3-Ethylpyridine: An In-Depth Commentary
Historical Development
Chemistry often moves in small steps, with setbacks and rare flashes of luck. 3-Ethylpyridine got its name in the early 20th century as synthetic methods for pyridines gathered steam. Traditional pyridine chemistry sparked curiosity for molecules that could tweak basicity, sterics, or reactivity by shifting a side chain. By targeting the third carbon on the pyridine ring, chemists discovered that changes in molecule layout could influence all sorts of properties—boiling point, solubility, even how sharply a substance smells. As petroleum and coal tar processing matured through the decades, scientists found more cost-effective ways to produce small nitrogen heterocycles. This expanded interest in pyridine derivatives for dyes, agrochemicals, and, eventually, pharmaceuticals—especially those with subtle ring modifications like 3-ethylpyridine. My own research experience tells me, those early years demanded endless patience for synthetic yields that we’d laugh at now, but they gave a solid foundation for today's efficient lab routines.
Product Overview
3-Ethylpyridine is a colorless to pale yellow liquid, with a flammable personality and an unmistakable pyridine-like, somewhat fishy odor. Its molecular formula is C7H9N, placing it among the alkylated pyridines which each stake out their own slice of reactivity and application. Today, it frequently serves intermediate roles in pharmaceutical building blocks and specialty chemical preparations. Across the board, folks handling it must respect its flammability and pungency, a detail impossible to ignore once you’ve uncapped a bottle in a poorly ventilated space. The substance often draws attention for its role in creating vibrant dyes, refined catalysts, and sometimes even flavors and fragrances—though its odor reminds everyone why careful filtering and purification matter in those fields.
Physical & Chemical Properties
At room temperature, you’ll find 3-ethylpyridine in a liquid state. It boils near 144°C and melts below -40°C, meaning it handles both laboratory and industrial processes involving standard pressures. The density hovers around 0.95 g/cm3. It mixes with typical organic solvents, such as ether and alcohol, but struggles to blend fully with water, a trait it shares with its cousins in the alkylpyridine family. This structure brings a nitrogen atom’s lone pair into play, granting the molecule basic characteristics and nudging it into hydrogen bonding reactions. That nitrogen, along with the ethyl side group, invites plenty of chemical interest—whether you want to protonate, oxidize, or halogenate it. In my experience, nobody forgets the sharpness of pyridine’s odor, and the ethylated form doesn’t improve that—but its distinct boiling point and stability often tip the scales for those who need specificity in synthesis.
Technical Specifications & Labeling
Commercial suppliers offer 3-ethylpyridine at purities around 98% to 99%, typically identified by the CAS Number 536-78-7. Containers arrive labeled for hazardous materials, usually featuring the UN number for flammable liquids, hazard pictograms, and statements cautioning users about inhalation and skin contact. Analytical data—NMR, GC, and MS spectra—back up quality guarantees, and suppliers often include storage guidance: keep containers tightly closed, away from open flames, strong oxidants, and direct sunlight. Handling requires goggles, gloves, and fume hoods; even a minor spill can unleash a stinging aroma through an entire lab, and the idea of improvising with PPE never sits well with seasoned chemists. Waste streams require isolation and safe incineration, not quick disposal down the drain. Regulatory databases flag 3-ethylpyridine for transportation risks, with safety sheets outlining symptoms linked to overexposure—another reminder that chemical curiosity demands respect, not shortcuts.
Preparation Method
Decades of work with pyridine synthesis showcase a few main approaches to 3-ethylpyridine. Earlier generations leaned on the condensation of simple aldehydes and ammonia sources, with variable yields. These days, the Chichibabin synthesis and side-chain alkylation of pre-formed pyridine lead the pack. In the alkylation method, pyridine meets ethyl halides in the presence of a base to generate mixtures, often followed by distillation and selective crystallization. Newer catalytic systems—think palladium or nickel complexes—promise better selectivity and greener credentials, a move welcomed by both industrial chemists and environmental regulators. My group once explored continuous-flow alkylation, with real-time monitoring, which made a typically slow and fussy synthesis more reliable. Temperature, base strength, and reaction vessel design all shape the outcome, so small changes add up fast. Optimizing the process isn’t just a question of making as much product as possible but also of avoiding side reactions, minimizing waste, and ensuring batch-to-batch consistency.
Chemical Reactions & Modifications
The ethyl group attached to the third carbon shuffles reactivity in thoughtful ways. At the nitrogen, 3-ethylpyridine behaves as a typical pyridine, grabbing protons or forming complexes with metals, a trait that brings catalytic uses within reach. Electrophilic aromatic substitution finds a tougher time here compared to pyridine itself, as the electron-donating alkyl tilts electron density around the ring. Oxidation ideas open up: strong oxidants target the ethyl group, driving it toward acetyl or carboxyl functionality—vital intermediates in both pharmaceutical and agricultural chemistry. Halogenation also calls for controlled conditions to avoid overdoing reaction runs. For custom molecules, teams often introduce substituents while leveraging the ethyl group’s directing influence, providing entry points for further synthesis. My most memorable laboratory moment came after coaxing a challenging oxidation to work with sub-stoichiometric catalyst, a feat that cut down cleanup headaches and verified the power of clever reagents.
Synonyms & Product Names
Literature and supplier catalogs list a range of synonyms for 3-ethylpyridine. The most direct is beta-ethylpyridine, reflecting the side chain location. Other references might shorten it to 3-EP or mention ethylpyridine without a positional qualifier, leading sometimes to confusion—those shopping for derivatives should always check CAS numbers. In the European chemicals market, systematic names like 3-ethyl-1H-pyridine surface in labeling. Researchers in flavor chemistry or catalysis studies may stumble across trade names or internal codes, though these rarely persist beyond niche applications. For practical purchasing or regulatory compliance, correct chemical identity matters more than tradition or short-hand. Even as technology improves labeling and traceability, old habits mean staff still double-check product codes, physical appearance, and basic spectra to sidestep supply chain mix-ups.
Safety & Operational Standards
3-Ethylpyridine poses familiar hazards of compounds in its class—flammability, volatility, and skin and mucous membrane irritation. Workplaces store it in ventilated, fire-resistant cabinets, away from oxidizers or acids. Technicians suit up with gloves, goggles, and often lab coats treated against permeation. The substance kicks out sharp vapor, easily inhaled, so experiments run under fume hoods—once, a careless colleague uncapped a flask outside one and got a day-long headache for the trouble. In emergencies, safety showers and eye washes line the nearest wall, and spill kits reside close to work benches. If spillage or inhalation happens, protocols push for quick evacuation and medical evaluation. Factory sites invest in real-time vapor monitoring, ensuring local air never exceeds exposure limits. Emergency teams train for both burns and respiratory distress, given the molecule’s flammable and toxic character—no operator worth their salt skips those annual drills.
Application Area
Industries reach for 3-ethylpyridine in a host of roles: intermediate in fine chemicals, precursor in agrochemical synthesis, scaffold for pharmaceutical discovery, and additive in some specialty polymers and dyes. In drug research, the molecule often serves as a starting point for creating unique nitrogen-containing scaffolds, offering pharmacokinetic properties not found from unsubstituted pyridines. Crop protection firms use it to build more robust pesticides and herbicides, banking on the molecule for reliable nitrogen insertion and custom-tailored volatility. Catalyst designers appreciate the ability to fine-tune basicity and steric bulk for industrial reactions. Researchers in fragrance and flavor sometimes flirt with 3-ethylpyridine’s less offensive derivatives to create smoky or nutty notes, always aware of the narrow margin between a pleasant whiff and an overbearing stench. In all these uses, the thread connecting discovery teams is an appreciation for small changes making a big impact on molecular function.
Research & Development
New uses for 3-ethylpyridine always ride the line between fundamental discoveries and application-driven breakthroughs. Synthetic chemists chase higher-yield, greener synthesis pathways, working to ditch expensive or hazardous reagents. Life sciences teams explore analogs bearing extra functional groups for use as enzyme inhibitors or imaging agents. Material scientists look at modifications to design corrosion-resistant coatings and think about how molecular tweaks improve thermal stability. These challenges spark collaboration: analytical scientists monitor reaction courses; process engineers scale up successful bench-top syntheses; computational chemists tweak predictive models of reactivity. In my own collaborations, success emerged from blending insights across sub-fields—often, it was the technician with sharp eyes on chromatograms that caught an unanticipated impurity, saving weeks of effort downstream.
Toxicity Research
Toxicology data for 3-ethylpyridine mirrors related pyridines. Studies indicate acute toxicity with low to moderate oral and inhalation exposure, producing symptoms like headaches, dizziness, or skin irritation. Rodent studies show LD50 values in the few hundred milligrams per kilogram range, with longer exposures suggesting organ targets including the liver and kidneys. Chronic data remain scant, but evidence argues for caution in repetitive or high-dose contexts. Regulatory authorities classify 3-ethylpyridine as hazardous for skin, eye, and respiratory exposure. My own safety training hammered in one point: even without catastrophic risks, repeated small exposures build over time. That awareness keeps research teams vigilant about glove changes, air sampling, and spill logs—nobody wants their work undone by overlooked occupational risks. Institutions back up this vigilance with routine limiting measures, including improved handling apparatus and air filtration.
Future Prospects
The next decade looks busy for 3-ethylpyridine. Green chemistry pushes in favor more sustainable feedstock and energy-efficient processes, with teams racing to replace harsh catalysts or cut down solvent waste. Drug designers eye additional modifications on the pyridine scaffold, hoping to unlock both better bioactivity and safety. Electronic materials research signals interest in nitrogen heterocycles for the next generation of OLED displays and flexible electronics. Artificial intelligence increasingly models new reaction pathways and predicts toxicity, aiming to weed out less promising candidates early. For all these ambitions, collaboration keeps progress on track—bring together chemists, engineers, and toxicologists, and novel applications or safer handling protocols follow. In my view, blending these perspectives remains the most likely source for breakthroughs, ensuring that 3-ethylpyridine transitions from an industrial intermediate into a backbone for future technology, health, and sustainability innovations.
What is 3-Ethylpyridine used for?
Seeing the Full Picture of 3-Ethylpyridine
3-Ethylpyridine rarely comes up at the family table, but it has found its way into labs and industry shelves for good reason. It's an organic compound tucked under the broad umbrella of alkylpyridines. This molecule doesn't headline product labels or TV ads, yet it shapes multiple fields behind the scenes. Understanding how it works and where it shows up offers real insight into the little molecules that touch our lives.
The Quiet Force in Pharmaceuticals and Synthesis
Researchers keep turning to 3-Ethylpyridine for a simple reason: it opens doors to more complex molecules. In pharmaceutical labs, chemists use it as a building block. Drug synthesis gets complicated fast. 3-Ethylpyridine forms part of that puzzle—helping craft compounds for potential antivirals, antifungals, and other materials that eventually support modern medicine. In these jobs, purity matters. Impurities cause cascading problems by throwing off reactions and wasting valuable time. The consistency of 3-Ethylpyridine means fewer surprises during scale-up from the lab bench to the plant floor.
I remember a close friend in grad school who chased down new anti-tubercular molecules. Many mornings started with a cup of bad coffee and a vial of 3-Ethylpyridine. Accuracy became their mantra. The tiniest slip in measurement, or sourcing material with hidden contaminants, sent experiments back to square one. Reliable sources made their breakthrough less a stroke of luck and more a question of stubborn persistence—one clean batch at a time.
Fuel Research and Catalysts
Petrochemical research leans on alkylpyridines too. 3-Ethylpyridine acts as a model in studies hunting for cleaner combustion, alternative fuels, and better catalysts. Researchers use it to probe chemical reactions or build more efficient pathways for refining crude oil. It’s not glamorous, but these tweaks to process chemistry ripple out to lower emissions or improve yields in big refineries.
Flavors, Odors, and Small Surprises
3-Ethylpyridine leaves subtle fingerprints on flavors and odors. Analytical chemists sometimes use it as a flavor or aroma marker, helping to pinpoint the presence of other molecules in substances like tobacco smoke or roasted coffee. Its scent catches trained noses and helps dissect what makes a flavor profile unique, which matters for food scientists and environmental testing alike.
Fact-Checking, Regulation, and Safety
Handling specialty chemicals demands trust and clear oversight. Google and other platforms aim to filter trustworthy advice, so people rely on credible sources. Material Safety Data Sheets, regulatory guidelines, and strong track records from producers help set the ground rules. Even skilled chemists check these before handling new batches, making sure to use gloves and work in ventilated spaces. Good manufacturing keeps problems out of downstream products, too—something regular folks benefit from every time they reach for a medicine or packaged snack on the store shelf.
Looking Forward
Innovation keeps pushing demand for building blocks like 3-Ethylpyridine. Synthetic chemistry, pharmaceutical advances, and environmental testing will keep it in rotation. As open science grows, sharing reliable findings and clear safety protocols matters even more. Industry and academia both need access to high-quality chemicals, steady supply chains, and above all—information they can stand behind.
What is the chemical formula of 3-Ethylpyridine?
Understanding 3-Ethylpyridine at Its Core
Chemistry class always brings up images of complex glassware and pungent odors. Back in university, I remember cramming for organic chemistry exams and sorting compounds not by their smell or color, but by their structure and formulas. Among those, pyridine derivatives seemed daunting at first glance. Their names roll off the tongue like a riddle, but they follow strict rules. 3-Ethylpyridine is no exception. The formula, C7H9N, shows exactly how chemists piece together this particular puzzle.
What’s Hidden In The Name?
Names in organic chemistry might sound confusing, though they carry more honesty than most people realize. “Pyridine” refers to a six-membered ring, much like benzene, but with one nitrogen replacing a carbon. Then comes the “3-ethyl” bit. That signals a two-carbon ethyl group clinging to the third position of the ring. With this basic information, chemists can sketch out the molecule without ever seeing it in person. The formula, C7H9N, lays out the building blocks: seven carbons, nine hydrogens, and one nitrogen. A straightforward count, but with big implications.
Where 3-Ethylpyridine Pops Up Beyond the Lab
3-Ethylpyridine matters for more than just test answers on an exam. It creeps into pharmaceutical research and industrial chemistry. Some scientists use it as a basic “skeleton” to build bigger, more complex molecules. It helps test reactions that need a little nitrogen punch without the quirks of a more elaborate ring system. In practice, I’ve seen it show up as a reference molecule for analytical chemistry standards and sometimes in the footprints of chemical manufacturing—a reminder that chemicals travel unnoticed in many corners of industry.
Importance of Knowing the Formula
It’s easy to brush off the whole business as academic trivia. Yet, knowing the precise formula matters. Laboratories depend on accuracy, not just for research but for safety and efficiency. A typo in a formula can lead to wasted resources or unsafe handling. The Environmental Protection Agency lists how compounds like pyridine and its derivatives behave, their potential to irritate skin or pollute waterways. Regulatory forms demand flawless documentation. Pair those rules with the importance of reproducibility in science and it’s clear: one letter or number out of place can mess up the entire chain.
How to Keep Chemistry Real
I learned early on that chemistry rewards careful work. I watched classmates scramble after a mistaken compound created an unexpected reaction. Mistakes stick in the mind and in the fume hood. These memories push me to double-check chemical structures and formulas, not out of fear, but from practical experience. It doesn’t take long for a misplaced group or number to spark real headaches.
For students learning the ropes, the best advice comes from repetition and actually drawing these molecules out. Reading the formula over and over again does not compare with sketching the ring and its side chains on paper. That kind of “muscle memory” sticks far longer than words on a screen or textbook.
Path Forward for Better Understanding
Chemistry shouldn’t scare anyone away—though it pays to respect how precise the field needs to be. More accessible explanations and hands-on study make a difference. For teachers and librarians, practical resources can bridge gaps faster than dense academic language ever could. Clear labels, updated safety sheets, and lab exercises help everyone stay sharp when dealing with something like C7H9N. And if all else fails, don’t be afraid to ask questions or look up a trusted chemical database for confirmation. Confidence grows from small, concrete steps, one formula at a time.
Is 3-Ethylpyridine hazardous or toxic?
Shining a Light on an Overlooked Chemical
Sometimes people stumble across names like 3-ethylpyridine and wonder what risks might hide behind the jargon. This compound, with its roots in the world of industrial chemistry, usually gets skipped in public conversations, but it turns up in labs and manufacturing. Curiosity about its danger level makes sense, especially with headlines about chemical safety growing louder every year.
Hazards That Come with Regular Handling
Anyone with science lab experience learns to respect chemicals with strong odors like 3-ethylpyridine. A sniff of this stuff leaves no doubt: it means business. Exposure can hit in different ways, yet the biggest problem shows up when safety steps get skipped. Touching the liquid might spark skin or eye irritation. Breathing its fumes leads to a burning sensation—or a cough that doesn’t quit until you walk far away.
The trouble isn’t just about immediate reactions. Studies testify that pyridine derivatives, especially those close to the structure of 3-ethylpyridine, pile up toxic effects after repeated contact. Over time, the liver, kidneys, and even the central nervous system show signs of wear and tear. The National Center for Biotechnology Information notes that pyridine-based chemicals can produce long-term organ damage when handled recklessly, making personal protective gear less of a suggestion and more of a requirement.
Is 3-Ethylpyridine a Major Health Threat?
Pyridines generally don’t attract as much fear as something like cyanide, but dismissing them would be a mistake. Research explains how inhaling pyridine or touching it too often carries long-term risk. Evidence points to carcinogenic potential in animals, which gives authorities reason to apply the precautionary principle around people. The European Chemicals Agency classifies 3-ethylpyridine as harmful if swallowed, inhaled, or absorbed through the skin. That warning connects directly with my own days working at a chemical plant—nobody wanted to test the safety limits.
Another point often missed: improper storage can worsen its hazards. Spills release vapors quickly, leading to headaches and dizziness for workers who linger nearby. I’ve seen cleanup crews suit up with respirators and gloves, making the urgency clear even to bystanders. It only takes a careless move to spark trouble for everyone in the room.
Using 3-Ethylpyridine Responsibly
Sensible rules can keep people safer. The use of chemical fume hoods, gloves, and eye protection shouldn’t get skipped for the sake of saving time. Working in a community college lab introduced this lesson fast—one botched experiment sent fumes running down the bench, and nobody ignored their safety glasses again. OSHA guidelines set clear exposure limits, and for good reason. In my experience, it’s best to stick to those numbers rather than push boundaries out of impatience or habit.
It helps when organizations invest in regular staff training. Posters and safety data sheets educate, but real discussions about incidents help people grasp what’s at stake. Strong policies should include rapid response for spills and regular equipment checks. I’ve seen teams prevent big problems just by practicing their response plan every few months. From there, toxic exposure drops, and health complications stay rare.
What Makes This Question Matter
Curiosity about the risks around 3-ethylpyridine isn’t just academic. Anyone working near chemical stocks or processing lines puts their health on the line, even if it doesn’t seem obvious at first glance. Taking the issue seriously, listening to the science, and respecting the lessons learned from years in the field: these habits go a long way toward reducing danger. In chemistry and life, shortcuts don’t often pan out.
How should 3-Ethylpyridine be stored?
The Risks We Live With
I remember the sharp, almost tear-inducing smell that clung to my gloves whenever I handled 3-Ethylpyridine in the lab. That gritty reminder—your senses can only take so much—keeps safety at the front of your brain. This liquid doesn’t belong near people not prepared for its bite. It’s flammable, and it can hurt skin and lungs quickly. Laboratories often keep it around, using it in synthesis or research, but comfort, convenience, and carelessness shouldn’t get mixed together. That’s where good habits, built from experience and backed by research, make all the difference.
Temperature Control
Don’t just stuff 3-Ethylpyridine on a random shelf by the window. Heat in a storage spot raises the risk of vapor buildup and fire. The flash point sits low, not much above room temperature. That’s tempting fate if you store the bottle near a radiator or direct sunlight. My worst close call came from a forgotten bottle left near a heat lamp—that room stank for days. Safer practice means finding a cool, well-ventilated place, far from ignition sources. A flammable liquids cabinet offers solid protection, with metal sides keeping heat out and locking doors limiting who gets inside. Flammable cabinets aren’t just for chem majors—they’re a must for anyone unwilling to roll the dice with fire.
Keep Oxygen and Water Out
3-Ethylpyridine doesn’t play nicely with open air or humidity. I learned early—wipe every drop from the neck after you pour, screw on the cap tight, and label the bottle with an open date. If the cap stays loose, oxygen makes its way in, risking slow changes or contamination. While not instantly deadly, unstable air-exposed pyridines corrode labels and can even degrade over time, raising costs and complicating tracking for anyone needing accurate records. Store the liquid in a bottle sturdy enough for chemicals and keep it shut unless you’re pouring.
Labeling Doesn’t Lie
The urge to skip labeling happens more than folks admit. Sharpies dry up or someone claims, “I’ll remember.” In crowded labs or shared spaces, that’s wishful thinking. Clear, bold chemicals labels prevent ugly surprises, and safety officers know where to look if something goes wrong. List the name, hazards, and the date. If the color shifts, vapor escapes, or a smell appears where it shouldn’t, a good label speeds up problem-solving. Label blindness leads to accidental mixing, and unlike mixing up your sugar and salt, the results can burn, poison, or blow up.
Personal Responsibility in Shared Spaces
Every time someone cuts corners on safety, the risk spreads to everyone else nearby. Years ago, I saw a new student toss solvents into an old cleaning-solution bottle, cap it, and walk out. It’s easy, in the rush of the moment, to forget the chain reaction one shortcut can spark. Encourage everyone—no matter how busy—to put things back right, respect compatible storage (keep oxidizers, acids, and bases far apart), and clean up small spills straight away. Chemists have a code: leave the space safer than you found it.
Building a Safer Routine
Storing 3-Ethylpyridine isn’t a plug-and-play task. Sensible routines grow from stories, close calls, and stubborn reminders. Cool, locked cabinets, clean labels, vigilant housekeeping—straightforward steps keep trouble at bay. If you’re still not sure where to start, reach for the chemical safety data sheet, talk to a trusted lab manager, or recheck your storage space before the next shipment. Safety, after all, builds one good habit at a time.
What are the physical properties of 3-Ethylpyridine?
Physical Details That Actually Matter
Picture a clear, colorless liquid with a sharp, slightly fishy odor—3-ethylpyridine isn’t something you’d want to spill near your workspace. Its aroma can linger in a lab, embedding itself deep into the memory of anyone handling it for long. That smell comes from the nitrogen atom in its ring. I remember the first time I opened a bottle: I learned straight away to work in a fume hood.
3-Ethylpyridine keeps things straightforward with its boiling point, sitting at roughly 155°C. Compared with water, the extra carbon chain nudges the temperature up. That makes it stable for basic distillation and gives it resilience in moderate heating. It doesn’t catch fire instantly, but reaching its flash point—about 45°C—takes only mild warming. In practice, that means no one with experience leaves its container open near sources of heat.
As for its density, 3-ethylpyridine weighs in at roughly 0.95 g/cm³, slightly lighter than water, so it won’t settle to the bottom if poured into an aqueous mix. Neither does it float on air, making ventilation in confined spaces pretty important. This property sometimes trips up students in labs, especially during early extractions—expect it to dissolve better in organic solvents than in water.
Solubility and Handling in the Lab
Working with this compound, I’ve seen that it dissolves quickly in common organics: ether, benzene, and chloroform blend with it easily. Water, on the other hand, only welcomes small amounts of 3-ethylpyridine. That can slow down reactions relying on mixing with aqueous solutions, so it’s not the go-to for anything water-heavy.
Lab veterans get used to its volatility. You can't leave the bottle open without the air becoming hard to breathe. Keep gloves and goggles on—this stuff stings on contact and sneaks into tiny scrapes. The label’s warning about “harmful if inhaled or absorbed through skin” earns every bit of its bold print.
Why Its Properties Dictate Application
Looking at its combination of solvent affinity and moderate boiling point, 3-ethylpyridine steps up in organic synthesis and research, especially with pharmaceutical precursors and agrochemical studies. Its liquid state at room temperature gives it flexibility, though the strict storage requirements stem from its volatility and odor. Over time, I’ve developed a real appreciation for sturdy screw-cap containers—one loose lid can stink out an entire bench.
Beyond the physical inconvenience, there’s an environmental consideration. Spills evaporate fast, so exhaust fans and closed transfer systems aren’t optional. Ignoring safety gear means dealing with headaches or worse. In settings without modern ventilation, those properties make it hard to justify routine use.
Safe Use Makes All the Difference
After years working with similar heterocycles, I’ve found the trick isn’t treating 3-ethylpyridine as some rare hazard; it’s about making its properties second nature. Respect the fumes, keep it corked, and use gloves that actually stand up to solvents. Students who remember its sharp bite rarely skip their PPE next time.
We can’t shape its boiling point or odor, but the right awareness in handling and storage keeps 3-ethylpyridine useful—not just another chemical on a crowded shelf. It’s proof that sometimes, a few simple properties shape everything about how we use a compound, from the fume hood to the waste drum.
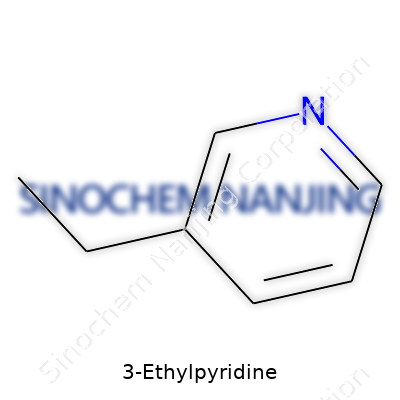
| Names | |
| Preferred IUPAC name | 3-Ethylpyridine |
| Other names |
3-Ethylpyridine beta-Ethylpyridine m-Ethylpyridine |
| Pronunciation | /ˌθriːˌɛθ.ɪl.pɪˈrɪd.iːn/ |
| Identifiers | |
| CAS Number | 536-78-7 |
| Beilstein Reference | 1109951 |
| ChEBI | CHEBI:89680 |
| ChEMBL | CHEMBL161532 |
| ChemSpider | 12156 |
| DrugBank | DB04109 |
| ECHA InfoCard | 03d675d7-d7d6-420c-9d2e-2ed18e8b478e |
| EC Number | 207-924-2 |
| Gmelin Reference | 7876 |
| KEGG | C06162 |
| MeSH | D017909 |
| PubChem CID | 13509 |
| RTECS number | UC3850000 |
| UNII | YL8I2Y0U6A |
| UN number | UN2522 |
| Properties | |
| Chemical formula | C7H9N |
| Molar mass | 107.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pyridine-like |
| Density | 0.944 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | 1.56 |
| Vapor pressure | 1.8 kPa (at 20 °C) |
| Acidity (pKa) | 5.86 |
| Basicity (pKb) | 5.82 |
| Magnetic susceptibility (χ) | -53.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.520 |
| Viscosity | 0.887 cP (20°C) |
| Dipole moment | 1.689 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 188.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 94.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3346.9 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H301, H311, H314, H331 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P370+P378 |
| Flash point | 68 °C (154 °F; 341 K) |
| Autoignition temperature | 480 °C |
| Explosive limits | 1.2–8.3% |
| Lethal dose or concentration | Lethal dose or concentration: **LD50 (oral, rat): 353 mg/kg** |
| LD50 (median dose) | LD50 (median dose): 160 mg/kg (rat, oral) |
| NIOSH | KW4025000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Ethylpyridine: Not established |
| REL (Recommended) | 100 |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
2-Ethylpyridine 4-Ethylpyridine Pyridine 3-Methylpyridine 3-Propylpyridine |

