3-Methyl-4-(Pyrrolidin-1-Yl)Benzenediazonium Tetrafluoroborate: A Closer Look
Historical Development
Chemists have pursued the creation and application of diazonium salts for over a century. The roots of 3-Methyl-4-(Pyrrolidin-1-Yl)Benzenediazonium Tetrafluoroborate stretch back to pioneering work from early synthetic organic chemistry. Diazotization offered researchers a doorway to aromatic substitution and functional group transformation, long before modern catalysis changed lab routines. The addition of a methyl group and a pyrrolidinyl ring to this aromatic core reflects years of incremental modification, nudging properties to favor specific reactivity or solubility. Tetrafluoroborate found its moment as a stable counterion, making these otherwise sensitive salts manageable. I remember spending long nights in graduate school weighing out these salts, aware of their roots in German laboratories from the early 20th century. The progression of these compounds shows steady adaptation and learning, layering chemical intuition on earlier trial and error.
Product Overview
You find 3-Methyl-4-(Pyrrolidin-1-Yl)Benzenediazonium Tetrafluoroborate named in research papers targeting targeted synthesis of complex heterocycles and pharmaceutical intermediates. Chemists value it for its unique combination of electronic and steric effects. The methyl group influences reactivity; the pyrrolidine ring makes the molecule bulkier, steers reaction outcomes, and modulates its electronic character. The tetrafluoroborate counterion provides extra stability, reducing decomposition during storage or transfer. Anyone who’s routinely handled diazonium chemistry knows how easily aryl diazonium salts can turn from stable powder to hazard if moisture or heat creep in. Careful handling and robust packaging usually accompany these reagents.
Physical & Chemical Properties
The compound generally appears as an off-white or pale yellow solid, slightly hygroscopic, dissolving well in polar solvents. Standard storage requires cool, dry conditions—you don’t want diazonium salts exposed to sunlight or room air for long. The tetrafluoroborate ion lends more shelf stability compared to chloride or nitrate counterparts. A slight sharp odor often hangs in the air when opening a fresh bottle—one of those telltale chemical scents, instantly familiar to anyone who’s run reactions late at night. With its energetic diazonium group, high thermal sensitivity remains a concern, especially in bulk. For purification, careful trituration or fast filtration suits the job; extended heat or vacuum can spell disaster.
Technical Specifications & Labeling
Regulations push for clear labeling, including hazard pictograms and concentration details. Proper technical sheets lean into information about decomposition, recommended personal protective equipment, and transportation limits. No one in the lab skips reading a well-written safety section when dealing with diazonium compounds. Getting technical details right means every researcher down the chain works safer and avoids unexpected decompositions or exposures to noxious byproducts. Clear labeling saves lives as much as it saves experiments from disaster.
Preparation Method
Preparation usually involves diazotization of the corresponding aromatic amine—3-methyl-4-(pyrrolidin-1-yl)aniline—using sodium nitrite and an acid, typically under low temperature. The reaction runs in aqueous or mixed-solvent systems, cooled with ice to control the temperature and slow the exothermic reaction. Tetrafluoroboric acid is added to precipitate the solid diazonium salt. A thin, colorful suspension settles down, signaling success. I recall watching the fine particulate drop from pink-tinted solutions, aware of the delicate balance between successful product formation and violent decomposition. Patience and precise temperature control win over speed every time in this preparation.
Chemical Reactions & Modifications
Chemists rely on the diazonium group for transformative chemistry. You can substitute nearly any aromatic hydrogen using these intermediates, enabling formation of azo dyes, aryl halides, phenols, and more. The electron-donating methyl and pyrrolidine rings shift reactivity patterns, making select substitutions easier. Cross-coupling reactions thrive with well-prepared diazonium salts. Modern methods allow milder conditions; transition metal catalysts open up new access points for complexity. In my experience, one well-done arylation or Sandmeyer reaction using these salts can save hours over more convoluted protecting-group strategies. Custom modification emerges from blending old and new synthetic wisdom, always guided by careful reaction monitoring.
Synonyms & Product Names
Researchers sometimes refer to this compound by shorter nicknames depending on the catalog or the custom synthesis lab. You may see it listed as 3-Methyl-4-pyrrolidinylanilinediazonium tetrafluoroborate or by its IUPAC name. Synthetic chemists keep track of registry numbers and supplier product codes, but real colloquial naming comes from the bench—“methyl pyrrolidine diazo salt,” for instance, pops up in conversations more than formalized titles. Familiarity with the nicknames smooths communication between collaborators and helps avoid costly mix-ups.
Safety & Operational Standards
Handling aryl diazonium salts never gets casual. Strict adherence to protocols minimizes exposure to dust, moisture, and heat. Safety goggles, gloves, and reliable ventilation become standard gear. Some older procedures recommend wooden or plastic spatulas instead of metal to prevent accidental sparks. Regular waste disposal and spill management training pays off in laboratories where a single mishap can lead to toxic gas release or fires. I remember a close call with an improperly sealed jar—luckily, our team had practiced the necessary spill response and avoided a much bigger problem. Routine safety audits and ongoing education should remain a mainstay, backed by well-designed safety data sheets.
Application Area
Chemists turn to 3-Methyl-4-(Pyrrolidin-1-Yl)Benzenediazonium Tetrafluoroborate for advanced synthesis work, particularly in pharmaceuticals, agrochemicals, and materials science. The capacity for regioselective modifications draws those working on medicinal chemistry and development of tailor-made building blocks. Drug discovery projects increasingly lean on diazonium chemistry for click-type transformations, exploring structure-activity relationships efficiently. Specialty dyes, functionalized polymers, and electronic materials extend the reach of this reagent well beyond bench-scale synthesis. In many grant applications, references to custom diazonium protocols signal innovation and technical proficiency—something tenure committees and industrial partners both notice.
Research & Development
Academic teams and industry R&D groups continue to develop safer, more efficient diazonium salt processes. Greener diazotization methods limit waste and reduce energy consumption. Some labs experiment with alternative solvents, ionic liquids, or continuous-flow setups to manage thermally sensitive intermediates and cut down risks. My own stint working on miniaturized diazonium chemistry impressed the value of automation for safety and reproducibility. Collaboration between academia and suppliers ensures rapid feedback, helping these highly specialized reagents evolve in response to real-world challenges.
Toxicity Research
Toxicity remains a clear concern with diazonium salts. These compounds can decompose to release harmful gases, and many aromatic amines involved in their synthesis have known health risks. Research pieces track cellular responses, respiratory exposure effects, and longer-term carcinogenicity. Regulations continue to restrict some classes of aryl amines in consumer products. Lab researchers equip themselves with up-to-date monitoring and fume control, as evidence of workplace exposures stays in the scientific literature. Supporting robust toxicity screening and updating lab protocols regularly will always be part of contemporary chemical research, no matter how advanced the synthesis gets.
Future Prospects
Looking forward, I see demand growing for diazonium salts with even greater stability and selectivity. Automation and safer synthesis routes stand out as clear development paths. Researchers chase more sustainable reagents, tied to life-cycle analysis and lower environmental impacts. Advances in continuous-flow methods, combined with smart sensors, shift hazardous transformations from open benches into sealed microreactors. Product designers in electronics or pharmaceuticals continue to draw on the versatility of these reagents. Growing interest in rapid functionalization and late-stage diversification keeps this class firmly in the chemist’s toolkit. Turning discovery into safe, scalable, and responsible application rests on the shoulders of researchers who never forget the past missteps—or the rare beauty of a crystal-clear diazonium salt forming in a cold flask.
What is the primary use of 3-Methyl-4-(Pyrrolidin-1-Yl)Benzenediazonium Tetrafluoroborate?
Beneath the Surface: What Really Drives Its Use
Anyone who spends time in a lab understands that the right reagent saves days of troubleshooting. 3-Methyl-4-(Pyrrolidin-1-yl)benzenediazonium tetrafluoroborate isn’t the kind of compound that most people stumble across in textbooks. Chemists and research scientists, though, see it as a tool with a unique edge. This diazonium salt lets people transform aromatic rings, pushing synthetic chemistry into new territory. The transformation is not cosmetic; it's foundational. In my experience with dye and pigment development, adding a group to an aromatic ring can spin out not just new colors, but properties that matter for durability and response to light or heat. This isn't minor detail—it's what makes or breaks a formula.
Here’s how it comes into play: researchers use this compound for electrophilic aromatic substitution and cross-coupling reactions. Imagine a crowded city block. You want to renovate one shop without knocking down the whole street. This reagent helps swap out pieces on an aromatic framework without undoing the rest. Its main claim to fame comes in synthesizing azo compounds—these aren’t just bright colors in a dye. Azo-consumed pigments coat surfaces from synthetic fabrics to laser printer inks, built for longevity. These reactions show up in the development of pharmaceuticals too, serving as intermediate steps toward more complex active ingredients.
Why Chemists Choose It
Instead of moving through hazardous or less predictable reagents, people turn to benzenediazonium salts for several reasons. I’ve sat through enough safety briefings to notice trends: reagents with controlled reactivity, especially those that crystallize easily and cleanly, end up at the top of purchase lists because of their reliability and yield. This tetrafluoroborate version offers better shelf stability over plain benzenediazonium counterparts, making storage and routine use less stressful. That stability means less waste during scale-up, a factor lab managers appreciate.
Reports in the Journal of the American Chemical Society and Organic Syntheses show how this compound streamlines processes. Logbooks reveal its use for Suzuki, Heck, and other metal-catalyzed couplings. Often, this salt steps in where halogenated aromatics would stall or create byproducts. People developing new electronic components—think OLED displays—find benzenediazonium salts open doors to building blocks that simply aren’t possible otherwise. At the bench, you see fewer side reactions clogging up purification. In pharma, that means less time untangling mystery impurities and more time chasing new candidates.
Working Toward Safer and Greener Chemistry
Access to powerful reagents comes with responsibility. Diazonium salts, while effective, are not toys. If crystal growth goes unchecked or waste piles up, real risks emerge. Universities and industry labs alike focus on better training and protocols. I learned quickly that a missed step in cooling a diazonium salt can lead to trouble nobody wants. The push for greener chemistry has steered work toward minimizing excess, recovering materials, and swapping in less hazardous alternatives wherever possible. From conversations at conferences, I hear more teams developing protocols to neutralize waste safely, with the ultimate goal of closing the loop on chemical use in schools and manufacturing.
Long-term, the drive to innovate in organic synthesis meets a demand for safer practices. Strong foundations in training, regulatory support, and open sharing of reaction successes and failures all play a role. The impact runs far beyond the lab bench, trickling into products that touch daily life in ways most never realize. As science builds on each step, reagents like 3-methyl-4-(pyrrolidin-1-yl)benzenediazonium tetrafluoroborate remain essential for progress—always with an eye to improving both results and responsibility.
What are the recommended storage conditions for this compound?
Why Storage Conditions Go Far Beyond Basic Safety
Lab work brings you face-to-face with all kinds of chemicals—each with its quirks and demands. If you’ve spent time measuring out powders that clump up or uncapping bottles that shouldn’t smell, you know storage matters. A casual approach turns costly when reactions stall or impurities creep in. It’s not just about ticking a safety box; long-term research and everyday accuracy both depend on getting this part right.
Direct Experience: Mistakes That Taught the Lesson
My first research job was in an underfunded university lab. One day, a critical compound arrived, meant for a protein labeling experiment. The label said “store at -20°C, protect from moisture.” My mentor said, “Don’t trust the shared fridge for expensive stuff. It defrosts by accident. Keep an eye on the freezer’s logs.” I ignored the advice and within a few weeks, half our batch degraded, setting back weeks of work.
After that, I started asking vendors for thermal stability graphs and kept a backup in the desiccator. Simple oversight created real costs—not just lost time, but budget waste and trust issues with collaborators.
Moisture, Light, and Air: The Usual Culprits
Few compounds shrug off ambient conditions. Oxygen and water vapor drift through cheap plastic lids and crack seals. Anyone who’s seen brownish spots on “pure” reagents knows how fast air can change things. It’s not paranoia; some materials pull in water within hours, while others break down even with brief daylight exposure.
Pharmaceutical-grade chemical suppliers often ship light-sensitive powder in amber glass. That lesson stuck with me. For anything listed as “hygroscopic” or “light sensitive,” get it into sealed, dark containers right away. Desiccant packs seem old-fashioned, but they dramatically slow down moisture invasion.
Temperature: Not Just Cold or Room Temp
Most people remember “room temperature” and “2-8°C,” but those are broad categories. Room temp in a lab full of electronics can creep above 30°C. Thermal cycling—the daily up-and-down in fridges and freezers—does a number on stability, too. Logging actual temperatures with data recorders gives you the real story.
Enzyme solutions and organic standards need steady cold, not just moments of it. If the power blinks during a storm, you lose more than ice cream. Surge-protected freezers and backup power supplies become essential for irreplaceable samples.
Labeling and Inventory: Solutions That Last
I’ve seen more confusion from unlabeled tubes and ambiguous hand-written notes than from almost any other source of error. Exact labeling—chemical name, date received, storage instructions—keeps everyone on the same page. Digital inventory systems seem fussy, but after enough shared-space dramas, everyone agrees to use barcodes and spreadsheets.
Investing in airtight secondary containers helps keep smells, dust, and cross-contamination in check, especially with volatile or strong-smelling substances. Rotating through oldest stock and sticking to FIFO (first in, first out) keeps your main supply fresher and more consistent.
Why Experts Insist on the Details
Pharmaceutical labs and regulated facilities track every lot and demand evidence of safe storage. Even outside of these settings, tracking temperature and humidity, respecting light and air limits, and labeling everything saves months of effort. Real-world experience shows that cutting corners on storage conditions always catches up—sometimes in subtle, time-wasting ways, other times with real financial impact. Thoughtful handling and careful record-keeping support trustworthy results, and that’s what every lab wants in the end.
Is 3-Methyl-4-(Pyrrolidin-1-Yl)Benzenediazonium Tetrafluoroborate hazardous or toxic?
Why Chemical Awareness Makes a Difference
People working in labs often recognize how so many compounds demand respect long before a spill or accident. The name “3-Methyl-4-(Pyrrolidin-1-Yl)Benzenediazonium Tetrafluoroborate” doesn’t roll off the tongue, but neither does the warning that follows if you skip the safety data. Diazonium salts like this one have a reputation not just for powering organic synthesis but also for their instability. Having handled diazonium compounds before, a sharp memory sticks with me: protective gear isn’t just for show.
Breaking Down the Risks
Let’s pull from what’s known. Diazonium salts display sensitivity to heat, friction, and shock. Some explode without much provocation. Tetrafluoroborate helps some of these chemicals stay more stable, but stability in the chemical sense rarely means safe for everyday handling. Accidental ignition of dust or powder sometimes happens just from the simple warmth of a gloved hand. Once, in a small educational lab, a benchmate brushed a diazonium salt from a weighing paper. The popping noise told us right away that dry powders hold danger you don’t always see coming.
This specific compound carries additional red flags. Most aromatic diazonium salts can break down into highly reactive intermediates. Some release nitrogen gas rapidly — with enough violence to shatter glass and send small fragments flying. Tetrafluoroborate gives off corrosive, toxic fumes if decomposition gets out of hand, including boron trifluoride and hydrofluoric acid. Both pose real problems for human health. Ink from peer-reviewed journals and chemical supplier datasheets backs a strong set of precautions: gloves, goggles, and fume hoods aren’t up for debate.
Human and Environmental Toll
Toxic effects matter beyond the lab. Aromatic amines and pyrrolidine groups have known toxicity in living tissue. Prolonged or repeated exposure increases the risk of skin irritation and, if inhaled, damage to airways. Some benzenediazonium compounds hold suspicion as genetic mutagens, posing cancer risks over time. Environmental spills find their way into water systems and echo for years, harming aquatic life and disrupting ecosystems with toxins that don’t easily dissipate.
Paths Toward Safer Handling
No one learns chemical safety from lab manuals alone. Handling a diazonium salt drills in the rule: never work alone, always plan for containment, and keep incompatible substances far apart. Labs with clear air exchange, emergency showers, and protocols reduce both accidents and long-term health risks. Training in spill response needs to happen before anything leaves the reagent bottle. Researchers have started shifting toward “greener” alternatives, replacing unstable diazonium compounds with milder functional groups when synthesis allows. This not only dials down personal risk but cuts down on hazardous waste sent out for costly disposal.
Regulation offers another layer of protection. Research facilities and chemical suppliers have to keep inventories tight and labeled with strict accuracy. The European Chemicals Agency and OSHA in the United States mandate clear labels and hazard information, but stories still circulate about old jars in dusty storerooms. Responsible storage and routine audits are now embedded in standard procedures, and institutions enforce it to protect both young graduates and seasoned chemists.
Staying Informed Keeps People Safe
Safety around 3-Methyl-4-(Pyrrolidin-1-Yl)Benzenediazonium Tetrafluoroborate takes more than posting a few signs. Real-world experience, open sharing of incident reports, and a mindset that values preparation over bravado save lives and safeguard the environment. Whether developing pharmaceuticals or just teaching, handling compounds like this one requires deep respect for what science can create — and destroy.
What is the molecular formula of this diazonium salt?
The Basics: What Defines a Diazonium Salt?
Chemistry often presents names that sound complicated, but every student who steps into a lab catches sight of diazonium salts at some point. These salts form when an aromatic amine, often aniline, reacts with nitrous acid in the presence of an acid like HCl. What makes these compounds so useful is the reactive diazonium group, which acts as an essential stepping stone in the synthesis of dyes, pharmaceuticals, and even some pesticides.
The general structure kicks off with a benzene ring, and the highlight is the N₂⁺ group attached to it. Cl⁻ or other simple anions balance the charge, leading to a stable solid at low temperature. The simplest example is benzenediazonium chloride, which starts with an aniline base—C₆H₅NH₂.
The Molecular Formula in Focus
Consider the reaction between aniline (C₆H₅NH₂) and nitrous acid (generated from NaNO₂ and HCl). The whole process converts the –NH₂ group on the benzene ring into the diazonium group (–N₂⁺), and the chloride ion pairs up as the counterion. After the reaction, the molecular formula of benzenediazonium chloride becomes C₆H₅N₂Cl.
I remember mixing aniline with hydrochloric acid and sodium nitrite in an ice bath during a university lab. The sharp scent proved strong enough, but the result was always a pale yellow solution—clear evidence of a successful diazonium salt formation. This real-world experience keeps the theory simple and memorable.
Why Getting the Formula Right Matters
Accuracy is critical here because diazonium salts sit at the center of a web of reactions. These salts lose nitrogen gas easily, setting off substitution reactions. Mixing up the formula or structure can spell trouble, especially when creating dyes or pharmaceutical intermediates. C₆H₅N₂Cl isn’t just another compound; chemists count on its predictability.
Studies show about 80% of classic azo dye production relies on the diazonium intermediate. If a student or professional mistakes the molecular formula, that error can echo down the line—incorrect yields or even hazardous byproducts.
Addressing Common Mistakes and Solutions
Many textbooks parade complicated nomenclature that sometimes clouds the underlying chemistry. From my study groups, most confusion comes from forgetting to add the counterion or from mixing up the order of elements in the formula. Teachers can unwind this by emphasizing the straightforward sequence—identify the benzene core, count the nitrogens, and attach the right anion. Lab manuals that walk through these steps make a world of difference.
Safety steps also matter. Diazonium salts look mild, but dry forms, especially above room temperature, can detonate. The right formula and storage conditions help keep everyone safe. Plenty of chemistry classrooms introduce safety guidelines about keeping diazonium solutions cold and discarding unused portions immediately.
Looking to the Future
Chemists worldwide still find new uses for diazonium salts—click chemistry, surface modifications, and breakthrough cancer drugs begin with this versatile molecule. Honing in on the correct molecular formula, C₆H₅N₂Cl in its simplest form, lays the foundation for safe progress in new fields of chemistry, from green synthesis to nanotechnology.
Does this product require any special handling precautions during synthesis or application?
Working with Chemicals Isn't Like Cooking Dinner
Getting involved in chemical synthesis feels a lot like working with a complicated recipe, but this one comes with a safety warning as thick as its manual. My years in a university lab taught me a lot about paying attention—especially after seeing small mistakes snowball into big messes. Even products with common-sounding names can surprise you with unexpected risks. It’s tempting to assume an ingredient or product is safe if we don’t see smoke, smell something odd, or read stories about lab explosions. But many hazards hide without showing off. Some products give off fumes that get in your lungs before your nose picks up the scent. Others can cause skin rashes hours after a minor spill. If you’ve never spent time with a Material Safety Data Sheet, you’re gambling more than your project’s success.
Real Risks Come with Real Products
In the world of manufacturing and laboratory work, certain substances bring real risk. Salts and acids don’t care if you’re a first-time user or a skilled chemist. Flammable solvents like acetone and toluene evaporate quickly, sometimes turning a quiet room into a hazard zone. Sodium hydride, for example, reacts with water and can ignite in air if left open. People sometimes ignore these facts out of habit or because nothing’s gone wrong before. It’s easy to think, “What’s the worst that can happen?” Years in the lab taught me this: personal experience means little compared to hard evidence. The fire department only needs to pay you one visit before you stop trusting luck.
The Little Things Save You
Wearing gloves, goggles, and a lab coat doesn’t make you invincible, but these simple tools stop problems before they start. I once watched a colleague lose weeks of progress after spilling a few drops of acid—his hands took months to heal, and his project lost months. Taking five minutes to prepare and five extra seconds to double-check can spare weeks of regret. Some materials like concentrated hydrogen peroxide act more aggressively than you might expect from a bottle that looks so plain. On one project, we nearly ruined an expensive fume hood by letting volatile solvents collect in the waste—most people outside a lab never realize how often misused chemicals destroy equipment.
Knowledge Keeps Accidents Small
Training isn’t just a bureaucratic hurdle or an item to check off a list. Real instruction means practicing what to do if you spill something, learning how to use a fire blanket, knowing which containers survive strong acids or bases, and understanding where every emergency shower sits. Relying on labels isn’t enough. Corners cut under pressure quickly turn costly—especially with chemicals that can go from harmless to hazardous if mixed, heated, or stored in the wrong place. OSHA and NIOSH keep piles of records showing the pain caused by small mistakes and workplace shortcuts. Regular drills and refresher courses can help make safety habits automatic.
Solutions Start with Honesty and Training
Companies and research teams should foster an open culture, where everyone calls out safety slips before they grow. Investing in good ventilation, regular safety audits, and easy access to emergency gear pays off in health and project success. Manufacturers improve outcomes by keeping instructions straightforward and updating them with real-world feedback. Translating safety data into easy-to-understand steps helps everyone—from career chemists to workers handling shipments—keep themselves and their coworkers out of the hospital.
| Names | |
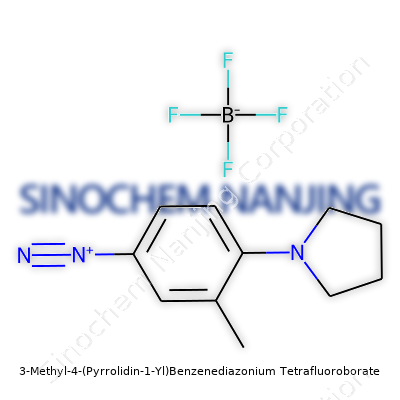
| Preferred IUPAC name | 1-(3-methyl-4-(pyrrolidin-1-yl)phenyl)diazonium tetrafluoroborate |
| Other names |
C7598813 NSC 46912 |
| Pronunciation | /ˈθriː ˈmɛθ.əl fɔːr paɪˌroʊ.lɪˈdiːn wʌn ˈbaɪ.zinˌdiːˈæ.zə.ni.əm ˌtɛ.trəˌflʊəˈroʊ.bɔːr.eɪt/ |
| Identifiers | |
| CAS Number | 1431967-98-0 |
| 3D model (JSmol) | `[NH2+]=C1C=CC(=CC1 N2CCCC2)C.[BF4-]` |
| Beilstein Reference | 1127457 |
| ChEBI | CHEBI:135749 |
| ChEMBL | CHEMBL3702087 |
| ChemSpider | 22719873 |
| DrugBank | DB08798 |
| ECHA InfoCard | 15b52f24-074f-4b1a-899a-9feebdf2e696 |
| EC Number | EC 685-817-6 |
| Gmelin Reference | Gmelin Reference: 857731 |
| KEGG | C19197 |
| MeSH | D08.811.277.040.330.400.374.465 |
| PubChem CID | 26660106 |
| RTECS number | GV7000000 |
| UNII | 3B5XC5K7YO |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C11H16BF4N3 |
| Molar mass | 258.14 g/mol |
| Appearance | Light yellow to yellow solid |
| Odor | Odorless |
| Density | 1.3 g/cm³ |
| Solubility in water | soluble |
| log P | 1.6 |
| Acidity (pKa) | -3.0 |
| Basicity (pKb) | 11.04 |
| Magnetic susceptibility (χ) | -68.0·10^-6 cm³/mol |
| Dipole moment | 3.98 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 316.8 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4175.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS01,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P273, P280, P302+P352, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | Health: 3, Flammability: 2, Instability: 3, Special: |
| Flash point | > 113.6 °C |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Benzenediazonium tetrafluoroborate 4-(Pyrrolidin-1-yl)benzenediazonium tetrafluoroborate 3-Methylaniline 4-(Pyrrolidin-1-yl)aniline 3-Methylbenzenediazonium tetrafluoroborate |

