3-Methyl-2-Pentanone: A Closer Look at Its Role and Risks
Tracing the Roots: Historical Development
Long before I ever came across 3-Methyl-2-pentanone in a laboratory supply catalog, mineral oil distilleries and early chemical syntheses in the 20th century paved the way for its discovery. Interest picked up not during fanfare but because organic chemists kept running into this compound as an unexpected side product from catalytic processes, especially those related to alcohols and ketones. Over time, improvements in distillation and understanding of catalytic rearrangements made it possible to isolate this compound in a pure form. The science world quickly saw the value in refining such chemical processes—not only for academic curiosity but for practical uses that matter in coatings, flavors, and inks.
What the Compound Actually Is
Look at most bottles in an organic lab, and you’ll find a host of ketones, each with a unique twist. 3-Methyl-2-pentanone does not boast glamorous publicity, but its structure—a six-carbon backbone, a ketone group stuck at the second carbon, and a methyl group on the third carbon—gives it a solid standing. That particular arrangement, compressed into C6H12O as a formula, shapes how it behaves both in synthesis and in the wild world of application. The odor can be compared to other solvents, a hint sweet and sharp, something you’d catch instantly during a bench-top experiment gone messy.
Physical & Chemical Legwork
The real workhorses in chemistry aren’t always showy. 3-Methyl-2-pentanone lands in this category because it turns up as a clear, mobile liquid with a boiling point hovering just above typical room temperatures (around 120°C). That might sound dull to most people, but for folks working with distillation columns or open containers, it means you have to pay attention to ventilation and temperature controls. Its modest polarity makes it a decent solvent for a range of organic substances. Besides, with a flash point around 25°C, storage calls for respect—a small spark could spell trouble, as those who have worked with volatile organics know all too well.
Technical Specifications & Labeling: Getting beyond the Sticker
A bottle of 3-Methyl-2-pentanone doesn’t exactly stand out on a shelf, unless you understand what’s behind its hazard markings. Labels carry more than just the name; they warn about flammability and inhalation hazards. Chemical suppliers base their purity ratings on GC-MS testing, showing chemists exactly how many tenths of a percent they are dealing with. Low water content, listed impurities, and material safety must be checked before any experiment. Compliance with labeling standards isn’t just red tape—it reassures those working with the material that they know what to expect from the contents.
How It's Made: Preparation in Practice
Ask any seasoned chemist about making 3-Methyl-2-pentanone, and many point to aldol condensation as the way forward, often starting with smaller building blocks like 2-pentanone and methylating agents. Acid- or base-catalyzed environments allow for rearrangements, sometimes with frustratingly low yields, pushing for repeated distillations and purifications. The process isn’t glamorous; it takes patience, a good eye for detail, and a respect for controlling reaction temperatures and byproduct formation. None of this comes easy without serious skill and reliable lab equipment, which highlights the importance of proper training.
What Happens Chemically: Reactions and Tweaks
Once on hand, 3-Methyl-2-pentanone proves itself versatile. Its ketone group means reduction, oxidation, and even alpha-halogenation reactions remain in play. Cross-coupling turns up often in synthetic planning. Nucleophilic additions, especially with Grignard reagents, open doors to various alcohol derivatives. Solvent choice and ambient conditions matter more than textbooks let on—something that only comes clear after hours of trying to clean up poorly separated layers in a sep funnel. Chemists returning to this compound often look for stable intermediates, big yields, and reasonable reaction times.
Aliases and Shelf Names
As is often the case in chemical supply, a compound never really owns just one name. In catalogs, 3-Methyl-2-pentanone shows up as Methyl sec-butyl ketone, MSBK, and sometimes even 3-Methylpentan-2-one. For regulatory discussions, the CAS number often trumps names. Keeping an eye out for these synonyms saves time and avoids expensive ordering mistakes—an easy trap for the unwary, especially for labs juggling dozens of isomers and similarly named reagents.
Staying Safe: Operational Standards and Hazards
Anyone who's poured chemicals long enough has learned that solvents like 3-Methyl-2-pentanone demand care. The solvent vaporizes easily and catches fire below average indoor temperatures, so proper ventilation and spark-free environments matter. Gloves rated for organic solvents, goggles, and chemical fume hoods aren't just suggestions—they belong in every part of handling this compound. Spills on the bench can mean more than a quick wipe; the fumes irritate the nose and throat, and prolonged exposure triggers headaches or nausea. The most reliable safety habit stems from treating every procedure as a potential risk—not out of paranoia, but because mistakes with flammable ketones stick with you.
Where It Ends Up: Application Areas
3-Methyl-2-pentanone doesn’t get the press of acetone or methyl ethyl ketone, but show up it does—in formulating paints, inks, and specialty coatings. Its solvency and volatility make it suitable for cleaning and degreasing agents in industrial use. Research labs sometimes use it to dissolve hydrophobic compounds where the more common ketones don’t quite work. Interestingly, the fragrance and flavor industries have experimented with it in trace concentrations, hunting for aroma notes that give products distinction. Its moderate boiling point also appeals to engineers designing recovery systems in closed-loop processes, reducing loss and improving recovery rates.
Chasing the Next Discovery: Research & Development
Ask researchers what keeps 3-Methyl-2-pentanone interesting, and many point to new catalytic methodologies, sustainability pushes, and solvent recycling innovations. Chemists stay busy modifying its core to build new intermediates for pharmaceuticals and agrochemicals. As green chemistry standards tighten, R&D groups hunt for ways to minimize byproducts by rethinking catalysts and closed-loop systems. Some have turned to bio-based feedstocks as starting materials, hoping to make production cleaner and less reliant on fossil-derived chemicals. Each improvement grows from both lab mishaps and lucky successes—a cycle familiar to anyone who’s tested methods with their own hands.
Weighing the Risks: Toxicity and Health Research
Years spent poring over MSDS sheets and inhaling solvent vapors (unintentionally, of course) drove the point home that 3-Methyl-2-pentanone isn’t as benign as it seems. Acute exposure in animals indicates central nervous system depression, headaches, dizziness, and respiratory irritation can occur above certain thresholds. Chronic exposure and bioaccumulation risks continue to be studied, with findings pushing for strict workplace exposure limits and engineered controls. The compound doesn’t get used directly in consumer goods and for good reason. Many practices in toxicology remind those in the field to respect what they’re handling and never take short-term comfort in the fact that severe adverse effects require high doses. Cumulative exposure stories shared over late nights in the lab often land as cautionary tales that shape future standards.
Looking Forward: Future Prospects
Change rarely comes easily, especially for niche compounds like 3-Methyl-2-pentanone. With sustainability driving much of today’s chemistry innovation, growing demand for greener solvents could either challenge or uplift its place in the chemical hierarchy. Some industry groups hope new production techniques will trim energy use and waste, drawing lessons from decades of emissions scandals and solvent recovery failures. Breakthroughs in catalysis could streamline its synthesis, making it more accessible without the cost and hazard burden that chokes off broader adoption. Safety improvements, whether by better PPE or smarter engineering controls, promise lower accident rates for the next generation of chemists. Keeping an eye on regulatory signals, safety data, and new process patents offers a map to where this solvent could go, both on a bench and in larger reactors. The story of 3-Methyl-2-pentanone reflects how even seemingly modest chemicals influence bigger trends in science, safety, and sustainability.
What is 3-Methyl-2-Pentanone used for?
Unearthing Its Role in the Industry
Everyday products often rest on the shoulders of strange-sounding chemicals. 3-Methyl-2-pentanone, sometimes called methyl sec-butyl ketone, doesn’t get much recognition outside of a chemistry lab, yet it’s woven through more industries than most realize. Chemically, it belongs to a family called ketones, which usually help other substances do their job instead of commanding the spotlight.
Workhorse Solvent in Industrial Labs
The major job for 3-Methyl-2-pentanone lands in the solvent category. Industrial coatings, paints, and adhesives rely on solvents to mix and apply smoothly. This chemical stands out because it dissolves both natural and synthetic resins, which makes it a favorite in paints and finishes aimed for wood, metals, and plastics.
Chemical plants also lean on it to help separate compounds. Strong solvency power is valuable for those aiming to replace more hazardous alternatives, like methyl ethyl ketone (MEK). Some of the usual standbys for cleaning and production sit on strict regulatory lists thanks to safety problems, and 3-Methyl-2-pentanone steps in to fill gaps while presenting fewer health or environmental concerns.
Printing, Inks, and the Push for Quality
Printing presses demand inks that flow cleanly and dry at a steady pace. Printers have found that this ketone helps with exactly that. Ink manufacturers add it because it carries colorants smoothly and speeds up drying on cardboard packaging or product labels without causing jams or smearing. Over years spent in product design, nothing derails a project more than ink problems. This solvent sidesteps many headaches, so ink companies lean on it for consistent quality.
Safety and Environmental Tradeoffs
People who handle industrial chemicals remember the slow shift away from leaded paints, mercury lamps, and chlorinated solvents. On paper, 3-Methyl-2-pentanone presents a lower risk for toxicity and environmental fallout compared to nastier cousins. Experts at the National Institute for Occupational Safety and Health (NIOSH) watch over compounds like these, flagging any health risks for workers. In research from the last decade, exposure limits tend to land higher for this solvent than some others in its class, signaling reduced acute toxicity.
Spills or fumes still call for care—nobody wants headaches, nausea, or air pollution drifting out of a paint shop or print plant. Responsible companies improve ventilation or install scrubbers, while others swap to water-based formulations when quality won't take a hit. There’s a lesson in those decisions. Small chemical tweaks—choosing a safer solvent—can ripple out to make entire workplaces and communities healthier.
R&D and the Future of Cleaner Industry
As factories push towards greener chemistry, researchers keep tweaking solvent formulas. Universities and large manufacturers pilot test safer blends, and that brings 3-Methyl-2-pentanone into more conversations. A better understanding of long-term effects, biodegradability, and emissions tracking could cement its spot or signal further change. Everybody benefits if industry starts cleaner and stays accountable.
I've watched a simple switch in solvents rescue small businesses from hefty fines or sick workers. With the right decisions, one compound can point the way to smarter chemistry and remind us that the tools we pick today shape the world for years to come.
What are the safety precautions when handling 3-Methyl-2-Pentanone?
Understanding the Real Risks
3-Methyl-2-pentanone might sound like just another chemical, but keeping your guard up is essential. This solvent features heavily in coatings, adhesives, and specialty cleaning. The challenge isn’t always obvious—exposure might come from skin contact, vapors in the air, or even accidental spills. With chemicals like this, too many have learned the hard way just how quickly a lack of protection can lead to health problems. Workers have reported headaches, nausea, and respiratory irritation after breathing its fumes for too long, and contact with bare hands brings skin irritation or worse.
Personal Protective Equipment — More Than a Hassle
Many folks shrug off PPE, especially during busy shifts. I’ve watched co-workers ditch gloves to handle small spills, thinking quick jobs call for shortcuts. Trouble is, 3-Methyl-2-pentanone passes through the skin. Gloves and goggles block exposure, and a decent apron or lab coat shields arms and clothes. You’ll want eye protection every time: a splash can lead to lasting irritation or even injury. It never feels worth that risk just to save a minute.
Ventilation Can’t Be an Afterthought
Small shops and crowded facilities rarely prioritize fresh air, but 3-Methyl-2-pentanone fumes build up quickly. Too often, you notice the headache coming on faster than the smell. I’ve seen old fans doing little in cramped corners, so local exhaust—like fume hoods or adequate open windows—makes a huge difference. Airflow matters most in warm weather or tight indoor spaces where vapors linger. Well-maintained equipment ensures those controls don’t become decoration instead of protection.
Safe Storage Takes Commitment
It’s tempting to stack chemical containers wherever there’s a free shelf. I’ve seen bottles balanced over breakroom fridges—never a good idea. 3-Methyl-2-pentanone should sit in a sturdy, labeled container in a cool, dry spot away from ignition sources. This stuff creates flammable vapors, so sparks from tools or static electricity can turn laziness into disaster. Good habits—double-checking lids, keeping flammable materials separate, and using dedicated storage—protect more than just property.
Spill Response Plans Save More Than Time
Accidents don’t wait for the safety officer to arrive. Clearing the area fast, grabbing the right spill kit, and using absorbent material like sand or commercial pads reduce the potential fallout. Workers with proper training know to seal off drains and ventilate the room. Left unchecked, a small spill grows into a legal and health headache. Regular safety drills pay off when everyone moves by instinct, not hesitation.
Training Sets the Tone
Every jobsite I’ve worked on, the mood around safety changes with the manager’s attitude. Regular training sessions keep knowledge fresh and let people ask about new risks or procedures. Posting clear instructions near workstations works better than burying them in a manual somewhere nobody checks. Whether it’s refresher tips or demonstrations, clear guidance cuts confusion and prevents “I didn’t know” after an incident.
Solutions Build a Culture of Care
Safer workplaces grow from daily habits. Workers need equipment that fits and gets replaced after wear. Facilities that invest in storage systems and working ventilation systems see fewer accidents. Most of all, safety grows from real conversations where people feel heard and supported, not rushed or blamed. In the end, respecting chemicals like 3-Methyl-2-pentanone means respecting each other—and that attitude saves lives.
What is the chemical formula and structure of 3-Methyl-2-Pentanone?
An Everyday Look at a Useful Ketone
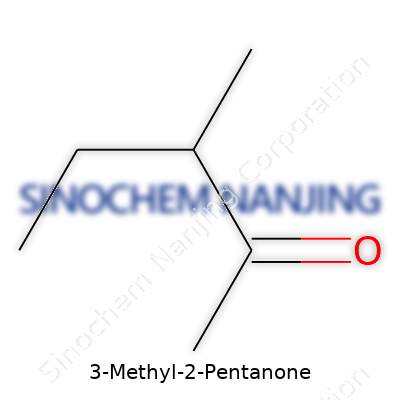
Plenty of folks outside chemistry labs may never hear about 3-Methyl-2-pentanone, but it helps shape our world from behind the scenes. Its chemical formula is C6H12O. Think of it as a six-carbon chain peppered with an oxygen atom and a little branch off the side. It offers a glimpse into how small structural changes in molecules can change their behavior and value.
The Shape That Matters
3-Methyl-2-pentanone belongs to a family called ketones, compounds recognized by a carbonyl group—a carbon double-bonded to an oxygen—nestled somewhere within the structure. In this one, you get a straight five-carbon chain, but the story doesn't stop there. At the second carbon, there’s an oxygen hanging on. The “3-Methyl” part means one carbon further down, on the third carbon, a small methyl branch juts out. Drawn simply, the structure goes as follows:
CH3-CH2-CO-CH(CH3)-CH2-CH3
Looking at this, you can see the backbone unspooling: two carbons, then that key carbonyl, a branch sticking off the third carbon, and finally a few more carbons to finish. These drawings are like a map for chemists, making sure no bonds or groups get forgotten during a reaction or product formulation process. The details matter when it comes to safety, storage, and how it reacts with other chemicals.
Why the Structure Influences Use
In my experience handling various solvents and intermediates, I’ve seen how even one small branch changes what a chemical can do. For 3-Methyl-2-pentanone, the methyl group not only nudges the boiling point a little higher than its straight-chain cousin 2-hexanone, it shifts the way it mixes with other liquids. This matters in industries looking for precise evaporation rates, like paint formulation or fine chemical extraction.
Keeping safety in mind, that same structure affects flammability and toxicity. Ketones often smell strong; workers exposed in manufacturing settings need good ventilation. Recognizing each compound’s unique branching and shape teaches us how to plan for proper protective gear, spill response, or transit regulations.
Addressing Challenges and Finding Better Paths
The chemistry world keeps moving forward, often relying on compounds like 3-Methyl-2-pentanone for synthesis and as a solvent. Yet, its use isn’t without bumps. For instance, health concerns linked to solvent inhalation push us to train employees, upgrade ventilation, and monitor exposure. Strong regulations set by groups like OSHA and the EPA shift companies to seek greener options or limit the purchase amounts for certain applications.
Finding alternatives that mimic the benefits of 3-Methyl-2-pentanone—quick drying times, effective solvency—gets trickier as regulations tighten. This sparks innovation. Some startups explore bio-based solvents from renewable sources. Others focus on engineering new compounds with fewer health or environmental risks, using knowledge drawn from traditional molecules as a blueprint.
The formula and structure of 3-Methyl-2-pentanone remind us how all choices in the lab have echoes outside of it: in paint shops, in cleaning routines, and in long-term public health. Staying informed and nimble with chemical handling and substitution can make a lasting difference, turning everyday expertise into smart, safer practices.
How should 3-Methyl-2-Pentanone be stored?
The Real Need for Safety with Chemicals
Few folks outside of labs or manufacturing shops give a second thought to names like 3-Methyl-2-Pentanone. This liquid, sometimes called methyl sec-amyl ketone, crops up in sectors like coatings, adhesives, and even pharmaceuticals. People who work with it know it comes with a sharp odor and evaporates quickly. Hazards aren’t just printed on a safety data sheet. You might not see vapor drifting, but its effects can sneak up—causing dizziness or trouble with breathing if ventilation stinks or if storage gets sloppy.
Understanding What 3-Methyl-2-Pentanone Does
This chemical acts as a solvent, breaking down or dissolving other substances. From first-hand experience in paint labs, you find out quickly it lights up easily. Even static can set off vapors. The risk isn’t just about flames—it creeps into headaches, nausea, or skin rashes after contact or inhalation. In workplaces without proper controls, accidents don’t just happen to folks who ignore the rules; they hit seasoned workers who get too comfortable.
Storing with Care: Practical Steps That Work
Doors to chemical storage areas shouldn’t swing wide for anyone wandering by. These rooms need to be locked, dry, and away from direct sunlight or heat sources. Temperatures matter. Heat speeds up evaporation and raises vapor levels, making even a small container more dangerous. If you’ve worked through a Texas summer, you know how fast garages and sheds can turn deadly.
Metal drums or high-quality chemical-grade plastic containers usually hold solvents like 3-Methyl-2-Pentanone. You never want to risk storing them in open or corroded cans. Vents and fume hoods count for a lot—just one whiff of concentrated vapor makes you realize how important moving air is.
Separation and Segregation: Keeping Chemistry Simple
Staff in larger companies follow a color-coded system for different hazards, and it saves headaches in emergencies. Mixing chemicals isn’t just theory—vapors from a nearby acid or oxidizer can spark a fire. Solvents live far from oxidizers and acids, often in separate cabinets. Even at home, putting cleaning chemicals and gasoline together spells trouble. Simple habits, like labeling and sealing containers right after use, save more messes than most realize.
Emergency Readiness: More Than a Fire Extinguisher
Fast action keeps small spills small. Absorbent pads, spill kits, and proper waste bins belong in any chemical storage area. Relying on a borrowed mop or old towels means tracking chemicals across floors or into drains. Fire extinguishers rated for chemical fires make a difference, but training matters more than just buying gear. Everyone in the building should know where exits sit and how alarms sound—not just the safety officer.
Reducing Risk in Everyday Practice
Regular checks keep surprises away. Inspecting seals, looking for drips, or hunting down labels that start to fade are habits built through training and experience. Talking openly about near-misses or slip-ups encourages new hires to speak up. Skipping steps because “we’ve never had a problem before” usually leads right to trouble.
People often think chemical spills and accidents only hit factories, but garages, art studios, and even cleaning closets pick up risk when solvents are stored carelessly. Proper storage of 3-Methyl-2-Pentanone isn’t about following every obscure regulation—it’s good sense and a way of looking out for friends, family, and co-workers.
Is 3-Methyl-2-Pentanone hazardous to health or the environment?
What 3-Methyl-2-Pentanone Means in Day-to-Day Life
3-Methyl-2-pentanone enters the picture mostly as a solvent in paints, coatings, and adhesives. Folks working in manufacturing or laboratories might cross paths with this compound, but the average person doesn't run into it at the grocery store or park. Its powerful ability to dissolve certain substances gives it value in industry, yet not every powerful solvent is harmless.
Risks Workers Face
Breathing in the fumes from 3-methyl-2-pentanone can irritate the nose, throat, and lungs. Prolonged exposure piles on new problems—headaches, dizziness, and even confusion. Years back in my first warehouse job, not enough attention was paid to good ventilation; headaches weren’t uncommon, even though most workers blamed dehydration or bad coffee. Companies today know more and can invest in safety, yet accidents and shortcuts still happen. Skin contact doesn’t lead straight to disaster, but rashes and irritation stack up without gloves. Safety Data Sheets advise workers to keep this solvent off their skin and out of their lungs for good reason: repeated exposure chips away at health over time.
Concerns About Air Quality
Much like other industrial solvents, 3-methyl-2-pentanone does not respect property lines. It evaporates easily, especially in a poorly ventilated plant. Once in the air, it contributes to the formation of ground-level ozone. The American Lung Association warns about ozone’s link to asthma and respiratory issues, especially for children or seniors. Factory workers see the most danger, but neighborhoods downwind from large plants may notice the impact too. If regulatory agencies set strict emission limits, companies may cut back. Monitoring equipment doesn't just gather dust; it keeps the worst emissions in check.
Environmental Impact Once It Leaves the Factory
3-Methyl-2-pentanone rarely sticks around outdoors for long. Sunlight and other chemicals in the air break it down fairly fast. Unlike heavy metals that settle and poison soil, it’s not a top concern for groundwater or crops. There’s no guarantee runoff won’t cause issues, though. Experience shows that industries tend to underestimate risk when waste disposal gets expensive. Companies have dumped solvents into waterways in the past, chasing short-term savings with long-term costs. Vigilant regulation and community watchdogs help keep waterways clean.
Better Practices and Solutions
Big factories now rely more on closed systems that trap fumes instead of letting them leak into the air. Automation helps by removing people from direct exposure—robots don’t develop rashes or lung irritation. Mandating proper masks, gloves, and ventilation makes a world of difference for human health on the floor. Switching to greener solvents comes up in meetings but doesn't always stick—managers push back because new compounds can mean higher costs or complicated approvals. Sometimes laws force change when wallet and habit slow it down. Knowing that people still cut corners drives home the need for regular inspections and worker training. Environmental groups and local governments calling out bad actors pressure companies to clean up their act.
Why Awareness Matters
Folks who work with chemicals like 3-methyl-2-pentanone need clear information—not just fine print or rushed safety videos. I’ve seen firsthand that real-life stories and practical know-how save more people than lists of technical hazards. People can't choose safety if nobody levels with them about real-world risks or solutions. If companies, governments, and communities work together, hazardous solvents don’t have to cause lasting harm. Sharing knowledge, setting real standards, and enforcing the rules—these steps matter for both livelihoods and communities downwind from the plant.
| Names | |
| Preferred IUPAC name | 3-Methylpentan-2-one |
| Other names |
Methyl sec-butyl ketone MSBK 3-Methylpentan-2-one sec-Butyl methyl ketone |
| Pronunciation | /ˈθriːˈmɛθɪl tuː ˈpɛntəˌnoʊn/ |
| Identifiers | |
| CAS Number | 565-69-5 |
| Beilstein Reference | 1209244 |
| ChEBI | CHEBI:34941 |
| ChEMBL | CHEMBL15351 |
| ChemSpider | 7443 |
| DrugBank | DB02171 |
| ECHA InfoCard | 100.011.369 |
| EC Number | 246-558-9 |
| Gmelin Reference | 80849 |
| KEGG | C06215 |
| MeSH | D008699 |
| PubChem CID | 7906 |
| RTECS number | SA9100000 |
| UNII | 6Z1GP13V76 |
| UN number | UN2397 |
| Properties | |
| Chemical formula | C6H12O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet minty ketonic |
| Density | 0.805 g/mL at 25 °C |
| Solubility in water | soluble |
| log P | 0.89 |
| Vapor pressure | 2.74 kPa (at 20 °C) |
| Acidity (pKa) | pKa ≈ 20 |
| Basicity (pKb) | pKb = 10.26 |
| Magnetic susceptibility (χ) | -7.97 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.404 |
| Viscosity | 1.04 mPa·s (20 °C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 282.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -302.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3406.1 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H319, H335 |
| Precautionary statements | P210, P261, P271, P280, P301+P312, P303+P361+P353, P304+P340, P312, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 42 °C |
| Autoignition temperature | 515 °C |
| Explosive limits | 1.2–8.0% |
| Lethal dose or concentration | LD50 oral rat 1600 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1600 mg/kg (oral, rat) |
| NIOSH | MV4700000 |
| PEL (Permissible) | 100 ppm (410 mg/m3) |
| IDLH (Immediate danger) | 300 ppm |

