3-Methylbenzonitrile: Digging Beneath the Surface
Historical Development
Chemicals with “benzonitrile” in their name trace their story back to early aromatic chemistry, when scientists first learned how to tweak rings and tinker with side chains. The journey toward 3-methylbenzonitrile—a molecule sporting both a nitrile group and a methyl on the benzene ring—reflects how curiosity, need, and technical know-how built the modern chemical toolbox. Nitriles entered the scene during the 19th century as chemists sorted out ways to convert aromatic hydrocarbons, understanding little about molecular arrangement beyond what boiling points and smells could tell. Over time, researchers figured out how adding different groups to the benzene ring shifted reactivity and produced brand-new families of compounds. That’s how 3-methylbenzonitrile found its niche: distinct enough to open applications beyond its big sibling benzonitrile, but accessible through the industrial-scale organic reactions that marked the last century of chemical development.
Product Overview
3-Methylbenzonitrile isn’t splashed across consumer labels or household cleaners, but anyone who’s worked in a chemical lab will recognize its sharp aroma and the tight bottle cap that signals something demanding respect. It often stands among the small family of niche intermediates that slip quietly into countless syntheses, especially where a sturdy aromatic backbone and a reactive nitrile group are needed. Chemists reach for it when crafting complex pharmaceuticals, tuning polymer properties, or building blocks that underpin dyes and specialty chemicals. Even though not a staple for the average person, industries, researchers, and industrial chemists depend on its predictable behavior under pressure, heat, or in the presence of aggressive reagents.
Physical & Chemical Properties
Folks new to the molecule might notice right away that 3-methylbenzonitrile comes as a colorless, sometimes slightly yellowish liquid, stubbornly holding onto its aromatic smell. The methyl group at the third position on the benzene ring makes it less straightforward than plain benzonitrile. That tweak gives unique reactivity, acidity, and electron distribution, a detail synthetic chemists—and graduate students sweating over NMR readings—come to value. The boiling point stands noticeably higher than many simpler aromatics, making distillation a chore that demands cooling water and patience. In a flask, it resists breakdown in light and air, holding stability under standard lab conditions, but reacts smartly with strong acids or bases, and forms tight bonds in organic synthesis.
Technical Specifications & Labeling
Chemical suppliers mark their bottles with detailed purity grades—anything below 97% can spell disaster for researchers chasing precision. Standard labeling distinguishes between technical, laboratory, and synthesis grades, giving buyers an idea about trace water or unexpected aromatics lurking in the mix. These details matter less to high-volume manufacturers and much more to those making micro-quantities for high-stakes reactions or analytical work. Labs receive materials labeled with hazard pictograms, underscores about vapor toxicity, instructions to minimize inhalation, and reminders about personal protective equipment. Even an old hand in the lab reminds themselves with a quick check of flammability and storage warnings, especially in academic and startup settings where mistakes gain attention fast.
Preparation Method
Direct preparation tends to lean on classical methods, often starting from toluene with a clever tweak—a bromination of the methyl group, then switching out bromine for a cyano group using copper catalysts. Chemists have come a long way since the first, clumsy attempts; these days, anyone with a modest setup and the right safety gear can whip up batches that beat commercial standards. Still, at scale, manufacturers jump through hoops to minimize byproducts and dial in reaction conditions to avoid over-bromination, prevent air oxidation, and purify the target compound in good yield. Waste disposal and solvent recycling remain annoying hurdles, shaping both cost and environmental footprint, and pushing R&D labs to dream up greener routes year after year.
Chemical Reactions & Modifications
The value of 3-methylbenzonitrile pops up in what one can transform it into. The nitrile group works like a chemical handle, ready to trade up for amides, acids, or amines through hydrolysis, reduction, or substitution. Medicinal chemists lean hard on the nitrile’s ability to connect with drug scaffolds, acting as a gateway into more elaborate ring systems or chiral centers. Its methyl group isn’t just decorative either; it steers reactivity to favor certain regioselective attacks, so creative chemists use it as a tactical tool when trying to guide tough reactions toward the products they want. More than just a stepping-stone, 3-methylbenzonitrile becomes a launchpad for everything from anti-fungal agents to photographic chemicals, sometimes forming the nucleus for more elaborate heterocycles in pharma pipelines.
Synonyms & Product Names
Step into the world of fine chemicals and product catalogs, and the molecule may show up as 3-cyanotoluene, m-tolunitrile, or by the less catchy “methylbenzonitrile (meta).” Each name reflects subtle distinctions in how chemists, suppliers, and industrial players track their products, usually influenced by the reaction being carried out or the starting material used. The modest jumble of names leads anyone in the field to double-check CAS numbers before closing a purchase order, since a missed digit could shift a project weeks off course.
Safety & Operational Standards
Working with aromatic nitriles takes more than just careful pouring—3-methylbenzonitrile demands gloves, goggles, and good fume hood ventilation. It’s not the kind of chemical that does visible harm on the spot, but enough gets on the skin or in the air, and headaches or sore throats often follow. A small spill during transfer, left untended, can turn an ordinary workspace into an all-hands emergency evacuation. Strict lab rules insist on secondary containment, precise labeling, and keeping incompatible materials (strong acids or oxidizers) stored apart. The chemical world has learned lessons too often the hard way, so even experienced chemists check procedures and keep material safety data sheets nearby, a routine that never feels like overkill.
Application Area
3-Methylbenzonitrile has carved out roles in making pharmaceuticals, specialty dyes, and custom chemical intermediates, where its aromatic framework lends both stability and the right sort of reactivity. Pharmaceutical chemists looking to build molecules that bind selectively across biological targets use the methyl and nitrile groups to fine-tune electronic properties and improve drug-like character. Dye chemists appreciate the way this compound’s structure lets them adjust shades or improve colorfastness in textiles. Polymer chemists integrate it to give plastics unique resistance to breakdown, or to create resins that handle heat and light with ease. Behind the headlines, countless research publications and patent filings cite 3-methylbenzonitrile as an essential ingredient in building the new and the vital.
Research & Development
For all its established roles, research teams keep looking for ways to improve both how 3-methylbenzonitrile gets made and what it gets turned into. A lot of R&D centers on reducing waste, speeding up reactions, and developing catalysts that work under milder, safer conditions. Sustainable chemistry projects focus on minimizing hazardous solvent use and designing more selective pathways so fewer byproducts need disposal. Recent years have seen a wave of work in catalysis and green chemistry, where researchers swap out legacy metals for reusable or “earth-abundant” catalysts. In the application space, ongoing research explores how subtle tweaks to the molecule unlock new pharmaceuticals and responsive materials—small adjustments in the methyl or nitrile placement shift biological activity in sometimes dramatic ways.
Toxicity Research
The toxicity of nitriles presents a long-standing challenge, drawing attention especially from those keeping an eye on occupational health and regulatory compliance. Though 3-methylbenzonitrile doesn’t top lists of acute hazards, repetitive exposure or poor handling have taught many labs what headaches or nausea can signal. Studies catalog effects in mammalian systems, monitoring liver and kidney function post-exposure and tracking metabolic pathways to see if breakdown products compound the risks. The chemical industry’s push for safety data relies on animal studies and environmental surveys, supporting workplace exposure guidelines and informing emergency response strategies. Some research has aimed at finding alternatives or safer derivatives, but, so far, careful handling and robust protocols offer the most consistent defense.
Future Prospects
The future of 3-methylbenzonitrile rides on two fronts: better synthesis and new fields of use. Efficiency improvements—especially those that cut costs, boost yield, or ease regulatory headaches—shape its industrial prospects. Academic and industrial labs are racing to find catalytic systems that streamline reactions, minimize toxic waste, and give more control over purity. On the horizon, possibilities include medical diagnostics, advanced materials, and chemical sensors, all leaning on the compound’s versatility as a building block. As environmental standards tighten, chemists will push for greener processes and safer alternatives, likely steering the molecule’s story in directions impossible to guess from an early textbook. Until something replaces the balance of reactivity and stability it offers, 3-methylbenzonitrile will keep finding work in places where chemistry’s complexity meets the stubborn needs of the real world.
What is 3-Methylbenzonitrile used for?
What It Is
3-Methylbenzonitrile may sound unfamiliar, but it means a lot to folks working in chemistry and manufacturing. It's an organic compound featuring both a methyl group and a nitrile group attached to a benzene ring. You’ll find it as a pale liquid, kind of sharp in odor, and surprisingly persistent in the world of chemistry. Its CAS number is 620-22-4, which makes tracking it in technical literature a bit easier for scientists.
Why Companies Value 3-Methylbenzonitrile
Ask someone at a specialty chemical supplier or a pharmaceutical lab about this compound, and they’ll mention its role as a building block. In the pharmaceutical space, scientists use it to create all sorts of potential drug candidates. The methyl group and nitrile functionality offer options for shaping and constructing more complex molecules. Sometimes, new medicines begin life from simple compounds like this.
I remember chatting with a university researcher who joked that the the real story of drug discovery involves a lot of these “useful yet boring” molecules, which get pieced together, changed, torn apart, and finally transformed into something that can save a life or help someone recover faster from illness.
Main Uses in the Real World
Organic synthesis often depends on chemicals that play supporting roles. 3-Methylbenzonitrile doesn’t grab headlines, yet it turns up in projects where chemists want to add a “nitrile” function to a molecule. The nitrile group gets converted to other things—a carboxylic acid, amide, or amine, to name a few. These changes lead to new flavors, dyes, agri-chemicals, and medicines. I once visited a flavor lab and saw this compound on the shelf, waiting to be fed into a synthesizer.
Dye manufacturers also rely on intermediates like 3-Methylbenzonitrile. Getting the chemistry right lets people produce brighter or longer-lasting colors in fiber and textiles. Sometimes, new specialty coatings or optical brighteners need exactly this type of building block to work well. A deep dive into patents for dyes and colorants will show its presence behind the scenes.
Safety and Environmental Perspective
Handling chemicals in this class doesn’t come without health and environmental risks. 3-Methylbenzonitrile isn’t the worst offender, but exposure in labs or factories brings the potential for eye or skin irritation. Proper gloves, goggles, and working in a fume hood can reduce issues. Regulations in many countries require industrial companies to keep strict control, track usage, and prevent release into wastewater or air. City water departments rely on this kind of vigilance to ensure residues don’t end up in drinking supplies or rivers.
Waste disposal is an ongoing challenge, especially in countries with growing chemical industries but weaker enforcement. Responsible manufacturers often set up recycling and incineration systems that reduce environmental load. Open communication about what’s in use, and clear public records, keep neighbors safer and give consumer advocates more trust in the system.
Looking Forward
The world’s appetite for advanced medicine, safer dyes, and versatile materials keeps demand for intermediates like 3-Methylbenzonitrile steady. Continued research into greener synthesis is a promising trend. Academic chemists and industrial firms alike keep hunting for lower-impact pathways using renewable feedstocks or more efficient routes, aiming for less waste and cleaner air.
It’s easy to overlook chemicals like this beneath the glamour of modern drug launches or high-tech textiles, but every finished product rests on solid, reliable chemistry. The story of 3-Methylbenzonitrile illustrates how true progress depends on the unsung links that hold big innovations together.
What is the chemical formula of 3-Methylbenzonitrile?
Understanding 3-Methylbenzonitrile: More Than Just a Set of Letters
People familiar with chemistry know the satisfaction of cracking a compound’s molecular puzzle. 3-Methylbenzonitrile, with the chemical formula C8H7N, gives both students and scientists a real reason to pay attention. Here’s a molecule with a benzene ring foundation, one methyl group clinging to the third carbon, and a cyano group stretching out from the first carbon. It’s easy to overlook such a small change in a molecular structure — a swapped group here, an added chain there — but these tweaks have pushed research, industry, and medicine in unexpected directions.
Why Does a Chemical Formula Like C8H7N Matter?
Lots of folks ask why anyone outside a lab would care about something like 3-Methylbenzonitrile. I’ve sat through nights reading research about substituted benzenes. In much of modern organic synthesis, every methyl and nitrile group changes a molecule’s behavior. Chemists use this compound as a vital starting point for making dyes, pharmaceuticals, and specialty materials. A researcher tuning a pharmaceutical’s performance can appreciate how shifting a methyl group on a benzene ring creates a new drug candidate from a simple chemical tweak.
A formula like C8H7N isn’t just trivia. It points to exactly how many carbon, hydrogen, and nitrogen atoms come together. With this information, chemists decide the solvents needed for purification, predict the compound’s reactivity, or even run computer models to test its performance. For instance, swapping out that methyl group onto a neighboring carbon atom makes a whole new molecule, and with that comes shifts in properties like melting point, boiling point, and toxicity.
Safety, Supply, and Staying Informed
Industrial demand often starts with molecules like 3-Methylbenzonitrile. Growing up around a family member running a small lab, I saw firsthand how the safety sheets always included the chemical formula. That practice stuck for a reason. Accidents become far less likely when everyone knows what each flask contains. When regulations change, authorities depend on unique formulas to track import and storage. Quality control and legal compliance hinge on smart, transparent records.
Looking for Better Ways Forward
People constantly search for greener ways to make and use these chemicals. Factories and startups now push for techniques that cut down on waste by using more efficient catalysts or recycling solvents in reactions using 3-Methylbenzonitrile. University teams drive this research, often funded by grants focusing on sustainable chemistry. The core remains the same: a compound with formula C8H7N, whose exact structure lets scientists test new ideas, minimize hazardous byproducts, and tighten safety controls.
Sharing knowledge and making data open benefits more than the scientific elite. Simple facts such as a chemical formula can carry a ripple effect. They empower students, help audit labs, and let workers spot inconsistencies in shipments before problems turn costly or dangerous. For anyone interested in the building blocks of science — or just aiming for a new approach to safety, efficiency, or innovation — knowing what goes into 3-Methylbenzonitrile brings more than formulas on a shelf. It keeps science moving forward, one group at a time.
Is 3-Methylbenzonitrile hazardous to health?
Understanding What 3-Methylbenzonitrile Really Is
Most folks don’t chat about 3-Methylbenzonitrile at dinner tables. The name alone can throw you off balance, but in simple terms, this is a chemical used mainly in specialty manufacturing, chemical research, and even in pharmaceuticals. Its structure puts a nitrile group onto a benzene ring and tacks on a methyl group, making it attractive in the chemistry world for building other things. What happens once it leaves the lab is where things start to matter for human health.
The Reality of Exposure at Work
Having spent time around chemical companies, I’ve seen labels that send a clear warning: these substances make the workplace safer or riskier. For 3-Methylbenzonitrile, the risks aren’t on most folks’ radar, but that doesn’t make them disappear. It isn’t like spilling table salt. Eye and skin contact, as well as inhalation, are the main ways someone might run into problems. Getting it on your skin can sting or redden the spot. Breathe in a little too much and you might notice your nose and throat feel irritated.
Workers mixing batches or loading barrels handle risk every day. One factor that can’t be ignored: just because something is rare doesn’t mean the risk turns zero. Most companies limit direct contact, use gloves, goggles, and proper fume hoods, but mistakes do happen. It isn’t a substance for home projects or casual use. Anyone who’s spent a late night mopping up a small spill and then double-checking the SDS sheet learns to respect chemical hygiene rules.
Long-Term Health and the Big Picture
Turns out, there’s limited data on what 3-Methylbenzonitrile does to health over years. Most of what we know comes from short-term exposure studies or close relatives in the nitrile family. Nitrile compounds, especially aromatic ones, have been linked to headaches, dizziness, and sometimes symptoms affecting the central nervous system. There just aren’t piles of research tracking effects over a lifetime, but that never means people should take their chances.
My colleague once described a cough that wouldn’t quit after routine work with aromatic nitriles. Wearing a respirator and doubling up on gloves helped, but it drove home that if companies cut corners, the cost comes straight out of workers’ health. Even tiny amounts floating in the air could build up over shifts, leading to repeated exposure.
Following the Science and Building Solutions
There’s never a substitute for good training and real ventilation. Relying on luck or “this is how we’ve always done it” leads only in one direction. Employers need to provide robust safety data sheets, regular checkups, and working safety gear. Workers need to speak up, report unsafe conditions, and insist on protective tools that fit right. I’ve lost count of times I saw disposable gloves used past their limit just to save a dollar or two.
Regulators like OSHA and the European Chemicals Agency pay attention to nitrile compounds and set limits based on available research. For 3-Methylbenzonitrile, guidelines recommend minimizing airborne concentrations and avoiding skin contact, even if a specific exposure limit isn’t always spelled out. In my experience, leaning on precaution beats waiting for someone else to prove harm.
Leaning Toward Caution Makes Sense
Chemistry drives modern life, but so does caution. Treating 3-Methylbenzonitrile with respect, using proven safety steps, and listening to people on the ground reduce health risks. This isn’t a chemical to fear blindly, but it won’t baby-sit those who take shortcuts either. If you find yourself working with it, the best move is to ask questions, learn the hazards, and take the rules seriously. There’s no reward in rolling the dice with your health or the health of those around you.
What are the storage conditions for 3-Methylbenzonitrile?
Everyday Handling and Basic Storage
3-Methylbenzonitrile might sound like something hidden away in labs, but it turns up in places that matter: chemical manufacture, research, and sometimes the pharmaceutical industry. In a world of volatile organic compounds, safe handling isn’t optional—it protects health and keeps facilities running smoothly. Anyone who’s spent time around chemicals knows that a little bit of care can dodge a mountain of trouble.
The Real Stakes Behind Chemical Storage
3-Methylbenzonitrile comes with risks if ignored. Vapors can irritate, and spills contaminate workspaces fast. Parenthetically, I once saw a bottle stored next to an oxidizer. A sharp supervisor caught the mistake, and disaster got avoided. Good storage practice isn't just paperwork—it's safety.
Key Requirements for Safe Storage
Let’s look at what actually keeps this compound stable. A cool, dry area usually does the trick since heat and moisture send chemicals down winding paths—sometimes leading to unwanted reactions. Placement in a tightly sealed glass or compatible plastic container makes all the difference. This keeps out humidity, limits air contact, and stops unwanted contamination.
Keeping chemicals with strong odors or those likely to react far apart from 3-Methylbenzonitrile helps prevent accidental cross-reactions. Safety data sheets call this “segregation,” but to someone putting away chemicals after a long shift, it means double-checking that nothing sits too close to acids, oxidizers, or bases. Those basics matter, because the nose alone won’t warn you about all possible hazards.
Fire and Ventilation Concerns
Combustibility poses a very real threat. According to the National Fire Protection Association, organic nitriles can ignite under the wrong circumstances. Facilities with adequate fire suppression and fume extraction systems reduce the odds of anything ugly breaking out. Nobody wants to find out if a spark from a piece of equipment will ignite vapors, so installing explosion-proof switches and making sure containers stay closed pays off.
The Human Element
In high school, my chemistry teacher drove home the point: learn the hazard labels, use proper gloves and goggles, and never rush clean-up or storage. Those lessons stick because chemical safety comes down to habit, not just regulations. Keeping 3-Methylbenzonitrile where temperature shifts don’t surprise it and away from light slow down degradation.
Climate Control and Labeling
Walk-in chemical storage spaces offer steady temperature and humidity year-round. Labels with purchase dates, concentration, and hazard warnings deserve respect. Knowing exactly what’s in a bottle, and how old it’s gotten, shrinks the odds of mystery spills and saves time in an inspection. Faded labels only invite confusion.
What Happens When People Cut Corners
Lax storage breeds headaches—unlabeled containers, expired or degraded chemicals, and risks to cleaning staff. Oversights add up. Audits and regular checks catch these mistakes early. That’s how labs and factories dodge fines and keep their records spotless.
Staying Ready for Emergencies
Prompt response to spills or leaks depends on awareness and training. Materials like absorbent pads, proper ventilation, and quick disposal routes take stress out of surprise incidents. Local authorities often audit chemical storage, and folks who prepare never scramble to prove compliance.
Building a Culture of Responsibility
Training goes far. Up-to-date material safety data sheets within arm’s reach make a world of difference. Open discussion about hazards, improvements, and near misses fuels a proactive work environment. With the stakes involved, every person shares responsibility for chemical safety—including the simplest step of shutting a lid tight.
What is the molecular weight of 3-Methylbenzonitrile?
What 3-Methylbenzonitrile Means in Everyday Science
Chemistry doesn’t always catch the public’s eye. But compounds like 3-Methylbenzonitrile show up in labs that shape the flavors, fragrances, and pharmaceuticals all around us. Knowing about its molecular weight unlocks accuracy in product formulation, ensures safe handling, and supports advancements in research. The molecular weight for 3-Methylbenzonitrile stands at 117.15 grams per mole. This number came from adding up the atomic weights for carbon (C), hydrogen (H), and nitrogen (N) in its structure—eight carbons, seven hydrogens, and one nitrogen.
Why Getting This Number Right Matters
When I’ve run chemical reactions in college labs or helped train new lab techs, I always emphasize precision. Labs use the molecular weight for basic tasks: figuring out how much chemical to weigh, confirming identity in quality control, and calculating yields. Think about a pharmaceutical manufacturer mixing two compounds. If someone uses the wrong value, a whole batch of medicine risks being off-target, which wastes resources and endangers patients. It’s a lesson in scale—lab slip-ups echo into the real world.
Errors often creep in through assumptions. Online sources or outdated textbooks sometimes list wrong information. I once saw a student lose a day’s work because an internet search gave a generic benzonitrile weight, not the 3-methyl variant. One small methyl group bumps up the molecular weight, which changes the outcome. Accuracy supports everything from safe dosage to intellectual property disputes. Facts matter at the bench as much as in boardrooms.
Supported by Science
The molecular weight for 3-Methylbenzonitrile traces back to its structure: a benzene ring, a methyl side group, and a nitrile group. Here’s the math:
- Carbon (C): 8 atoms × 12.01 = 96.08 g/mol
- Hydrogen (H): 7 atoms × 1.008 = 7.056 g/mol
- Nitrogen (N): 1 atom × 14.01 = 14.01 g/mol
Total: 117.15 g/mol
Tightly-checked databases like PubChem and the CRC Handbook agree. Published work from institutions like NIST also backs up this value. Accuracy in molecular weights lets chemical suppliers and pharmaceutical companies comply with regulatory standards. Small errors risk failed audits or product recalls—both carrying real financial fallout. No wonder so many people double-check by running these calculations themselves.
Practical Solutions: Building Lab Reliability
Good habits support data integrity. I learned quickly to cross-check information against official sources—never just a website summary. Investing in training pays off. Teams can run their own molar mass calculations instead of taking numbers at face value. Reliable reference materials, like certified reagents or analytical standards, cut the risk further. Mistakes slow down projects and breed distrust both within and outside the team. Science builds on details, so clear processes make the difference.
Up-to-date digital resources can support in-the-moment checkups. Software tools linked to chemical registries lessen copy errors and speed up workflow. Creating a lab culture where double-checking is routine keeps knowledge flowing. At the end of the day, disciplined chemistry isn’t just about numbers. It’s about earning trust—yours and the public’s.
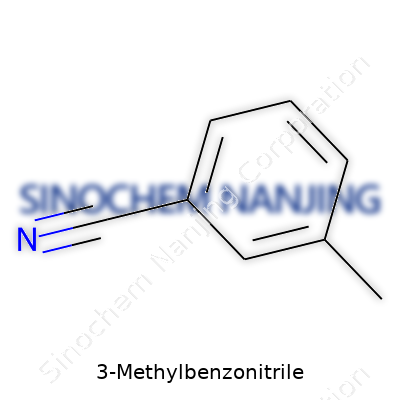
| Names | |
| Preferred IUPAC name | 3-Methylbenzonitrile |
| Other names |
m-Toluonitrile m-Cyanotoluene 3-Cyanotoluene |
| Pronunciation | /ˌθriːˌmɛθ.əlˈbɛn.zə.naɪ.tril/ |
| Identifiers | |
| CAS Number | [620-22-4] |
| Beilstein Reference | 1209238 |
| ChEBI | CHEBI:86175 |
| ChEMBL | CHEMBL3190011 |
| ChemSpider | 12393 |
| DrugBank | DB08794 |
| ECHA InfoCard | 17a8ec62-8c95-4d18-8c74-768f31737b41 |
| EC Number | 211-255-2 |
| Gmelin Reference | 793157 |
| KEGG | C01745 |
| MeSH | D018414 |
| PubChem CID | 6941 |
| RTECS number | GV8575000 |
| UNII | KN9E6O0A5A |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C8H7N |
| Molar mass | 117.15 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Odorless |
| Density | 1.005 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 1.93 |
| Vapor pressure | 0.17 mmHg (25 °C) |
| Acidity (pKa) | pKa = 24.8 |
| Basicity (pKb) | 15.10 |
| Magnetic susceptibility (χ) | -70.0 × 10−6 cm3/mol |
| Refractive index (nD) | 1.521 |
| Viscosity | 0.878 cP (20°C) |
| Dipole moment | 3.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 91.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –4633 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. Harmful if swallowed. |
| Precautionary statements | P261, P264, P271, P280, P301+P312, P304+P340, P305+P351+P338, P312, P337+P313, P405, P501 |
| Flash point | 74°C (165°F) |
| Autoignition temperature | 540°C |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 3-Methylbenzonitrile: **>2000 mg/kg (rat, oral)** |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Methylbenzonitrile: Not established. |
| REL (Recommended) | 10 g |
| Related compounds | |
| Related compounds |
Benzonitrile 2-Methylbenzonitrile 4-Methylbenzonitrile 3-Methylaniline 3-Methylbenzylamine |

