An Editorial Dive Into 3-Methyl-3-Pentanol: Unpacking a Little-Known Chemical Cornerstone
Tracing the Backstory: Historical Development
3-Methyl-3-pentanol stands as a somewhat quiet member within the family of tertiary alcohols. It cropped up on chemists’ radar during the steady expansion of organic synthesis in the early-to-mid twentieth century. As labs around the world steered their curiosity into the terrain of branched alcohols, 3-Methyl-3-pentanol became a case study in isomeric structure and reactivity. The drive to model and predict carbon frameworks led researchers in the UK, US, and Germany to sketch and tinker with this molecule. What once began as an academic puzzle about carbon branching now influences many applied fields, marking just how deeply foundational research can drift into manufacturing, fragrance, and solvent technologies.
Getting to Know the Stuff: Product Overview
Talk about a molecule that wears many hats. 3-Methyl-3-pentanol belongs to the tert-alcohol family, with a structure that looks simple but tells a more interesting tale: six carbons, all bound together with branches in exactly the right places to offer a suite of chemical properties. In daily industrial life, few outside the chemical trade would ever notice it, but formulators, synthetic chemists, and even some flavor developers see it pop up in specific commercial blends. Its slightly camphoraceous odor rarely sees the spotlight, but specialists value this niche attribute in certain flavor or fragrance compositions.
Physical & Chemical Properties: Getting Into the Weeds
3-Methyl-3-pentanol sticks to the colorless, clear liquid playbook at room temperature. Boiling arrives not far above that of water, yet volatility is nothing compared to light aromatics or ethers. Hydrophobic by design, mixing with water serves little use—solvents like ether or alcohol handle it better. With a vapor pressure tucked lower than many lighter organics, you don’t see it jumping into the air as aggressively as some of its counterparts. In flammability terms, it takes ignition risks seriously, but not at the explosive pace of ether or gasoline. Its molecular shape imparts notable resistance to oxidation, which keeps its shelf life healthy so long as it avoids strong acids or open flames.
Technical Specifications & Labeling
Regulatory frameworks push detailed labeling for 3-Methyl-3-pentanol. Any reputable laboratory supply bottles flag its contents as hazardous if ingested, inhaled, or exposed to skin for prolonged intervals. The industry uses recognized synonyms—like 3-methylpentan-3-ol or tert-hexyl alcohol—so those searching through records spot it under varied guises. Transport codes index it as a flammable liquid, so shipping practices emphasize grounding containers and limiting exposure to open air or ignition sources. Barcode and batch details rarely matter to the layperson, but for traceability, they streamline responses to contamination or product recalls, bolstering a safety-first approach.
How Chemists Cook It Up: Preparation Methods
Nobody synthesizes 3-Methyl-3-pentanol by accident—its origins are intentional. Grignard reaction steps into the scene, where 3-methyl-3-pentanone faces off with alkyl magnesium halides in the embrace of anhydrous ether. For those not steeped in bench chemistry, this means engineers add magnesium to an organic halide, turning it into a powerhouse nucleophile that clubs through carbonyl groups and sticks a new carbon on the chain. It’s not just a trick for this molecule; it’s a routine for making tert-alcohols worldwide. Waste management, an ever-present concern, makes chemists capture and treat effluents carefully, which shines a light on green chemistry initiatives creeping steadily into specialty alcohol manufacture.
Reactions and Tweaks: Chemical Playgrounds
At its core, 3-Methyl-3-pentanol resists most of the classic substitutions and additions that primary and secondary alcohols entertain. Its tertiary status shields its alcohol group from straightforward oxidation, keeping aldehydes or acids off the product list. Still, under the right acid catalyst, dehydration transforms it into branched alkenes, feeding the petrochemical pipe for higher-value intermediates. Reactivity with halides spins off intermediates for pharmaceutical ventures. You don’t see this molecule married off to just one end use; it’s a restless actor in the laboratory, willing to test out different scripts depending on who writes the reaction conditions.
Nicknames in the Chemical Industry: Synonyms & Product Names
Outside the realm of formal IUPAC nomenclature, people in labs toss around names like “tert-hexyl alcohol” or “3-methylpentan-3-ol.” The crowded world of branch-chain alcohols runs thick with aliases, and this one keeps up. Spotting it under these tags on a datasheet, catalog, or safety bulletin speaks to just how much naming conventions reflect historical precedent more than systematic trends. The folks who run chemical storerooms nod to both the traditional and systematic side, staving off mishaps due to mislabeling.
Safe Handling and Responsible Use: Safety & Operational Standards
No matter how familiar a substance gets, risk deserves respect. 3-Methyl-3-pentanol counts as flammable and slightly toxic. People who handle it run ventilation and don gloves, goggles, and sometimes even face shields during big pours or transfers. Spills command immediate attention, demanding cleanup with absorbents and evacuation of vapors. Storage boxes shut tight after use, shelving gets checked for ignition hazards, and chemical showers get tested regularly. As demand for environmental compliance grows, disposal ceilings and emission badges get lower year by year, pressing companies to train crew in spill prevention and chemical hygiene well beyond the law’s minimum.
Where Does it End Up? Application Area
3-Methyl-3-pentanol left its most famous mark as a chemical intermediate; you won’t spot it as the hero in household products. Its branching structure makes it a candidate for synthetic lubricants that need just the right blend of flow and evaporation. Fragrance chemists toy with it when chasing notes that stand out from common floral or fruity themes, seeking to flesh out herbal or camphorous backbones. Certain mainline pharmaceuticals quietly draw on its unique structure during multi-step synthesis, as do some customized pesticides where branching disrupts metabolism in target species. Even specialists in the plastics arena see value in its presence during monomer preparation, nudging polymer properties without overcomplicating end-process handling.
Staying Curious: Research & Development
Labs keep a steady curiosity reserve for structures like 3-Methyl-3-pentanol, especially in catalysis and green transformations. Synthesis teams tinker with biocatalytic options or solvent-minimizing methodologies looking to bump up yields without environmental baggage. Researchers exploring branched chain alcohols’ roles as biofuel additives give chemical engineers real puzzles: can an industrial byproduct carve out a role in renewable energy? Software-led retrosynthesis platforms now comb through databases and flag it as a possible building block for new drug candidates. This legwork doesn’t just stay in the lab—patent records over recent decades show a low but steady heartbeat of innovation, especially in niche solvents and custom pharmaceuticals.
Understanding the Risks: Toxicity Research
Work in toxicity screening for small organics underscores practical realities for 3-Methyl-3-pentanol. Acute exposure in animal studies flags mild to moderate systemic effects, mainly at doses rarely reached outside the laboratory or production site. The molecule’s tertiary structure hinders metabolic breakdown, which somewhat reduces the risk of reactive metabolites but extends persistence in biological tissues compared to many lower alcohols. Eyes and mucous membranes react quickly, so splash protection isn’t optional. Long-term toxicity studies still hit limits due to sporadic uses and small human exposure data pools. Medical teams keep antidotal therapy basic—oxygen, symptom management, and in major ingestion, activators for rapid elimination.
Peeking Over the Horizon: Future Prospects
Demand for greener processes will press researchers to examine every step of 3-Methyl-3-pentanol’s manufacture for lower waste and smarter input cycles. As regulatory agencies scrutinize chemical releases and residue levels, more attention is going into lifecycle assessments and alternative synthesis feeds, like biosourced ketones or electrocatalysis. Expansion into greener solvent blends or high-efficiency pharmaceuticals could transform this tertiary alcohol from a footnote into a keystone for specialty manufacturers. My own conversations with researchers digging into branched chain molecules point to a belief that the next leap forward may emerge from the intersection of computational modelling, genomics, and small-volume, high-impact chemical production. Far from being just another name on a solvent shelf, 3-Methyl-3-pentanol still holds more to discover, provided science keeps asking unconventional questions.
What is the chemical formula of 3-Methyl-3-Pentanol?
Breaking Down 3-Methyl-3-Pentanol
People working in labs or teaching high school chemistry run into organic compounds that sound like tongue-twisters. 3-Methyl-3-pentanol is one of those. Its chemical formula—C6H14O—might look simple on paper, but for anyone handling it or thinking about its uses, digging deeper really means something.
What This Formula Tells Us
Knowing the formula C6H14O tells you the compound holds six carbons, fourteen hydrogens, and a single oxygen. Add a pen to paper, and you sketch its structure: a five-carbon pentane backbone, with a methyl group stuck to the third carbon, and a hydroxyl (-OH) group hanging on as well. I spent too many afternoons hunched over lab tables drawing structures just like this, realizing every line and branch gave each compound a very different set of behaviors.
3-Methyl-3-pentanol falls under what folks call tertiary alcohols. That -OH group sits on a carbon atom already locked in with three other carbon atoms. This little difference stands out, especially for students or chemists, because it affects reactivity, polarity, and even safety. Tertiary alcohols, for example, don’t oxidize into aldehydes like their primary cousins. They act a little stubborn in some typical reactions.
Why Naming Still Matters
There’s a reason teachers hammer home IUPAC names and formulas. In my own time as a tutor, students mixed up 3-methyl-3-pentanol with 2-methyl-2-pentanol or other alcohols all the time. A single digit shift changes lab results, solvent selection, even toxicity profiles. I’ve seen labs halted because someone grabbed the wrong bottle, thinking “it’s just another pentanol.” Mistakes like that get expensive, fast.
Real-Life Impact of Getting It Right
This matters beyond textbooks. Take manufacturing; process chemists double-check these details before green-lighting any run. A formula mistake there doesn’t just shift a product’s scent or flavor—it could send batches to waste or turn a safe product into a safety hazard. Industry recalls tied to wrong ingredients or contaminants still make the news. That’s all about chemistry at its most basic.
The everyday person may think a formula is just a bunch of letters and numbers, but as chemical products keep showing up in everything from automotive fluids to pharmaceuticals, knowing exactly what’s inside can mean the difference between good results and medical emergencies. Even for students, the formula drills teach rigor, proof that chemistry is a language best spoken fluently and precisely.
Solutions: Better Practice, Less Error
Cutting back on costly mix-ups means making chemical literacy a habit early. Standard labeling, digital inventories, and using clear naming conventions save headaches. In the digital age, QR codes and safety datasheets help even more. It pays to triple-check those six carbons, the branching methyl, and that oxygen—every time.
So C6H14O isn’t just a formula to memorize. It’s a reminder that attention to detail in science shapes both small experiments and big industries. If you haven’t messed up a drawing or misread a label, you haven’t spent enough hours at the bench. Every good chemist and cautious student learns fast—miss the details, and you miss everything.
What are the common uses or applications of 3-Methyl-3-Pentanol?
Why Chemists Reach for 3-Methyl-3-Pentanol
3-Methyl-3-Pentanol sounds technical, but this clear, flammable organic compound pops up more often in lab solutions than in daily conversation. Chemists keep it around for its reliable performance in specialty synthesis jobs. Thanks to its structure—a six-carbon chain with a hydroxyl group and a single methyl branch—this alcohol brings unique value that straightforward compounds like ethanol or isopropanol can’t always deliver.
In the lab, it’s prized for forming esters, especially in organic synthesis where precision matters. Complex fragrances and flavors owe their zest to fine-tuned reactions, and 3-Methyl-3-Pentanol can jumpstart or steer those chemical changes. It’s less volatile than lighter alcohols, so controlling reactions becomes easier. In my own bench time, using solid intermediates like this helps get consistent purity when other alcohols give unpredictable results.
Behind the Scenes in Pharmaceuticals
It’s easy to overlook the supporting players in pharma. Chemists developing drugs often use 3-Methyl-3-Pentanol as a building block, modifying its backbone to craft formulations that fight infection or manage symptoms. That methyl branch gives drug developers more freedom when choosing how a molecule interacts with enzymes and cell receptors. I remember reviewing formulation recipes where this compound stood out because it offered a solid starting point for new therapeutic targets.
Not all uses show up in the final pill or syrup, but the workhorses in labs quietly keep the process moving. In some API syntheses, this alcohol participates as a reagent, serving as an intermediate. It helps shape molecules that might otherwise require more toxic or unpredictable chemicals. The trend in research efforts leans toward cleaner, safer procedures, and 3-Methyl-3-Pentanol keeps the door open for improvements in that direction.
Industrial Chemistry and Testing Labs
Industrial applications sometimes fly under the radar, yet 3-Methyl-3-Pentanol can help test other chemicals for purity or stability. Instrument calibration standards need chemical consistency—and this alcohol fits well in quality control protocols. Any lab that runs analytical instruments benefits from reliable reagents. The consistency of results matters when product safety and consumer trust sit on the line.
It also appears in specialty solvent blends. That balance of solubility and low reactivity lets manufacturers use it to dissolve stubborn organics, remove residues, or clean sensitive lab equipment. From my own troubleshooting, those cases when nothing else quite manages to dissolve remnants on glassware often call for mid-weight alcohols like this one.
Safe Handling and Responsible Use
As with many laboratory chemicals, care matters. 3-Methyl-3-Pentanol is flammable, and it can irritate the skin or eyes. Personal experience has shown me just how easy it is to forget goggles “for just a quick job,” but industry safety standards exist for a reason. Anyone handling it needs robust gloves, eye protection, and proper ventilation. EPA guidelines stress minimizing environmental discharge, and that fits with a broader push to keep labs accountable.
Looking Toward Better Alternatives
Practical discussions in research circles highlight the search for greener, safer reagents. There’s no wide consumer demand for 3-Methyl-3-Pentanol, but that doesn’t mean industry can ignore its impact. Synthetic routes keep improving, and pressure remains on both producers and users to reduce waste and environmental risks. Renewable feedstocks are gaining attention, which means even these specialty alcohols could get a sustainable upgrade in coming years.
For now, 3-Methyl-3-Pentanol does much of its work in the background, supporting chemical and pharmaceutical advances while safer handling and responsible sourcing continue to rise as priorities.
What are the safety precautions and hazards associated with 3-Methyl-3-Pentanol?
A Closer Look at the Hazards
3-Methyl-3-Pentanol shows up in plenty of chemical labs. This clear, sometimes sweet-smelling liquid sticks out because of its low boiling point and ability to dissolve certain compounds. Many scientists and industrial workers use it for synthesis work. Anyone who handles it should keep safety on their mind since there’s a real risk of eye and respiratory irritation if it goes airborne, and skin reactions are a real possibility too.
Direct contact with skin leads to dryness, irritation, even cracking with regular exposure. Breathing in its fumes never feels comfortable. Dizziness, headache, or a sore throat usually tell you someone isn’t getting enough ventilation. Major spills in an enclosed area push those dangers higher. Liquid splashed into eyes stings and can cause redness or tearing. Swallowing this stuff by accident triggers nausea and a burning feeling right down the throat. Long-term exposure, even at low concentrations, sometimes leads to nervous system symptoms—brain fog, decreased coordination, or even more severe effects.
Precautions Everyone in the Lab Should Take
Keeping 3-Methyl-3-Pentanol controlled starts with keeping it contained. I’ve seen too many rushed labs leave bottles wide open or fail to check their fume hoods. Even if it’s tempting to skip safety gear in a hurry, a decent pair of chemical-resistant gloves, splash goggles, and a fresh lab coat cut exposure way down. It’s smart to work under an active fume hood and keep containers tightly closed when not in use.
Spills don’t need to escalate into emergencies. Absorbing the liquid with sand or an inert absorbent and then sealing it in a safe disposal bag almost always does the trick. Those handling any size cleanup should keep gloves and goggles on—skin and eye contact causes most of the immediate injuries. Having an eyewash station within arm’s reach isn’t just nice to have; after seeing someone splash chemicals in their face and scramble around searching for water, I make sure to double-check one before starting work.
Fire and Storage Warnings
3-Methyl-3-Pentanol catches fire at a relatively low temperature. Its vapors form flammable mixtures with air, especially in small, poorly vented spaces. I’ve remembered to keep all sources of ignition far away and never plug in electronic equipment on the same bench where it’s poured. Storing it in a cool, well-marked flammable cabinet, away from oxidizers and acids, helps to prevent accidental reactions or fires down the line.
Responsible Handling and Better Awareness
The rise in chemical safety training over the last decade has changed how people view chemicals like this alcohol. Labs that take the time to review safety data sheets and run emergency drills lower the odds of both minor and major mishaps. Posting proper signage and keeping open lines of communication between all lab members go even further. It’s about looking out for everyone in the lab, not just checking a box next to “safety.”
Proper disposal steps up as a bigger issue with environmental rules tightening. Pouring solvent waste down the drain sticks with the old days. Instead, collected waste heads into sealed containers, gets labeled up, and ships out to certified disposal outfits. It helps limit environmental hazards and keeps the regulator’s stress dial turned way down.
Safer Practices Move Us Forward
Anytime I’ve seen a lab cut corners, small problems snowballed. Prioritizing good ventilation, basic PPE, smart storage, and clear procedures keeps accidents rare. As awareness grows, people in all roles—from students to senior researchers—treat chemicals like 3-Methyl-3-Pentanol with the respect they deserve. That mindset, more than any rulebook, keeps both the lab and the people in it safe.
What is the boiling point and melting point of 3-Methyl-3-Pentanol?
A Look at Boiling and Melting Points
3-Methyl-3-pentanol belongs to the family of tertiary alcohols, and its physical properties tend to generate curiosity among both students and professionals. In chemistry labs, accuracy always matters. For those seeking details, 3-methyl-3-pentanol melts at around -5 °C and boils close to 120–124 °C. These values don’t just fill tables in textbooks, they also carry weight in real-world uses; they guide safe storage, proper handling, and effective distillation.
Why Properties Matter for Real Work
Boiling and melting points aren’t random trivia. I worked in an academic lab where separation of compounds was daily business. Once, we dealt with a batch of tertiary alcohols. Distilling each required watching the temperature like a hawk—hitting 3-methyl-3-pentanol’s boiling range too soon meant losing valuable product. The right boiling point acts as a landmark in the process. This is especially true for those who handle such chemicals outside the classroom, for instance, in chemical manufacturing or forensic labs. If you don’t know the boiling point, you risk contaminating samples or endangering yourself and others by releasing vapors at the wrong temperature.
Melting points do more than show up on a certificate of analysis. Years ago, I learned firsthand that a liquid stored in a cold warehouse could solidify overnight. Opening containers or transferring chemicals at the wrong temperature created hassles nobody wanted. Melting points give everyone, from warehouse crews to researchers, a number for planning simple but crucial steps.
What the Numbers Really Tell Us
Look at the boiling range of 120–124 °C for 3-methyl-3-pentanol. This helps chemists separate it from similar alcohols, since each compound has its own boiling “fingerprint.” The melting point, at about –5 °C, means it usually stays liquid at room temperature, which makes it easier to handle and measure out. If storage areas drop below freezing, it’s time to check bottles for solidification and plan for gentle warming before use. These temperatures guide basic decisions about glassware, cooling baths, and even the best kind of thermometer to reach for.
Solving Problems Around These Properties
Common sense goes a long way, but not every workplace has all the answers right away. I’ve seen experienced technicians double-check references every time an unfamiliar chemical appeared. Safety data sheets help, but nothing beats having reliable reference books or access to reputable digital databases. Teams should run a quick check before ordering bulk amounts—does the warehouse have a cool or warm spot for chemicals near the freezing mark? Will any process step hit the boiling point and fill the air with fumes?
Some folks learn to use digital hotplate-stirrers with precise temperature settings specifically because of challenges with boiling points. Others make charts for common chemicals kept on hand. Sharing experience, even just pinning a list near the workbench, cuts down on errors and saves headaches. Sometimes the right lab habit is as simple as writing the boiling and melting points right on the bottle with a permanent marker.
Staying Safe and Productive
3-Methyl-3-pentanol looks like a standard chemical at first, but understanding its properties leads to fewer spills, better yields, and safer labs. Paying attention to details like boiling and melting points reflects a deeper respect for both the science and everyone who works with these substances. No amount of equipment replaces the knowledge that comes from reading, sharing, and remembering these small but critical facts.
How should 3-Methyl-3-Pentanol be stored and handled?
Recognizing What You’re Dealing With
3-Methyl-3-pentanol falls into that category of chemicals that most people won’t encounter every day. Used in labs and sometimes in industrial applications, it’s not famous, but it carries real risks. Over the years working in education, I’ve seen beginners underestimate substances that sound less intimidating than sulfuric acid or benzene. The trouble with alcohols like this one—they can lull you into thinking they’re “just” solvents. That kind of thinking leads straight to accidents.
Storage Demands Respect
Ask anyone who manages a chemical prep room about proper storage, and they’ll probably talk about ventilation and segregation for volatile liquids. 3-Methyl-3-pentanol behaves much like the more recognized alcohols: flammable and able to give off enough vapor to spark a fire if someone lets their guard down. That means storing it in a tightly sealed container, ideally made of glass or a compatible plastic—never a container made for food.
Anyone with a bit of experience will tell you never to leave this sort of chemical near heat sources or in direct sunlight. Regular room temperature is fine, so long as the storage spot keeps steady and cool. Dedicated chemical storage cabinets work best, especially cabinets designed to contain flammable liquids. Those orange or yellow safety cabinets aren’t just for show; they buy precious minutes in emergencies.
The Real Reason Segregation Matters
Some chemicals get along about as well as oil and water. 3-Methyl-3-pentanol should stay away from acids, oxidizers, and even some strong bases. Decades of chemical safety data show that storing incompatible chemicals together often ends in disaster, sometimes fatal. I always tell students that the chemical closet isn’t a random stack; there’s a logic to what sits where.
Labels and clear documentation help—sloppy handwriting and faded stickers have tripped up more than one seasoned worker. The Material Safety Data Sheet, often ignored until something goes wrong, spells out all those storage rules in black and white. Trust the MSDS. It isn’t just paperwork.
Personal Protection Counts for More Than Looks
Handling any solvent requires more than faith. Gloves rated for alcohols, eye protection, and good ventilation form the minimum. Splashing alcohol in your eyes or on your skin feels like fire. Fumes build up faster than you think. I remember a story from a colleague who once fainted after working in a shut fume hood—felt “fine” one minute and hit the floor the next. Take those ventilation warnings seriously. A decent fume hood or, for smaller jobs, a well-ventilated bench lessens the risk of headaches or worse.
Spills don’t wait for you to be ready. If a spill happens, blotting with paper towels just spreads fumes and risk. Absorbent pads designed for chemical spills do a better job. Once the mess is cleaned up, disposal should follow hazardous waste rules—never pour it down a drain, no matter how harmless someone says it is.
Managing Risk is a Community Thing
All these precautions aren’t just niceties from out-of-touch researchers. Experienced lab techs, teachers, and industrial workers share stories for a reason—they know what slips through the cracks. Training makes the difference. If you’re working with 3-methyl-3-pentanol, regular safety drills, updated labels, and accessible spill kits protect everyone. Safety culture grows stronger each time people double-check a lid or rethink how close those bottles sit to each other.
Trust science, trust protocols, and never trust a shortcut. Long years in labs and classrooms reinforce one fact: chemicals demand respect. Simple steps—store smart, handle with care, and don’t get complacent—keep accidents at bay.
| Names | |
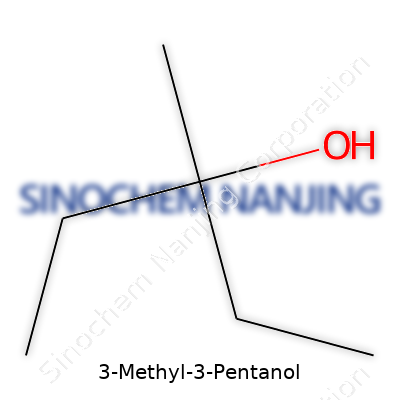
| Preferred IUPAC name | 3-Methylpentan-3-ol |
| Other names |
3-Methylpentan-3-ol tert-Butyl ethyl carbinol 3-Methyl-3-hydroxypentane |
| Pronunciation | /ˈθriːˈmɛθəlˈθriːˈpɛntənɒl/ |
| Identifiers | |
| CAS Number | 77-74-7 |
| Beilstein Reference | 1209218 |
| ChEBI | CHEBI:77598 |
| ChEMBL | CHEMBL16874 |
| ChemSpider | 7672 |
| DrugBank | DB03136 |
| ECHA InfoCard | 100.007.875 |
| EC Number | 200-474-7 |
| Gmelin Reference | 7877 |
| KEGG | C02311 |
| MeSH | D010029 |
| PubChem CID | 11535 |
| RTECS number | SA9275000 |
| UNII | UN1L962A35 |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID9020452 |
| Properties | |
| Chemical formula | C6H14O |
| Molar mass | 88.15 g/mol |
| Appearance | Colorless liquid |
| Odor | mild odor |
| Density | 0.817 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 0.89 |
| Vapor pressure | 0.93 mmHg (at 25°C) |
| Acidity (pKa) | The acidity (pKa) of 3-Methyl-3-Pentanol is approximately **16.1**. |
| Basicity (pKb) | Product 3-Methyl-3-Pentanol does not have a pKb value because it is an alcohol and not a base. |
| Magnetic susceptibility (χ) | −7.45 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.413 |
| Viscosity | 3.23 mPa·s (20 °C) |
| Dipole moment | 2.50 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 314.06 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -360.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3894 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 40 °C (104 °F; 313 K) - closed cup |
| Autoignition temperature | 411 °C (772 °F; 684 K) |
| Explosive limits | Upper 6.9%, Lower 1.0% |
| Lethal dose or concentration | LD50 (oral, rat): 5800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3500 mg/kg (rat, oral) |
| NIOSH | SN8750000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Methyl-3-Pentanol: 100 ppm (360 mg/m³) TWA |
| REL (Recommended) | REL: 100 ppm (410 mg/m³) |
| IDLH (Immediate danger) | 400 ppm |
| Related compounds | |
| Related compounds |
1-Pentanol 2-Pentanol 3-Pentanol 3-Methyl-2-pentanol 3-Methyl-1-pentanol |

