3-Hexanone: A Closer Look at a Versatile Chemical
Old Roots with Modern Value: Tracing 3-Hexanone’s Story
People often overlook the stories behind the bottles on lab shelves. 3-Hexanone, though less famous than other carbonyl compounds, plays a bigger role than many realize. Chemists first encountered it in the early 20th century while diving into the synthesis of various ketones, poking around with hydrocarbon chains to understand how structure shapes function. Back then, the big draw was finding chemicals that reacted predictably for making plastics, medicinal agents, and solvents. The emergence of 3-Hexanone in laboratory reports marked a turning point for certain industries, especially as researchers tinkered with chain length and branching to tweak properties. Its place in organic chemistry books tells the story of a building block rather than a headline star, yet that same modesty is what keeps it in demand.
What’s Inside the Bottle: A Practical Overview
So many products in research labs carry mouthful names and cryptic codes, but 3-Hexanone comes down to a clean, unbranched chain of six carbons sporting a ketone group at the third carbon. This sets the stage for reactivity and an approachable molecular weight, usually weighing in at 100.16 g/mol. The liquid’s mild, somewhat fruity odor often escapes in a lab, hinting at low molecular complexity. It shows up as a colorless, mobile liquid at room temperature—easy to mix with most common organic solvents but less at home in water. That little dash of nonpolarity explains a lot about why people prefer it as a solvent and reaction intermediate. Having handled it several times in the lab, I can say it is far less aggressive than acetone but still packs enough punch to dissolve compounds stubborn to milder solvents.
Physical and Chemical Character: What Sets 3-Hexanone Apart
Walking past the rainbow of bottles lining the lab, each with its label and quirks, I learned that small tweaks can mean big changes. With a boiling point that lands close to 127°C and a melting point well below freezing, 3-Hexanone stays liquid through a broad temperature range—handy during purification or while running reactions that need a non-volatile medium. Its refractive index holds steady, making it useful in certain optical measurements, and its density falls in line with other straightforward ketones. The molecule’s structure means it won’t engage in hydrogen bonding like alcohols or amines, so its vapor escapes more freely—which means fume hoods and good air flow in the workspace are your friends. Chemically, the carbonyl group on the third carbon is reactive, capable of playing into all the classic organic transformations—think reductions, condensations, or nucleophilic additions—in ways that many synthetic routes rely on.
Labeling and Technical Specifics: What to Watch in the Details
In my time around chemical storerooms, I noticed that clarity in labeling sidesteps both safety issues and research mix-ups. 3-Hexanone labels typically confirm purity, often above 98%, advertise and warn about flammability, and detail the nature of its hazards: flammable liquid, skin and eye irritant, avoid inhalation, and something about working in well-ventilated spaces. Its identification on shipping manifests falls under commonly recognized chemical designations, though its synonymous names—ethyl propyl ketone, hexan-3-one—appear often in academic and industry documentation. That subtlety hasn’t tripped me up yet, but it has caused confusion in less organized labs, which shows the need for clear, consistent naming and a good grip on the various ways chemists refer to even common compounds.
Getting There: How 3-Hexanone is Prepared
There’s some craft and a little luck in making 3-Hexanone efficiently. Most commercial sources rely on oxidation or dehydrogenation of parent alcohols like 3-hexanol, with catalysts such as copper or silver to help the reaction along. Older textbooks mention acetoacetic ester synthesis followed by hydrolysis and decarboxylation, while more modern plants stick to controlled oxidations—fewer byproducts, fewer headaches. Getting the right conditions—temperature, pressure, catalyst presence—makes the process a dance between speed, yield, and purity. Having run smaller-scale setups myself, I can vouch for the simplicity of the reaction in spirit, but keeping water and peroxides out becomes a never-ending cleaning ritual.
Chemical Tinkering: Reactions and Modifications
3-Hexanone opens doors to a palette of reactions. Its core ketone group welcomes nucleophiles, giving rise to secondary alcohols on reduction or more complex adducts in classic Grignard reactions. Aldol condensations work like a charm—given the right partners—and its carbon skeleton supports rearrangements that build up to more complex molecules. Chemists in the flavor and fragrance field play off these transformations, turning basic ketone functionality into layers of alluring scents. Some research teams chase after even more tailored modifications, like selective halogenation or forming oximes for further utility. The playbook here stretches from textbook basics to the creative edge of synthesis, with 3-Hexanone acting as both launching point and quiet workhorse.
A Web of Names: Synonyms in the Literature
Names matter in both daily lab work and when digging for literature. 3-Hexanone often answers to hexan-3-one, ethyl n-propyl ketone, or even 3-oxohexane. Over the years leafing through references, I’ve stumbled on these alternatives, and recognizing them speeds up data searches and avoids mix-ups (and awkward calls to the chemical supplier). This mix of nomenclature reflects differences in conventions between the IUPAC system and older common names, especially in catalog listings. For researchers or students, knowing all its aliases sidesteps wasted hours, especially in cross-disciplinary work between organic synthesis, environmental science, and pharmaceuticals.
Balancing Safety and Practicality: Standards in the Field
Running reactions and cleaning up spills taught me lessons about the gap between reading a safety sheet and real-world practice. 3-Hexanone falls under hazardous chemicals, thanks to flammability and potential skin irritation. Labs that follow good ventilation, consistent glove use, and reliable chemical storage keep accidents rare. Most regulatory agencies set clear guidelines for use—keep away from heat, avoid spark sources, eliminate unnecessary contact, and keep containers tightly closed. Ongoing training, clear SOPs, and easy access to clean-up stations make the work not only safer but smoother all around. In industrial settings, the stakes multiply because of larger volumes and open transfer operations. Teams that build safety into their daily rhythm protect both people and the projects they work on, especially with chemicals like this that blend utility with risk.
Stepping Outside the Lab: Real-World Applications
The uses for 3-Hexanone span well beyond lab benches. Industrial chemists often reach for it as a solvent in producing adhesives or resin formulations where evaporation rates and residue profiles matter. Researchers in the flavor and fragrance industries use it as a base for synthetic aromas, drawing on the subtle notes it can impart or amplify. Its role as an intermediate shows up in pharmaceutical manufacturing as well, where it gets built into more complex molecules or included as a reaction solvent. Agricultural science uses it to synthesize certain herbicides and insect repellents, where the predictability of its chemistry delivers reliable results. My own experience tells me that while it doesn’t get the spotlight, each new use-case keeps growing, as researchers uncover small ways to boost efficiency or develop alternatives to legacy solvents.
Expanding Horizons: Ongoing Research and Development
Every few years I see new studies aiming to improve synthetic routes or identify greener catalysts for making 3-Hexanone. Sustainability trends push industry and academia to search for renewable feedstocks or more energy-efficient reactions; even incremental improvements matter at large scale. There’s ongoing curiosity about how 3-Hexanone derivatives can serve as platform molecules for drugs, fragrances, or plastics. Cross-disciplinary projects, including computational studies or more sensitive analytical techniques, dig deeper into its reactivity and potential. Green chemistry pushes for process intensification, perhaps using bio-catalysts or flow chemistry systems to cut waste and improve yields. It’s this continuous tweaking and process reflection I relate to—often small, persistent changes drive the most reliable progress.
Looking Closely at Toxicity: Health Questions and Evidence
Handling 3-Hexanone, even in small quantities, sharpens your awareness of toxicity issues. Short-term exposure can cause headaches, dizziness, and irritation, making good ventilation and personal protective gear standard practice. Chronic studies on animals highlight potential neurotoxic effects at high doses, drawing parallels to related ketones like 2-hexanone, which carries more established risks. Occupational exposure limits help keep workers safe, but knowledge gaps remain regarding long-term low-dose exposure and interactions with other volatile organics. Regular monitoring, both of air concentrations and health outcomes, gives teams a way to catch trouble before it escalates. Tighter regulation and broader epidemiological studies could illuminate the subtler risks—until then, I always err on the side of caution, especially after seeing what lax safety has caused in older labs.
Where Things May Be Headed: Future Prospects for 3-Hexanone
The road ahead for 3-Hexanone ties into shifting priorities in both industry and research. As the world moves away from hazardous solvents and toward safer, greener processes, chemists keep searching for ways to refine its synthesis or even swap it out where better alternatives exist. Its core properties still make it attractive for specific reactions or product lines, and with research pushing toward bio-based feedstocks, the basic framework of 3-Hexanone could show up in more unexpected areas—from biodegradable plastics to smart drug delivery systems. Regulatory scrutiny will likely shape both availability and approved uses, especially as evidence around workplace exposure sharpens. From where I stand, innovation in both synthesis and applications keeps basic chemicals like 3-Hexanone relevant, resilient, and ready for whatever challenges the future cooks up.
What is 3-Hexanone used for?
Everyday Chemistry, Real Application
Step into a regular lab or flavor plant, and you might spot a clear liquid called 3-Hexanone. It isn’t a household name like salt or vinegar, but this chemical works behind the scenes in more ways than most folks realize. Think of the subtle notes in some foods, that underlying fruitiness in fragrances, or a specialty solvent that helps shape products before they hit the shelves.
The Flavor and Fragrance Connection
Pop open a fruit-flavored drink or toss a stick of gum in your mouth, and you’re tasting more than just basic sugar and citric acid. Small amounts of chemicals like 3-Hexanone fill out fruit and green aromas, making synthetic strawberry or melon flavorings taste closer to the real deal. Industry experts often turn to this molecule for that elusive balance—too much, and it’s off-putting; in the right proportion, it pushes products closer to nature.
Personal experience working with flavor houses showed me how picky they are about every ingredient. One wrong note can ruin an entire batch. Chemists measure 3-Hexanone out with respect, keeping in mind regulations set by food safety authorities. The U.S. Food and Drug Administration, among others, sets strict limits on how much can go into food, keeping public health at the center.
Role in Industrial Labs
Industrial applications tend to fly under the radar, but they matter. Companies blend 3-Hexanone as a solvent—it breaks down or dissolves other chemicals, helping everything from plastics to coatings come together in manufacturing. In research, it pops up in the synthesis of specialty compounds or custom treatments, and I’ve seen it used for separating mixtures or cleaning sensitive equipment. Its molecular structure makes it useful for working with polar and nonpolar compounds, sort of like a Swiss army knife in chemical research.
One of the big challenges with any solvent concerns safety. 3-Hexanone can cause irritation with direct contact or inhalation, so professionals use gloves, goggles, and fume hoods. Continuous exposure poses neurological risks, so organizations demand proper training and regular health checks. Regulatory authorities like OSHA keep a close eye on workplace standards, aiming to lower the chance of chronic health issues.
Safety, Impact, and the Road Ahead
The chemical supply world doesn’t work in isolation. With growing focus on sustainability, teams in chemical plants seek alternatives that have a lighter impact on health and the environment. Handling 3-Hexanone with care, using it only where necessary, and recycling solvents through distillation can help minimize risk. Innovators keep searching for substitutes that promise similar function with fewer safety trade-offs, but few have checked every box so far.
3-Hexanone may not get talked about much, yet the push for ethical sourcing and better transparency shines a light on these building blocks of modern industry. Transparency from suppliers, audits by buyers, and cooperation with regulators paint a picture of a field moving toward safer days. Each time a new regulation arrives or a greener alternative gets put to the test, everyone—from chemists to consumers—has another chance to raise the bar.
What is the chemical formula of 3-Hexanone?
Getting to Know 3-Hexanone
3-Hexanone has the chemical formula C6H12O. It belongs to a family of organic compounds called ketones, which often turn up in labs, manufacturing, and everyday settings. The structure holds six carbons in a row with a double-bonded oxygen sitting on the third carbon. Knowing this formula isn’t just about trivia—it means people can predict properties, safety rules, and even smells that come with the substance. Anyone who has set foot in a chemistry classroom has probably smelled how sharp and slightly sweet a simple ketone can get. These characteristics make even the most basic molecules part of real-world conversations, beyond the textbook.
Why Chemists Care About Simple Compounds
Many researchers look for shortcuts to understanding how building blocks like 3-Hexanone fit into bigger processes. I remember in college chemistry labs, using formulas like C6H12O gave us the information to mix, react, and measure. It helped make sure nothing dangerous could go wrong once reactions started heating up. The right formula means safety for students and workers. There's no room for guessing with flammable solvents, especially in the presence of open flames or sensitive equipment.
More Than an Academic Question
3-Hexanone doesn’t just stay behind lab doors. Its unique scent and solvency bring it into flavoring, fragrance, and even cleaning product manufacturing. People in these industries need to know the formula to predict interactions and stick to regulations. Food scientists and perfumers rely on molecular formulas like C6H12O to avoid unexpected byproducts that could affect quality or safety. In my own experience, even the simplest mislabel in the supply chain can lead to ruined batches or expensive recalls. So, understanding the actual chemistry behind even a small compound holds real financial and safety importance.
Addressing Challenges—Handling and Regulation
One issue raised by the widespread use of chemicals like 3-Hexanone is exposure risk. The U.S. Occupational Safety and Health Administration (OSHA) and the European Chemicals Agency (ECHA) post strict guidelines for working safely. 3-Hexanone can irritate skin and eyes, and inhalation isn’t pleasant. Fact-checking every bottle and practicing solid storage habits prevent accidents and health issues—something that anyone storing solvents at home or work can relate to. Some countries even list workplace exposure limits based on proven research, not just assumptions or broad recommendations. Over the years, stories of improper handling have made the news. They serve as reminders that chemistry impacts people directly, not just on paper but in busy labs and crowded factories.
Smart Solutions—Education and Error Reduction
Simple, thorough labeling systems and frequent hands-on safety drills solve more hazards than another pamphlet ever could. Early in my research days, drill practice often seemed like overkill, but watching new students fumble with similar-looking bottles changed my mind. Training brings fewer spills and improved confidence. Companies now invest in digital inventory systems and improved labeling—small efforts that save lives and dollars. For teachers and students, real-life case studies hit harder than abstract rules, so schools should show videos and invite guest speakers from industry. Sharing stories keeps memory fresh and tradition solid.
Final Thought—Chemistry on the Street
A formula seems simple until real-world pressure tests it. 3-Hexanone’s story proves a routine line of text, C6H12O, ties into bigger issues about safety, business, and the power of knowledge. Even the smallest molecules shape daily life, inside and outside the lab.
Is 3-Hexanone hazardous or toxic?
Everyday Chemicals: A Closer Look at Safety
Most people have never heard of 3-hexanone. Yet, it pops up in places you wouldn’t expect, from food flavors to labs in industry. Chemicals like this rarely get much attention until someone starts asking, "Is it safe?" That simple question sets up a whole web of responsibility. Folks want to trust the air they breathe at work and the products used in homes and businesses. So, 3-hexanone deserves a careful look, not just for chemists but for anyone working or living near it.
Understanding 3-Hexanone’s Profile
This liquid has a faint odor, a bit like acetone. Its main job for industry involves solvents, flavors, and fragrances. Lab reports show it evaporates easily, raising questions about inhalation at work. My time around shop floors and research benches taught me that hazardous doesn’t always mean deadly, but it always asks for respect.
Known Hazards and Health Effects
Studies in both animals and humans suggest high enough exposure can irritate eyes and lungs. Anyone spending too long in a poorly ventilated area with 3-hexanone may feel dizzy, headache, or even nauseated—classic signs of chemical exposure, not unique to this substance. Animal studies hint at possible liver and kidney strain after repeated or high-dose exposure, but that doesn’t mean a single whiff at a factory spells doom. Most evidence points to risk growing with longer, repeated exposure. Scientists at regulated toxicology labs have flagged these patterns for years and the chemical’s material safety data sheet tells employees to avoid skin contact and to watch for warning signs of overexposure.
Limits and Guidelines Keep Exposure Low
Regulatory agencies, like OSHA and the EPA, review chemicals like this and set air concentration limits for workplaces. At places I’ve worked, anything rated as a possible irritant got ventilation, splash guards, gloves, and supervisors reminded folks to wash up after handling. Those basic practices cut down on risk for everyday users. Factories and labs logged exposures, checked vent fans, and kept robust first aid at hand—protecting workers and catching problems before they grew.
Precaution and Knowledge Go Hand-in-Hand
Many people tune out chemical names, trusting that rules keep them safe. That trust only holds if everyone along the way pays attention. Chemical suppliers must label containers with hazard symbols and clear instructions. Employers must provide safety training; I remember team safety meetings where stories about “close calls” stuck with folks better than any rule book. It takes as much work to keep records up-to-date as it does to keep supplies ordered, but both matter for health.
Better Solutions for Safer Workplaces and Products
Finding alternatives makes sense where risk is highest. In places where 3-hexanone could trigger headaches or worse, switching to a less volatile, less irritating solvent reduces stress—on workers and everyone downstream. Automation and better ventilation systems cut down on open-air exposure, from factory to university lab. Simple ideas like closed transfer systems, splash-proof containers, and routine monitoring keep surprises rare. For home use, clear directions matter most. People need honest labels, and not just fine print about “irritants.” Clarity supports trust and helps users make safer choices.
Building a Safety-Minded Culture
Experience tells me that accidents often start with shortcuts or missing information. Managers and workers share the job of looking out for each other—open communication works better than top-down orders. Reporting near misses leads to real improvements, like swapping out worn seals or redrawing work routines. Investing in better safety now prevents bigger headaches later, for workers, companies, and the community. Small steps—like keeping storage areas labeled or insisting on good airflow—mean a lot in the long run, reducing risk for everyone exposed to chemicals like 3-hexanone.
How should 3-Hexanone be stored?
Understanding the Risks
Anyone who’s worked in a lab or on a production floor has crossed paths with solvents like 3-Hexanone. This clear liquid hides some problems beneath its mild look. It’s flammable, has a sharp odor, and its vapors can trigger headaches or worse. The number of stories I’ve heard about folks shrugging off ventilation safety because "it’s just another ketone" never fails to surprise me. Even seasoned techs sometimes undervalue the risks, probably because nothing dramatic happened—yet. The fact is, chemistry doesn’t take a day off. Mismanaging a compound like this usually comes back to bite you, sometimes in ways you don’t see right away.
Safe Storage: Real-World Approaches
Keep 3-Hexanone away from sparks, flames, or things that get hot. Direct sunlight is another enemy; I’ve seen labels bleach out completely on bottles kept near a window. Find a spot that stays cool and dry—a metal chemical cabinet with self-closing doors is a strong bet, especially one labeled for flammable liquids. This isn’t about satisfying someone's checklist; it’s about making sure little mistakes don’t snowball into major incidents.
Airflow in the storage area can make a difference. I remember helping clear out a back room with containers that reeked of solvent, where nobody thought twice about ventilation. Later, workers reported nausea long after the bottles left the shelves. Venting chemical storage and keeping the doors sealed when not in use make a world of difference for the people nearby. Chemical-resistant shelves and trays catch drips. Old towels and cardboard break down and soak up spills, turning a small leak into a real headache, so keep those out of reach.
Containment That Works
Plastic jugs just don’t cut it for most ketones, especially over time. Use containers made from materials that don’t react—often that means dark glass with screw caps, never pour-spout jugs from the hardware store. The right cap matters—loose lids or those with warped threads aren’t enough. Vapor-tight seals limit fumes, slow down evaporation, and keep accidental inhalation off the table.
Chemical labels deserve more respect, too. Use waterproof markers and slap on a clean, clear label with the name, date, and hazard statements. In a fast-moving workplace, it only takes one lazy day to end up with mystery bottles. Lost records aren’t just a paperwork problem; they can set the stage for mixing mistakes and dangerous reactions later.
Keeping People Safe
Locked storage discourages casual handling and careless grabs from folks who don’t know what they’re dealing with. Kids, visitors, and new staff don’t have business around volatile solvents by themselves. Safety gear—gloves, goggles, lab coats—should sit in plain sight nearby. I learned early that gear collects dust unless it’s easier to reach than the hazard itself.
Spill kits belong nearby, stocked with materials actual people feel confident using, or they won’t get touched in a real crisis. Regular training builds muscle memory for tough moments. People often roll their eyes at drills, but in tight spaces or under stress, routine practice makes cleanups faster and smarter, with fewer regrets.
Why It All Matters
Chemicals like 3-Hexanone store up trouble for the inattentive. They reward caution and consistency, not just compliance with codes but with habits forged from actual experience. Small changes—like a sturdy storage system or quick-glance hazard info—build up a safer workplace. Stories from emergency room visits and fire-rescue calls stick with you. Respecting basic safety rules around storage isn’t just good science; it’s looking out for yourself and the people counting on you.
What are the physical properties of 3-Hexanone?
Diving Into Appearance and Odor
3-Hexanone shows up as a clear, colorless liquid that doesn’t call much attention to itself on sight. Its odor, though, pulls you right in with a distinct, sweet, fruity, and slightly acetone-like smell. Anyone who’s worked in a chemistry lab recognizes that mark—a sort of fragrant punch, less sharp than acetone or ethyl acetate but lingering enough to remind you of solvents often used for cleaning or extraction. That familiar scent can float through a space before you realize a bottle’s uncapped on the bench.
Boiling Point and Volatility
This compound turns up with a boiling point right around 127°C (260°F). That places it in a band with many mid-length ketones—not so low that it flashes off at room temperature, not so high as to require special gear to distill. Its volatility means anyone pouring or measuring it ought to keep good ventilation in mind, since vapors can build fast in closed rooms. The vapor density sits heavier than air, drifting along countertops and settling in low spots, a fact that raises safety needs if spills or leaks pop up.
Solubility and Polarity
In water, 3-Hexanone barely dissolves—less than 1 gram per 100 milliliters at 20°C. The molecule includes a carbonyl group (C=O), which has a strong pull for polar solvents, but most of the carbon chain repels water. Still, this substance mixes well with organic solvents like ethanol, diethyl ether, and chloroform. Anybody putting it to use, whether in industrial applications or research projects, needs to grab compatible solvents and avoid mixing with water where full separation is important.
Density and Viscosity
If you weigh it out, 3-Hexanone lands with a density close to 0.82 grams per cubic centimeter at room temperature. It pours smoothly, a bit less runny than many lighter alcohols but still faster-moving than heavy oils or syrups. Handling measures—like transferring from one flask to another—go without much fuss, as it flushes through glassware efficiently.
Reactivity and Stability Hazards
This ketone doesn’t react with light or oxygen in the air nearly as much as aldehydes or peroxides, making it relatively stable on the bench. Despite this, the vapor can form explosive mixtures with air at the right concentration. Safety data upholds a flash point around 27°C (80°F), so that familiar lab routine—keeping heat and flame far from open containers—applies every time.
Why Physical Properties Matter
Getting a grip on the physical features of 3-Hexanone makes for real-world utility. In fragrance creation, those fruity notes get dialed up and slotted into formulations precisely because chemists know how volatile and dissolvable it is. In extraction processes, its blending traits help separate plant oils or flavors. Handling rules and personal experiences reinforce the point—these facts aren’t just academic, they shape safe, efficient, and creative use of chemicals in daily work.
Practical Safety and Community Sharing
Talking about 3-Hexanone’s physical makeup isn’t only for experts. Field workers, students, and small manufacturers all lean on trusted facts—boiling points, flammability, solubility—because they have skin in the game. Learning by trial sounds romantic until someone breathes in toxic vapor or sparks a fire. Safety sheets, university articles, and manufacturer labels all anchor their facts in real-world evidence, pooling years of lab experience into every safety tip and handling rule.
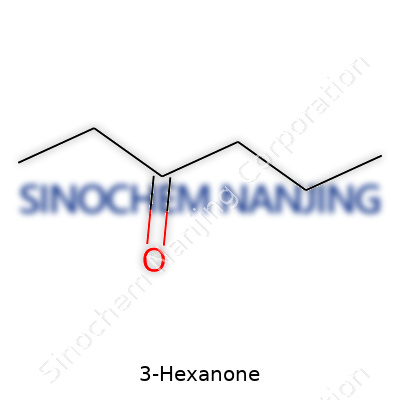
| Names | |
| Preferred IUPAC name | hexan-3-one |
| Other names |
Ethyl propyl ketone Ethyl n-propyl ketone Hexan-3-one |
| Pronunciation | /ˈθriːˈhɛk.səˌnoʊn/ |
| Identifiers | |
| CAS Number | 623-37-0 |
| 3D model (JSmol) | `3d://C(C)CC(C)C` |
| Beilstein Reference | 1200402 |
| ChEBI | CHEBI:16340 |
| ChEMBL | CHEMBL25434 |
| ChemSpider | 68252 |
| DrugBank | DB01941 |
| ECHA InfoCard | 100.059.408 |
| EC Number | 3.2.1.21 |
| Gmelin Reference | 82238 |
| KEGG | C01753 |
| MeSH | D006024 |
| PubChem CID | 8027 |
| RTECS number | MN9275000 |
| UNII | L2J1E80998 |
| UN number | UN 1226 |
| Properties | |
| Chemical formula | C6H12O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless liquid |
| Odor | Fruity |
| Density | 0.813 g/cm³ |
| Solubility in water | 11 g/L |
| log P | 0.89 |
| Vapor pressure | 2.8 kPa (at 20 °C) |
| Acidity (pKa) | pKa ≈ 20 |
| Basicity (pKb) | 6.74 |
| Magnetic susceptibility (χ) | -8.41 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.410 |
| Viscosity | 0.657 mPa·s (25 °C) |
| Dipole moment | 2.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 319.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -285.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3545.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H319, H335 |
| Precautionary statements | P210, P280, P305+P351+P338, P304+P340, P312 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 41 °C |
| Autoignition temperature | 515 °C |
| Explosive limits | 1.2–7.7% |
| Lethal dose or concentration | LD50 oral rat 1670 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1670 mg/kg |
| NIOSH | HK7070000 |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 41.7 |
| IDLH (Immediate danger) | 800 ppm |

