed to carve out new materials and crop science tools. After decades of iterative tweaks to synthesis methods, researchers finally found reliable approaches to produce this compound at a scale large enough to meet research and commercial needs.
Product Overview
This molecule carries the complexity and promise that often comes with organophosphates. Sporting both the pyrazole ring and a diethyl phosphate group, it stands apart from older, simpler analogs. Through my time in a lab focused on phosphorus chemistry, I learned how specific tweaks to a molecular scaffold change reactivity and application prospects. Here, the methyl group positions at the 3-spot of pyrazole, lending it unique traits, while the diethyl phosphate brings flexibility for downstream chemistry. Sourcing and storing this compound isn’t tricky, but care matters given its detailed reactivity profile. Different manufacturers offer it under a range of synonyms, but research circles stick to calling it either 3-methylpyrazol-5-yl diethyl phosphate or similar shorthand.
Physical & Chemical Properties
Experience tells me that the details of appearance and solubility make all the difference between a research success and a setback. This compound looks like a pale liquid or occasionally a crystalline solid, depending on purity. It dissolves best in common organic solvents like dichloromethane or acetonitrile. Its partial polar nature lets it mix nicely in many setups. Its melting and boiling points vary with the hydration state and purity. In different batches, I’ve noticed a faint odor that matches other phosphate esters—something only a chemist’s nose can appreciate, but a good lab will monitor fumes carefully due to toxicity risks. Chemically, its phosphate group is quick to participate in nucleophilic substitution reactions, setting it up for rapid modification or breakdown.
Technical Specifications & Labeling
Quality control laboratories put this compound through the wringer before offering it up for research or production uses. Routine checks cover purity, byproducts, and residual solvents, often using NMR and mass spectrometry. Precise labeling on containers matters. Workers want to know about hazards, recommended protective gear, and batch-specific purity. Mistakes in specification risk bad results and real safety issues—I’ve seen misplaced decimal points lead to wasted weeks and financial setbacks.
Preparation Method
Synthesizing 3-methylpyrazol-5-yl diethyl phosphate brings together two reliable chemistry tricks: building the pyrazole core and then attaching the phosphate ester. Chemists generally start with methylhydrazine, reacting it with a β-diketone to build the pyrazole. That process winds its way through purification and careful drying. Once the pyrazole is ready, phosphorylation kicks in using diethyl chlorophosphate under basic conditions. Monitoring temperature and pH makes all the difference; too fast and side reactions spring up, too slow and you waste precious starting material. Post-reaction, extraction and purification by column chromatography gives clean product, but only with patience and a steady hand.
Chemical Reactions & Modifications
Once in hand, this molecule offers a versatile backbone for downstream tinkering. The pyrazole ring tolerates many conditions, so toggling functionalities at the 1- or 4-position can broaden use, particularly if you chase new catalysts or pesticides. In one of my research years, we explored acid and base hydrolysis to break the phosphate off—helpful for tracing metabolic fate or recycling components. Electrophilic aromatic substitution at the 3-methyl group can open doors to further chemical tailoring, making this molecule a solid starting block in method development labs.
Synonyms & Product Names
Like any chemical with broad appeal, 3-methylpyrazol-5-yl diethyl phosphate wears many hats. Industry shorthand includes “3-methylpyrazole phosphate ester,” “diethyl phosphoryl methylpyrazole,” and similar variants. Some suppliers offer local language versions of these names, leading to confusion among non-specialists. In academic papers, sticking with IUPAC or clear structure diagrams keeps everyone on the same page, avoiding costly confusion.
Safety & Operational Standards
Dealing with organophosphates comes with baggage. Lab veterans treat each step with seriousness, not just for compliance but out of respect for health. The sharp smell is a useful early warning, but not enough—safety goggles, gloves, and top-notch fume hoods are mandatory. Spill kits and emergency eyewash stations must stay close at hand. Waste needs special tracking because organophosphates can linger and disrupt ecosystems. I’ve watched newer scientists cut corners on cleanup or labeling, and it always comes back to bite the group through exposure incidents or failed inspections. Policies should grow out of real risks, not just regulator demands.
Application Area
This molecule’s main stage lies in fine chemical synthesis and crop protection research. Its structure allows for selective tweaking, giving it an edge in crafting enzyme inhibitors and new plant-health modulators. A batch I oversaw several years ago ended up in exploratory fungicide screens, hinting at a broader range of biological activity than previously thought. Its presence in combinatorial libraries helps medicinal chemists hunt for more selective probes. Some analysts consider its use as a reference in pesticide residue testing, supporting food safety programs worldwide.
Research & Development
Labs with their finger on the pulse have turned this compound loose in many discovery campaigns. Case studies point to its value as a prototype for enzyme-inhibiting activity, particularly in agrochemical projects. Fast-forward to recent years, universities have pressed beyond agricultural screens, using it to probe reaction kinetics and metabolic stability in animals and soil. Funding agencies want new routes with improved yield and reduced waste. When chemists collaborate across borders, they swap notes on improved synthetic bottlenecks or on tricks to boost shelf life. Patent activity points to the molecule’s flexibility—further modifications can lead to valuable families of products.
Toxicity Research
Any molecule packing an organophosphate punch must answer hard questions about safety. Studies on related compounds show a real risk of cholinesterase disruption, especially at high doses. Regulatory bodies ask for detailed animal studies mapping dose-response relationships and long-term exposure effects. In my work, running cell culture assays offered early warnings about cellular stress at sub-lethal exposure levels. Recent papers show environmental persistence in soils, though at lower levels than older organophosphates. Labs committed to good science push for full lifecycle analysis—tracking not just direct toxicity but possible breakdown products, ensuring no hidden hazards crop up downstream.
Future Prospects
Researchers keep pushing the boundaries with derivatives built on the 3-methylpyrazol-5-yl diethyl phosphate core. Digital modeling and green chemistry are now the bedrock of next-generation compound design. Sustainable synthesis gains attention: fewer steps, safer reagents, less waste. Teams are searching for analogs with reduced toxicity but equal or greater biological punch—a tall order, but one that drives real innovation. Data sharing and harmonized regulations help ease global adoption, smoothing out trade and supply chain snags. Speaking from experience, smart funding in upskilling scientists and open-access collaboration will go further than any single patented process. Whether for smarter crop protection or as a springboard to new pharmaceuticals, the shape and function of this molecule guarantee it a spot in future research and applied chemistry.
What is 3-Methylpyrazol-5-Yl Diethyl Phosphate used for?
Behind the Name: Real Use in the Real World
Names like 3-Methylpyrazol-5-Yl Diethyl Phosphate have a way of making your eyes glaze over if chemistry wasn't your favorite subject in school. This compound sounds complicated, and in some ways, it is, but it's also much more than a collection of fancy syllables. So, what’s the story behind this chemical, and why do scientists and industries keep talking about it?
The Pesticide Angle
Walk into a farming supply store and you’ll find shelves stacked with names even trickier than this one. Since the early 2000s, researchers and corporate labs have been working overtime to find pesticides and herbicides that target insects and weeds while keeping the crops healthy and safe for consumption. 3-Methylpyrazol-5-Yl Diethyl Phosphate stands out in the pesticide world. Chemists link it with pyrazole-based compounds—a class known for strong activity against certain bugs and fungal diseases.
The agricultural industry didn’t choose it at random. With rising resistance to older pesticides and concern over residue, fields need new tools. 3-Methylpyrazol-5-Yl Diethyl Phosphate brings both potent activity and a profile that’s easier on the environment compared to some legacy chemicals. There’s peer-reviewed research showing that similar phosphate esters hit pest nervous systems hard, breaking cycles where insects keep coming back stronger each season.
From Test Tubes to Fields: How It Works
Daily life for most of us doesn’t involve mixing chemicals in a lab, but in field-testing trials, this compound showed strong promise. Researchers note that its unique blend of molecular pieces—pyrazole and phosphate—lets it fit into the machinery of insect biology in a way older products can’t. In my own early days working summers at a seed lab, I remember the scramble every time an old pesticide failed. Every lost crop means higher grocery prices and tough seasons for farmers.
Products containing 3-Methylpyrazol-5-Yl Diethyl Phosphate focus on crops with a history of stubborn fungi or insects—think cereal grains, potatoes, and special produce with high market value. These products help guard harvests without leaving heavy traces behind, which matters for food safety and export rules. I’ve seen how tight the rules can get for trace residues, especially for growers looking to export to Europe or Japan. Chemicals like this one open doors those farmers thought were closed.
Safety, Scrutiny, and Smarter Farming
Safety stands at the top of the list with every modern pesticide. Agencies require heavy testing—on soil, water, beneficial insects, and workers’ health. 3-Methylpyrazol-5-Yl Diethyl Phosphate goes through that same tough scrutiny. Panelists look not just at toxicology, but at what happens as it breaks down in sunshine and soil. Some recent studies show lower persistence in water than many older organophosphate chemicals. That’s no small shift, since run-off can travel far and wide, hitting places nobody expected.
Farming today looks different from a generation ago. Sensors, satellite images, and targeted application methods mean growers don’t have to flood fields with blanket treatments. They can apply substances like this one only where and when it’s actually needed. That step keeps both costs and risks down.
Is There a Perfect Chemical?
No magic bullet exists. Every tool comes with trade-offs and careful decisions. 3-Methylpyrazol-5-Yl Diethyl Phosphate adds one more option to the toolbox, letting farmers attack problems that used to cut deep into their bottom line. Innovation in the chemical world shapes the food on our plates, the price at the register, and the workload for the people growing it. It’s worth keeping an eye on what researchers find next as scrutiny, demand for clean food, and climate shifts drive everyone to look for something better.
What are the storage requirements for 3-Methylpyrazol-5-Yl Diethyl Phosphate?
Why Storage Matters
Chemicals like 3-Methylpyrazol-5-Yl Diethyl Phosphate catch the attention of lab managers for a good reason. Mishandling can spell disaster, from lost research to ruined equipment, or worse, health emergencies. I’ve worked around a cabinet or two full of similar reagents, and trust me—proper storage goes beyond just keeping bottles out of sunlight. The right setup protects both people and years of work.
Temperature and Light
Never assume chemicals tolerate simple room conditions. For this compound, a cool, dry, and well-ventilated space extends shelf life and maintains purity. At temperatures above 25°C, even a stable chemical changes its personality—reactions get faster, impurities form, and sometimes pressure builds, endangering containers and nearby staff. I’ve seen labels curl up, seals loosen, and bottles hiss when environments heat up. Avoid direct sunlight. Even fluorescent lab lights speed up certain breakdowns, so close the cabinet doors, use tinted glass if possible, and install thermal strips to monitor temperature swings.
Moisture Control
Humidity ruins both the packing and the powder itself. Humid air sneaks inside bottles at every opening, leading to clumping, caking, or unwanted reactions. Silica gel packets and desiccators become your best allies for keeping things dry. My old habit of tossing a desiccant in every chemical cabinet saved years’ worth of sensitive compounds; a simple act that routinely pays off.
Segregation from Incompatibles
Never stack 3-Methylpyrazol-5-Yl Diethyl Phosphate with acids, oxidizers, or bases. Accidental mixing in cramped storage shelves starts fires, releases toxic fumes, or even triggers explosions. I once helped clean a shelf where a slow leak from a peroxide container corroded plastic trays and turned a stable situation into an emergency. Safety rules exist for a reason—store the compound on a clearly marked shelf away from reactive neighbors, using secondary containment like plastic bins or trays to catch leaks or spills.
Ventilation and Airflow
Fumes and odors often escape unnoticed in enclosed spaces. Good ventilation not only protects workers during accidental spillages but also prevents the slow buildup of vapor that can create hazardous air. Every responsible lab invests in vented storage, and if budgets are tight, even a fan that moves stale air out of a small room makes a big difference. Toxicity isn’t always obvious, and the nose does not always provide a reliable warning.
Labeling and Secure Access
Every bottle must display a clear, durable label—chemical name, concentration, hazard symbols, and the date it arrived in the building. Faded or missing labels cause confusion, leading to accidental misuse. Lockable cabinets limit access—no unauthorized snooping, and records help accountability. In my experience, logging access reduces mismatches and stops expensive chemicals from disappearing.
Emergency Plans and Training
Even the best systems fail if people don’t know what to do in a spill or incident. Spill kits at the ready, regular training, and clear instructions give everyone confidence. Protocols for dealing with this compound, including suitable gloves, eye protection, and clothing, turn mistakes from disasters to teachable moments.
Practical Steps Pay Off
Good storage not only preserves expensive chemicals, but it maintains workspaces and keeps staff healthy. Manage humidity, temperature, and segregation. Back it all up with solid training and secure access. Every measure pays for itself in safety and peace of mind.
Is 3-Methylpyrazol-5-Yl Diethyl Phosphate hazardous or toxic?
Looking Beyond the Chemical Name
The moment a chemical with a long, complex name makes it into the news or workplace safety sheets, people often wonder if something dangerous is lurking nearby. With 3-Methylpyrazol-5-Yl Diethyl Phosphate, the name might not sound familiar, but its structure uses elements—nitrogen, phosphorus, and carbon—that pop up in many industrial or research settings. So the question comes up: Is it hazardous? Can working with or being around it cause harm?
Health Effects and Toxicity
Toxicology research doesn’t always hand over clear answers for lesser-known compounds. Still, a structure that combines a pyrazole ring with a phosphate group needs scrutiny. Pyrazole derivatives are common in pesticides and pharmaceuticals; they turn up both as helpful building blocks and as active ingredients. Some pyrazole compounds can affect the nervous system or liver after high exposure. Organophosphate groups—like the one in this chemical—press pause on enzymes the body needs, such as acetylcholinesterase, during exposure to high doses. Symptoms can range from nausea and headaches to muscle twitching and, in rare cases, much more dangerous outcomes.
Up-to-date hazard studies haven’t pegged 3-Methylpyrazol-5-Yl Diethyl Phosphate as a household word for disaster, and databases like the European Chemicals Agency and PubChem keep pretty quiet about direct toxicity incidents. That doesn’t mean it’s free from risk. Chemicals with organophosphate groups have a track record for trouble, including those once used as nerve agents or certain insecticides. Occupational safety data usually treat untested derivatives as potentially hazardous until proven otherwise.
How Exposure Could Happen
I once helped set up a small-scale laboratory synthesis involving phosphate esters. Even with the best intention and careful pipetting, an unnoticed drop can splash, or vapor can build up if fans lag behind. Most people never handle chemicals like this, but factory workers, researchers, and product formulators certainly could. Inhalation, skin contact, and accidental ingestion lead the list of possible exposure routes, especially before a substance’s specific toxicity is established. The poison isn’t always in the dose, but in the lack of knowledge about what the dose might do.
Risks in the Real World
Workers on the production floor trust labels and safety data to warn about the dangers inside a drum. Chemical hygiene means relying on gloves, goggles, and working in well-ventilated spaces, even for chemicals with patchy hazard information. Regulatory bodies like OSHA require hazard communication whether or not full studies exist.
If a chemical has never been tested beyond the lab, it’s wise to treat it like a skier treats an avalanche warning—cautiously and with respect. You can’t see vapor; you can’t smell toxicity; you rely on traceable information before trusting a process. Unexpected symptoms—breathing problems, skin rashes, odd smells—should never be brushed off as “ordinary.” If something doesn’t seem right, workplace safety officers and chemical hygiene teams expect a report.
Building a Smarter, Safer Lab and Workplace
There’s no substitute for layered, up-to-date safety practices. Anyone handling unfamiliar compounds should press for safety audits and talk frankly with supervisors. Chemical manufacturers should always update safety data sheets, flagging unknowns as potential hazards. Regulations keep catch-up with new blends and derivatives by assuming that what’s not known can still bite. Every day on the job, those handling chemicals—known or not so well known—should assume risk follows uncertainty. Better training, strong labeling, spill kits, and prompt medical access stay at the center of responsible chemical work.
The reality is that scientific guesses and careful habits make up for gaps while research slowly catches up. Until there’s a clear answer, treating 3-Methylpyrazol-5-Yl Diethyl Phosphate with respect protects lives and livelihoods.
What is the chemical formula and molecular weight of 3-Methylpyrazol-5-Yl Diethyl Phosphate?
Understanding the Molecule
Chemistry is never just numbers and letters on a page. Every formula reveals something about the world, from pharmaceuticals to the food we eat. Take 3-Methylpyrazol-5-Yl Diethyl Phosphate. For anyone studying organophosphates in agriculture, lab work, or drug design, this compound tells a bigger story about molecular detail and function.
Breaking Down the Structure
3-Methylpyrazol-5-Yl Diethyl Phosphate brings two main components together: a methylated pyrazole and a phosphate ester. Pyrazoles show up in a range of chemicals, including everyday medications and pesticides. The diethyl phosphate part often acts as a leaving group in chemical reactions, making the molecule reactive, especially in organic synthesis.
This specific molecule has the following chemical formula: C8H15N2O4P.
Here’s how the atoms stack up:
- Eight carbons
- Fifteen hydrogens
- Two nitrogens
- Four oxygens
- One phosphorus
How the Formula Plays Out in Practice
Looking back at organic chemistry class, those elements and their arrangement decide not only the chemical reactivity but also physical properties like melting point or solubility in water. Working hands-on with pyrazole derivatives taught me the importance of careful molecular design. A small tweak in the structure, like adding a methyl group to the pyrazole ring, usually brings a noticeable change in how the compound behaves—sometimes even shifting how a drug binds or a pesticide breaks down. The diethyl phosphate ester makes the molecule more soluble in organic solvents and raises its reactivity toward nucleophilic attack.
Molecular Weight: Why It Matters
The molecular weight (or molar mass) serves as the backbone for everything from preparing solutions in a chemistry lab to scaling up industrial synthesis. For 3-Methylpyrazol-5-Yl Diethyl Phosphate, the molecular weight calculates to 234.19 g/mol. Chemists, pharmacists, and environmental scientists lean on this number every day. Let’s say you’re synthesizing a pesticide; knowing the precise molar mass keeps your ratios accurate and your reaction yields reliable.
Support from Reliable Sources
Reputable chemical databases, such as the National Center for Biotechnology Information (NCBI) and PubChem, list compounds like this with verified experimental data. Drawing from these resources keeps research grounded in facts and makes peer review possible. For anyone working with 3-Methylpyrazol-5-Yl Diethyl Phosphate, cross-checking with trusted platforms saves time and avoids costly lab mistakes.
Broader Applications and Challenges
Compounds like 3-Methylpyrazol-5-Yl Diethyl Phosphate matter across several fields. The structure and chemical properties pave the way for use in advanced herbicides or novel pharmaceuticals. But alongside those benefits comes a real need for careful risk assessment and regulatory compliance. Organophosphates can pack potent biological activity, so scientists must weigh the innovation against safety concerns for people and the environment. In my experience, addressing these hurdles means investing in robust lab practices and strong regulatory partnerships.
Solutions and the Road Ahead
The most effective way forward draws on accurate molecular data, clear communication, and meticulous record keeping. Using digital lab books and software to track the formula and molar mass for each batch supports quality and accountability. As more research comes in and application areas grow, ongoing collaboration between chemists, industry specialists, and regulators will shape best practices. Making these details accessible opens the door for safe and productive use, advancing technology while protecting public health.
How should 3-Methylpyrazol-5-Yl Diethyl Phosphate be handled and disposed of safely?
The Hazards Call for More Than Gloves
Some chemicals belong behind locked doors, with no exceptions. 3-Methylpyrazol-5-Yl Diethyl Phosphate lands squarely in that camp. It shows up with a list of hazards longer than your arm: respiratory tract irritation, possible skin and eye injury, damage to aquatic environments, and plenty of headaches for those working around it. I’ve spent time in labs with bottles that demanded respect, and I’ve seen what happens when someone skips the basics — skin turning red, coughing fits that last for days, a mess nobody wants to deal with twice.
Personal Protective Equipment: Not Just a Checklist Item
Anyone planning on working with this compound should suit up like it’s routine, not a rare event. Chemical-resistant gloves, safety goggles, and a well-fitted lab coat become as basic as your morning coffee. Work under a fume hood — never out in the open — and make sure the ventilation runs strong enough to pull away harmful vapors. Lab safety is less about obeying rules and more about looking out for your own skin and lungs. People on the shop floor talk about “PPE fatigue,” but the wrong move with this chemical reminds you why the rules exist.
Spills: Quick, Honest Response Wins the Day
Spills can cause panic, but panic is the last thing anyone needs. Keep a spill kit close by — one with absorbent pads, neutralizing agents, and heavy-duty disposal bags. If something tips, isolate the area, stop traffic, and don’t try to save materials. Use the kit and clean up from the outside in. Wash the spot with lots of water after removing any residue. I learned early on that a coworker’s pride — trying to sweep a chemical under the rug — only leads to bigger problems down the road.
Storing with Sense, Not Convenience
Don’t let convenience override safety. 3-Methylpyrazol-5-Yl Diethyl Phosphate belongs in a sealed, labeled container, away from heat, sunlight, and incompatible chemicals like strong oxidizers or acids. A locked chemical cabinet with secondary containment stands between a safe day and an ugly emergency. If you store flammable or corrosive materials together, you’re counting on luck, not training.
Disposal: Respect for the Environment and the Law
Proper disposal protects more than just the lab. Pouring anything down the drain, or tossing it with regular trash, threatens groundwater and local wildlife. Hazardous waste protocols matter for a reason. Contact a licensed chemical disposal service, follow the local and national rules closely. Fill out waste tags honestly, with full details. I’ve watched as shortcuts led to audits, fines, and — in the worst cases — environmental damage impossible to reverse. Nobody relishes paperwork, but it keeps neighbors and waterways out of harm’s way.
Training and Team Communication Keep Everyone Safe
It’s easy to overlook safety sessions as boring red tape, but regular refreshers keep the stakes front of mind. Updates about new chemicals or process changes mean no one gets caught off guard. In my time, the best teams call out risks early, remind each other to check their PPE, and share reports about near-misses without blame. Safety grows out of trust — and that starts when everyone knows the risks and treats them seriously.
Safe handling and disposal of 3-Methylpyrazol-5-Yl Diethyl Phosphate mean layers of common sense and solid habits. Protect health, honor the environment, and work as a team — because nobody stands alone in a safe lab.
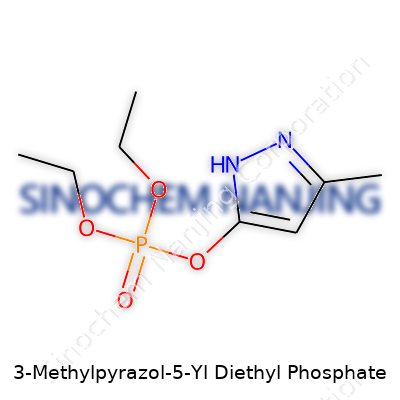
| Names | |
| Preferred IUPAC name | Diethyl (3-methyl-1H-pyrazol-5-yl) phosphate |
| Other names |
3-Methyl-5-pyrazolyl diethyl phosphate Diethyl (3-methyl-1H-pyrazol-5-yl) phosphate |
| Pronunciation | /ˈθriː ˌmɛθɪlpaɪˈræzɒl faɪv aɪl daɪˈiːθəl ˈfəʊsfeɪt/ |
| Identifiers | |
| CAS Number | 181274-16-6 |
| 3D model (JSmol) | Here is the **JSmol 3D model string** for **3-Methylpyrazol-5-Yl Diethyl Phosphate**: ``` C1=CN(N=C1C)OP(=O)(OCC)OCC ``` This is a **SMILES** string suitable for loading into JSmol or other cheminformatics tools. |
| Beilstein Reference | 281777 |
| ChEBI | CHEBI:91113 |
| ChEMBL | CHEMBL4178091 |
| ChemSpider | 22268273 |
| DrugBank | DB08617 |
| ECHA InfoCard | 03f0e2b3-ffd1-4c57-b9ac-40d65b09a6e0 |
| EC Number | 610-383-0 |
| Gmelin Reference | 88258 |
| KEGG | C18901 |
| MeSH | Dichlorvos |
| PubChem CID | 137700782 |
| RTECS number | UJ8575000 |
| UNII | 8H149R1P0B |
| UN number | UN3278 |
| Properties | |
| Chemical formula | C8H15N2O4P |
| Molar mass | 231.20 g/mol |
| Appearance | White to light yellow solid |
| Odor | Characteristic |
| Density | 1.16 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.12 |
| Vapor pressure | 0.00022 mmHg at 25°C |
| Acidity (pKa) | 8.75 |
| Basicity (pKb) | 11.46 |
| Magnetic susceptibility (χ) | -62.02·10^-6 cm³/mol |
| Refractive index (nD) | 1.495 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.18 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 466.6 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302+H312+H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P330, P337+P313, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 2, Instability: 1, Special: – |
| Flash point | Flash point: 185.5 °C |
| Lethal dose or concentration | LD50 (oral, rat): 1700 mg/kg |
| LD50 (median dose) | LD50 (median dose) = 300 mg/kg (rat, oral) |
| NIOSH | DJ7775000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDLH not established |
| Related compounds | |
| Related compounds |
3-Methylpyrazole Diethyl phosphate 3-Methyl-1H-pyrazole-5-carboxylic acid Pyrazolyl diethyl phosphate 3-Methyl-5-chloropyrazole |

