3-Hydroxy-2-Butanone: Far More Than Just Another Chemical
Historical Development
Decades ago, chemists stumbled onto 3-Hydroxy-2-Butanone, better known in certain circles as acetoin, during their work with fermentation products. Fermentation in food production shed light on this compound as a natural byproduct of bacterial metabolism, especially in traditional products like yogurt or butter. Research from the mid-1900s led to rapid growth in both biological and synthetic production routes. Its scent, reminiscent of freshly baked bread or butter, charmed food scientists and flavor designers. By tracing its roots through food, biotech, and chemical industries, people can watch the journey of a simple molecule moving from a mere laboratory curiosity to a mainstay in food chemistry and industrial biotechnology.
Product Overview
Ask anyone working in food flavoring or microbial engineering, 3-Hydroxy-2-Butanone hardly counts as exotic. It anchors the buttery aroma in many products. Its synthesis doesn’t strain the imagination: controlled fermentation using certain strains of bacteria churns this molecule out efficiently. In the early days, extraction from natural sources set the pace, but cost and scalability gently nudged the world toward fermentation and chemical synthesis. It’s the sort of compound that quietly slips into various niches—flavors, fragrances, and even cosmetics—without demanding much attention.
Physical & Chemical Properties
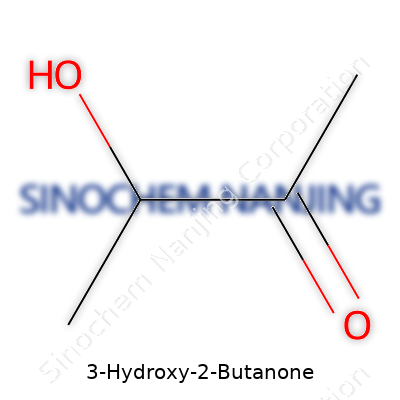
This molecule carries a straightforward formula: C4H8O2. Structurally, it balances a hydroxyl group and a ketone group, both of which handle widespread chemical reactivity. In the flask, 3-Hydroxy-2-Butanone presents itself as a colorless to pale yellow liquid with a pungent, buttery scent. It dissolves well in water, making it a favorite for food scientists and chemists alike. The boiling and melting points sit within a range that eases both storage and practical handling, and its volatility supports use in flavors and fragrances where rapid release into the air carries aroma effectively.
Technical Specifications & Labeling
Any batch bound for the food or pharmaceutical industries requires strict adherence to purity standards, given the potential for off-flavors or toxicity in the presence of contaminants. Regulatory bodies such as the FDA set clear benchmarks, such as minimum purity thresholds that typically reach above 99 percent. Labels must show chemical identity with the standard nomenclature and relevant safety instructions. For any company shipping and receiving it, recognizing the less familiar names—acetoin or dimethylhydroxyacetone—remains crucial. Some producers run additional analysis to detect trace metal contaminants or volatile organic residue, reflecting mounting quality pressures from both legislation and public expectation.
Preparation Method
My own real-world experience leans toward fermentation, both from work in academic and industrial settings. The process involves bacteria from the genera Bacillus or Klebsiella, which convert simple carbon sources such as glucose into 3-Hydroxy-2-Butanone with impressive yield. Optimizing the feedstock, tweaking the microbial strains through genetic engineering, and precisely controlling fermentation conditions (temperature, pH, oxygen flow) have all stacked the production deck in favor of high efficiency, cost savings, and sustainability. Chemical synthesis has not vanished—traditional chemical reduction routes starting from diacetyl still exist—just that biological production today responds better to the market’s green chemistry demands.
Chemical Reactions & Modifications
Outside its native form, 3-Hydroxy-2-Butanone jumps into a suite of chemical transformations. Thanks to its dual functional groups, it partners up with mild oxidizers or reducers, shifting toward diacetyl or 2,3-butanediol. In larger facilities, these reactions feed into broader manufacturing pipelines, generating everything from solvents to potential precursors in pharmaceutical syntheses. Researchers continue to work on more energy-efficient and selective methods, hunting for catalysts that cut down on waste and environmental impact.
Synonyms & Product Names
Plenty of people know this molecule as acetoin, and under International Union of Pure and Applied Chemistry (IUPAC) rules, it shows up as 3-Hydroxybutan-2-one. Some call it dimethylhydroxyacetone, pointing at its chemical makeup. Cross-referencing becomes a headache for both newcomers and experienced handlers—so remembering the most common terms eases confusion in busy labs or on industrial loading docks.
Safety & Operational Standards
Handling 3-Hydroxy-2-Butanone shouldn’t fuel anyone’s nightmares, provided safety basics get proper attention. Direct contact with concentrated material sometimes causes mild irritation, and inhaling high concentrations, especially as vapor during processing, triggers headache and nausea in sensitive people. The industry follows established rules—gloves, goggles, and good ventilation make up the holy trinity of lab and plant safety. Labels need to include hazard statements, usually with warnings about possible irritant properties. Environmental protection agencies keep tabs on discharge and atmospheric release, recognizing that even benign food flavors, when emitted in bulk, can disrupt local ecosystems or contribute (albeit minutely) to air quality issues.
Application Area
Chefs and food technologists depend on 3-Hydroxy-2-Butanone for its key role in dairy flavors, processed baked goods, and countless “buttery” products. Drop the name in a cosmetics lab and perfumers nod; they use it for masking off-odors and lending a creamy note to high-end fragrances and lotions. In biotechnology, this compound’s metabolic pathway serves as a benchmark for strain selection or GMO optimization, lending molecular biologists a reference point for precision fermentation. Environmental researchers use it as a signature of microbial activity in soils and composts, a subtle fingerprint of fermentative life beneath the surface.
Research & Development
Ongoing research probes both the metabolic pathways that produce 3-Hydroxy-2-Butanone and the broader industrial possibilities it opens up. Genetic engineering led to smarter microbial strains, able to pump out higher titers, tolerate harsher fermentation environments, or use less refined feedstocks. Green chemistry fans search for faster purification steps, lower-energy reaction routes, and circular economy models that reclaim waste. On the analytical side, chromatographers and spectroscopists advance ever more sensitive detection methods, those able to track down vanishingly small traces in environmental samples or complex blends.
Toxicity Research
Ask toxicologists, and the verdict on 3-Hydroxy-2-Butanone remains cautiously reassuring: used at standard food-additive or fragrance levels, it shows low acute toxicity. Long-term testing in animal models did not throw up red flags for carcinogenicity or chronic systemic effects. There’s a margin of safety, especially at the concentrations seen in processed foods or perfumes. That said, as with any volatile organic compound, inhaling large amounts makes nobody feel good, and in industrial settings, chronic exposure to vapors above recommended limits raises questions about respiratory irritation or subtle neurological symptoms. Regulatory agencies expect manufacturers and users to uphold workplace monitoring and protective practices, always keeping one eye on future toxicological data.
Future Prospects
The road ahead for 3-Hydroxy-2-Butanone stretches beyond food and flavor. Bioplastics research circles it as a pivotal building block, with metabolic pathways redesigned to generate downstream monomers or more complex chemical intermediates. Synthetic biology, particularly CRISPR editing and high-throughput screening, pushes the boundaries of what engineered microbes can achieve in terms of yield, cost, and feedstock flexibility. Startups pursue "carbon negative" fermentation cooled by renewable energy, aiming to sidestep fossil input altogether. Regulatory developments could push for even stricter safety and labeling standards, forcing suppliers to continually innovate. As global diets shift, natural and "clean-label" flavor demands will keep this unassuming chemical in the spotlight. Continued research, tighter safety regulations, and ongoing innovation in production methods hold the keys to making sure the world keeps benefiting from 3-Hydroxy-2-Butanone, without falling into complacency.
What is 3-Hydroxy-2-Butanone used for?
More Than a Sciency Name
3-Hydroxy-2-butanone goes by a less intimidating name in everyday life: acetoin. It’s got that warm, buttery smell baked goods and dairy products give off fresh from the oven or straight from the churn. I’ve found its sweetness hard to miss, even as someone who doesn’t spend all day in a lab. Walk into a bakery, breathe deep, and thank acetoin for that cozy hit. Many food producers lean on it for authentic flavor and aroma—think of that “buttery” popcorn at the movies or the rush of scent inside a pastry shop.
Inside Processed Foods and Flavors
People often ask why foods taste so much richer or smoother than homemade versions. The answer, more often than not, includes compounds like acetoin. The food industry uses it in sugar confections, ice creams, yogurts, baked goods, and even drinks. Labels may call it “natural flavoring.” Research from the European Food Safety Authority classifies it among compounds safe for human consumption, so long as it stays within regulated amounts. And safety matters: I pay extra attention when feeding my own family, and transparency helps me trust what’s on the shelf.
Secret Ingredient in Fragrances
Perfumers and cosmetics makers value 3-hydroxy-2-butanone, not just for a buttery aroma but how it blends with floral and fruity notes. It brings out a warm, mellow background, helping scents linger on skin or fabric. Industrial flavors and cosmetic chemists report its wide use for perfumes, body sprays, and hair products meant to run closer to comforting, familiar scents. With more folks leaning into “natural-smelling” personal care, acetoin’s role just keeps growing.
Everyday Chemistry: Beyond Tasting and Smelling
Walk beyond the kitchen or perfume aisle, and you’ll find this molecule in serious places, too. Chemical manufacturers add acetoin to create other compounds—or they tap it for its usefulness as a solvent. It’s far from the most complex molecule in the toolbox, but its solubility helps dissolve or modify sticky, oily, or waxy bases. Some cleaning products, plastics, and adhesives start with small amounts of acetoin.
Pharmaceutical Touchpoints
A few years back, scientists highlighted acetoin’s role in the body’s own bacterial dance. Certain probiotics actually create this compound. Medical researchers are looking at these connections in new health products, pushing for more natural fermentations. Smarter use of acetoin could help improve medicines, supplements, and functional foods, especially those built on “gut health.”
What Matters to the Consumer
Most shoppers never see “3-hydroxy-2-butanone” on a package label. I’ve learned that tracing the source, and knowing which regulations shape ingredients, seems overwhelming—but the payoff is real. Food-grade acetoin passes through tough quality controls and risk assessments in the US, EU, and other markets. Transparency, sourcing, and clear communication let parents, cooks, and small business owners enjoy better flavors while minimizing uncertainty.
Safe Use and Potential Pitfalls
No ingredient is totally without risk. Food chemists, regulators, and toxicologists agree on limits for how much acetoin food and cosmetic makers should use. Excess can cause irritation or unexpected allergic reactions, though real cases are rare with regulated use. As science moves, researchers keep a close watch, updating the guidelines if new evidence appears. Whenever I see updated ingredient rules, I read them close, knowing that constant review keeps the marketplace safer for everyone.
Building Better Foods, Smells, and Products
What stands out about 3-hydroxy-2-butanone isn’t just the chemistry. It’s about bringing moments of comfort—a meal, a scent, a healthy dose—without sacrificing trust or wellbeing. Responsible industries, independent scientists, and everyday users shape how its story unfolds. Keeping questions alive and information upfront invites safer innovation, which helps make food tastier and daily products more enjoyable.
Is 3-Hydroxy-2-Butanone safe for human consumption?
Getting Familiar with 3-Hydroxy-2-Butanone
For folks who pay close attention to food labels or work in kitchens, 3-Hydroxy-2-Butanone might ring a bell as acetoin. This compound brings a creamy, buttery aroma and taste, showing up in everything from baked goods to yogurts. The food industry grabs it straight from fermentation tanks, where certain bacteria work their magic, or whips it up in labs. Many call it natural if a microbe makes it, although that word has lost much of its meaning after reading enough packaging.
Safety Research and Real-World Exposure
Powerful organizations like the US Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) have already spent years picking apart its safety. These oversight teams looked at animal and cell research, as well as how much people actually eat daily. The FDA folded acetoin into its category of GRAS—meaning "generally recognized as safe." The Joint FAO/WHO Expert Committee on Food Additives has reached similar decisions after reviewing data going back decades.
Setting regulations isn’t an exact science. Companies present data, experts comb through it, and over time, opinions harden into rules, sometimes shifting years later if a red flag appears. For acetoin, the overwhelming pattern tells the same story: no sign of harm at the levels you’ll ever find in food, even desserts loaded with it for flavor. That reassurance comes from years of culinary habits and detailed lab work, not marketing.
Reason for Caution: Too Much of Anything
Even safe-sounding ingredients come with a catch. Chugging a bottle of flavoring—acetoin included—could overwhelm the body, same as downing a full salt shaker or eating a family-sized bag of gummy bears. There’s a number for everything. Doses used in baked goods hover at a fraction of what ever showed harm in animal trials. Still, food makers must stick to recommended limits. Digestive upset and irritation show up long before anything worrisome, according to existing studies.
From a cook’s point of view, using products where flavor comes from fermentation instead of factories offers peace of mind. Regular folks do not walk out of supermarkets with bottles labeled “3-Hydroxy-2-Butanone” anyway.
The Need for Ongoing Oversight
No food additive gets a free pass forever. Science doesn’t stand still, and my experience tells me that nutritional debates almost never close for good. With acetoin, watchdog groups continue poking and prodding. A handful of studies on e-cigarettes—totally different from food—have raised more questions, since inhalation might not tell us much about eating. Still, it pays to keep an open mind and welcome more research, especially with products that land on dinner plates worldwide.
Clear Labeling and What Consumers Can Do
Trust grows when food packages tell the truth. Acetoin sometimes hides behind “natural flavor” on labels, which leaves shoppers scratching their heads. Simple ingredient lists help regular folks make confident choices. For those worried about chemical-sounding names, seeking out brands that use traditional fermentation rather than chemical synthesis offers a path forward. I’ve met plenty who read every word on a box and avoid anything they can’t pronounce, and that works for them.
Moving Toward Balanced Choices
In kitchens and at tables around the world, flavor rules. Thanks to oversight and longstanding use, acetoin hasn’t shown up as a troublemaker in food. No one ingredient tips the scales—the full diet picture matters far more. Keeping eyes open for updates, asking questions, and reading labels closely remains the best advice. Safety isn’t set in stone, but experience and evidence leave room for confidence today.
What are the storage conditions for 3-Hydroxy-2-Butanone?
Understanding the Risks and Responsibilities
3-Hydroxy-2-butanone, better known as acetoin, often pops up in different industries: food flavoring, fragrance, and as an intermediate in various chemical processes. As someone who’s worked around chemical storerooms and small labs, I can say firsthand that storing this compound safely matters far more in practice than it does on any one safety data sheet. Too much confidence in shelf-stable containers has led to ruined batches and sometimes ruined equipment—no one wants to learn that lesson the hard way.
Temperature is Everything
Acetoin can handle room temperature, but that doesn’t mean tossing it in any old cabinet will do. Direct sunlight or fluctuating heat can speed up breakdown or even stir up hazardous vapors. I’ve seen storerooms where the air gets stuffy in summer and that air can cause bottles to sweat, labels fade, or even pressure to build up inside sealed containers.
Cool rooms—generally between 15–25°C—offer a far more stable environment. Keeping things constant matters, especially in regions where seasons swing hard. For operations with high purity requirements, refrigeration brings extra peace of mind, though you don’t want to place it near food stored for human consumption. It’s not about paranoia. It’s about cutting down the odds of mishaps and surprise contamination.
No Room for Moisture or Air
Acetoin is hygroscopic to an extent, meaning it attracts moisture from the air. That little fact sneaks up on those new to chemical storage. I’ve watched coworkers mistakenly crack open a container in a humid storeroom only to return to a bottle of cloudy, compromised material a few weeks later. Moisture dulls the compound, sometimes making it useless for sensitive work.
Tight, air-resistant sealing stops this trouble before it starts. Glass or high-grade plastic containers fit with solid screw caps (not corked, not loosely pressed-on) keep out most of the unwanted vapor. In bulk settings, metal drums lined with inert materials give another layer of protection. Containers shouldn’t be filled all the way to the top—in case of pressure buildup—but it’s not the place for lazy lids or cut corners.
Safe Storage Means Safer Work
Leaving acetoin near sources of ignition or acids and oxidizers invites unnecessary hazards. In labs and shops I’ve worked in, good habits—separating organic and inorganic chemicals, logging bottle movements, using marked, purpose-built shelving—pay off again and again, especially during annual inspections.
Even in small quantities, a bottle stored near bleach or other reactive substances can trigger dangerous reactions. A simple rule: keep incompatibles several shelves apart or, better yet, in different storage cabinets. It takes maybe a minute longer but beats scrambling to contain a spill or fighting off headaches from unexpected fumes.
Label Everything and Keep it Updated
A faded label or a reused bottle invites disaster down the road. I’ve seen confusion grow in cramped spaces. Permanent markers, dated tracking sheets, and consistent naming systems make it easier for everyone, especially night shift workers or new trainees stepping in for the first time. It doesn’t take fancy inventory management—just solid, repeatable habits.
Strict adherence to guidelines from OSHA and contemporary safety standards isn’t bureaucratic red tape—it stopped a lot of avoidable accidents where I’ve worked. Training doesn’t end after orientation, either. Refresher drills help keep old-timers and new staff sharp, especially on what to do with expired or damaged supplies.
Better Storage, Fewer Surprises
Taking a stronger approach to acetoin storage always pays off in the long run—less waste, fewer accidents, and smoother operations. Investment in well-ventilated spaces, properly lined containers, and constant monitoring builds a work environment that doesn’t turn risky every time something gets busy. Years of trial, error, and improvement in both small labs and larger supply rooms prove that the little details of storage shape the results on every level.
What are the physical properties of 3-Hydroxy-2-Butanone?
Getting a Grip on the Basics
Everyday chemistry doesn’t always feel impressive until you meet something like 3-hydroxy-2-butanone. Folks in flavor science and production call it acetoin, and some people have spread it all over kitchen counters—more than they realize—since this stuff pops up in butter, yogurt, and even fresh strawberries. But beyond taste and aroma, understanding its physical properties means you can handle it properly, spot it in the lab, and keep your workplace safe. That’s as much about health as it is about precision.
Liquid that Looks Like Water, Smells Like Cream
I remember the first time I prepared a batch for a food science project: it looked almost like plain water. 3-Hydroxy-2-butanone pours as a clear, almost colorless liquid. Watch it glint in the light, and if you put your nose to it, the sweet, creamy scent is unmistakable. That buttery, slightly caramel hint hangs on clothing, clings to stir rods, and can turn a dull afternoon in the lab into dessert-hour. Still, this is no plaything.
Temperature and Handling
This liquid boils at about 148 degrees Celsius. You can actually see it bubble for a good while on a gentle hotplate, reminding you it won’t rush into the air like something with a lower boiling point. Now, for freezing points, it solidifies near -20 degrees Celsius, which means most folks won’t find it freezing during regular storage or handling. Its melting point actually makes it handy for storing at room temperature, unlike some volatile solvents. You won’t have to lock it in a freezer just to keep it liquid.
How It Mixes and Moves
One memory sticks: a single drop dissolves right away in water. Acetoin’s solubility is high in water and alcohol, so whether you’re trying to mix it with a culinary extract or work it into a chemistry demonstration, you see clean solutions without lumps or streaks. In a classroom, this rapid mixing lets you skip long stirring sessions.Density hovers near 1.02 grams per cubic centimeter at room temperature. Pour some into a measuring tube, and you’ll find it weighs almost the same as water. For a chemist, that means it won’t separate out if you shake it into an aqueous mixture. This feels simple, but it saves hassle and helps in recipes and research projects where precision counts.
Potential Hazards
Given its sweet scent and food applications, folks sometimes underestimate acetoin. Still, safety sheets warn about eye and skin irritation. Gloves and goggles should stay close at hand, especially during a spill. In the past, I’ve seen over-enthusiastic students forget about labeling bottles—mixing it up with harmless water. This isn’t a disaster, but proper tagging and awareness go a long way.
Why These Details Matter
Researchers, manufacturers, and flavor technologists all benefit from knowing these properties. Fact: 3-hydroxy-2-butanone’s high solubility makes it a go-to for blending flavors and stabilizing aromas in processed foods. Understanding its boiling point means safer distillation and less worry over flammable vapors. If someone wants to work with this compound, they need to have clear guidelines for storage, handling, and mixing—practices that keep workplaces safe and food grade applications consistent. That’s real-world chemistry at work, driven by both respect for the material and the facts of its behavior.
How is 3-Hydroxy-2-Butanone synthesized or produced?
What Goes Into Making 3-Hydroxy-2-Butanone?
Most folks know 3-Hydroxy-2-Butanone as acetoin. This little molecule smells a lot like butter and pops up in fermented foods, baked treats, and even some tobacco products. The thing is, getting pure and high-quality acetoin isn’t as simple as walking into a lab and mixing a couple of random chemicals together.
In the lab or factory, one common way to produce acetoin starts with fermentation. I’ve spent enough time around breweries to know that nature can turn simple sugar into all sorts of interesting compounds, and this is another example. Certain bacteria—mainly strains like Bacillus subtilis and Klebsiella pneumoniae—will eagerly chew through glucose, leaving acetoin as one of the byproducts. Over the years, scientists have fine-tuned the fermentation process to boost yields. Limiting the air supply, controlling the temperature, and keeping the pH steady help the bacteria focus on making acetoin rather than waste.
People often ask, “Why not just stick to old-school chemical synthesis?” Well, that route’s still on the map. The diacetyl reduction pathway remains one of the chemical approaches. In simple terms, chemists take diacetyl—which itself can be made by oxidizing butanone—and use a reducing agent. Hydrogen gas with a metal catalyst or even sodium borohydride can convert diacetyl to acetoin. This approach delivers a fairly precise product, and the technology behind it doesn’t change much year to year. The catch? You need to handle the chemicals carefully, and the process produces some waste that takes thoughtful management to avoid environmental problems.
Why Does the Production Method Matter?
Some folks in food and flavor industries want “natural” labels, and a fermentation route using bacteria checks that box. It also helps reduce the reliance on petrochemicals. On the other hand, pharma and fine-chemical sectors might look for absolute purity, so a chemical approach becomes pretty attractive. Safety isn’t just about product quality—there’s worker safety, waste stream management, and the push to meet stricter regulations about emissions.
Fermentation, when managed well, puts less strain on resources and might even use leftovers from other food processing. That’s something I think more companies should pay attention to. As a consumer, it feels right knowing that the buttery scent in your yogurt or baked bread came from a process that’s cleaner and a bit more sustainable.
Room for Improvement
No production line runs perfectly. Fermentation tanks are expensive, and keeping microbes happy means round-the-clock monitoring. Chemical reduction offers fine control but relies on reagents that aren’t always easy to source, and disposal drives up costs. Some research labs have experimented by tweaking bacterial genes, pushing yields higher and sidestepping the slowdown that sometimes happens from byproduct buildup. Companies investing here aim to strike a balance—more efficient, less expensive, and safer for both workers and the planet.
I’ve seen firsthand how pressure for cleaner chemistry pushes factories to rethink old habits. Whether it’s swapping out a harsh solvent or capturing byproducts for reuse, these shifts often begin on a small scale and spread as the business case becomes clear. For acetoin, the next wave will likely lean even more on biology. There’s a growing recognition that what’s better for the earth often turns out to be better for the bottom line, too.
| Names | |
| Preferred IUPAC name | 3-Hydroxybutan-2-one |
| Other names |
Acetoin Acetyl methyl carbinol Dimethyl glycol ketone Methylacetylcarbinol 3-Hydroxybutan-2-one Ketoin |
| Pronunciation | /θriː-haɪˈdrɒksi tuː bjuːˈteɪnoʊn/ |
| Identifiers | |
| CAS Number | 513-86-0 |
| Beilstein Reference | 1209240 |
| ChEBI | CHEBI:16642 |
| ChEMBL | CHEMBL14258 |
| ChemSpider | 54606 |
| DrugBank | DB04112 |
| ECHA InfoCard | 100.007.900 |
| EC Number | 1.1.1.4 |
| Gmelin Reference | 8828 |
| KEGG | C00410 |
| MeSH | D017220 |
| PubChem CID | 651 |
| RTECS number | EL8975000 |
| UNII | ES5B6IMG9O |
| UN number | UN1233 |
| CompTox Dashboard (EPA) | DTXSID4021620 |
| Properties | |
| Chemical formula | C4H8O2 |
| Molar mass | 88.11 g/mol |
| Appearance | colorless to pale yellow liquid |
| Odor | Butter odor |
| Density | 0.989 g/cm³ |
| Solubility in water | soluble |
| log P | -0.41 |
| Vapor pressure | 0.4 mmHg (20 °C) |
| Acidity (pKa) | 12.8 |
| Basicity (pKb) | pKb = 8.2 |
| Magnetic susceptibility (χ) | -8.9×10^-6 |
| Refractive index (nD) | nD 1.438 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 264.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –370.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1246.7 kJ/mol |
| Pharmacology | |
| ATC code | A16AX10 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 58 °C (closed cup) |
| Autoignition temperature | 385°C |
| Explosive limits | 5.7–17% |
| Lethal dose or concentration | LD50 oral rat 2463 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2.7 g/kg (rat, oral) |
| NIOSH | HN9625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Diacetyl 2,3-Butanediol |

