3-Methyl-1-Butanethiol: An Editorial Perspective
Historical Development
Decoding the story of 3-Methyl-1-Butanethiol lands us deep in the heart of 20th-century organic chemistry, right alongside discoveries that shaped everyday experiences of smell and flavor. Researchers examining the volatility and peculiar aroma behind certain cheeses, hops, and even skunk spray, uncovered this molecule among others responsible for powerful sensory experiences. Layers of research in agricultural, food science, and olfactory biology have been devoted to understanding its properties, origins, and safety implications. Over time, the molecule shifted from an off-note in food to a touchpoint for quality control, process monitoring, and product formulation. The journey involves curious university labs, frustrated brewers trying to diagnose stray flavors, and plenty of late nights with chromatographs and flasks. Without this science, brewers and perfumers would have far fewer tools to tackle flavor problems.
Product Overview
3-Methyl-1-Butanethiol belongs to the organosulfur family, a group notorious for producing some of the most potent odors around. The compound's reputation in the food and beverage world is well established—it’s infamous among beer makers for contributing to “lightstruck” or “skunky” flavors in poorly stored bottles. Other industries see both a villain and a muse, as the same low-threshold odorant that ruins one product becomes a valued component in another, especially in flavor arts and fragrance chemistry. The delicate balance comes in containing, controlling, or creatively applying it, challenging R&D teams to push their analytical and technical know-how. Whether in the air outside a brewery, a perfume lab, or research clean room, 3-Methyl-1-Butanethiol earns respect for its impact at parts per billion concentrations.
Physical & Chemical Properties
Chemically, 3-Methyl-1-Butanethiol stands out thanks to its structure: a five-carbon chain tipped with a sharply aromatic thiol group. At room temperature, it shows up as a colorless to pale yellow liquid, sporting an unmistakably strong, pungent smell reminiscent of onions, garlic, or even a whiff of cooked cabbage—those who’ve handled it in the lab don’t soon forget. The molecule’s low boiling point points to its volatility and explains why its odor travels so effectively. High affinity for forming hydrogen bonds gives it chemical flexibility, while the sulfur atom at its core opens possibilities for oxidative reactions, basic thiol reactivity, and participation in complex aroma networks. In flavor chemistry or environmental studies, this reactivity can solve mysteries about food spoilage, environmental impact, or aroma masking. It’s easy to lose track of how powerful some molecules are until they cross one’s path in pure form.
Technical Specifications & Labeling
Handling 3-Methyl-1-Butanethiol isn’t for the casual chemist. Any operation involving this compound requires airtight labeling, specified concentration limits, and strict record-keeping under laboratory standards. Certificates of analysis speak directly to purity and potential contaminants, especially in research and production settings. Laboratory containers must carry hazard statements addressing toxicity, volatility, and flammability under the Globally Harmonized System, just to keep handlers and facilities safe. Commercial shipments follow additional protocols under chemical transport regulations. For consumers in industries like food or fragrance, transparency in labeling ensures that trace components are tracked and fully disclosed according to regulatory thresholds. I’ve learned over the years that serious problems in process safety often arise from missing or outdated labels—one small oversight with a compound as potent as this one can trigger major headaches across a business or research department.
Preparation Methods
Producing 3-Methyl-1-Butanethiol calls for precise organic synthesis, usually by integrating thiol groups into branched carbon backbones. Common lab-scale syntheses often use the addition of hydrogen sulfide to 3-methylbutanal or derivatization strategies employing Grignard reagents, all under controlled, inert conditions to keep oxidation or contamination at bay. Scale-up moves into trickier territory because of the odor and hazard profile; continuous-flow setups, scrubbers, and containment procedures are a must. It’s fascinating to note how old-school synthetic chemistry, combined with modern safety and green chemistry efforts, have enabled these reactions to be run more efficiently, with lower emissions. Waste handling and post-processing also shape the success of these preparations, as no one likes a persistent sulfur smell lingering in the workspace or escaping outdoors.
Chemical Reactions & Modifications
The thiol group in 3-Methyl-1-Butanethiol gives it a distinct reactivity profile, lending itself to oxidation into sulfoxides or sulfones and reactions with diverse electrophiles. It can undergo thiol-ene reactions—useful in clicking together molecular building blocks—or even serve as a precursor for larger, more complex sulfur-containing molecules. Within brewing and food processing, chemical modifications arising during storage or heating can create off-flavors, complicating quality assurance. In analytical chemistry, tracking these reactions leads to better detection techniques and even predictive models for spoilage and product stability. The molecule also provides a springboard for research into sulfur cycling in environmental or metabolic systems. Any chemist who’s worked on flavor stability in the food tech sector knows that tracking each step where a mercaptan may transform or degrade is mission-critical for a consistently enjoyable product.
Synonyms & Product Names
Among specialists, 3-Methyl-1-Butanethiol might also appear in print and discussion as isopentyl mercaptan, isopentyl thiol, or by its IUPAC name, 3-Methylbutane-1-thiol. In some flavor and fragrance catalogs, it may go under slightly different trade names, most derivatives referencing its backbone structure. Regardless of the label, the hallmark odor always tips off professionals to its presence. For process engineers, keeping abreast of these synonyms avoids confusion during inventory checks or supplier negotiations, since one misplaced name could send the entire procurement process sideways. You see how even the mundane work of tracking chemical names ends up tangled in the quality and traceability demands of today’s supply chains.
Safety & Operational Standards
Real-world handling brings safety concerns front and center. Regulatory bodies require that anyone working with this compound observe strict precautions due to its acute toxicity, volatility, and negative sensory impact at trace levels. Standard operating procedures lay out detailed personal protective equipment requirements, air filtration, and spill protocols. Proper ventilation remains non-negotiable—nobody wants an accidental release to halt an entire day’s work. Industry standards, such as those outlined in OSHA guidelines or the EU’s REACH program, outline specific controls, which in my experience become part of daily routines in any competent lab. People underestimate just how persistent mercaptan odors can be—without rigorous training and discipline, a single drop can contaminate a research site or production line, leaving everything smelling like a bad day in the kitchen for weeks.
Application Area
Any talk of 3-Methyl-1-Butanethiol’s real-world role finds itself stretched across a broad map—from food science and beverage quality control to olfactory research, fragrance formulation, and environmental diagnostics. Brewers worry about it as the chief culprit in the “skunked” notes of sunstruck beer, prompting packaging innovations and chemistry deep-dives into light protection. In contrast, some flavorists actively use the compound in microdosed blends to simulate ripe cheese, tropical fruits, or savory broths, banking on its authenticity at low levels. Environmental engineers track mercaptan emissions to diagnose leaks or industrial off-gassing, using advanced detection technology tuned to this molecule’s signature. Over the years, each application field discovers new wrinkles—one man’s contaminant is another’s asset, depending on context, dosage, and creativity.
Research & Development
On the research front, 3-Methyl-1-Butanethiol keeps attracting interest from chemists, sensory scientists, and engineers. Developing better detection techniques—think gas chromatography-mass spectrometry with refined detectors—remains a driving goal, especially as industries demand tighter contaminant controls. Some labs dive into biosynthetic pathways, seeking to disrupt or enhance natural mercaptan production for improved food quality or engineered scents. Research publications over the last several decades outline countless innovations: enzyme-catalyzed modifications, micro-encapsulation for controlled release, and green chemistry approaches to synthesis and neutralization. Investments in research often tie back to consumer preferences—nobody forgets a spoiled batch of ale or a ruined perfume, and those mishaps inspire better technology, better training, and smarter prevention.
Toxicity Research
Scientific literature lays out the human toxicity profile with sobering clarity. Even at low exposure, 3-Methyl-1-Butanethiol triggers strong reactions in the respiratory tract and can induce headaches, nausea, or worse symptoms with prolonged exposure. Chronic exposure risks remain under close scrutiny, particularly for workers in confined spaces or processing plants. Regulatory science demands rigorous risk management: establishing exposure limits, providing clear route maps for spill containment, and mandating regular medical checks for at-risk personnel. In my own encounters, ignoring these warnings leads to both immediate and long-term consequences—the persistent odor is only the start, with subtle health effects manifesting after repeated contact. Toxicology research underpins every operational decision around the compound, as workplace safety and environmental management cannot afford any compromises.
Future Prospects
Looking forward, 3-Methyl-1-Butanethiol’s narrative is poised for new chapters shaped by advances in detection, synthesis, and sustainable management. Green chemistry offers promising leads in minimizing harmful emissions and creating safer derivatives, while digital process controls promise smarter interventions during production or storage. Regulatory authorities will continue pressing for lower permitted exposure and trace levels, prompting even greater investment in monitoring technology. On the application side, new research in sensory science hints at opportunities for more precise control over flavor, aroma, and consumer enjoyment—potentially transforming what’s now a contaminant into a creative asset. The molecule’s story now sits at an intersection, where science, safety, and creativity pull in new directions. For anyone invested in quality, innovation, or safety, this is a compound worth watching as priorities and possibilities keep evolving.
What is 3-Methyl-1-Butanethiol used for?
A Compound With a Powerful Punch
3-Methyl-1-Butanethiol isn’t a name many folks kick around at the dinner table, yet the smell certainly visits. This compound carries the kind of aroma you associate with aged cheese or even skunk spray. It packs quite a punch, even at very low concentrations, making its presence known with that distinctive, strong, sulfurous scent.
Where You’ll Encounter 3-Methyl-1-Butanethiol in Daily Life
Most people brush past its signature odor in foods and in nature. Certain cheeses, like cheddar or gruyère, owe a good chunk of their deep flavors to this molecule. It’s a natural byproduct of fermentation. Every time a cheddar wedge makes your kitchen smell like a dairy cave, 3-Methyl-1-Butanethiol is in the mix, helping create those flavors cheese lovers crave.
This compound’s sharp, skunky aroma also pops up in beer. Some craft brewers use it to add complexity to their beverages, especially in styles chasing after that “funky” profile. Its presence shapes beer from simple barley pop into a layered, heady experience.
Heavy Lifting in Fragrance and Flavor Work
The food and fragrance industries tap 3-Methyl-1-Butanethiol not only for pungency. Its power to influence taste and aroma is outsized compared to many other molecules. It finds its way into flavor enhancers made for processed foods, giving products the distinct bite many consumers associate with savory treats or gourmet snacks. Adding even a drop in a big batch makes a difference.
Tracking Skunks: From Nuisance to Utility
Beyond kitchens and factories, researchers and wildlife experts watch this compound for a different reason. The spray from a skunk, famous for clearing backyards in a hurry, relies heavily on 3-Methyl-1-Butanethiol. That same reek works as a natural animal repellent, driving off predators and curious pets. Manufacturers sometimes use similar chemicals to craft animal repellents for gardens and sheds. It’s not just a natural trick; it turns into a tool.
Challenges in Handling and Safety
Pungent chemicals like 3-Methyl-1-Butanethiol demand respect. Exposure in pure form can be overwhelming. Factories use strict safety protocols. Workers wear gloves, use ventilated hoods, and store even modest amounts in tightly closed containers. I’ve seen first-hand in flavor labs how one open vial can send the entire building scrambling for fresh air. Regulators such as OSHA set strict exposure limits for sulfur-containing chemicals, protecting people from health risks like respiratory irritation or headaches.
Potential Solutions and Future Uses
Scientists keep hunting for ways to use this molecule without the headaches—literally and figuratively. Better ventilation, closed systems, and new synthetic alternatives help control exposure. Research into odor-masking solutions can boost processing safety and meet public complaints about smells in urban settings. Using this compound more thoughtfully, and only where its specific punch is necessary, means both nature and people can benefit without unpleasant side effects.
Understanding where and how 3-Methyl-1-Butanethiol shows up helps industries and consumers make smarter choices. After years around food science and nature trails, I’ve learned that even the sharpest smells often serve a direct purpose—whether it’s making cheese memorable, beer complex, or gardens a little less tempting for raccoons at night.
What is the odor or smell of 3-Methyl-1-Butanethiol?
A Sharp Encounter
Nobody needs to spend hours in a chemistry lab to realize people react strongly to smells. One whiff of 3-Methyl-1-Butanethiol and you learn that lesson right away. People use words like “skunky,” “garlicky,” and “sweaty sock” for a reason. This chemical strikes nerves in more ways than one. Years ago, while cleaning out an old storage room behind a college organic lab, I caught a hitchhiking puff of this compound. The experience lands somewhere between rotten onions and the tail end of a crowded locker room.
The Science of Stink
3-Methyl-1-Butanethiol fits into a family called thiols. These molecules grab onto noses with clawed hands, even at low concentrations. The sulfur atom, key to their structure, gives them a powerful odor punch. For 3-Methyl-1-Butanethiol, the aroma registers at parts-per-billion. Some describe it as the archetypal “skunk spray.” That's no accident. Skunks use a ble nd of thiols—our chemical here takes a starring role.
From Facilities to Food—And Beer Disasters
This isn’t just lab trivia. Breweries fear this molecule. 3-Methyl-1-Butanethiol can sneak in and turn a prized craft lager into something nobody wants to drink. When beer gets “lightstruck”—exposed to light with hops present—3-Methyl-1-Butanethiol forms from reactions between iso-alpha acids and sulfur sources. Drinkers call the result skunky. Many have tossed an otherwise clean pint after a single sniff.
In the world of flavor chemists, compounds like this represent a challenge and a tool. Some foods depend on trace levels—think certain cheeses, or even durian fruit. Add too much, and repulsion overrides nuance. Perfume specialists know to avoid powerful thiols unless searching for shock value.
Why Care About Smells Like This?
Odors affect more than comfort. Workers in sewer management, water treatment, and certain factories run into 3-Methyl-1-Butanethiol more often than the average person. Odorous leaks spark quick investigations because even tiny amounts can signal busted pipes or chemical spills. Testing in the air often means using advanced, expensive gear. Scientists track levels in cities and workplaces to ensure public safety.
People don’t ignore intense smells. Repeated, low-level exposure to offensive chemicals leads to real distress and health complaints. I have seen folks suffer headaches and nausea just walking through rooms with thiol buildup. Medical research backs up these observations: strong, unpleasant odors can trigger migraines and harm mood, even if the molecules themselves aren’t toxic at exposure levels found in the wild.
Managing the Problem
Industrial settings deploy scrubbers, carbon filters, and fresh air ventilation for odor control. Lifecycle assessments now factor in the “odor footprint” of chemicals as part of environmental stewardship. Municipalities enforce strict discharge standards to keep unwelcome scents from drifting into neighborhoods.
A solid step forward involves improving detection methods. Digital noses and sensor networks allow for early warning and quieter communities. For industry, training on odor recognition helps workers spot leaks faster, reducing complaints and risks.
More Than Just a Nuisance
Modern life means dealing with the whole spectrum, from floral to foul. The story of 3-Methyl-1-Butanethiol highlights that even fleeting wafts can have economic, workplace, and emotional consequences. Addressing these isn’t about chasing perfection, just practical respect for a sense that fuels memory, safety, and—sometimes—laughter.
Is 3-Methyl-1-Butanethiol toxic or hazardous?
A Potent Chemical with a Powerful Stench
3-Methyl-1-butanethiol catches attention fast, mostly because of its eye-watering smell—think burning tires or rotten cabbage that never left the fridge. The world of chemicals has a few that make you take notice if they slip in the air, and this one tops the list. It shows up in the flavor and fragrance industry, labs, and even gets used as a warning odorant for gas leaks. But everyday folks rarely run into it outside of labs, plants, or certain industrial settings.
Toxicity: What the Research Says
I spent some time in a university chemistry lab, and the safety data for thiols was never far from reach. 3-Methyl-1-butanethiol packs a punch not just for your nose. Direct contact with skin or eyes leads to severe irritation. Even a splash brings burning and redness, and a whiff in the air burns the nose, throat, and lungs. Headaches and dizziness can kick in quickly, sometimes before you realize it. Chronic or repeated exposure risks more severe symptoms, like respiratory problems or, in rare cases, liver or kidney effects.
Toxicological databases and safety sheets from reliable sources—like the European Chemicals Agency (ECHA) and PubChem—report that thiols aren’t just about nasty odors. They call these compounds toxic in moderate doses and hazardous upon inhalation, ingestion, or skin exposure. Animal studies show that high enough doses of 3-methyl-1-butanethiol bring real risk: liver and kidney damage, plus central nervous system depression. These findings shaped how chemists and workers handle these substances—lab coats, gloves, fume hoods, and training.
Hazards Inside and Outside the Lab
Back at my lab bench, fume extraction systems sucked away toxic vapors, and safety showers stood close by. Out in the world, plant workers who deal with odorants and additives need similar protections. Workplaces treating or producing thiols carry legal obligations under OSHA’s chemical safety rules. Safety Data Sheets (SDS) for 3-methyl-1-butanethiol make it clear—never skip personal protective equipment (PPE) like gloves and goggles. Spills demand immediate response, with evacuation in case of high vapor concentrations. The chemical’s flammability ramps up the danger, making it a double threat: it can catch fire almost as fast as it sickens you if mishandled.
Regulation and Solutions: Respect for Safety Protocols
Across my career, ignoring chemical safety always spelled trouble. Local and international regulations treat 3-methyl-1-butanethiol as a controlled hazard. Makers and users must label, store, and transport it with care. Training makes all the difference—knowing how fast symptoms can sneak up, having spill containment ready, and using proper ventilation help keep people safe.
For smaller businesses and staff outside chemical manufacturing, guidance from agencies like OSHA and NIOSH provides checklists: ventilation, respiratory protection, emergency plans. Investing in these safeguards costs less than dealing with accidental injuries and sick leave. Good communication across teams prevents slip-ups when handling anything as toxic and malodorous as this thiol.
Why Stay Vigilant?
Even with experience, I read every updated hazard sheet like it might save a life. 3-Methyl-1-butanethiol’s dangers teach respect for chemistry’s power. It’s too easy to overlook how quickly exposure harms, especially if you’ve never experienced chemical burn or inhalation injury. Staying informed, staying cautious, and respecting protocols keeps labs, factories, and workers healthy — and that’s worth the effort every single day.
What is the chemical structure or formula of 3-Methyl-1-Butanethiol?
Breaking Down 3-Methyl-1-Butanethiol
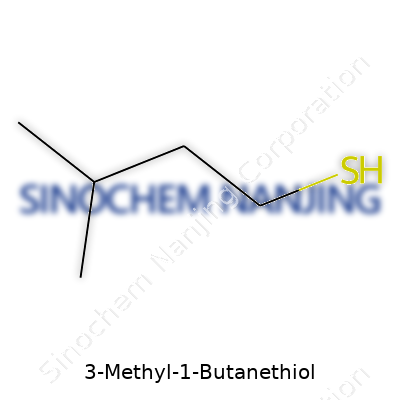
3-Methyl-1-butanethiol stands out for its scent and its use as a reference in flavor and fragrance work, but its value goes deeper than its sharp odor. This small molecule carries the formula C5H12S, which means five carbon atoms, twelve hydrogens, and a single sulfur. The way these atoms hook together shapes not just how we smell and encounter it, but the way it behaves in our world.
Picture the backbone — a chain of four carbon atoms. At the tail end, a thiol group (–SH) attaches, giving the compound its class-defining feature: a strong, often unwelcome aroma. Add a methyl group to the third carbon, and you get that three-dimensional zig-zag pattern chemists map out. No fancy tricks. Just basic chemistry, reminding us how sulfur changes everything.
Real-World Presence: From Nature to Labs
Anyone who has spent time around grains or fermentation knows how this molecule pops up in nature. It appears in malted barley, especially after malting or roasting. Some brewers develop a love-hate relationship with it. On one hand, a trace adds a characteristic note to ale. On the other, more than a whiff overshadows the whole brew.
This compound’s unmistakable scent helps scientists simulate spoilage in controlled settings. It teaches students and researchers about sulfur chemistry and volatile organic compounds. Give a whiff of 3-Methyl-1-butanethiol to anyone, and they’ll remember the lesson.
Health and Safety Concerns
Sulfur compounds go straight to our instincts; people react fast because our noses read them as a warning sign. 3-Methyl-1-butanethiol reminds us of the safety checks needed in labs and workplaces. Sulfurous smells mark the presence of low-level toxins or signal equipment leaks. Regular training that covers chemical storage, handling, and ventilation isn’t optional. Over the years, I’ve seen newcomers ignore these lessons and regret it. Gloves and fume hoods aren’t just for show.
Spill a drop of this thiol on your clothes, you’ll spend all day apologizing for the stench. Even cleaning crews can struggle to clear the air. The sulfur stickiness isn’t just about odor; exposure at higher levels can irritate skin and eyes. Having material safety data sheets for chemicals on hand never feels like overkill when something pungent hits your nose.
Addressing Challenges and Finding Solutions
Reducing unwanted byproducts in brewing or food production takes long-term focus. Training and better quality control play a big part. Analytical instruments that detect sulfur compounds give instant feedback during production, saving effort down the line. It comes down to maintaining awareness and following protocols, not shortcuts.
On a larger scale, adapting processing methods or choosing different barley strains limits the buildup of 3-Methyl-1-butanethiol. Removing these off-odors during food engineering offers more control over flavor and safety. Listening to experienced workers and researchers makes a bigger difference than new equipment alone.
Social and Environmental Impact
Take too many shortcuts with thiols, and communities near factories notice. Strong sulfur odors create complaints, disrupt business, or lead to regulatory fines. Engaging with neighbors before issues build up, using filters and carbon scrubbing, prevents escalation. Real relationships come from honesty and preparation, not just compliance paperwork.
3-Methyl-1-butanethiol might seem like a footnote to some, but its chemistry reaches far into real problems and practical solutions that touch health, industry, and daily life.
How should 3-Methyl-1-Butanethiol be stored and handled safely?
Understanding the Material
3-Methyl-1-butanethiol carries a strong, pungent odor that no one ignores in a lab or industrial setting. People who work with chemicals like this quickly learn that half-measures never cut it. From personal experience, even a small spill lingers in the air and sticks to fabric. Just a whiff signals the need for tight controls. Most often, this compound shows up in organic synthesis and flavors work, so even those familiar with strong-smelling reagents can get caught off guard.
Importance of Proper Storage
Keeping this material under control is essential for both health and reputation. Some years back, an unsealed container turned an entire research wing into a place where hardly anyone wanted to breathe. Not only did people complain, but local authorities got involved. Beyond odor, the chemical poses real flammability risks. Its low flash point means it ignites easily. People responsible for chemical stocks never store it near open flames, sparks, or sources of heat. Proper chemical storage cabinets, clearly marked for flammable substances, offer peace of mind.
Temperature and Ventilation Matter
Temperature control plays a big part in safe storage. Air conditioning or temperature-monitored spaces reduce the risk of spontaneous ignition. Years of lab work reinforce this: never trust a “cool corner” on a summer day. Storage rooms need good ventilation. Any accidental release should vent out and not linger. Fume hoods and local exhaust systems catch vapors and protect everyone nearby.
Handling Precautions
Hands-on experience reminds me that even small leaks or spills spread odor quickly. Gloves aren’t optional. Disposable nitrile gloves give better protection than basic latex. Always check for holes before use. Eye protection stands as another must. Spills splash, especially when pipetting or measuring. Safety goggles prevent painful lessons. No one should ever sniff or waft to identify this compound—it leaves a burning sensation and nausea.
Equipment Needs
Glass and compatible plastics work best with thiols. Metal containers often corrode and contaminate contents. Making sure all containers have tight, chemical-resistant seals keeps fumes locked away. Labels should stay clear, with hazard warnings in visible print.
Spill Response and Emergency Measures
Training drills show that quick response keeps minor spills from spiraling. Spill kits with absorbent materials and neutralizers sit close at hand in labs handling this chemical. Workers trained in containment steps control the spread. Waste collection doesn’t go down any normal drain. It goes directly to hazardous waste facilities. Rooms need clear exits and eyewash stations within reach.
Looking Ahead: Building Good Habits
No one can cut corners with a chemical like 3-methyl-1-butanethiol. Briefings and ongoing training remind everyone—new hires and old hands alike—about best practices. Building a safety culture means caring for each other, not just following checklists. The smarter labs run regular reviews and encourage questions. Reducing mistakes takes teamwork, respect for the material’s power, and a willingness to double-check every step.
| Names | |
| Preferred IUPAC name | 2-methylbutane-1-thiol |
| Other names |
Isopentyl mercaptan Isoamyl mercaptan 3-Mercapto-1-butanol Isoamylthiol |
| Pronunciation | /ˈθriː ˈmɛθɪl wʌn ˈbjuːteɪnˌθaɪɒl/ |
| Identifiers | |
| CAS Number | 96-03-7 |
| Beilstein Reference | 1331563 |
| ChEBI | CHEBI:52053 |
| ChEMBL | CHEMBL3634114 |
| ChemSpider | 57277 |
| DrugBank | DB11349 |
| ECHA InfoCard | 03c6dfcd-837c-4ea9-b4d7-8e20ff5afa87 |
| EC Number | 209-073-7 |
| Gmelin Reference | 30254 |
| KEGG | C08298 |
| MeSH | D013314 |
| PubChem CID | 15606 |
| RTECS number | EI8575000 |
| UNII | K76W6S7K2J |
| UN number | UN2347 |
| Properties | |
| Chemical formula | C5H12S |
| Molar mass | 106.21 g/mol |
| Appearance | Colorless to pale yellow liquid with strong unpleasant odor |
| Odor | Unpleasant, garlic-like |
| Density | 0.819 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 1.89 mmHg (at 25 °C) |
| Acidity (pKa) | pKa = 10.6 |
| Basicity (pKb) | pKb = 3.81 |
| Magnetic susceptibility (χ) | -74.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.434 |
| Viscosity | 0.85 cP (20°C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 246.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -168.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3635.8 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS07 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H314, H331, H335, H410 |
| Precautionary statements | P210, P261, P271, P273, P280, P301+P310, P305+P351+P338, P304+P340, P312, P330, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-2-0-~W |
| Flash point | 38 °C (100 °F) |
| Autoignition temperature | 220 °C |
| Explosive limits | 1.4–7.0% |
| Lethal dose or concentration | LD50 rat oral 9.5 mg/kg |
| LD50 (median dose) | LD50 (median dose): 9 mg/kg (rat, oral) |
| NIOSH | XN8575000 |
| PEL (Permissible) | Not Established |
| REL (Recommended) | REL: 0.5 ppm (1.5 mg/m³) |
| IDLH (Immediate danger) | IDLH: 2 ppm |
| Related compounds | |
| Related compounds |
1-Butanethiol 2-Methyl-1-propanethiol 2-Butanethiol 2-Methyl-2-propanethiol |

