3-Hydroxybutanal: More Than Just a Chemical
Historical Development
Few people outside a chemistry lab spend much time thinking about 3-Hydroxybutanal, or, as many know it, acetaldol. Folks have tinkered with it since the late nineteenth century. In that time, research communities pieced together its value step by step, starting with foundational work by August Kekulé and the wave of synthetic organic chemistry that swept through Europe. Back in those days, practical chemistry wasn’t about making a quick buck or dazzling with nanotechnology; it was a puzzle, something that called for rudimentary glassware and a good nose for what happens when you let aldehydes mingle. Today, it remains one of those compounds that gets rediscovered every decade, doing a lot behind the scenes in labs and factories worldwide, often without a headline.
Product Overview
3-Hydroxybutanal presents itself less as some flash-in-the-pan curiosity and more as a workhorse for synthetic organic chemists. This compound acts as a bridge in reactions that yield a host of products, from flavors to pharmaceuticals. No chemist expects to see something dramatic just by looking at it – it's usually a colorless or slightly yellow liquid. Most importantly, it behaves reliably, which matters much more in a research setting than any superficial trait. Its value lies in its ability to facilitate aldol condensations, providing raw material for further transformation. In day-to-day terms, it helps scientists string small molecules together, building blocks on the way to more complex targets.
Physical & Chemical Properties
If you pop open a bottle of 3-Hydroxybutanal, you’ll notice a sharp odor. This isn’t something you'd invite to dinner, but in the hands of a skilled chemist, that smell indicates a familiar aldehyde backbone. It comes with a molecular weight of 88.11 g/mol, and its density sits just shy of water, making it easy to handle and measure out. It mixes well with polar solvents, giving it an edge in reaction setups that rely on even mixing. The reactivity profile draws heavy influence from its aldehyde and alcohol groups. With both on board, it offers a playground for chemists chasing more elaborate transformations; the molecule steps right up, ready to react at either end.
Technical Specifications & Labeling
From a practical standpoint, handling specifications mean more to the working chemist than any sales pitch. Any bottle of 3-Hydroxybutanal asks for tight labeling, clear hazard warnings, and a handy reminder that this volatile material can irritate skin, eyes, and lungs. On the technical sheet, you’ll see purity percentages, water content, and color – all indicators of whether you’re starting with something that won’t mess up your synthetic route. Even in top research settings, there’s no glamour in sloppily labeled bottles or poorly stored material. Detailed labels, accurate batch numbers, and tightly managed shelf lives contribute to safety – and reproducible results.
Preparation Method
Synthesizing 3-Hydroxybutanal often involves a hands-on condensation reaction between acetaldehyde molecules. The classic approach, known as aldol addition, involves adding a bit of base to get the reactants moving. Once formed, the product can head into isolation by distillation or extraction, depending on whether water needs removing or anything else needs tweaking. There’s an old-school simplicity in this process. It hasn’t changed much in a century, and that's a testament to its reliability. The ability to shape a versatile intermediate molecule from such basic feedstocks remains an important tool in any self-respecting organic chemist's playbook.
Chemical Reactions & Modifications
Chemists have found plenty of ways to put 3-Hydroxybutanal to work. Most pathways involve using its reactive groups to build bigger, more complicated molecules. The hydroxyl group opens the door to dehydration, giving rise to crotonaldehyde, a precursor that leads in many directions, especially in industrial chemistry. Alternatively, the molecule can undergo further oxidation or reduction, morphing into anything from alcohols to acids. Each modification demands close attention to reaction conditions, since small changes in pH, temperature, or catalyst can swing yields in a big way. In practice, these transformations form the backbone of many research and industrial syntheses – whether you’re after bulk chemicals, specialty aromatics, or pharmaceutical precursors.
Synonyms & Product Names
Walk into a storeroom and you’ll see this compound sold under several names, and keeping track of them helps avoid confusion. Alongside “3-Hydroxybutanal,” you’ll run into “acetaldol” and sometimes “beta-hydroxybutyraldehyde.” Some older texts go with “aldol” straight up, thanks to its place in the classic aldol reaction. This tangle of names speaks to its broad historical use, as well as a tendency among chemists to shorthand terms. For safety and procurement, recognizing all these names ensures nobody orders the wrong material – and no one ends up using the wrong compound in a rushed late-night synthesis.
Safety & Operational Standards
Lab safety with 3-Hydroxybutanal can't be brushed aside. With strong odors and proven irritant qualities, anyone handling it should suit up with gloves, lab coats, closed-toed shoes, and decent ventilation. Long-term exposure leads to risks no professional should ignore. Proper storage in tightly sealed containers reduces evaporation and protects anyone entering the storage room. In my own experience, failing to follow those standards often results in headaches – literally – for everyone in the workspace. Here, safety isn’t about bureaucracy; it’s about running a functioning lab without putting people at risk. Flammable, volatile chemicals need a great deal of respect, and protocols around spill cleanup and waste disposal have become non-negotiable in responsible settings.
Application Area
3-Hydroxybutanal has no trouble finding work across fields. In industry, it provides a route to crotonaldehyde and 1,3-butanediol, both important intermediates in the manufacture of rubber chemicals, resins, and even flavors. Pharmaceutical companies have long seen its potential as a building block, since its two functional groups open up reaction options that streamline synthesis steps. Outside large-scale operations, smaller specialty labs often reach for it as an intermediate for more bespoke compound preparation. Being able to anchor so many transformations in one starting material reflects its practical power. The food industry sometimes tests derivatives as flavoring agents after proper downstream modifications.
Research & Development
Despite being a classic, 3-Hydroxybutanal keeps showing up on cutting-edge research benches. Academic groups focus on new catalytic pathways to shape its reactivity, aiming to save energy or cut down on waste. Research into selective modification, new solvent systems, and greener synthesis methods often takes this molecule as a starting point. Much of the current excitement revolves around sustainable chemistry, and the compound’s simple structure makes it a strong candidate for developing new, less hazardous reaction conditions. Industry isn’t standing still either; companies chase ways to scale up production more efficiently while limiting byproducts. Progress on these fronts won’t grab mainstream headlines, but matters a great deal to those aiming to modernize chemical processes.
Toxicity Research
Research reveals that 3-Hydroxybutanal comes with toxicity that chemists take seriously. Skin contact and inhalation cause irritation, especially for workers repeatedly exposed to vapors without protection. Lab animals exposed to large doses develop respiratory symptoms and central nervous system effects. While it has not shown widespread environmental persistence, accidental release in concentrated form puts stress on waste treatment systems. For those working daily with this compound, strict exposure controls and regular air quality monitoring lower risks. Many of the toughest lessons about chemical safety came after ignoring warning signs from animal studies or early worker health reports. Pushing for newer, safer practices can prevent future problems – an approach that benefits both employees and the environment.
Future Prospects
As chemistry shifts toward greener processes, demand grows for intermediates like 3-Hydroxybutanal that offer both versatility and established safety protocols. Research groups keep chasing processes that cut out harsh reagents and improve atom economy, setting the stage for this compound to remain relevant in sustainable pathways. There’s room for advances in continuous flow processing and improved analytics, opening doors for real-time monitoring and reduced waste. More efficient uses in pharmaceutical synthesis sit on the horizon, promising shorter workflows and fewer chemical steps. While some compounds fade into irrelevance, 3-Hydroxybutanal hangs around, repeatedly earning its place as both a workhorse and a touchstone for new directions in chemical manufacturing.
What is 3-Hydroxybutanal used for?
What is 3-Hydroxybutanal Used For?
3-Hydroxybutanal, known by some as aldol, usually shows up in labs and factories as a key starting material. If you've worked in the chemical field or with organic synthesis, you’ve probably seen how it forms when acetaldehyde goes through an aldol reaction. That’s pure chemistry—simple, direct, and practical. But its uses stretch far beyond the glassware.
Industrial Importance
Look around the plastics world and you’ll see traces of 3-hydroxybutanal hiding in everyday products. The big draw? You can turn it into 1,3-butanediol through hydrogenation. 1,3-Butanediol stands out as a backbone for many plastics, polyurethanes, and even polyester resins. If you’ve ever dealt with coatings or adhesives, you know these components give flexibility, durability, and a smoother finish. That starts with the transformation from 3-hydroxybutanal.
Factories running large-scale chemical processes don’t choose their feedstocks lightly. They want reliable reactions with clear end-products and low waste. This chemical delivers. Every chemical engineer I’ve spoken with on the production floor knows how finicky scaling can get. A compound like 3-hydroxybutanal offers a straightforward path with well-understood reactions. That’s gold in manufacturing, where predictability often saves both time and money.
Role in Flavors and Fragrances
Not everyone realizes this compound also gives rise to crotonaldehyde. Picture the world of flavors and fragrances, where complexity and subtlety matter. Crotonaldehyde stands at a central spot, giving rise to the sweet, fruity, or nutty notes added to food products and perfumes. For those of us concerned about what’s inside packaged foods, every ingredient on the label matters. The raw materials feeding into those flavors deserve just as much scrutiny as the final product.
Pharmaceutical Synthesis
No serious chemist overlooks the versatility of 3-hydroxybutanal. In pharmaceutical development, it’s used to craft specialty molecules. Complex drugs often mix simple carbon chains in just the right way—3-hydroxybutanal provides a flexible carbon framework. Whether you’re developing sedatives, cardiovascular treatments, or vitamins, an accessible compound like this enables efficient routes to your target. Many research chemists rely on its straightforward chemistry to quickly build and test new drug candidates.
Health, Safety, and Environmental Considerations
Every large-scale user knows chemical safety can’t be ignored. 3-Hydroxybutanal is reactive and can irritate skin or eyes, demanding thoughtful handling. Early in my lab days, I saw a spill cause a daylong headache for a team that skipped the right protective gear. As a result, strict training and double-checks around storage and handling routines became our norm.
Environmental concerns have grown louder over the years. Waste management regulations continue tightening, pushing factories to capture or recycle byproducts like those from aldol reactions. Chemical makers who practice transparency and community dialogue set a strong example. Labs and manufacturers can do better by investing in closed-loop processing and improved storage, rather than cutting corners.
Future Directions
As industries look for greener, safer processes, suppliers are exploring new ways to make and use 3-hydroxybutanal with fewer harmful byproducts. Some researchers invest in bio-based methods, starting with fermentation rather than petroleum. This shift helps reduce carbon footprints and waste outputs—changes that benefit workers and neighborhoods. By swapping out old catalysts for newer, less toxic options, waste streams shrink and downstream hazards fade.
The practical legacy of 3-hydroxybutanal continues to grow because chemists and engineers push for more responsible and efficient uses. Keeping an eye on new research and regulatory shifts will guide safer, more adaptable uses of this key compound.
What is the chemical structure of 3-Hydroxybutanal?
Breaking Down the Name
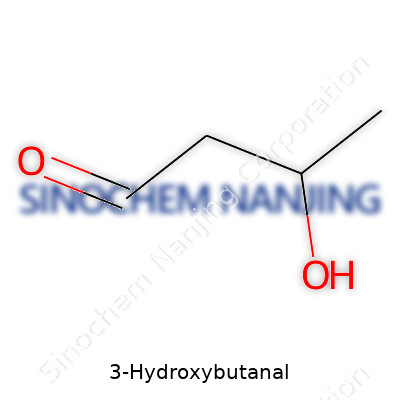
3-Hydroxybutanal looks like a mouthful, but its name gives clues about what this molecule actually is. Anyone who’s taken part in a chemistry class might see the pattern here—“hydroxy” says there’s an –OH group somewhere in the chain, and “butanal” means there’s a four-carbon backbone with an aldehyde group at the first carbon. So, putting that together, you get a molecule with a hydroxy group at carbon three and an aldehyde at carbon one.
The Structure Itself
Drawing this one out brings up a simple skeleton: CH2OH–CH2–CH(OH)–CHO. That’s four carbons in a row. The end has an aldehyde (–CHO). Starting from that side, counting over three carbons, there’s an –OH attached to carbon three. It feels like a basic leap in organic chemistry, but this compound shows up in interesting places, despite being pretty simple at its core.
Where You Might See 3-Hydroxybutanal at Work
Many folks might not recognize 3-Hydroxybutanal under its more technical name. It’s often called “aldol,” known to chemists for being the product of an “aldol addition” between two molecules of acetaldehyde. This reaction forms an important link in organic chemistry labs and industry, especially during the creation of more complex chemicals. The structure brings both a reactive aldehyde and a reactive alcohol together, making it act like a springboard toward building bigger molecules. This dual function can lead to everything from useful solvents to pharmaceutical intermediates.
Why Its Structure Matters
There’s real-world value in understanding 3-Hydroxybutanal’s layout. The way the molecule is put together gives it the skills to participate in a wide variety of reactions. The alcohol group on carbon three gives a spot for hydrogen bonding, influencing how this molecule mixes with water or other solvents. The aldehyde at the end signals both more reactivity and a tendency to participate in condensation and polymerization. I’ve seen firsthand how quick it can turn into larger compounds—which plays a role in both helpful industrial chemistry and accidental, messy lab bench problems if someone leaves a solution exposed for too long.
Environmental and Safety Considerations
Every time someone works with 3-Hydroxybutanal, either in a teaching lab or at an industrial scale, safety comes up. The aldehyde group brings some volatility, and the whole molecule can cause irritation on contact. From my own work, even small spills bring out a strong, pungent smell that’s hard to ignore. Using gloves and working in a fume hood keeps everybody safe. Proper storage and disposal stop unnecessary reactions—especially since this compound likes to turn into polymers or break down under the wrong conditions.
Moving Toward Better Practices
Keeping a handle on chemicals like 3-Hydroxybutanal calls for both knowledge and practical safeguards. Researchers develop safer derivatives or more stable formulations. Industry tracks air quality and manages waste streams to reduce risk to workers and the environment. For students and professionals, it boils down to respecting both the structure and the properties it brings—never just looking at the labels, but understanding the real stories these simple chemical drawings tell.
Is 3-Hydroxybutanal safe to handle?
Understanding What 3-Hydroxybutanal Is
3-Hydroxybutanal, often called acetaldol, comes up in organic chemistry and industrial labs as an intermediate. Plenty of folks working in research and chemical processing bump into it. The clear liquid doesn’t carry any color, but it doesn’t have a friendly scent either. It’s a stepping stone in making more complex chemicals and can sometimes show up under different names.
What Makes It Risky
Chemists learn early on: don’t judge a chemical by its appearance. 3-Hydroxybutanal can go through self-condensation if left on the shelf too long or exposed to certain conditions. That means it gets thicker or can even gum up equipment. You don’t want that in a reactor or piping system.
But the real concern isn’t just gunk. This stuff is known to cause severe irritation to the eyes and skin. If it splashes or spills, the burning sensation will make the point crystal clear. Inhaling its vapors brings headache and throat irritation. I’ve met colleagues who underestimated a “routine” compound, only to end the day with stinging eyes and a story that sticks.
Evaluating Data—What Science Says
There’s history here. Scientists measured the oral toxicity of 3-hydroxybutanal years ago. The LD50 (the dosage that causes death for half of a group of lab animals) hovers at moderate toxicity levels. In short, it won’t kill you after a drop or two on the arm, but it isn’t harmless.
Safety boards, like OSHA and the European Chemicals Agency, don’t give it a gentle pass. They warn about handling in places with proper ventilation, keeping it away from open flames since it doesn’t take much to catch on fire. Breathing masks and gloves come up every time in the documentation.
Lessons from the Lab
In my early career days, I saw a veteran treat every liquid as if it had teeth. That habit pays off, especially with reactive compounds like this one. I always recommend a lab coat that actually closes, safety glasses that don’t leave the eyebrows exposed, and gloves rated for organic solvents. Not every chemical demands the full gear, but 3-hydroxybutanal sure does.
If the smell of acetaldol starts drifting, the fume hood should be running. That one detail turns a headache episode into a non-event. Handling it directly without protection easily ruins a workday. Eventually, I learned to label bottles clearly and double-check storage—some folks keep these bottles too warm, which only speeds up trouble.
Simple Steps for Safer Work
Safety training sometimes feels like a bureaucratic hoop but rarely fails to spot trouble before it happens. Refresher training on 3-hydroxybutanal helps everyone to remember which gloves work best and how to store it. Even the small step of keeping material safety sheets within reach makes a difference during a spill.
For those who oversee a team, checking stockrooms and ensuring the chemical isn’t old or exposed to air makes a big impact. Once a container shows crystals or thick residue, it’s time for careful disposal—not cutting corners.
Addressing Hazards for the Future
Workplace safety depends on knowledge and daily habits. 3-Hydroxybutanal won’t do harm unless folks ignore the basics. Keeping labs well-ventilated, using the right protective gear, and teaching respect for even the “routine” substances holds the real key. Mistakes add up fast, so building a culture where every compound gets the attention it deserves keeps people safe day after day.
How should 3-Hydroxybutanal be stored?
3-Hydroxybutanal Storage Isn’t Just Chemistry—It’s Safety
Working with chemicals like 3-hydroxybutanal, also known as Acetaldol, turns the spotlight on storage more often than people realize. Left out on the lab bench, this colorless oily liquid can ruin your project and harm anyone nearby. It has a low flash point, reacts with oxygen, and doesn’t exactly give polite warnings before causing trouble. I’ve handled aldehydes over the years—one spill, one bad container, and the cleanup could take hours, not to mention the safety risks involved.
Temperature and Ventilation Count
Talk to anyone in a real-world lab, and you’ll hear stories about containers swelling or popping open. 3-Hydroxybutanal breaks down when it sits out at room temperature and doesn’t even need direct sunlight to start reacting. At my first lab job, we found a bottle starting to sweat after a weekend. That led to a scramble for the fume hood and some hard-earned lessons on climate control. Storing this compound in a dedicated refrigerator (not the lunch fridge!) set just above freezing offers good protection. That cooler temperature slows down decomposition and stops vapor formation.
Choose the Right Container—It Matters
Glass makes the best choice for storing this chemical. Polyethylene or polypropylene works in a pinch, but glass gives you a clear view if something forms on the bottom or if the material inside starts turning yellow. Make sure the caps fit tightly—every lab I’ve worked in keeps a stash of PTFE-lined caps for just this reason. Metal containers invite disaster, since 3-hydroxybutanal can corrode some metals and promote unwanted reactions. A leaky or contaminated bottle can turn a storage shelf into a hazard zone before anyone notices.
Labeling Protects More than Your Work
Unclear labels lead to the kind of mix-ups that get entire experiments tossed and people sent to the campus doctor. If you’re storing 3-hydroxybutanal, the label should spell out exactly what’s inside, flag potential hazards, and carry a clear date. It’s one of those simple habits that save time and prevent arguments. In my personal experience, even a highlighter can mean the difference between grabbing the right bottle and handling a surprise.
Fire Risk Calls for Good Judgment
Flash point below 70°C means that this chemical goes up in flames faster than you’d expect. Keep it away from heat sources, open flames, or sparking electrical equipment. Flammable storage cabinets with solid vents or explosion-proof refrigerators rank high on the smart list for any workplace dealing with hazardous organics. I’ve watched fire drills turn into real emergencies from poor storage, and that’s not the kind of excitement you want in your daily routine.
Spill Response Should Never Wait
If a container tips or leaks, act quickly. No one enjoys bench cleanup, but with 3-hydroxybutanal, every minute counts. Equip storage areas with absorbent pads and have a clear spill plan. Even experienced chemists hesitate when a pungent smell hits their nose, so fresh air and the right tools save more than just experiments. Safety goggles and gloves belong on your hands, not just your supply list.
Reaching Out Makes Storage Safer
No one manages chemical storage alone. Working with a safety officer or an experienced lab manager brings in perspective the textbooks miss. Newer chemists benefit from walkthroughs showing which storage locations dampen risk. Storing chemicals properly means less waste, fewer accidents, and workdays that end safely. It all comes down to respect—both for the chemical itself and for the people around you.
What are the main applications of 3-Hydroxybutanal in industry?
An Overlooked Chemical Workhorse
Most people outside the lab probably haven't heard much about 3-Hydroxybutanal, even though it quietly powers many processes behind the scenes. Also known as Acetaldol, this simple molecule carries an outsized role in both chemical manufacturing and pharmaceuticals. Factories often prize building-block chemicals like this for their flexibility — and with 3-Hydroxybutanal, versatility truly defines its story.
Chemical Synthesis: More than a Starting Point
Factories use 3-Hydroxybutanal as a stepping stone to more complex molecules. During my work with chemical supply chains, I saw how its unique structure makes it easy to tweak, break down, or expand. This has made it a top pick for making 1,3-butanediol and crotonaldehyde, both regular features in large-scale organic syntheses. Big chemical companies use it to streamline production and minimize excess steps, which helps keep costs low and yields high.
3-Hydroxybutanal’s role extends further as a precursor in the plastics industry. Once processed into 1,3-butanediol, it paves the way for certain polyurethanes and plasticizers. These compounds find their way into everyday products — from flexible pipes to coatings — proving that sometimes the raw materials matter just as much as the flashier finished goods.
Medicines and the Fine Balance of Synthesis
My conversations with pharmaceutical chemists reminded me how sensitive drug synthesis can be. Small impurities or inefficient starting chemicals often lead to expensive clean-up. 3-Hydroxybutanal, thanks to its predictable reactions, gives researchers more control in making active pharmaceutical ingredients. Its use as an intermediate lets teams fine-tune molecular structures without introducing unwanted byproducts.
For instance, it pops up in the early stages of making sedatives, antibiotics, antimicrobial agents, and sometimes even certain heart medications. While few patients would recognize it by name, the compound has made important drugs safer and more affordable in the long run. In a world where medicine prices get plenty of scrutiny, that practical impact means a lot.
Fragrances, Flavors, and Farm Fields
The world of flavors and fragrances also sees regular demand for 3-Hydroxybutanal. Its mild reactivity allows for gentle conversion into aroma-rich aldehydes and alcohols found in food flavorings or perfumes. I’ve seen finished perfumes and flavor manufacturers rely on such intermediates to mimic the natural scents and tastes of fruits or flowers — a detail many of us take for granted.
Farmers and agri-businesses use plenty of crop-protection chemicals each season. Some key herbicides and plant-growth regulators start their journeys with 3-Hydroxybutanal. Companies pick it for its reliability and straightforward conversion into molecules that control weeds or boost yields. These farm applications keep food more abundant and affordable.
Challenges and Sustainable Solutions
Every chemical, no matter how useful, brings its own hazards. 3-Hydroxybutanal can cause skin and eye irritation; safe handling stays a top priority for workers in both small labs and large plants. Industrial guidelines set by groups like OSHA and the International Chemical Safety Cards help, but there’s always room for improvement.
New research into greener chemistry aims to cut down on hazardous byproducts and waste during production. Bio-based feedstocks and energy-saving reactions could limit environmental harm. Moving forward, industry leaders seek suppliers who invest in such safer and cleaner production.
Even chemicals that rarely grab headlines often shape daily life and industrial trends. 3-Hydroxybutanal shows how targeted improvements in one link of a supply chain can ripple out — keeping costs reasonable, supporting innovation, and pushing us all toward cleaner solutions.
| Names | |
| Preferred IUPAC name | 3-Hydroxybutanal |
| Other names |
Aldol Acetaldol beta-Hydroxybutyraldehyde 3-Hydroxy-1-butanal |
| Pronunciation | /ˌθriː.haɪˌdrɒk.siˈbjuː.tə.næl/ |
| Identifiers | |
| CAS Number | 107-89-1 |
| Beilstein Reference | 1772086 |
| ChEBI | CHEBI:15601 |
| ChEMBL | CHEMBL14357 |
| ChemSpider | 33197 |
| DrugBank | DB04208 |
| ECHA InfoCard | ECHA InfoCard: 100.003.244 |
| EC Number | 1.1.1.30 |
| Gmelin Reference | 7798 |
| KEGG | C05998 |
| MeSH | D016229 |
| PubChem CID | 7907 |
| RTECS number | EL5950000 |
| UNII | C5176WHH9E |
| UN number | UN1148 |
| Properties | |
| Chemical formula | C4H8O2 |
| Molar mass | 88.11 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | fruity |
| Density | 0.953 g/cm³ |
| Solubility in water | soluble |
| log P | -0.47 |
| Vapor pressure | 0.342 mmHg (at 25 °C) |
| Acidity (pKa) | 13.16 |
| Basicity (pKb) | 3.58 |
| Magnetic susceptibility (χ) | -44.0e-6 cm³/mol |
| Refractive index (nD) | 1.4392 |
| Viscosity | 2.87 mPa·s (at 20 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -403.65 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -565.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319 |
| Precautionary statements | P210, P260, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 58°C |
| Autoignition temperature | 210°C |
| Explosive limits | 3.1–15.8% |
| Lethal dose or concentration | LD50 oral rat 2460 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 2460 mg/kg |
| NIOSH | HB7700000 |
| PEL (Permissible) | 50 ppm |
| REL (Recommended) | Hazard Statements: H302, H312, H315, H319, H332 |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
3-Hydroxybutyric acid Crotonaldehyde Butyraldehyde Acetoin |

