3-Isopropyl-5-Methylphenyl N-Methylcarbamate: A Closer Look
Historical Development
Back in the twentieth century, chemical researchers began exploring carbamate compounds in the hunt for substances that could regulate pests without the environmental burdens of earlier solutions. The synthesis and practical study of compounds like 3-Isopropyl-5-Methylphenyl N-Methylcarbamate lifted the prospects for safer agricultural chemistry. Chemists noticed that derivatives of N-methylcarbamate interacted differently with biological systems; some proved safer, less persistent in the environment, and more effective for controlling certain insects. The molecule’s structural specifics emerged out of iterative organic synthesis—trial, error, analysis, and optimization. At one point, this class of chemicals drew attention due to breakthroughs in pest management and interest from public health researchers seeking new options beyond organophosphates.
Product Overview
3-Isopropyl-5-Methylphenyl N-Methylcarbamate belongs to the carbamate class of insecticides. Its structure delivers selectivity: the basic backbone—a carbamate ester—links to an aromatic ring, tweaked with isopropyl and methyl groups. The resulting compound displays moderate volatility and targets specific enzymes. Its primary recognized function involves inhibiting acetylcholinesterase, causing accumulation of acetylcholine in insect synapses. This disrupts neural processes essential for survival. Users found its action sufficiently rapid to address pest outbreaks before populations rebounded, which simplified timing the application. This feature earned it a place on shelves alongside better-known carbamates.
Physical & Chemical Properties
Diving into its physical attributes, 3-Isopropyl-5-Methylphenyl N-Methylcarbamate forms a crystalline or powdery solid at room temperature. The compound doesn’t dissolve well in water, yet shows readiness to mix with various organic solvents—acetone and ethanol come to mind. Chemically, the carbamate linkage resists slow hydrolysis unless met with acidic or basic conditions. Its melting point lands in the moderate range for carbamate esters. A faint, characteristic odor sets it apart from other unmodified aromatics. These tangible characteristics dictate handling, storage, and best practices for formulation in field products. Its molecular formula, C12H17NO2, and corresponding molecular weight aid technicians in calibration and quality control.
Technical Specifications & Labeling
Commercial-grade 3-Isopropyl-5-Methylphenyl N-Methylcarbamate often arrives with specified purity above 97%, limiting the amount of residual solvents, unreacted precursors, or trace contaminants. Labels provide key data: batch number, production date, recommended storage conditions, and hazard pictograms. Safety Data Sheets include the CAS number and hazard statements reflecting current regulatory guidance. Pesticide regulators require these details for approval, and field operators depend on them for safe handling. Manufacturers often package the technical material in impervious, tightly sealed containers to keep out moisture and prevent accidental release.
Preparation Method
Synthesis typically starts from 3-isopropyl-5-methylphenol. This aromatic base gets alkylated or acylated to attach the carbamate moiety. The process requires precise temperature control and measured doses of reagents like methyl isocyanate or pre-formed N-methylcarbamoyl chloride. The selection of solvent—such as dichloromethane or toluene—depends on scale and practical aspects of separation. After reaction, the solution is washed and cooled. Crystallization follows, with filtration isolating the product. Industrial labs often run purity checks using chromatographic techniques, and may tweak steps for better yields or fewer byproducts.
Chemical Reactions & Modifications
The fundamental carbamate ester structure brings flexibility for modifications. Hydration under strong acid or base splits the ester, reducing activity. N-demethylation changes its toxicity profile. Mutations to aromatic ring substituents (more methyls, different alkyl groups) shift target insect spectrum, duration, and absorption. Some labs experiment with substitutions to develop derivatives that break down more quickly in soil or water. Other reactions—like oxidation—prove less relevant for field use, but remain useful for forensic or environmental analysis tracing breakdown products.
Synonyms & Product Names
3-Isopropyl-5-Methylphenyl N-Methylcarbamate circulates under various names in chemical catalogues and agricultural records. The IUPAC name outlines its structure; the synonym "Promecarb" often appears in trade articles and international inventories. Chemistry texts sometimes abbreviate or use older numbering conventions, but the N-methylcarbamate suffix reliably marks its functional group. Regulatory agencies might reference different identifiers in official listings, creating some confusion for those outside the field, but pesticide operators tend to stick to one or two trade names in routine speech.
Safety & Operational Standards
Safe use rests on understanding risks. Like many carbamates, this compound can harm mammals if absorbed in significant quantities. Acute exposure leads to nerve disruption, with symptoms ranging from headaches to more severe neurological effects. Regulatory frameworks shape the boundaries for concentration in workplace air, allowable residues on crops, and cleanup standards. Workers handle the powder with gloves, goggles, and respirators in enclosed spaces. Emergency procedures rely on well-documented protocols developed from earlier pesticide accidents and poisonings. Detailed training, locked storage areas, and routine air sampling together reduce the threat of mishap. Regulatory updates roll out as research uncovers more precise limits or links with long-term effects.
Application Area
Crops facing threats from insects with known resistance to other classes often receive 3-Isopropyl-5-Methylphenyl N-Methylcarbamate as a targeted treatment. Its specificity for insect acetylcholinesterase draws interest in situations where broad-spectrum agents might kill more than intended. In some public health scenarios—mosquito control, for example—programs deploy it to break cycles of transmission for vector-borne diseases. Certain specialty crops, grown in greenhouse or high-humidity conditions, benefit from controlled spraying schedules based on this active ingredient. Distributors align product formulations with the equipment farmers already use, so integration moves smoothly into established routines.
Research & Development
Scientists continue pushing boundaries with 3-Isopropyl-5-Methylphenyl N-Methylcarbamate. Advances in organic synthesis refine the route, aiming for higher yields and less waste. Analytical chemists test for trace impurities with sharper instruments—liquid chromatography-mass spectrometry now delivers clarity that once took days. Biologists explore structure-activity relationships to nudge the molecule’s profile, hoping to boost selectivity, reduce mammalian toxicity, and shorten persistence in soil. Field trials with newer variants probe resistance patterns and track crop health. Environmental scientists map breakdown products through water and soil, surfacing considerations for future stewardship.
Toxicity Research
No one in agriculture glosses over the risks. Multiple studies involve dosing lab animals, monitoring symptoms, establishing no-effect thresholds and calculating acceptable daily intake. Discrepancies between species pop up—rodents may tolerate more than birds or bees. Reports from the field add real-world depth: accidental spills, overexposure scenarios, and residue findings push changes in labeling or allowed usage. Independent researchers and regulatory authorities both weigh in, comparing this carbamate’s short half-life with the older, longer-lasting chemicals. Experts track shifting patterns in pest resistance that could encourage overuse, reminding practitioners to rotate chemistries.
Future Prospects
The next stage for 3-Isopropyl-5-Methylphenyl N-Methylcarbamate rides on multiple fronts. Synthetic chemists tweak existing structures, aiming for compounds with faster natural degradation and optimized insecticidal action. Regulators and policy shapers push for safer coatings, new delivery systems, and updated risk assessments. Farmers and crop managers face pressure to cut pesticide load, swinging attention toward precision applications enabled by drone technology or AI-driven data modeling. Public opinion about chemical residues, pollinator health, and ecological impact pushes product developers to build and test alternatives. Academic and commercial labs keep pressing for solutions that give effective pest control without leaving an environmental footprint behind.
What is 3-Isopropyl-5-Methylphenyl N-Methylcarbamate used for?
A Look at Its Purpose
3-Isopropyl-5-Methylphenyl N-Methylcarbamate is a name that sounds more at home in a laboratory than in daily life, but this chemical has carved out a space in modern agriculture. Farmers rely on it for its power as an insecticide, tackling pests that threaten crops like rice, potatoes, and a range of vegetables. You can think of it as one of those behind-the-scenes tools that help keep food production stable and yields high.
Why Farmers Turn to Carbamate Insecticides
In my time spent visiting rural farming regions and talking with growers, I’ve heard the same concern repeated: insects don’t just nibble, they devastate whole fields when left unchecked. 3-Isopropyl-5-Methylphenyl N-Methylcarbamate belongs to a larger family known as carbamates. These work by interfering with an enzyme called acetylcholinesterase in insect nervous systems, causing paralysis and eventually knocking down pest numbers. For crops facing a wave of aphids or leafhoppers, this chemical buys precious time and protects months of work.
The Balance Between Pest Control and Safety
No story about agricultural chemicals is complete without mentioning their double-edged nature. While these compounds stand between hungry bugs and a season’s profits, they bring challenges that can’t be ignored. Regulatory bodies like the United States Environmental Protection Agency and the European Food Safety Authority keep a watchful eye on these products. These organizations evaluate health risks to farmworkers, potential residue on harvested crops, and effects on beneficial insects like bees.
Many growers have switched to personal protective gear, enhanced training, and careful sprayer calibration. I’ve seen firsthand how these habits make a difference—nobody wants to risk their health, nor do they want a crop load quarantined because of residue failures.
Sustainability and Future Directions
Farmers today work under a microscope. Consumers demand food grown with fewer chemicals, and everybody in the industry knows the stories of rivers tainted and pollinator populations shrinking. Integrated pest management (IPM) practices involve rotating chemical classes, planting pest-resistant varieties, using trap crops, and releasing natural enemies like parasitoid wasps. These strategies stretch the life of current tools and shrink the environmental footprint.
Innovation in synthetic chemistry and biotechnology keeps the industry evolving. Research centers try to develop new actives with shorter environmental lives or break down swiftly in sunlight or rain. Community workshops bust old myths—one I recall had a crop adviser walk a group through reading a pesticide label, stressing the importance of time intervals and drift reduction. Education acts as a sturdy pillar supporting safer, smarter use of farm chemicals.
Protecting Crops, Communities, and the Planet
3-Isopropyl-5-Methylphenyl N-Methylcarbamate remains one tool in a big toolbox. Growers, scientists, regulators, and communities keep searching for that balance. Crop protection draws on careful planning and respect for the long-term health of the land. Real progress will depend on communication, transparency around use, and a strong commitment to sustainable agriculture practices.
Is 3-Isopropyl-5-Methylphenyl N-Methylcarbamate safe to handle?
What’s in a Name?
3-Isopropyl-5-methylphenyl N-methylcarbamate belongs to a class of chemicals called carbamates. These compounds have shown up for years in pest control. Formulators admire their effectiveness against insects in fields, gardens, and greenhouses. But let’s not skirt around the hard truth: many in this chemical family walk a fine line between helping crops and posing real risks to humans and wildlife.
Stories from the Field
Farmers and greenhouse workers often recognize the sharp, chemical smell of carbamates. I’ve seen hands that break out in rashes, eyes watering, and even folks feeling faint after mixing certain sprays. It’s no big secret among those who handle these materials directly—there’s an edge of respect and caution in every action. Protective gear becomes second nature for them. Lab workers, too, are taught to avoid skin contact and inhalation.
Why the Risks Stack Up
Research points at carbamates acting as nervous system disruptors. Exposure, even for a short period, can trigger nausea, headaches, muscle twitches, and sometimes much more serious effects if not caught early. Scientific literature stresses that these side effects are not exclusive to lab accidents; accidental spills or careless handling can lead to emergencies even for those outside the industry, like children or pets in treated areas.
In the past decade, the World Health Organization and the EPA placed several carbamates—including some close cousins of 3-isopropyl-5-methylphenyl N-methylcarbamate—under tougher review. The pattern is consistent: studies keep showing that routine or high-level exposure brings health risks. Fish and birds often take a hit too, with runoff from fields leading to trouble in nearby rivers and woodlands. For anyone who values safe food, clean water, and intact wildlife, these chemicals deserve honest scrutiny.
What Experience Teaches
People sometimes assume that if a product is on the market, it must be “safe”—but that doesn’t tell the whole story. The fine print on every container makes it clear: wear gloves, avoid inhaling the dust, wash everything after use. Emergency protocols exist for a reason. A neighbor once shared how he overlooked basic steps during a busy morning spraying and spent hours with dizziness and blurred vision. He’s far from alone in learning safety lessons the hard way.
Practical Steps Forward
No one can ignore that modern farms count on strong chemical tools. Still, there are ways to cut down risk. Closed mixing systems, full PPE, and proper training keep accidents in check. For home gardeners or hobbyists, favoring natural options or seeking out biological controls makes sense. Information matters; labels often include emergency contacts and detailed safety data. Reading them is not optional, it’s crucial.
Communities near large-scale applications push for buffer zones and clear public notifications, mapping out where and when chemicals are used. Clean-up procedures, spill management kits, and easily reachable first aid also help. Medical staff in rural clinics should brush up on signs of chemical exposure, too, not just for farmworkers but for kids who might play near treated areas.
Weighing Benefits and Hazards
It’s never just about the chemistry. The conversation around 3-isopropyl-5-methylphenyl N-methylcarbamate reveals bigger questions. People care about health, food, and the land they rely on. That means every handler, policymaker, and neighbor plays a part in shaping how these chemicals fit into daily life. Safety doesn’t get handed out; it gets built, action by action.
What are the storage requirements for 3-Isopropyl-5-Methylphenyl N-Methylcarbamate?
Handling Chemicals with Respect
Many of us deal with chemicals at work, in laboratories, or on the farm. Storing chemicals like 3-Isopropyl-5-Methylphenyl N-Methylcarbamate means paying close attention to the basics: safety, protection from the elements, and careful labeling. This compound, often used as a pesticide, requires the same respect as any other potentially hazardous material. Parents, teachers, farmers, and warehouse workers all rely on secure storage to avoid health risks and accidents.
Why Storage Practice Makes a Difference
This carbamate isn't just another substance sitting on a shelf. Direct skin or eye contact can bring health problems. Inhalation or ingestion carries even greater risk. A warehouse near my old job failed to keep pesticides locked up, and a janitor ended up in the hospital with respiratory issues. No one wants to deal with a spill or accidental poisoning. Using common sense and basic precautions offers a strong start.
Choosing the Right Space
This chemical shouldn't sit in just any storeroom. The storage area should remain cool, dry, and well-ventilated. The room needs solid locks—it's never wise to give just anyone access. Strong sunlight or high humidity encourage chemical breakdown. Ventilation helps disperse vapors. Somebody once stored a drum in a metal shed next to a heater; the pressure build-up blew the lid off. Smart storage saves headaches and reduces long-term damage to both people and property.
Keeping Containers Tight and Secure
Original containers always offer the best protection, especially with those tamper-proof seals and manufacturers’ safety labels. Avoid makeshift bins or boxes. Sealed containers cut down on spills and prevent harmful vapors from escaping. Chemical-resistant shelves—never wood—can keep leaks in check. You don’t want drips or corrosion eating away at shelving or flooring. If anything leaks, keep absorbent material and spill kits close. Response time matters.
Preventing Mixing and Cross-Contamination
Some chemicals shouldn’t mix. Store 3-Isopropyl-5-Methylphenyl N-Methylcarbamate well away from acids, alkalis, and food. Segregated chemical storage keeps volatility low. A friend once had to clean up after an acid and a pesticide got mixed in a rainstorm. It’s a recipe for trouble—and a reason to inspect storage regularly for any damaged packaging or moisture creeping in.
Clear Labeling and Documentation
Labels fade, get scratched, or just fall away over time. Double-check labels every few months and replace as needed. Store information sheets close to the chemical itself. If someone new shows up—a new hire, a maintenance crew—nobody has to guess what’s inside that barrel or box. Keeping inventory sheets updated will let managers see at a glance what’s on site, and when new shipments arrived. During a health inspection, this organization can make the difference between a fine and a compliment.
Disposal and Responsible Access
Unused or expired material belongs in a chemical disposal site, never tossed down the drain or into the trash. Only authorized personnel—people who understand the risks—should have keys or codes to the storage area. This keeps children, pets, and untrained adults out of danger and helps build a safer workplace culture.
Final Thoughts on Safety
Storing 3-Isopropyl-5-Methylphenyl N-Methylcarbamate safely isn't just a box you check for compliance. It’s a day-to-day responsibility that touches everybody from field hands to warehouse technicians. Careful storage policies prevent incidents that harm people, property, and the environment. Clear routines, regular inspections, and straightforward housekeeping habits save time, money, and sometimes lives.
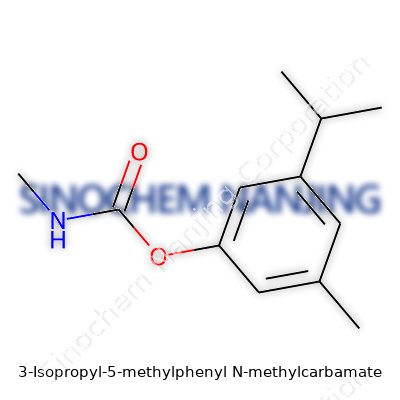
What is the chemical structure of 3-Isopropyl-5-Methylphenyl N-Methylcarbamate?
A Closer Look at the Compound
3-Isopropyl-5-methylphenyl N-methylcarbamate stands out in the world of organic chemistry not just for its complex-sounding name but for its practical relevance in crop protection. People sometimes lose sight of the fact that the way atoms arrange themselves can have ripple effects across healthcare, agriculture, and even local economies. This particular compound serves as the active ingredient in some carbamate-based insecticides, which turn heads both for pest control and for the regulatory scrutiny they attract.
The Chemical Anatomy
Here’s how the atoms link up: The base structure is a benzene ring, which people might think of as a simple hexagon in chemistry class. Add a methyl group (a single-carbon branch) at the 5-position, and you have a slight twist—a small but influential change. Swing over to the 3-position, and the isopropyl group (two carbon branches attached to a central one) takes its spot. Both branches, small and chunky, tweak the physical and biological behavior of this molecule.
Another core element is the carbamate group. It latches onto the benzene ring through the nitrogen on the N-methylcarbamate side chain. Imagine a link bridging the ring to a small chain that carries an extra methyl group, oxygen, and nitrogen. The full chemical formula lands at C12H17NO2. The structure packs bulk and reactivity in just the right way for it to work as intended, whether in a petri dish or in the field.
Significance in the Real World
The presence of both isopropyl and methyl branches fine-tunes the solubility, volatility, and overall effectiveness against insect pests. In practical use, this means a farmer has access to a tool that can knock out certain bugs without hammering the whole ecosystem. Still, every time this compound gets spread, people start asking questions—how does it break down, what long-term footprints does it leave in the soil, or in food?
Back during my early days consulting for agronomists, a lot of time was spent digging into the behavior of carbamates like this one. One client shared a story: after a routine application, the lab results showed traces of the compound way beyond the initial treatment zone. Even a slight tweak in the chemical backbone, whether an extra methyl or isopropyl group, can change how long the residue sticks around.
Risks and Solutions
The real challenge comes from the potential health risks, both for the field workers and for folks living nearby. The mechanism by which carbamates work—they block an enzyme called acetylcholinesterase—can cause issues in mammals as well if exposure passes certain thresholds. Misapplication or accidental overuse can spell trouble, making education and oversight non-negotiable. Monitoring programs, transparent labeling, and training for safe handling cut down on risk, but that’s not always enough. It’s critical to keep updating guidelines based on how fast the science moves and how local communities are affected.
Some forward-thinking researchers are digging into biodegradable versions or alternative compounds that fit the same bill but break down faster and with fewer side effects. Until those reach the market, understanding the chemistry stays top priority for anyone who needs to weigh the benefits against the trade-offs. The design of 3-isopropyl-5-methylphenyl N-methylcarbamate is a masterclass in the power and consequences of small changes in molecular structure.
How do I dispose of 3-Isopropyl-5-Methylphenyl N-Methylcarbamate properly?
Why Safe Disposal Needs Attention
3-Isopropyl-5-Methylphenyl N-Methylcarbamate, often called by its trade names, slides under most people's radars, yet it’s in some common pesticide formulas. Not every household runs into a canister of this stuff, but those in agriculture or old gardening shops might find some. Strong chemicals like this can hurt more than just bugs. Humans, pets, birds, and waterways can all suffer from careless handling.
Risks Behind the Label
Toxicity ranks high for some carbamate pesticides, which act fast in both pests and accidental victims. A little in the wrong spot can spell trouble for kids and animals. Pesticide residue clings to soil and seeps into nearby streams. I remember digging through an old storage shed, finding dusty, leaking pesticide bottles tucked behind rusty nails and paint cans – the story played out in many farm towns. Some cases in the news tell of fish kills downstream from illegal dumps. Poison control centers have files on emergency calls from well-meaning folks who poured “a little extra” down the drain, thinking water treatment plants will clean everything up. Most can’t.
What Laws and Guidelines Say
Government rules set by the Environmental Protection Agency, plus state regulations, block most household and business owners from tossing hazardous waste into regular garbage or drains. Labels on the can or bag never stretch enough – print gets tiny, instructions turn vague. The EPA groups N-Methylcarbamates as pesticides with tight restrictions. Disposal needs follow the letter, not just the spirit, of these laws. Otherwise, penalties can run high if someone gets hurt or environmental damage surfaces.
Practical Steps for Safe Disposal
Best advice: treat any leftover like industrial poison. Don’t dump it, burn it, or bury it. In cities and counties, household hazardous waste events welcome old chemicals, bottles with unreadable labels, and half-filled containers. Waste handlers at these sites know what steps to take and often accept pesticides without extra charge. Agriculture supply stores in rural areas sometimes join state “Clean Sweep” programs and collect farm chemicals one weekend a year.
Leaving it in storage racks up risk. Heat, cold, and leaks lead to accidents. Once, I saw a local hazmat crew draining a garage with cracked jugs of long-banned weed killer – the homeowners breathed pesticide fumes for weeks, not realizing the source sat behind a toolbox. No one needs that worry.
Cleaner Community Habits
Communities grow safer when disposal keeps up with changing loads of chemicals and waste. Schools, gardening groups, and neighborhood leaders educate neighbors about drop-offs. Never try to disguise old pesticide containers in household trash. Think about how even small spills reach storm drains, travel to streams, and show up in fish and drinking water. Cities with take-back days see lower rates of chemical accidents and cleaner water in neighborhood streams.
Support networks help, too. Local extension offices answer questions year-round, and state agriculture departments update lists of approved disposal locations. A quick call saves a lot of trouble. In my experience, a five-minute check with the local solid waste office gave me a pickup date and peace of mind.
Looking Out for Each Other
Handling toxic pesticides safely means looking out for the health of families, neighborhoods, land, and waterways. Proper disposal isn’t just a rule – it’s a responsibility we share. Prevention beats cleanup every time, especially with chemicals whose dangers last longer than the memory of their use.
| Names | |
| Preferred IUPAC name | 3-isopropyl-5-methylphenyl N-methylcarbamate |
| Other names |
Propoxur Baygon Blattanex Unden Bolfo Selecron |
| Pronunciation | /ˈaɪsəˌproʊpɪl faɪv ˈmɛθəlˌfiːnɪl ɛn ˈmɛθəlˌkɑːr.bəˌmeɪt/ |
| Identifiers | |
| CAS Number | 17087-03-1 |
| 3D model (JSmol) | `jmpalCnejoJOcjQ` |
| Beilstein Reference | 1201076 |
| ChEBI | CHEBI:81973 |
| ChEMBL | CHEMBL2007617 |
| ChemSpider | 21589511 |
| DrugBank | DB08667 |
| ECHA InfoCard | 03bffbcd-03bf-41ff-9ba1-0a243b43e8fa |
| Gmelin Reference | Gmelin 82122 |
| KEGG | C18533 |
| MeSH | D002388 |
| PubChem CID | 10153186 |
| RTECS number | NZ8575000 |
| UNII | 18D22V580K |
| UN number | UN2757 |
| CompTox Dashboard (EPA) | DTXSID4076080 |
| Properties | |
| Chemical formula | C12H17NO2 |
| Molar mass | 207.27 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.05 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 2.9 |
| Vapor pressure | 0.000018 mmHg at 25°C |
| Acidity (pKa) | 11.1 |
| Basicity (pKb) | 12.38 |
| Magnetic susceptibility (χ) | -61.8 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.536 |
| Viscosity | Viscosity: 1.68 mPa·s (at 25 °C) |
| Dipole moment | 2.06 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -437.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7416.7 kJ/mol |
| Pharmacology | |
| ATC code | N05CM11 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes moderate eye irritation. Avoid contact with skin, eyes, or clothing. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H332: Harmful if inhaled. H400: Very toxic to aquatic life. |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P312, P330, P363, P391, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 102 °C |
| Autoignition temperature | 455 °C |
| Lethal dose or concentration | LD50 oral (rat) 100 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 50 mg/kg |
| NIOSH | SKC64720 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Isopropyl-5-Methylphenyl N-Methylcarbamate: Not established |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
Carbaryl Propoxur Aldicarb Fenobucarb BPMC Methiocarb Isoprocarb |

