3-Methyl-2-Butanone: Science, Safety, and Societal Value
Looking Back: The Evolution of 3-Methyl-2-Butanone
Stepping into the world of ketones, 3-Methyl-2-Butanone stands out as a workhorse in industrial chemistry. My first exposure to this compound came during a university organic lab, where its sharp, ether-like aroma was impossible to ignore. It arrives with a long track record, tracing roots to early research on ketonic solvents in the mid-1900s, as the need for alternatives to diethyl ketone or acetone began surfacing in both industrial and academic circles. Around this time, innovation and necessity pushed chemists to examine each isomer’s unique behavior, and 3-Methyl-2-Butanone found its niche, particularly in extraction, flavoring, and even as an intermediate for further synthesis. The compound sprang not from sudden discovery, but from generations of hands-on work addressing industry demands for solvents that balanced reactivity with selectivity.
What Sets 3-Methyl-2-Butanone Apart
3-Methyl-2-Butanone doesn’t grab headlines like industrial titans toluene or acetone, but its features carve out critical roles. You notice its colorless liquid form, an unmistakable fruity scent, and volatility on par with other simple ketones. Where it really flexes its muscle is in its relatively high solvency coupled with low water miscibility, making it ideal for applications requiring selective solubility. The technical jargon boils down to this: it does a job that other ketones struggle with, especially when handling hydrophobic compounds during flavor or fragrance formulation. As a moderately polar solvent with a boiling point near 92 degrees Celsius, it sits between acetone and methyl isobutyl ketone, carving out a comfortable spot for niche uses.
Down to the Science: Properties and Technical Markers
Take a closer look at 3-Methyl-2-Butanone, and you find that it carries a chemical formula of C5H10O, molar mass of 86.13 grams per mole, and a density hovering around 0.805 grams per cubic centimeter at room temperature. Its vapor pressure and flash point make it a fire risk, not unlike most simple ketones, so responsible use weighs heavy on anyone handling it. Regulatory agencies keep a close eye on such properties, ensuring chemical manufacturers stamp containers with hazard icons and risk phrases according to GHS and regional standards. Purchase any batch, and you spot the telltale warnings about vapors, skin contact, and safe storage away from ignition sources. This sense of caution isn’t bureaucratic overkill—these specs have built-in wisdom, mostly written in the ink of previous accidents.
From Lab Bench to Factory Floor: Production and Preparation
Early methods for 3-Methyl-2-Butanone preparation relied on traditional oxidation techniques, transforming 3-methyl-2-butanol through controlled reactions with strong oxidizers. Today’s best practices focus on balancing yield with cost, turning to catalytic dehydrogenation or oxygenation in finely tuned reactors. If you step into any modern chemical plant, you see adherence to process safety, closed systems, and regular monitoring—not because regulation demands it, but because every chemical engineer learns quickly that loose process control leads to lost product and costly cleanup. Lab syntheses can look simple in organic chemistry textbooks, but scaling up always exposes unexpected hazards and environmental concerns. The old days of dumping waste or ignoring small leaks are over; the field now leans on lifecycle thinking, closed-loop production, and rigorous emissions monitoring to stay both profitable and responsible.
Chemical Flexibility: Reactions and Modifications
In chemical synthesis, 3-Methyl-2-Butanone is more than a solvent—it acts as a valuable intermediate in producing flavors, pharmaceutical agents, and even specialty polymers. Its ketone group welcomes nucleophilic addition, while the methyl groups influence regioselectivity in more complex transformations. Chemists learned to use this compound for Grignard reactions, acylations, or even as a precursor for 3-methyl-2-butanol by reduction. The trick lies in controlling side reactions, especially since branching in its carbon skeleton nudges certain rearrangements. A student might first recognize its chemistry during a simple reduction or oxidation, but the real-world impact settles in as you spot it in patent filings and process patents, quietly enabling progress in materials and specialty chemicals.
The Many Faces of 3-Methyl-2-Butanone: Names and Synonyms
Every chemist who has flipped through catalogs or old literature knows that one compound can have a half-dozen names, and 3-Methyl-2-Butanone is no exception. People might call it methyl isopropyl ketone or MIPK, depending on the context. Lab inventories sometimes spill over with abbreviations or legacy terms, so newcomers often need a decoder ring just to keep track. This scattergun naming only reinforces the need for standardized labeling and universal CAS numbers, which the profession relies on to cut confusion and prevent dangerous mix-ups in storage or usage.
Keeping the Workplace Safe
I never forgot the scent of 3-Methyl-2-Butanone clinging to lab gloves, and that memory always sparks a reminder about safety. Its vapors can irritate eyes and nose in small amounts, and higher levels pack a punch to the central nervous system—not quite as harsh as some chlorinated solvents, but enough to warrant solid precautions. Laboratories and factories face clear guidelines: proper ventilation, flameproof storage cabinets, and well-trained staff equipped with goggles and gloves. The compound’s low flash point and ready evaporation make it less forgiving during summer work or in confined spaces. We’ve come far since open benches and casual handling; industry standards and worker training now keep most accidents at bay, but compliance isn’t just about ticking boxes—it’s about sending everyone home in one piece.
Beyond the Lab: Where 3-Methyl-2-Butanone Matters Most
You encounter 3-Methyl-2-Butanone in settings few people recognize: food technology, where flavor houses use it in strict, minute concentrations; coatings and ink formulations; even pharmaceuticals seeking just the right balance of polarity for intermediate steps. Not every solvent or intermediate gets regulatory clearance for the food supply, so its limited but meaningful use there points to the compound’s relatively well-understood toxicology profile—especially when weighed against aromatic alternatives. Its reach stretches further into specialty adhesives and extractive metallurgy; the solvent’s selectivity helps separate value from waste, whether that means flavors from botanicals or metals from ore. Regulations on allowable levels keep industry on its toes, since consumer safety hangs in the balance.
Driving Progress: Research, Development, and Innovation
The science of 3-Methyl-2-Butanone hasn’t stood still. Researchers keep probing its qualities for green chemistry solutions, especially its biodegradability and alternatives to petroleum-based sources. Ongoing projects examine catalysts that lower production temperatures or new separation techniques that reduce emissions. My own experience in an industrial research lab taught me that solvent research never ends—some critics dismiss compounds like 3-Methyl-2-Butanone as legacy chemicals, but innovation often means making an old molecule do new tricks with cleaner, safer processes. Sustainable feedstocks, process intensification, and digital process monitoring all hold promise for minimizing the environmental cost while maximizing industrial value.
The Toxicological Puzzle
Toxicity work on 3-Methyl-2-Butanone paints a picture both reassuring and cautionary. Animal studies show low acute toxicity, with higher exposure levels yielding respiratory irritation, headaches, or narcotic effects. Chronic studies remain rare, making long-term effects less defined. Regulatory agencies scrutinize occupational exposure limits and update guidelines when new data surfaces. Workers in close contact carry the most risk, especially where management cuts corners or ignores airflow requirements. One lesson comes clear: constant attention to exposure data, personal protective equipment, and effective incident reporting matters for keeping risk as close to zero as possible. Industry-wide adoption of real-time monitoring and data-driven safety audits could further reduce unknowns, offering better health protections as future research fills in current gaps.
Looking Ahead: Future Prospects for 3-Methyl-2-Butanone
As the chemical industry marches toward sustainability, compounds like 3-Methyl-2-Butanone face new scrutiny. You see pressure for greener solvents, tighter emissions, and transparent supply chains. Some researchers test biosynthetic pathways, aiming for fermentation-based production instead of fossil-derived feedstocks. Process engineers look for more efficient reactors and recovery systems, slashing both resource use and waste. While some industry consultants forecast a gradual decline as new alternatives enter the market, the compound’s unique blend of solvency, volatility, and reactivity guarantees it won’t fade away quickly. Its future likely hinges on how well producers adapt—switching to circular economy models, investing in green chemistry, and showing regulators and the public that small, incremental improvements add up over time. My own gut tells me that as long as society keeps demanding safer, greener, and more specialized chemicals, 3-Methyl-2-Butanone will remain in the toolkit, always up for reinvention.
What is 3-Methyl-2-Butanone used for?
What 3-Methyl-2-Butanone Does in the Real World
Walk into a paint shop or a manufacturer’s floor and the air often carries a distinct, sharp, somewhat sweet aroma. That's the fingerprint of industrial solvents, and 3-methyl-2-butanone is one that often shows up if you look at the ingredient labels. Used mostly as a solvent, this compound plays a practical role in producing everything from fast-drying paints to specialty coatings.
Manufacturers like 3-methyl-2-butanone because the compound thins other chemicals quickly. It speeds up the drying process for paints and coatings, which helps factories boost production. This speed also reduces the need for energy-hungry ovens or fans. Wood shops use it in lacquers to get smooth finishes in less time, shifting more products out the door each day. If you're handling leather or synthetic rubber goods, this chemical shows up in the adhesives that bond soles to shoes or linings to upholstery. Basically, if something needs to stick and stay stuck, there’s a good chance that this chemical or its cousins have played a role.
Why Chemists Keep Choosing It
Experience in industrial labs teaches you to respect solvents that mix well and evaporate at just the right pace. 3-methyl-2-butanone stands out for its balance—fast enough to matter, slow enough to work with—making jobs more reliable and less nerve-wracking. As a technician, I saw paint booths avoid runs, drips, and uneven color because the solvent helped lay down coatings evenly. It felt good to rely on something that improved the workday for everyone on the floor.
On top of that, this molecule isn't picky about what it dissolves. It handles oils, greases, and resins with ease. Research in chemical engineering journals backs up its versatility, with tests showing the solvent pulling its weight in diverse applications from lab reagents to graphic inks.
The Health and Environmental Conversation
Alongside the benefits, there’s no avoiding responsibility. Exposure to 3-methyl-2-butanone fumes can irritate eyes or lungs, especially in places with poor ventilation. The National Institute for Occupational Safety and Health (NIOSH) points out the need for solid controls, and that matches what I’ve seen in well-run shops: air filtering units, fresh-air masks, and training that teaches people what to do if spills happen. It’s common sense, but it stops real harm on real people.
This chemical slips into waterways if nobody pays attention to waste handling. Facing up to this problem, companies invest in better containment and treatment. Technologies like activated carbon filters or chemical traps catch solvents before they escape. As regulations tighten—especially in regions focused on worker and environmental health—factories upgrade equipment or swap in safer alternatives where possible. Government agencies push for more data and less pollution, so responsible managers partner with environmental engineers and chemists to reduce exposure risks for both crews and communities.
Finding Better Ways Forward
Industries can keep using 3-methyl-2-butanone more safely by focusing on process improvements. Closed-loop solvent systems recover and recycle most chemicals on site, slashing waste and cost. Digital controls track usage and leak detection, turning spills into rare events instead of weekly cleanups. Chemists keep searching for bio-based or lower-toxicity substitutes, and some are already starting to work on production lines for less demanding applications.
In short, 3-methyl-2-butanone plays a quiet but vital part in the products people see every day—from polished floors to glossy packaging. The smartest way forward is to keep innovating both how we use it and how we protect ourselves and the planet in the process.
Is 3-Methyl-2-Butanone hazardous or toxic?
Getting to Know 3-Methyl-2-Butanone
3-Methyl-2-butanone doesn’t make headlines like mercury or lead, but anyone involved in science, manufacturing, or even art restoration could end up working with it. I remember the smell from a spray paint lab—sharp, almost fruity. This compound, often used as a solvent or in extraction, turns up in plenty of specialty chemicals. Before rolling up sleeves to use it, most folks want to know: is it safe?
Understanding the Risks
The thing about organic solvents—especially ketones like 3-methyl-2-butanone—is they pack some punch. OSHA and NIOSH list ketones among substances that cause irritation when inhaled or splashed on skin. Records show folks exposed to high vapors end up with headaches, dizziness, and dry throats. I once had to step back from a poorly ventilated workspace, eyes tearing up from fumes. Safety data sheets confirm eye and respiratory irritation as real issues. Long-term exposure brings a risk of central nervous system effects, even organ damage.
It doesn’t stop there. If spilled on skin, this liquid gets absorbed fairly easily, leading to redness and discomfort. Lab safety drills taught me never to ignore a small splash. Ingesting this chemical might burn your insides, and health agencies point out enough could damage kidneys or liver. Working with it in an enclosed space without a proper mask or gloves invites problems. The faint, sweet odor isn’t a reliable warning; olfactory fatigue sets in quickly, making leaks and overexposure hard to spot.
Fire Hazards and Environmental Concerns
Beyond personal health, fire is a real danger. 3-methyl-2-butanone ignites at low temperatures, sometimes just from static sparks or a poorly grounded electrical outlet. Firefighters I’ve spoken to rate chemicals like this at the top of their hazardous material charts in industrial blazes. Once spilled, it evaporates fast, which means vapors hang around and drift—potentially filling a room with an invisible, explosive cloud.
Then there’s the issue of disposal. Solvents like this run into wastewater, tempting fate with both the environment and the law. EPA guidelines urge against letting such chemicals go down ordinary drains. Toxicity extends to aquatic life, meaning careless disposal can harm rivers and fish long after the factory lights turn off.
Solutions: Safer Use and Responsible Decisions
Nobody wants to lose productivity, but simple safeguards cut risks sharply. Fume hoods, sturdy gloves, and fitted goggles do more than check boxes—they protect eyes, lungs, and skin from acute harm. Good ventilation and routine air quality checks make a real difference. Among coworkers, sharing tips and observations often helps spot trouble before any damage happens. I always appreciated a team that called out anything out of the ordinary right away.
Substituting less toxic solvents stays at the top of prevention wish lists. Careful storage, clear labeling, and proper waste handling shore up the last line of defense. Local hazardous waste drives or industrial pickup services keep chemicals like this far from storm drains and backyard incinerators.
Weighing the benefits against the costs, taking these chemicals lightly creates headaches—sometimes literal. Anyone thinking of working with 3-methyl-2-butanone should put safety and respectful handling first, long before any real damage occurs. That mindset saves more than just paperwork in the end.
What are the storage requirements for 3-Methyl-2-Butanone?
Why Storage Matters for Chemical Safety
Anyone who spends time in a laboratory or storage facility knows that chemical safety doesn’t only involve handling. Safe storage makes all the difference, especially with volatile substances like 3-Methyl-2-Butanone (also called methyl isopropyl ketone). I remember the first time I worked with this compound in a small industrial lab: the facility manager stressed the risks that come from improper storage, and for good reason. Safety incidents traced their way back not to handling errors, but to overlooked storage basics.
Physical Risks Linked with Volatility and Flammability
3-Methyl-2-Butanone releases flammable vapors at room temperature. Placing containers in cool, well-ventilated spaces goes a long way toward minimizing fire hazards. Laboratory guidance, including OSHA and NIOSH documents, highlights that temperatures above 30°C dramatically increase vapor pressure—small mistakes in temperature control add up. Keeping this chemical away from direct sunlight, heaters, and open flames lowers the chance of sudden ignition. I’ve seen firsthand how a sunny window can warm a storage shelf past safe levels in just an hour.
Combustible materials should always stay separated. I once witnessed the aftermath of a poorly organized storeroom where oxidizers and flammable solvents sat together—the risk wasn’t hypothetical. Organized shelves, clearly labeled containers, and good housekeeping keep incompatible chemicals apart and reduce the chance of catastrophic reactions.
Material Selection for Containers
Not all storage containers offer the same level of protection. Strong, tightly sealed containers made from materials that resist ketones hold up best. Polyethylene and specific metal alloys tend to outlast other types, like ordinary plastics, which crack or degrade after repeated exposure. The caps must seal vapor-tight; loose lids let vapors seep into storerooms, creating hidden hazards. I’ve found that investing in quality containers makes day-to-day life safer, easier and averts unexpected cleanup jobs.
Ventilation and Detection Are Essential
Ventilation keeps air fresh in storage spaces, dispersing vapors before they can reach dangerous concentrations. Good design sends any escaped fumes outdoors, not into adjacent work areas. In older buildings, I’ve helped add simple mechanical fans, which can bring dramatic improvements in air quality. Installing vapor detectors provides early warnings of leaks—these sensors cost less than the expense of an emergency, and they offer real peace of mind.
Security, Documentation, and Emergency Planning
Keeping 3-Methyl-2-Butanone under lock and key helps prevent theft or misuse, but documentation matters even more. Tracking inventory and noting container ages helps spot problems before they grow. Written emergency procedures—posted close to storage areas—give everyone clear guidance in case of fire or spill. Practicing spills clean-up and fire response drills transforms a checklist into a team’s instinctive response.
Long-Term Resilience Calls for Continuous Review
Conditions change over time: storerooms age, staffing levels fluctuate, and policies shift. Regular audits catch small issues before they grow large. In my experience, nothing beats a walk-through with fresh eyes—checking for crowded shelves, expired reagents, or blocked vents. This kind of hands-on inspection uncovers risks that paperwork alone will miss.
What is the chemical formula and molecular weight of 3-Methyl-2-Butanone?
Chemical Make-Up and Why It Matters
3-Methyl-2-butanone, known among chemists as methyl isopropyl ketone, shows up in more places than you might expect. The chemical formula is C5H10O. Sitting on the shelf, 3-Methyl-2-butanone doesn’t look like much—a clear, colorless liquid. Its molecular weight checks in at 86.13 g/mol. Knowing this number isn’t just about memorizing facts for a test. Molecular weight helps decide how much vapor this chemical will produce and how fast it leaves a surface. It’s woven into daily lab calculations, helping researchers stay safe and get their math right on the first try.
Real-Life Importance in Industry and Research
Handling chemicals like this in a lab, one slip with the math or a missed label can turn a day sideways. I’ve measured out ketones like 3-Methyl-2-butanone for routine extractions and missed the correct mass by trusting an old datasheet, only to realize later how a single digit can mess up an experiment. In manufacturing, a slight mistake in the calculation can ripple through an entire batch. For context, global chemical production lives and dies by details like these. If the molecular weight gets misused, a process might not just stall—it could release too much vapor, risk a fire, or slow down output.
3-Methyl-2-butanone helps clean, degrease, and dissolve in industrial settings. Some manufacturers pick it over other solvents because of how it interacts with resins or coatings. Its size and structure let it do jobs that larger or smaller molecules struggle with—meaning a company might lean on this chemical in adhesives or tough-to-clean electronics. The formula, C5H10O, gives quick insight into its volatility and how it might bond or mix with other ingredients.
The Science Behind the Numbers and Safety
Chemical formulas don’t just sit in textbooks. Picture it: five carbons, ten hydrogens, one oxygen, linked up to give 3-Methyl-2-butanone a flashpoint that’s lower than water’s boiling point. That property cranks up the need for proper ventilation, grounded containers, and good labeling. I’ve worked in labs where a poorly stored solvent meant closing up shop to let the fumes clear out. These situations waste time and raise the risk of accidents. The right number—the molecular weight—gives emergency responders, industrial workers, and teachers the details they need to react fast and keep everyone healthy.
Moving Toward Solutions
Keeping up with current chemical data makes a difference. Updated safety datasets ensure that teams don’t stumble over avoidable mistakes. Digital inventory tracking helps flag odd storage trends or excessive vaporization in real-time. Students and industry trainees should get hands-on practice with resources like PubChem and ChemSpider, where the formula C5H10O and the molecular weight 86.13 g/mol stay accurate to the latest research. I’ve seen that investing in smart chemical storage, employee training, and open communication shaves down risks and keeps projects on track. The field needs attention to detail, not just in measuring, but in making sure everyone has the right numbers at their fingertips.
How should 3-Methyl-2-Butanone be handled in case of a spill or exposure?
The Real Hazards in a Bottle
3-Methyl-2-butanone is not just another chemical sitting on a warehouse shelf. Its sharp smell gives away its strength, but it’s the risk to health that really matters. A splash on the skin or a lungful of its fumes can mean nausea, headaches, and even more serious effects after enough contact. It acts fast and lingers in the air, which makes paying attention during storage and use a basic rule for anyone working around it.
Pay Attention: Where Mistakes Happen Most
I’ve watched spills unfold in busy labs, where people carry on without proper gloves, thinking nothing much of a drip. In a few minutes, headaches start and the air feels heavier. Some workers feel the urge to keep cleaning with a towel or mop, thinking it’s no big deal. That’s where trouble begins. Chemicals like 3-Methyl-2-butanone never wait for second chances. If you ever see a label with that name, remember it can burn more than your skin — it can damage lungs and even irritate the eyes in seconds. According to the CDC, direct exposure can irritate tissues fast, and breathing vapors impacts the nervous system, leading to confusion or dizziness.
Step One: Protection Over Panic
Safety starts before the spill. Lab coats, nitrile gloves, chemical splash goggles, and a face shield make all the difference. Fume hoods should not be decorations — they cut down the risk of breathing in dangerous vapors. Long sleeves keep skin covered, and rubber boots guard feet if things get messy. I once saw someone try to clean a similar solvent without gloves, and the results landed him in urgent care.
Spill Response: Act Fast, Act Smart
At the first sign of a spill, open windows and kick on exhaust fans. Any step that moves those fumes away from faces helps prevent inhalation. Next, dry sand or an approved absorbent powder works best to contain the spread. No one should grab floor mops or cotton towels, since those can spread the chemical. Shovel the sandy mix into a metal drum — plastic bins sometimes react poorly with certain solvents. Never let this stuff near a drain; groundwater contamination hurts ecosystems and risks legal trouble.
If the skin gets exposed, running water does more good than panic. Strip away affected clothing quickly, and keep flushing the area. For a splash in the eye, eyelid held open, a steady rinse for at least fifteen minutes can keep damage at bay. Fresh air is the answer for anyone feeling dizzy or nauseous, and if symptoms drag on, a trip to the ER beats toughing it out. According to OSHA, quick action saves the most people from lasting harm after chemical exposure.
Long-Term Lessons
Training rarely captures the gravity of a bad spill. Having walked through accident reports and seen what happens when complacency wins, I know most incidents stem from skipped steps and overconfidence. Documenting near-misses and reviewing emergency plans make a difference as much as any fancy gear. Clear signage, proper labeling, and regular reminders about personal protective equipment turn safe handling from policy into habit.
Disposal demands just as much thought. Used absorbents and waste get sealed tight in proper containers. Leaving barrels in the open out back invites weather and spills, which leads to fines and environmental headaches. Good record-keeping and prompt removal keep both people and nature safe.
Why It All Matters
The cost of safety feels small next to the real price of a mishap. An exposed worker or poisoned stream never belongs in the budget. Fact sheets and training matter most when everyone approaches every small bottle with respect. Handling 3-Methyl-2-butanone right means nobody needs to learn safety the hard way.
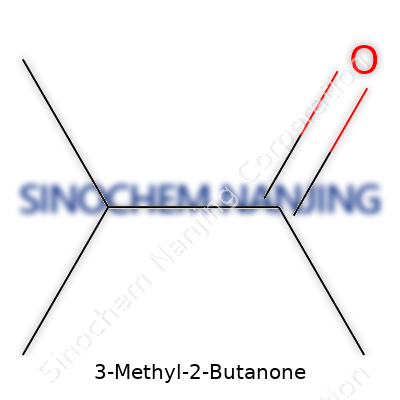
| Names | |
| Preferred IUPAC name | 3-methylbutan-2-one |
| Other names |
3-Methylbutan-2-one Methyl sec-propyl ketone 3-Methyl-2-oxobutane Isopropyl methyl ketone MPK |
| Pronunciation | /ˈθriːˈmɛθəlˈtuːˈbjuːtəˌnoʊn/ |
| Identifiers | |
| CAS Number | 563-80-4 |
| Beilstein Reference | 1209247 |
| ChEBI | CHEBI:34249 |
| ChEMBL | CHEMBL14319 |
| ChemSpider | 7276 |
| DrugBank | DB02141 |
| ECHA InfoCard | 03c67e6d-9996-4218-97eb-81ac02a2e85e |
| EC Number | 201-134-4 |
| Gmelin Reference | 5837 |
| KEGG | C06158 |
| MeSH | D008772 |
| PubChem CID | 7903 |
| RTECS number | EL9825000 |
| UNII | K7MM4GL8PT |
| UN number | UN2397 |
| Properties | |
| Chemical formula | C5H10O |
| Molar mass | 86.13 g/mol |
| Appearance | Colorless liquid |
| Odor | Sweet, mint-like |
| Density | 0.805 g/mL at 25 °C |
| Solubility in water | 14 g/100 mL (20 °C) |
| log P | 0.8 |
| Vapor pressure | 4.6 kPa (20°C) |
| Acidity (pKa) | 14.7 |
| Basicity (pKb) | pKb = 7.80 |
| Magnetic susceptibility (χ) | -8.49×10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.398 |
| Viscosity | 0.54 mPa·s (20 °C) |
| Dipole moment | 2.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 234.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -216.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2413 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P261, P271, P280, P304+P340, P312, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | '2-3-0' |
| Flash point | 14 °C |
| Autoignition temperature | 515°C |
| Explosive limits | 1.4–9.6% |
| Lethal dose or concentration | LD50 oral rat 2350 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2,730 mg/kg (rat, oral) |
| NIOSH | KN9825000 |
| PEL (Permissible) | 200 ppm (590 mg/m3) |
| REL (Recommended) | 50 ppm (175 mg/m3) |
| IDLH (Immediate danger) | 3000 ppm |

