3-Ethoxyaniline: A Comprehensive Commentary
Historical Development
The journey of 3-ethoxyaniline traces back to the progress in organic chemistry during the 19th century, when scientists learned how to manipulate aromatic rings and push the boundaries of synthetic routes. In university research labs, students often come across stories of aniline derivatives playing significant roles in the dye industry boom, sparking advances not just in textiles but also in medicine. The discovery of 3-ethoxyaniline — an aniline with an ethoxy group tucked into the meta-position — marked a small but important pivot. This compound never drew the limelight like its parent aniline or para-substituted cousins, yet it found its way into laboratory inventories. Over generations, chemists have valued the structure for its versatility, linking it to broader developments in pharmaceuticals, agrochemicals, and even polymer science.
Product Overview
3-Ethoxyaniline, belonging to the family of ethoxylated anilines, gets recognized by professionals for its clear to yellowish appearance and strong aromatic odor. Factories producing specialty chemicals list 3-ethoxyaniline as a core intermediate for making a range of compounds, often packaging it in sealed amber bottles to protect from light and air. Prices fluctuate with the availability of starting materials and demand in downstream industries, especially in Asia’s burgeoning markets. In real-world practice, labs order 3-ethoxyaniline not just for its direct reactivity, but as a valuable building block for custom syntheses.
Physical & Chemical Properties
3-Ethoxyaniline appears as a pale liquid, sometimes exhibiting light brown tinges as it ages or absorbs moisture. With a boiling point above 240°C and a melting point that rarely concerns most users thanks to its perpetual liquid state at room temperature, the compound has a density around 1.03 g/cm³. Its molecular formula, C8H11NO, translates to a weight of about 137.18 g/mol. 3-Ethoxyaniline dissolves in organic solvents like ethanol and ether, but struggles to mix with water, echoing the behavior of similar aromatic amines. The amino group’s basicity and the ethoxy’s electron-donating effect tweak both its reactivity and shelf stability, giving synthetic chemists a flexible toolkit.
Technical Specifications & Labeling
Producers measure 3-ethoxyaniline purity using gas chromatography, aiming for a minimum of 98%. Labels spell out storage needs— dark, cool environments with tight lids to prevent oxidation. GHS-compliant labels warn about toxicity, with pictograms for health hazards, and instructions for safe handling. Material Safety Data Sheets help researchers understand incompatibilities with strong oxidizers and acids. For regulatory traceability, bottles carry batch numbers, manufacturing and expiry dates, as well as the recognized CAS number (536-90-3). Companies packing this chemical must meet shipping requirements for aromatic amines, observing restrictions for certain modes of transit.
Preparation Method
Labs and factories favor the Williamson ether synthesis to make 3-ethoxyaniline, starting with 3-nitrophenol as the substrate. The process involves converting 3-nitrophenol to 3-ethoxynitrobenzene using bromoethane in the presence of a base, followed by reduction of the nitro group to an amino group with iron filings and hydrochloric acid or catalytic hydrogenation. This classic sequence continues to serve as a case study in undergraduate labs, illustrating both aromatic substitution and selective reduction in a straightforward workflow. Some academic groups explore greener modifications, replacing traditional solvents and reagents with more sustainable alternatives, but the tried-and-true route remains dominant in commercial settings.
Chemical Reactions & Modifications
The ethoxy and amino groups introduce interesting patterns in reactivity. The amino group provides a nucleophilic point for diazotization, acylation, or coupling with electrophiles, enabling the formation of dyes or pharmaceuticals. Electrophilic aromatic substitution targets often land at the para position relative to the amino group, unless steric or electronic effects intervene. In custom syntheses, 3-ethoxyaniline undergoes N-alkylation or protection-deprotection steps, or serves as a substrate for Suzuki and Heck cross-coupling, thanks to the activating effect of the ethoxy group. These modifications expand the molecule’s versatility, fueling innovation in medicinal chemistry and materials science.
Synonyms & Product Names
3-Ethoxyaniline appears in catalogs under different names, including m-ethoxyaniline, 1-ethoxy-3-aminobenzene, and 3-aminophenetole. Some literature mixes historical and commercial terms, highlighting the importance of cross-checking synonyms in chemical databases. For shipping and regulatory purposes, suppliers rely on CAS numbers to avoid confusion, especially in global markets where naming conventions sometimes diverge. No matter the name, the core structure remains constant, but clarity in communication helps prevent costly errors in research and manufacturing.
Safety & Operational Standards
Handling 3-ethoxyaniline requires respect for both its health hazards and reactive potential. Exposure can irritate the skin, eyes, and respiratory tract. Gloves, goggles, lab coats, and fume hoods become standard practice for anyone working with this aromatic amine. Real stories from industrial settings remind us that inhalation or skin absorption incidents disrupt work and bring legal consequences, prompting facilities to retrain teams regularly. Spills call for immediate action: absorbents, proper labeling, and safe disposal. Waste disposal follows local and national regulations, usually mandating collection in tight-sealed containers for incineration at licensed plants. Chemical hygiene policies, reinforced by periodic audits, give labs a culture of vigilance that protects employees and the wider community.
Application Area
The practical uses of 3-ethoxyaniline stem from its dual functional groups and stable aromatic core. In pharmaceuticals, it serves as a key intermediate for synthesizing painkillers, antihistamines, and other active ingredients. Agrochemical labs use derivatives as fungicide and herbicide scaffolds, taking advantage of the molecule’s modifiable sites. Dye manufacturers exploit its coupling abilities, producing colors that become embedded in fabrics and plastics. Polymer chemistry also relies on such intermediates to modify backbone structures and introduce new features. Research in specialty coatings explores 3-ethoxyaniline’s potential for imparting corrosion resistance or adhesion. Each sector brings its own challenges for purity, reactivity, and scale, illustrating the compound’s flexibility in the chemical world.
Research & Development
In the world of academic and industrial R&D, 3-ethoxyaniline provides a launching pad for the exploration of new molecular architectures. Graduate students design synthetic routes using it as a nodal point, seeking out routes to bioactive compounds or advanced materials. Journals share breakthroughs in modifying the ring system to optimize drug potency or environmental performance. Instrumentation advances, such as high-throughput screening and advanced NMR, speed up discoveries. Innovation often occurs where multidisciplinary teams work — chemists, engineers, and computational scientists collaborate to map reaction pathways and predict outcomes. Companies push for green chemistry approaches, aiming for higher yield, reduced waste, and renewable reagents. These projects bring lasting value, shaping both the scientific literature and the products reaching consumers.
Toxicity Research
Toxicologists investigate the effects of 3-ethoxyaniline on cells, tissues, and living organisms, noting the compound’s irritant and potentially mutagenic properties. Animal studies reveal dose-dependent effects, supporting calls for careful exposure limits in manufacturing and lab settings. Chronic exposure carries risks for liver and kidney functions and may cause sensitization reactions. Analytical chemists focus on detection and monitoring strategies, using GC-MS and HPLC methods to quantify trace amounts in workplace air and process streams. These efforts help regulatory agencies set occupational exposure limits, and they drive companies to improve containment and protective measures. Continued publication and transparency in toxicity data arm the scientific community with evidence for safe handling guidelines.
Future Prospects
As industries move toward greener processes and stricter safety standards, 3-ethoxyaniline’s role will continue evolving. Researchers explore bio-based routes for its synthesis, leveraging renewable biomass to replace petroleum starting materials, aiming to reduce the chemical footprint of large-scale production. Advances in process engineering give hope for more efficient and selective reactions, which translates into cost savings and less waste. Biomedical research continues to surprise, as new derivatives based on 3-ethoxyaniline show promise against infectious diseases or as part of novel diagnostic tools. Sustainable practices, transparent safety records, and open collaboration between stakeholders set the stage for the next generation of innovations built around this surprisingly versatile aromatic amine.
What is 3-Ethoxyaniline used for?
Behind the Lab Doors: Where 3-Ethoxyaniline Shows Up
Walking through a chemical plant or research lab, you spot shelves crowded with bottles labeled in sharp marker. Among them, 3-ethoxyaniline pops up more often than most folks may guess. It isn’t a household name, but to anyone stepping into dye chemistry, pharmaceutical research, or specialized materials, it rings a bell.
3-Ethoxyaniline is an aromatic amine that serves as a starting block for making other stuff. In places focused on color, it's used to synthesize dyes and pigments. Synthetic dyes give color to just about every textile, and many of the deeper shades need an aromatic amine foundation to anchor them in fabric. Industry experts talk about persistent, vibrant dyes that don’t fade in the wash, and 3-ethoxyaniline stands in the recipe for quite a few of these.
Connecting the Dots in Pharmaceutical Development
The story does not stop at dye houses or textile labs. Chemical companies use 3-ethoxyaniline as an intermediate in pharmaceutical research. It acts as a scaffold to build therapeutic agents. Medicinal chemists rely on its structure while developing drugs, testing new molecules for everything from anti-inflammatory properties to cancer studies. The value in this compound rests on its flexible structure and ease of swapping chemical groups. A small change in its structure might shift a molecule from benign to life-saving.
Safety matters in pharmaceutical work, and 3-ethoxyaniline brings challenges. Reports show concerns about its toxicity, so careful handling stays standard—proper lab coats, good ventilation, regular toxin screening. Safety manuals often mention 3-ethoxyaniline alongside reminders to double-check gloves and fume hoods. Exposure can cause headaches or skin irritation. The message: don’t play fast and loose with safety, no matter how well you know your chemistry.
Material Science and Beyond
Polymer researchers also lean on 3-ethoxyaniline. They need compounds that can integrate easily and let them tweak the resulting polymer’s properties. Imagine engineers aiming for a specialty plastic with resistance to heat or specific reactivity for electronics. Compounds like 3-ethoxyaniline help trial and error—the kind that leads to breakthroughs in flexible electronics or specialized coatings for wires.
Environmental Impact: Considering the Endgame
No one in industry ignores the waste issue. Disposal and breakdown of chemicals like 3-ethoxyaniline trouble plant managers and regulators. Some byproducts linger in water if not caught and treated. Over the past decade, regulations have pushed for cleaner synthesis and responsible disposal. The push comes from science and from public demand. Environmentalists want clear records and proof that chemicals get neutralized, not dumped. Some startups experiment with greener synthesis, swapping hazardous reagents for safer ones or improving filters at the source.
Looking Forward: Smarter Chemistry, Tougher Regulations
As chems get smarter, companies try to get more out of every molecule. 3-ethoxyaniline will keep showing up in new research. To keep industry safe and eco-friendly, open data sharing, stricter regulatory oversight, and strong workplace safety culture all matter. The story of 3-ethoxyaniline isn’t about just one use—it’s about building dyes, drugs, and future materials, while making sure the next generation inherits clean water and safe jobs.
What is the chemical structure of 3-Ethoxyaniline?
Understanding 3-Ethoxyaniline
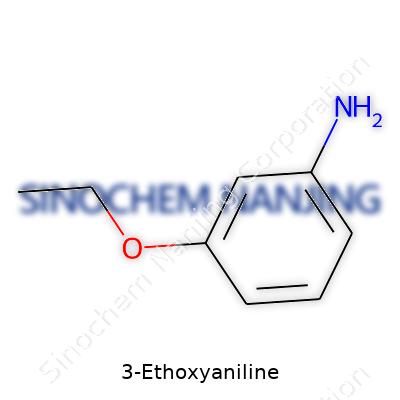
3-Ethoxyaniline draws attention among aromatic amines for its subtle shift in structure that opens up several practical uses and risks. Its backbone builds on an aniline core, carrying an amino group (–NH2) directly linked to a benzene ring. What sets it apart is the ethoxy group (–OCH2CH3) attached at the third carbon, counting from where the amino group sits. This layout shapes both its chemical behavior and a surprising amount of its role in industries or labs.
Looking at the Chemical Structure
To get a clear image, imagine numbering the carbons on the benzene ring starting with the carbon bonded to the amino group as position 1. At position 3, shift over to find the ethoxy group branching away. Each group alters electron flow in the ring, changing how the molecule reacts in synthesis and with other chemicals.
Benzene’s ring structure consists of six carbon atoms with delocalized π electrons, granting stability. An amino group at position 1 acts as an electron donor, raising activity for substitutions on the ring. The ethoxy group at position 3 influences reactivity even more, increasing the molecule’s potential as an intermediate in making dyes, pharmaceuticals, or specialized polymers. These additions shift physical properties, such as solubility in organic solvents and boiling point, compared to plain aniline.
Why the Structure Matters
3-Ethoxyaniline, through its layout, becomes a flexible tool for chemists. In the dye industry, the combination of electron-donating groups lets manufacturers adjust shade and stability for a range of applications. The amino group welcomes further reactions, like acylation or diazotization, to build colorful azo dyes. As a pharmaceutical intermediate, the ethoxy group’s presence controls how easily the molecule fits into synthesis pathways or interacts with biological targets, affecting both potency and safety.
There’s a lesson tucked in here on chemical safety. Anilines often ring alarm bells for toxicity. The presence of both the amino and ethoxy group in 3-ethoxyaniline doesn’t erase all danger. Studies note that exposure—even at low levels—can lead, over time, to health risks like methemoglobinemia. These risks are often overlooked in smaller labs or workshops, so workers should wear gloves and operate under good ventilation. Labels and safety data sheets detail risks, but in practice people take shortcuts. Some educators or chemists recall the sharp smell and lingering stains it leaves, a reminder to clean up quickly to avoid skin contact or breathing in vapors.
How to Improve Safety and Transparency
Safety takes more than regulations—it requires a mindset. Sharing lab experience helps new chemists respect these chemicals. Training sessions that include real stories about near-misses drive the point home, much more than dry rules or generic warnings. Companies and universities should keep teaching up to date, making sure new users understand how structure links to hazard. Greater access to transparent hazard data, in plain language, helps everyone on the floor work smarter and safer. Simple steps, such as using proper labeling and PPE, mean fewer health scares and more trust in workplaces big and small.
What are the safety precautions when handling 3-Ethoxyaniline?
Why Care Matters With Chemicals
Anyone who’s spent time in a chemistry or manufacturing lab understands that handling chemicals takes focus and respect. 3-Ethoxyaniline isn’t exactly a household name, but it’s not rare in research labs or certain production floors. I once saw a batch spill because someone got hurried while moving it from a flask. We learned the hard way that small mistakes can put health and safety on the ropes.
Potential Hazards
3-Ethoxyaniline brings a handful of risks to the table. Data suggests that exposure can irritate the skin, eyes, and respiratory tract. The liquid can seep through gloves not built for chemical duty. Vapors can trigger coughing, dizziness, or headaches. Breathing in enough of the fumes for a stretch could lead to longer-term problems—an issue far too many chemical techs only realize after the damage has started.
University research groups and chemical producers both classify this compound as harmful if swallowed or inhaled. The irritant label might sound mild, but anyone who’s felt their skin burn or watched a rash grow knows that even “mild” doesn’t mean harmless. There are reports of allergic reactions and, with repeated or heavy exposure, possible effects on blood or liver function.
Smart Handling = Fewer Problems
Making safety a habit starts long before opening a bottle. Gloves need to be chemical-resistant; standard lab gloves will not cut it. Think nitrile or neoprene, and always check the thickness rating. I once tried to save a few bucks using thin gloves, and the regret outlasted the skin irritation.
Lab coats or aprons shield your clothes and skin. Splash-proof goggles matter even if you think you’re just weighing solids or making a quick transfer. A bit of splatter happens faster than most realize, especially with volatile liquids. Good ventilation keeps the air clear. A fume hood isn’t just for looks—it actively pulls harmful vapors away from your face. Keeping respirators or masks at the ready gives extra peace of mind, especially on days bosses push ambitious workloads.
Spills and First Aid
Spill kits should not gather dust in a corner. Absorbent pads and neutralizing agents deserve a permanent spot in the lab. A spill on the floor increases slip and chemical contact risk. Grab the kit, contain the area, blot up the liquid, and clean everything with an approved solvent. Even small spills demand respect.
Accidents happen, even for careful workers. Wash any skin exposure right away with soap and plenty of running water. If 3-Ethoxyaniline gets in your eyes, rinse nonstop for fifteen minutes and get medical attention. Breathing in vapors may require a trip to fresh air or even emergency care in serious cases. Don’t just “walk it off.”
Better Training, Safer Results
Knowledge keeps labs safer. I believe every team should organize refresher courses every few months. Sharing close calls or mistakes fosters a culture where folks look out for each other. Signage, clear labeling, and updated safety data sheets stay vital, no matter how routine the job becomes.
In the end, chemicals like 3-Ethoxyaniline demand respect. Following tighter procedures and admitting mistakes make workplaces a lot safer. No bottle is worth risking your liver, eyesight, or lungs.
What are the physical properties of 3-Ethoxyaniline?
The Substance at a Glance
3-Ethoxyaniline doesn’t pop up in everyday conversation unless you spend time in a lab, but it deserves more attention for its distinct characteristics. With a chemical formula of C8H11NO, this compound lands in the group of anilines, the oily, benzene-based group best known for their role in dye and pharmaceutical production. At room temperature, 3-Ethoxyaniline often appears as a light yellow to brownish liquid, a clear sign that it's a stable organic molecule, not some mysterious crystal or powder hiding in a dusty jar.
Boiling and Melting Points Matter
This liquid has a boiling point in the ballpark of 241-243°C. Reaching that temperature without modern equipment is unlikely, locking this compound safely in the liquid state under most conditions. Its melting point comes in at around 1-3°C, which puts it just above the freezing mark for water. That small span between liquid and solid makes storage pretty straightforward in any basic facility. You’re not going to find this material turning solid in anything except a cold room or the fridge, which gives it an advantage for those handling and shipping it in various environments.
Solubility and Consistency
3-Ethoxyaniline shows off a trait seen in other anilines—it doesn’t mix too well with water. Instead, it prefers organic solvents like ether, alcohol, or acetone. That tendency reveals a lot about the molecular structure. The ethoxy group provides some flexibility, but the benzene ring keeps it largely non-polar, nudging the molecule away from water and more toward other solvents. Anyone blending or using this material in synthesis can count on its reliability; it won’t suddenly decide to dissolve while working with aqueous solutions. This trait can be important for creating selective reactions or preparing intermediates where solvent choice decides success or failure.
Density and Appearance
The density hovers around 1.03 g/cm³, which puts it just a slight notch above water. This could fool folks not used to handling it, as it doesn’t float or sink drastically. In practice, you notice its earthy smell pretty quickly—a common feature among related compounds. It’s not overpowering, but it lingers, hinting at the aromatic amine foundation.
Stability and Reactivity
By design, 3-Ethoxyaniline doesn’t break down under ordinary light or air exposure. It can start to oxidize if left open for long periods. That process darkens the material and can reduce its usefulness in finer applications. Using airtight, amber glass bottles and keeping containers away from strong sources of heat helps. People who work with similar chemicals know that extra care during storage pays off, both for safety and longevity.
Handling Practicalities and Safer Practices
Health and safety matter when working with aromatic amines. While 3-Ethoxyaniline isn’t among the most hazardous of its group, it does warrant gloves and goggles, especially when transferring or mixing. The chemical can cause irritation on contact with skin and mucous membranes. Good ventilation and immediate cleanup for drips or spills save a lot of headaches later. As someone who’s handled plenty of similar substances, it’s best not to get complacent—fume hoods and proper disposal processes make lab life much smoother.
Thoughts on Opportunities for Safer Use
Better packaging, clear labeling, and updated safety sheets support anyone involved in processing or logistics. Revisiting training every so often helps remind everyone of the basics. Regulations already nudge facilities in the right direction, but there’s always room to keep improving. Sharing what works—and what doesn’t—across teams and industries goes further than top-down mandates ever will.
How should 3-Ethoxyaniline be stored?
Why Storage Matters in Real Labs
3-Ethoxyaniline isn’t a chemical you leave lying around without a plan. I’ve worked in labs where someone once left just a small bottle of this stuff next to strong sunlight by mistake—by Monday morning, the cap had swelled and the air stank. Those moments teach you quick lessons on why safe storage isn’t just a detail, it’s a safeguard for both people and experiments.
What 3-Ethoxyaniline Can Do, and Why It Needs Care
This compound plays a part in dye production, organic synthesis and research. It looks harmless enough—a clear to slightly yellow liquid—but labels only tell half the story. Its vapors irritate the eyes, nose and throat, and skin contact can cause itching or rashes. Even a splash on the lab bench can linger if it’s not cleaned up fast. From experience, letting little things slide in chemical storage invites real trouble later, not just lost material but plenty of headaches when it comes to cleanup and safety reviews.
Storing It Right: Real World Tactics
You won’t find many old-timers stacking bottles of 3-Ethoxyaniline next to acetone. Organic amines like this tend to oxidize in air and react if stored near acids or oxidizing agents. I always use strong, tight-sealed glass bottles—HDPE works too—marked clearly with hazard labels. Curtains never go near my chemical shelving. Heat speeds up breakdown, making the compound unstable and even more pungent. Keep storage spaces cool and shaded. Rooms that stay between 2°C and 8°C offer the best balance—too warm, and you risk off-gassing, too chilly and the bottle could crack or leak.
Organization isn’t just about being tidy. Mixing incompatible chemicals leads to mix-ups at best, clouded bottles or even minor fires at worst. Separate shelves or cabinets for organics and acids keep mistakes from happening. In crowded college labs, I’ve seen well-meant shortcuts lead to bad spills that ruined an entire week’s research.
How to Handle Spills—And Why They Happen
Any time a bottle leaks, you deal with it straight away, not after lunch. Open windows or use fume hoods. Absorb spills with sand or vermiculite, not paper towels. Small details matter: plastic gloves crack with repeated exposure, so double up if you know you’ll be handling the stuff for a while. My hands have tingled long after ignoring that rule.
One overlooked trick: check expiration and batch numbers every few months. Old bottles turn more yellow and are harder to pour, and if a bottle’s been open and closed dozens of times, that seal wears out. Preventative care sometimes gets skipped in busy labs, but records and regular checks keep stock fresh and safe.
Thinking About People—Not Just Bottles
All these storage tips go beyond protecting glassware. They shield anyone who works the benches, runs the waste, or even just walks through the storage room. I’ve seen businesses fined not because of some huge spill, but because their storage areas failed basic checks. Practicing solid chemical storage should be second nature in labs.
Every bottle of 3-Ethoxyaniline becomes a reminder: the right storage keeps labs safer, research smoother, and everyone breathing a little easier.
| Names | |
| Preferred IUPAC name | 3-ethoxyaniline |
| Other names |
m-Anisidine 3-Aminophenetole 3-Ethoxybenzenamine |
| Pronunciation | /ˈiː.ˌθɒk.si.əˈnɪl.iːn/ |
| Identifiers | |
| CAS Number | 536-90-3 |
| Beilstein Reference | 1208753 |
| ChEBI | CHEBI:76136 |
| ChEMBL | CHEMBL16214 |
| ChemSpider | 75287 |
| DrugBank | DB08264 |
| ECHA InfoCard | ECHA InfoCard: 100.005.495 |
| EC Number | 202-361-1 |
| Gmelin Reference | 81855 |
| KEGG | C01781 |
| MeSH | D015759 |
| PubChem CID | 74020 |
| RTECS number | BY8575000 |
| UNII | 39X9A3F57G |
| UN number | UN1992 |
| Properties | |
| Chemical formula | C8H11NO |
| Molar mass | 137.18 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | amine-like |
| Density | 1.022 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 1.46 |
| Vapor pressure | 0.0965 mmHg (25°C) |
| Acidity (pKa) | 4.90 |
| Basicity (pKb) | 10.04 |
| Magnetic susceptibility (χ) | -54.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.561 |
| Viscosity | 1.09 mPa·s (at 25 °C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 213.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -74.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3158 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause allergic skin reaction, toxic to aquatic life. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H319 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P363, P403+P233, P405, P501 |
| Flash point | 80 °C |
| Autoignition temperature | 385°C |
| Explosive limits | Explosive limits: 1.2–7.0% |
| Lethal dose or concentration | LD50 (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2420 mg/kg (rat, oral) |
| NIOSH | SN8575000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5-10% |
| Related compounds | |
| Related compounds |
Aniline 2-Ethoxyaniline 4-Ethoxyaniline 3-Methoxyaniline 3-Ethylaniline |

