Editorial Commentary: The Changing Landscape of 3-Chloropropene
Historical Development
Most chemical stories start in the lab, driven by pure curiosity or practical need. 3-Chloropropene, sometimes called allyl chloride, traces a path through chemistry that's neither obscure nor trivial. Decades back, researchers noticed its ability to serve as a reactive building block. Early formulations, produced through chlorination of propylene, showcased how industry can transform something as basic as propylene gas into a compound with a far reach into the synthetic world. Historically, production rode the wave of demand for epichlorohydrin and specialty polymers, and by the middle of the 20th century, plants in the US and Europe started churning out metric tons, pushing safety officers and researchers to document effects, risks, and applications. This chemical didn’t pop up overnight—it earned its standing through consistent utility, market interest, and a long journey from small-batch synthesis to global commodity.
Product Overview
3-Chloropropene often gets credit for its versatility in organic synthesis. Chemists see a three-carbon chain with a chloride at one end and an unsaturated double bond at the other—a combination that tempts experimentation. The plant manager might check storage tanks holding a clear, flammable liquid, while a formulator looks at it as a stepping stone to resins, water treatment solutions, or pharmaceuticals. This isn't an obscure laboratory chemical gathering dust; it's an active ingredient in many industrial processes, valued for the way it opens molecular doors. Buying this means purchasing a set of possibilities, but every drum also comes packed with responsibility.
Physical & Chemical Properties
Any technician who opens a shipment notices the sharp odor and volatility. 3-Chloropropene boils just above room temperature, and it evaporates fast enough to demand good ventilation. Orange flames flare up easily if vapors catch a spark. As a liquid, it weighs more than water and spreads out thin when spilled. That double bond in the middle of the molecule creates reactivity, making it more than just a chlorinated solvent. It dissolves in many organic solvents but not in water, and it’s dense enough to sink. For most uses, this dual personality—part allyl, part halide—drives both value and risk.
Technical Specifications & Labeling
Shipping drums or containers must carry labels that admit no mistakes. Fire hazard, skin absorption risk, inhalation danger: these warnings mark genuine hazards, not regulatory excess. Standards require purity above certain thresholds, usually verified by gas chromatography. Impurities change the way it behaves in downstream chemistry. Technicians don’t just trust a product sheet; they pull samples and test before every big run. Each shipment carries identification, and operators check every drum for leaks since the vapor can build up quickly. Clear labeling, clear records, clear training—these are basics for anyone trying to keep workers, equipment, and communities safe.
Preparation Method
Chlorination of propylene transforms what’s basically a feed gas into 3-Chloropropene. In practice, that means bubbling chlorine through propylene in the presence of light or a catalyst, keeping temperatures under precise control. Poorly controlled reactions lead to a messy mix of byproducts, so operators pay close attention. Chemists sometimes investigate alternative routes—say, via allyl alcohol transformations—but the propylene route dominates commercial production. These processes scale to hundreds of tons, and engineers tweak parameters to squeeze out as much yield as possible without sacrificing purity. All along, safety remains top-of-mind, since a mix-up can mean hazardous releases or worse.
Chemical Reactions & Modifications
With that reactive double bond, 3-Chloropropene serves as a jumping-off point for a world of reactions. Manufacturers convert it into epichlorohydrin through reaction with hypochlorous acid and subsequent dehydrochlorination. This route feeds strong demand for epoxy resins. Synthetic chemists appreciate how the allylic chloride group allows nucleophilic substitution, creating a modular scaffold for pharmaceuticals or specialty polymers. Each modification, though, comes with its own safety challenges, since both the starting material and many of its derivatives retain considerable toxicity or flammability. These properties demand handling protocols, personal protective equipment, and careful monitoring throughout every stage.
Synonyms & Product Names
In chemical catalogs or safety data sheets, 3-Chloropropene might appear as allyl chloride, 1-chloropropene-3, or occasionally as 3-chloropropylene. Old-timers still use “allyl chloride” out of habit, reflecting the molecule’s most prominent use. No matter what label sits on a drum, it’s the same reactive, pungent three-carbon liquid, and anyone familiar with its hazards knows to keep a respectful distance—regardless of the name on the invoice. Synonyms tell a story of how one chemical travels through multiple industries, acquiring reputations, associations, and regulations as it goes.
Safety & Operational Standards
Anyone working around 3-Chloropropene keeps safety up front. Air monitoring equipment stays in high-risk areas, with ventilation systems running year-round. Regulations demand training for all workers, emphasizing how quickly inhalation or skin contact leads to acute symptoms—eye irritation, coughing, burning sensations. Fire departments urge storage in explosion-proof environments, and transfer systems include redundancy in case of leaks or spills. Operating guidelines built from decades of experience, and tough lessons remind everyone what can go wrong. Plant tours show elaborate containment—spill berms, dikes, and chemical-resistant gear aren’t accessories, they’re the norm. Managers treat every close call as a reason to revise rules and retrain teams, because complacency breeds accidents.
Application Area
Most of the world’s 3-Chloropropene converts into epichlorohydrin for epoxy resin production. These resins, in turn, show up in wind turbine blades, boat hulls, adhesives, circuit boards, or industrial coatings. That’s only the biggest slice. Down in specialty labs, chemists use it to create pesticides, pharmaceuticals, and water treatment compounds. Plastics researchers depend on it to introduce reactive sites into polymers. The molecule’s structure invites modification, allowing innovation in industries from medicine to energy storage. Even so, each new use runs up against a wall of safety and environmental scrutiny. Consumer demands, worker advocacy, and tight regulations all play a role in shaping what’s possible and what isn’t.
Research & Development
Investigators in research labs target new syntheses for minimizing byproducts or cutting energy usage. Some push alternative, greener methods—perhaps by leveraging biocatalysts or safer chlorine substitutes. Other teams study ways to capture and convert waste streams, aiming to reclaim and reuse every drop to reduce both cost and emissions. Molecular biologists investigate the mechanisms by which 3-Chloropropene causes harm in cells, producing data that informs both policy and protective gear design. Collaboration across departments and institutions brings cross-pollination of ideas, with shared publications driving improvement on both the scientific and operational fronts. Progress doesn’t emerge overnight, but the steady pace of inquiry builds real value over time.
Toxicity Research
Hard experience and detailed studies show 3-Chloropropene brings acute and chronic risks. Animal testing and incident records indicate it acts as an irritant at low levels, and at high exposures it affects the nervous system and possibly the liver. Chronic exposure correlates with elevated cancer rates in laboratory animals. Most chemical workers know stories about exposure symptoms—headaches, cough, even nosebleeds—long before they see the published studies. Regulatory bodies keep lowering acceptable exposure limits, prompting industries to invest in better containment, faster leak detection, and more robust personal protection. Researchers continue to look for biomarkers of exposure, aiming to catch effects before symptoms escalate. Public awareness keeps researchers and regulators on their toes; this is not a chemical that anyone can afford to take lightly.
Future Prospects
Demand for 3-Chloropropene continues to ride trends in plastics, wind energy, and electronics. As society leans into renewable resources, applications in composite materials and specialty polymers may widen, but only if production and handling keep pace with expectations for safety and environmental stewardship. Ongoing research into green chemistry could make this old standby less hazardous—at least in the way it’s produced and processed. There’s also pressure to recycle byproducts, limit emissions, and develop safer alternatives for workers and consumers. Regulators in the EU and North America watch advances closely, moving to restrict or control uses as new findings emerge. Companies able to blend innovation with responsibility will be best placed to keep 3-Chloropropene relevant. The path forward lies in smarter processes, safer plants, and honest transparency with the public.
What is 3-Chloropropene used for?
What’s 3-Chloropropene?
3-Chloropropene, which a lot of folks know as allyl chloride, stands tall in the world of industrial chemicals. If you ever walked through a big chemical plant, you’d probably catch a whiff of it somewhere. This colorless liquid might look plain, but it’s got plenty of uses packed into every barrel. Back in college, I had my nose in its chemistry, and I saw just how much impact a single compound can have across industries.
Industry Relies On 3-Chloropropene
Talking with friends who work in manufacturing, I started to see the full picture. 3-Chloropropene plays a big role in making plastics, especially epichlorohydrin. Epichlorohydrin matters because it’s the backbone of epoxy resins. These resins end up everywhere — boats, car parts, flooring, and even aircraft. Epoxy’s legendary stickiness and toughness come right from reactions that spin out of basic chemicals like allyl chloride.
Moving over to the agrichemical world, it’s pretty clear farmers wouldn’t have the same crop protection tools without 3-chloropropene. A chunk of well-known pesticides start their lives with it. In fact, researchers searching for new, less risky crop treatments still rely on its chemistry. Some of these pesticides help protect staples like wheat and rice, which puts food on millions of tables. That puts a real-world value on what might look like a lowly chemical formula.
Everyday Items Trace Back to This Molecule
If you’ve grabbed a plastic bottle or a length of vinyl siding, you’re more connected to 3-chloropropene than you think. It acts as an ingredient for making allyl alcohol and allyl isothiocyanate. That flavor of horseradish and mustard? You can trace some of that chemically-crafted kick all the way back to allyl chloride. In water treatment plants, this compound finds its way into making certain flocculants—agents that clump together dirt and make clean water flow from our taps.
The Downsides Can’t Be Ignored
I can’t skip over the health side. From my own research and experience with chemical safety, I know how important it is to use strong protection. Inhaling vapors or getting 3-chloropropene on your skin can cause burns or much worse. Science backs this up: several safety boards and medical journals highlight the risks tied to exposure, including a potential to cause cancer in animals. Proper ventilation, clothing, and handling are taught in labs for a reason.
Environmental rules keep a close eye on its use. Processing plants have to follow careful steps to keep spills low and air clean. Communities living near chemical sites often raise fair questions about safety, which pushes producers to invest in better containment and monitoring.
Room for Safer Alternatives
Some researchers look to alternatives every year, hoping to find a chemical that works the same with less risk. A few companies have already started leaning on greener methods—using less hazardous feedstocks when available. Regulations continue tightening, prodding businesses to keep safety and health in focus. As industries march forward, they carry the responsibility to balance the huge value of 3-chloropropene with the need for health and safer air, water, and soil.
One thing always stands out: the products around us hide surprisingly complex stories. Next time you spot a shiny new epoxy or munch some spicy mustard, think about the wild chemical journey that started with a little liquid called 3-chloropropene.
What are the safety precautions for handling 3-Chloropropene?
Understanding the Real Risks
3-Chloropropene, sometimes called allyl chloride, doesn’t get talked about much outside of labs and chemical plants. For those who handle it, though, its risks are serious. Breathing its fumes can bring on headaches, dizziness, or much worse — some people have landed in the hospital after heavy exposure. Touching it without protection burns skin almost before you know it. Even one splash in your eye can mean lasting damage. I’ve seen folks drop their guard with substances they thought they understood, only to find out the hard way that chemicals like this are full of nasty surprises.
Personal Protective Equipment: Non-Negotiable
Comfort plays no role in choosing gear around 3-chloropropene. Proper gloves — butyl or viton over latex or nitrile — keep your skin out of harm’s way longer than standard materials. Eye protection should cover the sides, not just the front. Face shields offer a smart second line when there’s splash risk. For the lungs, a tight-fitting respirator rated for organic vapors does the job. I’ve noticed new hires sometimes roll their eyes at all this, but old-timers know the value of good gear. No shortcut here has ever improved anyone’s workday.
Ventilation and Spills
Fresh air moves hidden fumes out fast. Closed rooms or poor airflow let dangerous concentrations form quickly, sometimes before you realize anything’s wrong. Open windows often can’t cut it; mechanical fume hoods or localized extraction do what open doors can’t. Adults in the room catch the sharp smell faster than kids would, and symptoms might go unnoticed until it’s too late. So, check those fans and repair broken hoods instead of “making do.”
Spills demand respect. No one wants to trigger an evacuation, but ignoring a puddle on the bench invites trouble. I once saw a colleague ruin his day by spreading a tiny spill with his sleeve, carrying the chemical where it didn’t belong. Sand, absorbents labeled for chemicals, and sealed containers fit for hazardous waste make cleanup safe. Tossing paper towels into the regular garbage just spreads the risk.
Storage and Handling: Care Every Step
Storing 3-chloropropene safely avoids a lot of grief. Tightly sealed containers block both evaporation and accidental spills. I’ve seen bottles stored next to acids or bases, which spells out real trouble in bigger facilities. Tanks and bottles should sit in cool, well-ventilated spots, far from direct sun or heat. Permanent labels help keep everyone honest; faded ink and mystery containers never improved safety. Double-checking before pouring or mixing often stops mistakes before they grow. For some, it feels like overkill, but a few seconds pausing now gives far better odds later on.
Training and Emergency Plans
Regular, hands-on drills give better results than just reading a manual once. Knowing where the emergency eyewash is — and actually pulling the lever in practice — beats theory every time. Response doesn’t come from memorizing rules; muscle memory and clear instructions save seconds, not policies on the wall. If a team tests their plan a few times a year, slip-ups get caught before someone gets hurt. I’ve worked with groups who never trained, and the confusion during a real scare was painful to watch. Experience trumps paperwork every time.
Responsible Practices Make a Difference
Working around 3-chloropropene rewards those who take it seriously. A few key habits — good gear, decent ventilation, smart storage, real training — cut harm down to almost nothing. I’ve learned from both mistakes and careful teachers: the safest labs and shops aren’t lucky, just thorough.
What is the chemical formula of 3-Chloropropene?
Getting to Know 3-Chloropropene
On paper, chemistry can seem like a world of numbers and odd-sounding names. 3-Chloropropene, though, has a straightforward side and a clear recipe for how its structure comes together. Its chemical formula is C3H5Cl. This string of letters and numbers tells quite a bit about what the molecule brings to the table and the way it interacts with the world of industry, research, and even the environment.
What the Formula Says
C3H5Cl means we’re dealing with three carbon atoms, five hydrogen atoms, and one chlorine atom. Picture the carbon atoms forming a short, three-piece chain. Attach a double bond between the first and second carbon atoms, and you get the “propene” backbone. Put a chlorine atom at the far end of the chain, and you’re looking at 3-chloropropene. That single chlorine switch changes the way this molecule behaves, giving it uses beyond what plain old propene can do.
Why It Matters in the Real World
For many, listening to chemical talk just seems far removed from daily life, but this molecule has an impact. 3-Chloropropene finds use in the world of industrial chemistry, cropping up in the production of other chemicals like allyl chloride, which leads to making plastics, pharmaceuticals, and pesticides. Factories don’t whip up these batches without planning; they weigh the risks and handle this compound with respect because the chlorine atom in the formula makes 3-chloropropene toxic if mishandled. Exposure can irritate the eyes and lungs, and long-term contact means health officials keep it on their watch lists.
The Human Side of Chemical Safety
This isn’t just trivia for chemistry majors or people in lab coats. I’ve spent time in industrial settings, and safety rules always come down to basic knowledge; it’s about knowing what's in the drum or the pipe. A clear formula gives that information in shorthand, keeping everyone safer. If there’s a leak or a spill, you need to know exactly what you’re dealing with at a glance. Chlorinated compounds, including 3-chloropropene, need special containment and ventilation because of the health and environmental risks if released.
Taking Responsibility with Chemicals
Over the years, I've seen regulations grow stricter. Groups such as OSHA and the EPA draw clear lines around storage, transport, and disposal of chemicals like 3-chloropropene. Modern equipment offers remote detection sensors and better airflow management. The benefit goes beyond compliance; it protects workers and the communities living near chemical plants. Continued research points to greener alternatives and safer processes, but that doesn’t mean we can let our guard down. The chemical formula isn’t just numbers—it's a warning and a tool.
Looking Ahead
New technology holds promise for safer substitutes and more efficient reactions. Until that day arrives, people in the field continue to focus on health and environmental safeguards, using both experience and science. As someone who values practical knowledge, seeing information like C3H5Cl on a safety sheet doesn't just label a substance—it spells out a responsibility shared by everyone who ever steps foot near a drum or works in product development.
How should 3-Chloropropene be stored?
Why 3-Chloropropene Matters to Handle Carefully
3-Chloropropene, often called allyl chloride, doesn’t mess around. In the chemical industry, its sharp odor and high volatility mean risk, not just in theory but right in the warehouse and lab. I remember working in a plastics facility during my grad school years, and our safety officer eyed any bottle of this stuff like it could explode on a whim—which, given enough heat or a loose cap, wasn’t so far-fetched.
This chemical catches fire easily. The vapors spread fast, hugging the floor since it's heavier than air, and any spark could set off a nasty scene. A report by the World Health Organization pointed out serious health issues linked to inhaling or touching this liquid, from dizziness to damage to the liver and kidneys. I’ve seen emergency drills turn into real headaches just because one small leak got ignored.
Practical Storage Steps
Storing 3-Chloropropene starts with location. Keep it in a cool, dry spot—think well-ventilated chemical storage rooms or dedicated outdoor lockers far from ignition sources or sunlight. Even standard warehouse lighting can give off heat; every small detail counts. Workers often prop open doors without thinking, but with this stuff, that's asking for trouble if a spill happens. Proper ventilation stops dangerous vapors from concentrating and drifting into occupied areas.
Sturdy containers make a big difference. Stick to tightly sealed metal drums or glass bottles with chemical-resistant seals—polyethylene and polypropylene can break down over time. I’ve seen loose-fitting lids lead to sticky situations; vapors escape, smells linger, and suddenly everyone’s got a headache. Mark each container clearly, using bold labels and the right hazard warnings, so nobody grabs something they shouldn't.
Inventory controls don’t just mean keeping track of how much you have. They also mean regular checks for leaks, corrosion, or swelling containers—especially after hot days or rough transport. Regulatory bodies like OSHA recommend secondary containment, such as spill trays or bund walls. These aren’t just rules for the sake of rules. They stop small accidents from becoming major disasters. In my first industry job, we caught a small leak simply because secondary containment caught a strange puddle before anyone got hurt or equipment was ruined.
Protecting People and Places
Access limits cut down on risk. Only trained staff should handle the chemical, using gloves and face shields—not just the disposable ones from your local hardware store but specialty gloves rated for solvents. Emergency eyewash stations and showers must sit close to storage zones. A co-worker once told me those features seemed like overkill until he needed an eyewash station after a splash. After that, he was the biggest champion for safety gear on site.
Fire suppression gear stands ready at any serious facility. Because water doesn’t put out allyl chloride fires—in fact, it can spread the flammable liquid—ABC or CO2 fire extinguishers should always be close at hand. Check that extinguishers stay within arm’s reach and get serviced regularly. More than once, I’ve heard stories of safety teams pulling out expired equipment in a crisis, when it’s already too late.
Better Solutions Start with Accountability
Real safety goes beyond checklists. Training keeps everyone sharp, and drills—actual hands-on practice—give workers the skills they need to handle a spill or fire calmly. Before every shift, reviewing emergency info and knowing exactly where exits and safety showers are can make the difference between a scare and a tragedy. Companies can also work with local emergency responders to create a game plan for worst-case scenarios, not just hope everyone’s careful.
What are the physical properties of 3-Chloropropene?
The Tangible Face of 3-Chloropropene
I’ve spent a fair share of time around volatile chemicals, and there’s something memorable about working with 3-chloropropene, also called allyl chloride. This liquid catches attention right away with its sharp, pungent smell—impossible to miss, especially if you’ve ever handled it in a barely ventilated lab. Unlike some industrial liquids that prance around the lab at a medium pace, this one likes to dash: it evaporates at room temperature with a boiling point of about 44°C, which means leaving it uncapped for a few minutes grows expensive, and risky, extremely quickly.
It pours slick and almost colorless, a little like old-school gasoline. Anyone stuck cleaning up a spill learns its density clocks in just above water, somewhere around 0.94 g/cm³. This looks subtle, but makes itself understood when you’re scraping it off a benchtop, where it doesn’t sit stubborn and heavy like denser organics. Still, it will roll right off a hydrophobic surface—a pain to track and corral if you’re in a hurry.
The Wildcard of Volatility
The biggest headache with 3-chloropropene is how easily it takes to the air. Vapor pressure rockets up to about 600 mmHg at room temperature. The stuff moves fast, finds cracks in containment, and likes to remind you to double-check your respirator fit. In practice, even seasoned technicians know to work it in a fume hood and to never let a bottle linger open. One study I read placed its flash point at −28°C, a pretty corrosive reminder that it can ignite at colder temperatures than water freezes. Lightning quick, but not something you want in the wrong place.
Solubility in Practice
Getting 3-chloropropene to mix with water proves a fool’s errand. It barely dissolves at all—less than 3 grams in a liter. That stubbornness shows up most painfully during environmental spills; it hangs out from water supplies instead of dispersing quickly, making cleanups drawn out and complicated. Toss it in with ethanol, ether, or acetone, though, and you barely need to stir. This makes it popular as a solvent and intermediate. The downside: it’ll slip through plastic and rubber, chewing them up and forcing you to reach for glass or specific chemical-resistant polymers.
Reason for Caution—and a Path Forward
You don’t spend years around chemicals without developing respect for the feistier ones. 3-chloropropene demands attention not just because of how easy it is to mishandle, but because inhaling it causes eye-watering, dizziness, and worse with long exposure. The CDC and OSHA both highlight the risk of chronic effects, mentioning nerve and liver damage. Proper storage means cool, dry places, steel or glass containers, and enough ventilation to keep people from taking it home in their lungs.
Nothing replaces safe handling training. Every chemist I know who’s worked with 3-chloropropene has a story about a close call—a whiff that lingered too long, a splash near the glove cuff. Labeling, routine checks, and never getting complacent keep everyone healthy. While hazard mitigation sounds like a chore, it boils down to respecting the stuff and using tech—like automated handling or air sensors—where things get overwhelming. The takeaway: 3-chloropropene’s unique set of physical characteristics serve as both its calling card and its challenge, one best faced with preparation and care.
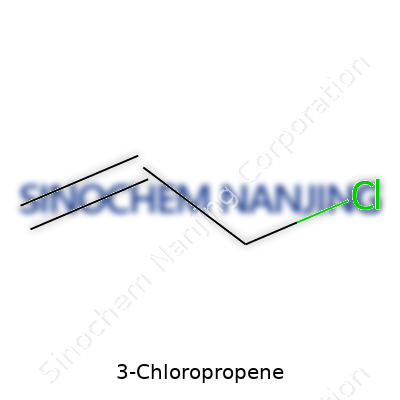
| Names | |
| Preferred IUPAC name | 3-chloroprop-1-ene |
| Other names |
Allyl chloride 1-Chloro-2-propene 3-Chloropropylene |
| Pronunciation | /ˌθriːˌklɔːrəˈprəʊpiːn/ |
| Identifiers | |
| CAS Number | 107-05-1 |
| 3D model (JSmol) | `JSmol('C=CCCl')` |
| Beilstein Reference | 1718735 |
| ChEBI | CHEBI:36937 |
| ChEMBL | CHEMBL149735 |
| ChemSpider | 8674 |
| DrugBank | DB14096 |
| ECHA InfoCard | DTXSID2021422 |
| EC Number | 204-070-3 |
| Gmelin Reference | 1163 |
| KEGG | C01457 |
| MeSH | D002789 |
| PubChem CID | 7914 |
| RTECS number | RR3325000 |
| UNII | 18TO4T3CI7 |
| UN number | UN1113 |
| Properties | |
| Chemical formula | C3H5Cl |
| Molar mass | 76.54 g/mol |
| Appearance | Colorless liquid with a pungent odor |
| Odor | Sharp, suffocating odor |
| Density | 0.857 g/mL at 25 °C |
| Solubility in water | Soluble in water |
| log P | 1.99 |
| Vapor pressure | 178 mmHg (20°C) |
| Acidity (pKa) | 15.7 |
| Basicity (pKb) | 11.99 |
| Magnetic susceptibility (χ) | -0.79×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.412 |
| Viscosity | 0.34 mPa·s (20 °C) |
| Dipole moment | 1.93 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 296.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -58.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −2040 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H315, H319, H331, H335, H341, H350 |
| Precautionary statements | P201, P210, P261, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2 |
| Flash point | -35 °C |
| Autoignition temperature | 430 °C |
| Explosive limits | 2.8–14% |
| Lethal dose or concentration | LD50 oral rat 90 mg/kg |
| LD50 (median dose) | 280 mg/kg (rat, oral) |
| NIOSH | SK8245000 |
| PEL (Permissible) | 1 ppm (3 mg/m3) |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 40 ppm |
| Related compounds | |
| Related compounds |
Allyl chloride Propene 1-Chloropropane 3-Chloropropanol Epichlorohydrin |

