3-Methylbutanal: Its Role, Science, and Where It's Headed
Historical Development
The story of 3-methylbutanal traces back to advances in organic chemistry and food science research from the early 20th century. Scientists working to pinpoint the sources of flavor and aroma in foods stumbled upon this compound in malt, chocolate, bread, and cheeses. I remember reading about early analytical chemists who relied on sensory panels and primitive gas chromatography columns. Their straightforward goal: find out why certain foods carried a malty, nutty, or toasted note. It’s fascinating to see how this work was not just about satisfying curiosity—it shaped the flavorings industry, and paved the way for the regulatory frameworks that guide food additives now. This background matters because it marks the shift from intuition-based flavor creation to a scientific approach, ultimately improving consumer safety and flavor authenticity.
Product Overview
3-Methylbutanal, also known by various synonyms in scientific literature, forms part of the family known as aldehydes. It’s a clear, colorless liquid at room temperature, with an odor most people would describe as malty, chocolate-like, or even slightly fruity. Today, this molecule finds strong demand in both the food industry as an aroma component, and the fragrance sector, as well as in some specialty chemical applications like pharmaceutical intermediates. Companies synthesizing flavors or reformulating chocolate alternatives know the impact even trace levels of 3-methylbutanal can have on taste. Its practical role goes beyond food; it appears in tobacco processing, perfumery, and the development of certain solvents or fine chemicals.
Physical & Chemical Properties
If you roll up your sleeves in any modestly equipped chemistry lab, it’s easy to spot how 3-methylbutanal stands out. Its boiling point hovers around 90–92 °C, and it sports a relatively high vapor pressure—so it evaporates quickly and spreads its scent even at low concentrations. Density measures lean close to 0.8 g/cm³. This volatility is what makes it pop out in sensory evaluations or GC-MS analysis. It blends well with other savory or roasted notes, supporting both background flavors and headspace aromas. Its solubility leans towards organic solvents, and it has a knack for integrating with oils and emulsions, making it straightforward to work into processed foods and fragrances alike.
Technical Specifications & Labeling
In real-world production, technical specs demand attention to minute contaminants. Users expect purity thresholds well above 95% for reliable sensory performance and safety compliance. Labeling conventions stick to IUPAC or common synonyms, and allergen status must be stated per region. Regulatory agencies like the FDA and EFSA categorize 3-methylbutanal under GRAS (Generally Recognized as Safe) for food use, but the emphasis lands squarely on batch traceability and pre-market review. In practice, this means rigorous sensory checks, testing for unwanted impurities, and tight labeling—vital not just for regulatory harmony but for consumer confidence and shelf-life stability.
Preparation Method
Most commercial 3-methylbutanal comes from controlled oxidation of 3-methylbutanol, itself a byproduct from fermentation processes. Speaking from experience in an academic context, bench-top methods mirror industrial ones at small scale: you feed air or oxygen over the alcohol, using mild catalysts, and collect the aldehyde after distillation. Biotechnological routes attract growing interest. Some companies explore genetically optimized yeast or bacterial fermentation, which could drop production costs and carbon footprint. Even so, old-school chemistry still dominates the volume market because it’s reliable, scalable, and easy to monitor with current technology.
Chemical Reactions & Modifications
The chemistry behind 3-methylbutanal isn’t glamorous, but it offers flexibility. Aldehydes in general bring reactivity. In labs, you see it reduced to the corresponding alcohol or further oxidized to the acid—these byproducts play different roles in food and fragrance formulations. Condensation reactions hook it to other small molecules, creating more complex aroma structures. This reactivity extends risk but also opens doors for nuanced control. The same ease of transformation that gives it power in a recipe or perfumery blend demands careful handling in production, and good process oversight to prevent unwanted side reactions.
Synonyms & Product Names
Browsing through chemical catalogs or regulatory listings, it’s clear the molecule goes by several names: 3-methylbutanal, isovaleraldehyde, and β-methylbutyraldehyde lead the list. Each synonym flags a different context: scientific publication, industrial bulk sale, or consumer-facing product. Ingredient lists, material safety sheets, and flavor labeling consistently refer to either ‘isovaleraldehyde’ or ‘3-methylbutanal’, depending on the jurisdiction. This variety in naming can trip up even seasoned professionals, underlining the value of CAS number checks and multi-lingual regulatory advice.
Safety & Operational Standards
Handling aldehydes brings practical safety needs. 3-Methylbutanal vapors can irritate eyes and mucous membranes even at low parts-per-million levels, so chemical workers rely on gloves and decent ventilation. In bulk transport, storage tanks get extra attention to avoid leaks or vapor build-up, since the low boiling point makes for easy evaporative loss. In regulated markets, storage aligns with flammable liquids standards, and production facilities need spill controls and exposure monitoring. From a food safety angle, reliable traceability and periodic purity checks ensure that what goes into a product doesn’t surprise consumers downstream.
Application Area
Step into any flavor lab or processed food plant and you’ll bump into 3-methylbutanal somewhere. Its biggest fans come from the chocolate, nut, malt, and baked goods sectors—where its aroma boosts authenticity or adds richness. Perfume blenders lean on it for specialty nutty or “gourmand” scents. It occasionally pops up in imitation tobacco blends or to mask off-notes in pharmaceuticals. In research, its significance extends into sensory analysis, where scientists use it as a marker for studying flavor formation and food stability.
Research & Development
Much of today’s R&D fights for better ways to make or control 3-methylbutanal in foods. There’s real momentum in developing yeasts and bacteria that pump out more of the aldehyde during fermentation, or that help convert off-flavor precursors into more desirable profiles. Analytical chemists keep pushing for more sensitive and precise methods to monitor it—down to parts per billion—in everything from cocoa nibs to hop-dried beer. The knowledge gained feeds into cleaner labeling, reduced off-flavors, and new market trends like “natural fermentation-derived” flavor ingredients. Tech advances, like miniaturized detectors, could soon put routine quality checks into the hands of small producers, not just big labs.
Toxicity Research
Toxicologists and regulatory scientists have clocked thousands of hours parsing the risks associated with aldehydes. Most current studies point to low toxicity at the doses consumers actually encounter. Still, lab animals given extreme amounts exhibit mild liver stress, so global safety assessments keep a close watch on maximum allowable intake—often at the microgram per kilogram level for finished foods. This aligns with experience reported in food industry incident reports: improper handling or mixing can occasionally lead to flavors that breach threshold levels, but rare are the documented acute health effects at approved use rates. All the work here supports ingredient safety re-evaluations and justifies periodic re-appraisal as food science marches on.
Future Prospects
Looking forward, biotechnology and green chemistry stand ready to transform how 3-methylbutanal gets made and managed. Fermentation-based roads cut down on waste streams, lower energy costs, and pilot plant results hint at ways to tune the flavor more precisely than chemical synthesis allows. Food companies search for near-identical substitutes or more stable analogs that perform better in high-heat processing or extended shelf life products. Research continues into safer packaging to slow 3-methylbutanal loss or unintended reaction. Regulatory harmonization, global safety evaluation, and advanced analytics stand poised to give both producers and consumers a clearer picture of what’s in that bar of chocolate or loaf of bread. This molecule’s journey ties in with consumer push for transparency, safer food, and tastier experiences on a global scale.
What is 3-Methylbutanal used for?
The Hidden Ingredient in Your Favorite Foods
3-Methylbutanal shows up in places most folks never expect. Open a package of cheese or take a bite of dark chocolate—there’s a good chance you’re tasting it. This molecule carries a nutty, malty aroma that works wonders in food and drinks. Instead of thinking about complex formulas, picture the scent of toasted bread or the rich smell when coffee roasts. Many of those flavors come from 3-Methylbutanal. In food science, it’s valued for making products smell and taste more inviting.
Cheese makers often monitor this compound closely. During cheese fermentation, certain bacteria produce it naturally, and the result adds depth to the final flavor. I remember visiting a local creamery years back. The cheesemaker explained how tweaking the aging process could turn the rind from bland to buttery, just by influencing the bacteria responsible for 3-Methylbutanal production. The difference between a plain brie and a top-shelf version often comes down to small amounts of this molecule. Adding it directly isn’t common in traditional cheese, but the principle holds true. In mass-market snacks or chocolates, the synthetic version can help replicate the deep notes missing from industrial processing.
Chocolate companies use 3-Methylbutanal as well. Its aroma profile pairs well with cacao, making bars smell richer. Food scientists lean on it to bring up those desirable roast and caramel notes. In my own experience baking at home, fresh bread or cookies coming out of the oven fill the air with similar scents. I later learned that many of those aromas link back to 3-Methylbutanal, part of the Maillard reaction—the same complex chemical process that turns food brown and delicious when it’s baked or fried.
Crafting Scents in the Fragrance World
Perfume makers take a lot of cues from the kitchen. 3-Methylbutanal shows up in fine fragrances as well, adding a nutty roundness to base notes. On a tour of a fragrance lab, I once had the chance to sniff pure samples. It’s startlingly strong and almost oily, but blended with woody or spicy elements, it mellows and deepens the scent. For perfumers, this provides a way to mimic the smell of aged wood, tobacco, or bread. Scent research studies—such as those published in journals like Flavour and Fragrance Journal—often list 3-Methylbutanal among the powerful aroma compounds critical to natural, complex smells.
Beyond Food and Fragrance: Industry Uses
Some industrial labs also use 3-Methylbutanal as a chemical building block. Small quantities help make pharmaceuticals, where it forms part of more complicated drug molecules. It also acts as an intermediate step in producing some flavors and scents for everyday goods. While these uses matter less to the public, the fact that a compound found in cheese ends up in certain medications highlights how chemistry connects different industries. The World Health Organization has cataloged its safety profile, and regulatory bodies keep a close watch to ensure it doesn’t end up in products at unsafe levels.
Safety, Regulation, and the Future
Concerns crop up when a compound appears so widely. Too much 3-Methylbutanal can be toxic—something workplaces monitor closely. The FDA and European Food Safety Authority set guidelines for how much is allowed in food and fragrance. Scientific groups keep tracking its effects through animal studies and exposure assessments. Keeping these rules updated matters. It’s easy to take flavor and scent for granted, but safety demands vigilance.
Moving forward, food scientists keep searching for ways to boost natural flavor in processed food without overusing artificial additives. Modern fermentation techniques and careful sourcing could let companies rely less on synthetic boosters. At the same time, regular testing and transparent ingredient labeling help give peace of mind for folks who want to know what’s in their food and products.
Is 3-Methylbutanal safe to handle?
Understanding 3-Methylbutanal in Everyday Terms
Anyone who’s ever walked through a food processing plant or dabbled in flavor chemistry has likely heard about 3-Methylbutanal. This compound pops up naturally in various foods and beers, offering that tangy malty note bakers and brewers like chasing. People don’t talk about it as much as other chemicals, but its scent always turns heads, especially in fresh bread or chocolate-making environments. As someone who’s spent a fair amount of time figuring out how these flavors come together, I’ve seen workers grow cautious around 3-Methylbutanal because it straddles the line between natural ingredient and chemical hazard.
Known Risks Taken Seriously
Despite the friendly aroma, 3-Methylbutanal shouldn’t be taken lightly in bulk. Direct exposure leads to irritation; eyes and skin feel the sting, airways get scratchy, and breathing in vapors can set off coughing fits. The flashpoint sits pretty low—people with experience in the lab respect its flammability. Breathing plenty of fresh air always makes sense, whether mixing large amounts or just cleaning up spills. Gloves, goggles, and good ventilation aren’t optional. It’s like handling strong cleaning products; just because you find some of them under your sink doesn’t mean they’re safe in all situations or in big quantities.
Clear-cut Facts From Studies
The scientific community has studied this compound. 3-Methylbutanal occurs in food, but concentrated forms raise red flags. The US National Institute for Occupational Safety and Health (NIOSH) lists it as hazardous if inhaled or if it touches the skin. No data supports cancer concerns yet, but laboratory tests on animals turn up signs of irritation and—in hefty doses—organ strain. European regulations classify it as hazardous, with labels for skin, eye, and respiratory tract irritation. Most guidance advises minimizing exposure and sticking to small application areas with proper protective gear.
The Importance of Context and Scale
There’s a world of difference between the whiff you catch from fresh bread and exposure in a chemical facility. I’ve watched folks become complacent, thinking “it’s natural, what’s the harm?” The harm shows up when mishandling concentrated forms. Any chemical used in food science or manufacturing—natural or not—calls for understanding and respect. The same compound that gives flavor can also irritate the lungs or ignite if someone forgets basic safety steps.
Practical Precautions That Work
Training pays off. Workers need clear rules—keep containers sealed, store in cool ventilated spaces, and never skip gloves or eye protection. Good ventilation keeps vapor levels down, especially in hot or crowded rooms. Know how to clean up spills safely—no one wants an accident lingering in the air. Emergency eye wash stations and shower access don’t take up that much space, but they make a world of difference during a mishap. Experienced supervisors run safety drills because muscle memory takes over during real accidents. I’ve watched lives saved by habits drilled during slow shifts rather than frantic reactions during emergencies. Simple steps bring peace of mind to everyone, from seasoned researchers to the newest temp worker.
Supporting a Culture of Safety, Not Fear
Knowledge and preparation beat worry. 3-Methylbutanal lives in a gray area—part of your favorite foods, but still a chemical hazard behind the scenes. The right approach sets a workplace apart. Calm, well-trained workers handle even the trickiest compound safely. People who stay curious, who ask questions, and who respect every little warning sign make for a crew that goes home safe, every time.
What is the chemical formula of 3-Methylbutanal?
Getting to the Root: Recognizing 3-Methylbutanal
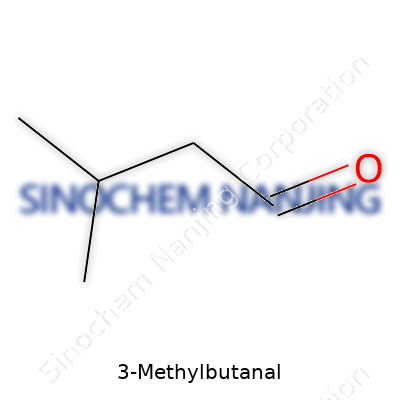
3-Methylbutanal, often popping up in conversation around flavorings or fermentation, carries the chemical formula C5H10O. Anybody who has spent time in a lab or paid attention to food chemistry has probably crossed paths with this compound. It belongs to the family of aldehydes, a group well known for their impact on aroma and taste. The structure has five carbon atoms, ten hydrogen atoms, and a single oxygen atom, which all stack together to create a compound with unique reactive potential.
Scent, Taste, and the Human Touch
People connect with 3-Methylbutanal most often through their noses: it’s one of the key compounds that give bread its freshly baked aroma. Brewers, bakers, and even folks making fermented sausages rely on it for depth in flavor. The molecule forms during the breakdown of amino acids like leucine—basic building blocks of protein—by yeast or bacteria. It’s not just some dry academic point: taste and smell drive food experience, and 3-Methylbutanal steers results in kitchens and food labs.
Safety, Sensation, and Small-Scale Chemistry
Before anyone tosses around chemicals for fun or business, safety ranks high. C5H10O, while present in minuscule natural amounts, can be an irritant at higher levels. This matters since industrial settings and research facilities sometimes use it in concentrated form. Safety data sheets flag potential hazards, reminding workers to wear gloves and use good ventilation. This extends beyond labs; food technologists who add flavorings to enhance processed foods must also consider regulations and thresholds.
Facts Behind Food and Fragrance
The International Fragrance Association lists 3-Methylbutanal among compounds approved for use in perfumes and flavorings, with standards in place to avoid overexposure. Research papers, such as those in the Journal of Agricultural and Food Chemistry, back up its significant presence in fermented foods and beverages. Anyone in the flavor industry recognizes its heavy lifting in contributing nutty, malty notes, especially in chocolate, cheese, and beer.
Facing the Challenges: Science and Regulation
Sometimes the issue isn’t just scientific, but practical: how do you balance flavor impact vs. regulatory safety? Food chemists work with the FDA and EFSA rules, staying under daily intake limits that scientific studies support. Quality assurance labs spend hours measuring these compounds to make sure products stay consistent and safe. Doing this work takes more than just knowing the formula—it demands constant monitoring and advanced testing tools.
Solutions on the Table
If taste quality dips, or if unexpected amounts show up, teams look back at fermentation conditions. Changes in temperature, pH, or bacterial strains can swing the levels of 3-Methylbutanal higher or lower. Sensor technology, along with machine learning models, now helps find the sweet spot in real time. Regular audits, employee education, and staying plugged into food safety research all play a part in making sure this chemical stays an asset, not a liability.
What are the storage requirements for 3-Methylbutanal?
Why Proper Storage Matters
3-Methylbutanal isn’t just another chemical name to skim past on a safety data sheet. It’s a substance that finds itself in the flavor and fragrance industries, and anyone who has spent time around these sectors knows how strict handling rules matter. It’s volatile, known for its distinctive, pungent odor that can quickly fill a whole room. Spills or improper storage aren’t just an inconvenience—they can create safety problems, impact product quality, and rack up spiking costs.
The Concrete Storage Conditions
3-Methylbutanal calls for cool, well-ventilated storage away from any sources of heat or ignition. In practice, this means keeping it in a chemical storage fridge or dedicated cabinet—not some dusty, overcrowded shelf in a back room. Most chemical suppliers recommend a temperature below 25°C. Realistically, a stable, low-temperature environment slows down its degradation and limits the buildup of dangerous vapors.
Ventilation isn’t just on the checklist—it keeps the room from turning into a headache zone. Open flames, sparks, or even equipment that gets warm during use should never share space with 3-Methylbutanal. The stuff can ignite at a low flash point, around 45°C, so the risk extends beyond the rare chemical lab accident.
Packing and Container Choices
Anyone who’s ever handled aldehydes knows how tightly you need to seal them up. Airtight, corrosion-resistant containers made from materials that don’t react with aldehydes are the only real option. Glass is reliable here, but some plastics react or break down, especially if stored for long periods. You hear about leaky seals more often than you’d think—one mistake can mean ruined stock, lost time, or even an emergency response.
Labels also matter far more than most people realize. The information needs to be legible and resistant to chemical smudging or peeling. I remember walking into a storeroom early in my career to find a dozen almost-identical bottles, old marker ink faded to gray—nobody wants to play “guess the vapor” with volatile organics.
Risks You Can’t Ignore
Exposure isn’t something to brush off. 3-Methylbutanal vapors irritate eyes, skin, and the respiratory tract. If a container isn’t well sealed, or storage areas aren’t monitored, even short exposure can raise alarms in a workforce. A friend in quality control told me about a plant shutdown triggered by improper aldehyde storage; the clean-up cost far outweighed any savings from skipping proper procedures.
Proper storage cuts down on shipment rejections, preserves product value, and, most importantly, protects workers. Employers who ignore established guidelines, like those outlined by the National Fire Protection Association (NFPA) and the Occupational Safety and Health Administration (OSHA), set themselves up for bigger headaches down the line.
Actionable Solutions
Keep regular inventory checks, and don’t trust memory when it comes to chemical stocks. Training every staff member who might access the storage area can cut down on accidents that come from a simple misunderstanding. Installing vapor alarms or even basic temperature monitors helps catch problems before they snowball. Keeping personal protective equipment—nitrile gloves, goggles, lab coats—within arm’s reach ensures nobody skips safety steps in a rush.
For busy workplaces with high throughput, a digital chemical management log makes it easier to track expiry dates, storage temperatures, and cleaning cycles. In my own experience, the fastest way to make safety the standard is to show how much time and money good storage habits actually save.
Storing 3-Methylbutanal isn’t an abstract compliance box to check. It’s about respect for the reality of risk—and a guarantee of quality for every batch that eventually hits the market.
What are the potential health effects of exposure to 3-Methylbutanal?
Understanding 3-Methylbutanal in Daily Life
Plenty of folks have never heard of 3-Methylbutanal, but this compound turns up in more places than most realize. You’ll find it in the aroma of some cheeses, cocoa, and certain beers. Food industries use it for flavoring, and it pops up in a few perfumes to add that nutty, chocolatey smell. Having worked in a lab where food safety mattered, I learned to respect even the smallest trace chemicals. People often forget that just because something smells nice, it doesn’t always mean it’s entirely harmless if inhaled or touched often enough.
Short-Term Health Effects: What Science Tells Us
I once handled a batch of flavor chemicals that included 3-Methylbutanal, so I paid attention to how my body reacted. At high concentrations, the strong, sweet odor can overwhelm your nose and eyes, leaving you coughing and, for some folks, dealing with a touch of nausea. According to research and toxicology reports, inhaling a lot of this compound in a poorly ventilated space irritates the eyes, throat, and respiratory tract. Lab workers and factory staff sometimes experience dizziness or headaches when levels get too high. The science lines up with what I’ve seen in workplace incident reports—excessive exposure leads to discomfort and acute symptoms that fade when you leave the area.
Long-Term Health: A Less Clear Picture
Evidence on what happens with low-level exposure over months or years gets sketchy. Regulatory bodies like OSHA and NIOSH classify 3-Methylbutanal as a chemical worth monitoring, but the data set isn’t as deep as with heavy hitters like benzene or formaldehyde. Some rodent studies indicate possible issues with long-term exposure, such as mild effects on liver and kidney tissue, but these came from doses much higher than typically found in the food industry. In my own experience, I’ve seen vigilance about gloves and goggles, not because this compound is especially dangerous at everyday levels but because you never want to risk a slow build-up or accidental spill.
Vulnerable Groups and Real-World Risks
People with asthma or allergies may react sooner and more severely to even moderate airborne concentrations. Children tend to breathe more air per pound than adults, boosting their risk if they get caught where a spill happened or in areas with lots of flavoring agents in the air. I recall one factory audit where a batch mishap sent fumes through the production floor; the most affected were workers with a history of breathing problems. Protecting employees starts with quick action—ventilating the space, evacuating if someone starts coughing uncontrollably, and using chemical-specific respirators instead of cloth masks.
Steps Toward Healthier Workplaces and Safer Products
From what I’ve seen, practical solutions begin with basic hygiene: always work in well-ventilated areas and stick to safety protocols. The industries using 3-Methylbutanal can cut down on accidental exposures by keeping storage containers sealed tight, maintaining air quality, and training staff to spot early symptoms. For the average consumer, risks stay low in foods and perfumes, but folks working with this chemical daily deserve real transparency and up-to-date Material Safety Data Sheets. More independent studies on low-level, chronic exposure would clear up lingering doubts, especially as flavor and fragrance use grows. Until then, good science and better safeguards let everyone breathe a little easier around this potent-smelling compound.
| Names | |
| Preferred IUPAC name | 3-Methylbutanal |
| Other names |
Isoamyl aldehyde Isovaleraldehyde 3-Methylbutyraldehyde |
| Pronunciation | /ˌθriːˌmɛθɪlˈbjuːtən.æl/ |
| Identifiers | |
| CAS Number | 590-86-3 |
| 3D model (JSmol) | `3d:JSMOL-c1cc(C)ccc1` |
| Beilstein Reference | 635923 |
| ChEBI | CHEBI:16170 |
| ChEMBL | CHEMBL31824 |
| ChemSpider | 6193 |
| DrugBank | DB04161 |
| ECHA InfoCard | 18e79c41-ebe4-4403-8fea-4e67bc5f1c04 |
| EC Number | 200-636-0 |
| Gmelin Reference | 871659 |
| KEGG | C05925 |
| MeSH | D008772 |
| PubChem CID | 31253 |
| RTECS number | EL8575000 |
| UNII | 8VZQ9R28W1 |
| UN number | UN2379 |
| Properties | |
| Chemical formula | C5H10O |
| Molar mass | 86.13 g/mol |
| Appearance | Colorless liquid with a strong, pungent, fruity odor |
| Odor | fruity, chocolate, pungent |
| Density | 0.802 g/mL |
| Solubility in water | slightly soluble |
| log P | 0.97 |
| Vapor pressure | 3.67 kPa (at 20 °C) |
| Acidity (pKa) | 14.72 |
| Basicity (pKb) | 14.43 |
| Magnetic susceptibility (χ) | -53.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.387 |
| Viscosity | 0.738 cP at 25 °C |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 234.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -184.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2586.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | -18 °C |
| Autoignition temperature | 234 °C |
| Explosive limits | Explosive limits: 1.7–11.0% |
| Lethal dose or concentration | LD50 (oral, rat): 730 mg/kg |
| LD50 (median dose) | LD50 (median dose): 630 mg/kg (rat, oral) |
| NIOSH | ENQ7790000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.06 ppm |
| Related compounds | |
| Related compounds |
Isovaleraldehyde Isovaleric acid Isovaleryl chloride 3-Methylbutan-1-ol 3-Methylbutyl acetate |

