The Ongoing Story of 3-Chlorobenzotrifluoride: Science, Industry, and Safer Practices
A Look Back: The Historical Journey
Years ago, chemical innovation opened new doors in industrial and pharmaceutical processes. 3-Chlorobenzotrifluoride arrived on the scene during a period of rapid expansion in organic chemistry, catching the eyes of researchers for its unique trifluoromethyl group, which set it apart from more conventional chlorinated aromatics. Laboratories in Europe and North America experimented extensively, noticing its value as a stable yet highly modifiable building block. As environmental scrutiny started rising over persistent pollutants, this compound became more noticeable in technical circles, given its role in synthesizing less persistent options compared to polychlorinated biphenyls and related unwanted byproducts. It never quite reached household-name status, but industry veterans see it as a quiet workhorse in manufacturing routes where adaptability counts.
What Sets 3-Chlorobenzotrifluoride Apart
In my experience, 3-Chlorobenzotrifluoride serves as a useful intermediate for a reason: it packs a certain chemical punch without the volatility of lighter aromatics. Its molecular formula, C7H4ClF3, might sound like a mouthful, but this small molecule carves a wide path. Its role spans pharmaceuticals, agrochemicals, and advanced polymers, and I've run across it both in small R&D quantities and in scale-up settings. Some call it 3-chloro-α,α,α-trifluorotoluene, others stick with the systematic name, but everybody knows the slightly biting smell and clear, colorless appearance. It resists breakdown under ambient conditions, which makes storage and handling less worrisome than more reactive analogs.
Physical & Chemical Traits in Practice
Operators recognize this liquid by its strong aromatic odor and its flash point in the mid to high 60s Celsius. It doesn't mix well with water, favoring organic solvents instead, and that's saved a lot of headaches in extraction operations. I've seen teams use standard PPE and chemical fume hoods for even modest quantities, a nod to its volatility and possible inhalation hazards. The compound holds a sweet spot for thermal stability without resisting all reactions—critical for downstream derivatization. Its density and viscosity mean gear won't clog in typical flow reactors, and in purification, the moderate boiling point lets labs separate it from volatile side products without blowing budgets on vacuum distillation.
Technical Specifications and Proper Labeling
Accuracy in labeling matters, especially once drum-sized quantities enter multi-step syntheses. Clear hazard pictograms, proper hazard statements, and traceable batch numbers make a real difference in warehouse operations and regulatory compliance. MSDS and GHS systems, if kept updated, give downstream users a fighting chance to stay ahead of workplace accidents or shipment mix-ups. Surface details—purity levels, isomer ratios, and contamination limits—are not just formalities. They dictate yield and product stability, which impacts profit margins and research integrity. I've seen R&D budgets eaten up by poorly labeled intermediates, causing wasted days in troubleshooting and analysis reruns.
Preparation Methods That Shaped the Industry
Manufacturers typically reach for chlorination of benzotrifluoride under controlled conditions, using catalysts or UV light to direct the reaction at the meta position. This synthesis may sound straightforward, but anyone who's run it at scale knows the balancing act required to suppress byproduct formation and minimize worker exposure. Selectivity matters—excess reagents drive up costs and create disposal headaches. The preparation sequence laid the foundation for many modern halogenated building blocks, and improvements over the years, such as flow chemistry adaptations, continue to take root in plant operations. Researchers and process engineers regularly revisit throughput and emission control, seeking lower environmental burdens and greater economic returns.
Chemical Tinkering and Downstream Adjustments
This compound stands out for its resilience, yet also for the functional possibilities unlocked with minor tweaks. Nucleophilic aromatic substitution and coupling reactions, among others, transform 3-Chlorobenzotrifluoride into advanced ingredients for herbicides or life-saving drugs. Grignard and organolithium approaches leverage the reactivity of the trifluoromethyl group or the meta chlorine, enabling pharmaceutical chemists to craft new scaffolds that resist metabolic breakdown or improve target affinity. Production sites often maintain substantial stocks of this compound precisely for its utility in such transformations, replacing more toxic or less stable intermediates in legacy protocols. In my own work, switching to this intermediate reduced unwanted impurities, allowing final products to meet modern purity and safety bars.
What’s in a Name? Synonyms and Product Identifiers
Throughout industry circles, this compound navigates multiple identities. Some catalogs list it as m-Chlorobenzotrifluoride, others prefer Benzotrifluoride, 3-chloro, and a few reference the CAS number 98-16-8 as the primary marker. The chemical marketplace rarely rewards inconsistency in naming conventions, and more than one procurement order has stumbled because of ambiguous requests. I always remind new colleagues: keeping tabs on synonyms keeps labs running and projects on schedule. Miscommunication over such small details creates outsized costs in a world where supply chains already walk a thin line.
Staying Safe and Meeting Standards
Safety can never be left as an afterthought. Direct skin or inhalation exposure, even with less aggressive chemicals like 3-Chlorobenzotrifluoride, remains a known occupational risk. Real-world incidents—splashes, spills, or leaks—underscore the need for gloves, goggles, and ventilation as daily routine. The chemical resists biodegradation, so compliance teams push for tight containment and waste management practices. Regulatory scrutiny from agencies such as OSHA, REACH, and local authorities shapes operational protocols, from restricted emissions to periodic spill drills. Emergency response plans, on-site sensors, and ready spill kits are the real shields against production downtime and employee injury.
Where This Compound Leaves Its Mark
The application scene stretches far beyond textbooks. Agrochemical plants rely on this chemical for herbicide and fungicide intermediates. Drug synthesis depends on its ability to anchor complex molecules together, supporting innovation in treatments for cancer and infectious diseases. Engineering teams embed its derivatives in specialty polymers with high chemical resistance. Paint and coatings manufacturers build weather-resistant products on its backbone. From my vantage point, the capacity to anchor trifluoromethyl groups with a meta chlorine opens up application windows unavailable through simpler chlorinated benzenes. You don't need wild leaps in technology—just a reliable building block, and 3-Chlorobenzotrifluoride provides that.
Pushing the Boundaries: New Research and Development
Chemists keep stretching what this molecule can do, chasing greener and more cost-efficient pathways. Academic labs explore catalytic cycles that use less hazardous starting materials, while industry pilots invest in renewable solvents and closed-loop purification, all aiming to cut both cost and footprint. These efforts draw from decades of practical lessons, not just idealistic targets, and they often reveal practical ways to minimize emissions or improve product shelf-life. Industry-university partnerships test advanced sensors and real-time monitoring to catch problems at their source, a move I see reducing both employee risk and material loss in recent projects.
What Toxicity Studies Teach Us
Every chemical deserves a closer look before large-scale use. Long-term toxicity research shows that, although 3-Chlorobenzotrifluoride doesn't carry the severe acute hazards of legacy solvents, repeated or uncontrolled exposure has its risks. Liver and kidney effects appear in high-dose animal studies, underlining the need for push-button sampling systems and closed batch operations. Environmental impact reviews point to slow breakdown rates, which pushes stakeholders to revisit waste treatment plans and consider alternatives that close the lifecycle. Sustainable chemistry isn't just buzz—it's a practical push from real toxicity lessons learned over years of use and study.
The Road Ahead for 3-Chlorobenzotrifluoride
Future prospects likely depend on how well the industry and regulators can balance performance, safety, and environmental stewardship. Green chemistry could chart new routes for making and consuming this intermediate, shrinking both waste and exposure along the way. Advanced catalysis and digital process monitoring hold promise to curb the biggest pain points: unwanted byproduct formation, fugitive releases, and off-spec batches. Customers in healthcare, crop science, and materials will keep pulling for innovation, but only those suppliers who prove both consistent quality and genuine dedication to environmental duties will stay trusted. Watching this chemical's journey, both as a practical tool and a marker of evolving safety culture, has convinced me that it will remain relevant, but only for those willing to face the real-world trade-offs head-on.
What are the main uses of 3-Chlorobenzotrifluoride?
What Makes 3-Chlorobenzotrifluoride Stand Out
Many folks outside the chemistry world haven’t heard of 3-chlorobenzotrifluoride. In labs and on factory floors, this compound often performs behind the scenes. Its chemical stability and resistance to harsh conditions draw attention from industries searching for reliable tools in their arsenal.
The Backbone of Agrochemicals
Farmers and food producers count on this compound indirectly, since it is used as a building block while making herbicides and fungicides. Its chemical structure helps in piecing together more complex molecules that protect crops. The fact remains, global food supplies depend on modern pesticides, and this compound forms a key stitch in that safety net.
Global pesticide manufacturing ramps up each year, as growers face pests evolving new tricks. 3-chlorobenzotrifluoride finds use in crafting products like fluazinam and other fungicides, which means healthier crops and fewer spoiled harvests. Some researchers remain cautious about overuse and its effect on resistance—so the chemistry needs careful management. Not all substitutes offer the same results.
Making Colors That Last
I spent a few summers working at a textile mill. Dye vats ran every day, pouring out colors which only stuck because dyes didn’t break down too fast. Many of those dyes carried stories that started with specialty chemicals like 3-chlorobenzotrifluoride.
It’s more than a splash of pigment. The compound provides stability for the color molecules, meaning shirts, canvas, and carpets keep their color after washing and sun exposure. Manufacturers depend on materials that don’t fade in warehouses or on the store shelf, raising the bar for consumer goods.
Upgraded dyes using this compound require fewer touch-ups and less material waste—so clothing doesn’t get tossed for looking old before its time. That reduces landfill load and cuts costs for customers in the long run.
The Paint and Coating Advantage
On job sites, I’ve watched painters crack open cans of specialty coatings for metal and concrete. They often trust these paints and sealants to last through winter freezes or heavy foot traffic. 3-chlorobenzotrifluoride frequently acts as a chemical intermediate, either making the raw materials for resins or serving as a solvent base for custom coatings.
Modern automotive paints lean on this compound for the same reasons. High-gloss, weather-resistant finishes protect investment in vehicles and bridges. Skipping the right ingredients leads to rust, fading, or early failures. With so much infrastructure investment at stake, this chemistry quietly saves money and energy.
Safety and Environmental Points
Of course, no story about industrial chemicals feels complete without mentioning risk. Production and use require trained workers, controlled environments, and constant review. Researchers keep investigating safer disposal and better recycling, since trace amounts can harm waterways or wildlife over time.
Safer alternatives show promise for some uses. Green chemistry campaigns push for biodegradable options where possible, though the high performance of 3-chlorobenzotrifluoride means replacements must do more than just “work well enough.” Regulation tightens each year, forcing factories to keep emissions low and track every step in the chain.
Looking Ahead
Farms, paint shops, and factories all rely on this hard-working chemical. Experience shows that careful use, combined with new science and strict oversight, helps balance its benefits with health and environmental needs. Research continues, aiming for chemicals that deliver reliability without lasting environmental consequences.
What is the chemical formula and structure of 3-Chlorobenzotrifluoride?
The Makeup of 3-Chlorobenzotrifluoride
3-Chlorobenzotrifluoride goes by the chemical formula C7H4ClF3. This means the molecule contains seven carbon atoms, four hydrogen atoms, one chlorine atom, and three fluorine atoms. Chemists often use shorthand names for chemicals, but the formula shows what the molecule consists of in practical terms. Knowing the pieces of a compound helps in figuring out its behavior and how it might be used.
The structure packs some interesting features into a single molecule. At its core sits a benzene ring—a six-carbon atom ring with alternating double bonds, a common platform in organic chemistry. On this ring, a chlorine atom attaches at the third position if you start counting from the carbon where the trifluoromethyl group is attached. That trifluoromethyl group (–CF3) tacks itself onto the first carbon, with its three fluorines offering chemical stability and affecting how the molecule interacts with other substances.
Structure: More Than Just a Formula
It can be misleading to look at chemical formulas and think they tell the whole story. To really know what this substance is, think about the way each atom hooks onto the aromatic ring. One side of the ring has –CF3, three fluorine atoms bonded tightly to a single carbon. On the other side, at what chemists call the meta position, a chlorine atom stands out. Everything else on the ring is filled with hydrogens. Visualizing this makes it easier to predict reactivity, toxicity, and environmental impact.
In my own experience working with halogenated aromatics, seeing that trifluoromethyl group tells me the chemical won’t break down easily in the environment. The carbon-fluorine bond is one of the strongest in organic chemistry, so once 3-chlorobenzotrifluoride leaks into soil or water, it sticks around for years. The chlorine atom at the meta position also makes it an interesting coupling partner in organic synthesis. You can swap it out in reactions to build more complex molecules, which comes in handy for making herbicides, dyes, and specialty chemicals.
Why Structure Matters for Real-Life Use
Chemists and safety specialists analyze chemical structure before using or handling new materials. With 3-chlorobenzotrifluoride, the traits of the structure demand some respect. The trifluoromethyl group’s presence changes flammability, volatility, and performance. In manufacturing, health, and environment, a single added atom can mean more risk or new utility. Chlorinated aromatics have gotten a bad reputation thanks to pollution scandals, with long-term studies showing that similar molecules persist in the food chain, building up slowly.
People working in chemical plants need to keep good data sheets nearby to ensure safe storage. If spills happen, the fluorine-carbon bonds tell responders this is not something nature can clean up easily. This drives discussions around safe disposal. In a lab, 3-chlorobenzotrifluoride unlocks routes to tougher, more stable molecules. I have seen it used in coupling reactions, especially in pharma research, to build molecules that have both high potency and high stability. Chemists understand that the same traits making it stable and useful can create regulatory headaches around safety and disposal.
Charting a Responsible Path Forward
Solutions start with strong process controls in the plant. Regular audits, real-time sensors, and employee training all make a difference. For labs, small-scale reactions with proper containment cut down safety risks. In storage, keeping this chemical away from heat, open flame, and incompatible substances remains basic protocol. Resourceful waste management—sometimes through high-temperature incineration where possible—can stop persistent environmental buildup. All this takes awareness and a sense of responsibility. The chemical’s value in synthesis, research, and manufacturing is clear, but the environmental and health stakes call for careful and informed use at every step.
What are the storage and handling precautions for 3-Chlorobenzotrifluoride?
Understanding the Risks
3-Chlorobenzotrifluoride doesn’t show up in home pantries. It usually lands in chemical plants, research labs, or manufacturing facilities. This chemical acts as a strong solvent and pops up often as a building block in making pharmaceuticals, agrochemicals, and specialty coatings. Its dangers hit more than just a bad smell—this stuff’s flammable, can irritate airways and skin, and doesn’t mix well with a sloppy approach. If spilled, vapors can build fast and cause breathing trouble for anyone unlucky enough to be nearby. Fire risk cranks up in confined spaces. Once, I saw a drum leaking in a warehouse; without immediate action, the harsh odor spread quickly, making eyes water and noses sting. Folks scrambling into respirators showed how even a small oversight could threaten everyone’s health.
Storing Chemicals So People Go Home Safe
Chemical storage rooms shouldn’t double as break rooms. 3-Chlorobenzotrifluoride prefers cool, dry, and well-ventilated surroundings. Temperatures above 30°C can set off enough vapor for a blaze, especially if electrical equipment sparks. You don’t want to stack these containers near oxidizers, acids, or anything heat-producing. Fire codes say flammable liquids need special metal or safety-rated cabinets, far from sunlight or machinery that runs hot. I once watched workers set up shelves away from workstations, with spill trays underneath every drum. It seemed tedious, but a minor leak got caught in a tray one morning, and nobody needed emergency showers.
Always check every drum or container on arrival. Look for rust, dented lids, or oily marks—these weak spots can break open fast. Keep a clear label with hazard warnings intact. Emergency contact info and chemical details should be right there on the front. Routine inspections cut down on surprises. A friend told me about a forgotten canister in the back of their storeroom; it split open during summer heat and prompted an expensive cleanup. Small habits keep emergencies off the clock.
Handling with Heads-Up Awareness
No shortcuts fit chemical safety. Gloves that resist solvents, goggles, and lab coats or coveralls serve as basics, not extras. Proper chemical training can’t be a one-and-done; everyone who handles this solvent should understand spill response and first aid for chemical exposure. I always remember the team at a small industrial lab—they rehearsed their response on a slow Friday, then nailed it when a valve sprang loose weeks later. No confusion, no panicked calls, just teamwork.
Pouring or transferring 3-Chlorobenzotrifluoride should never happen near open flames or in cramped places without air movement. Static sparks sound far-fetched until you hear about a fire in a warehouse where someone skipped grounding procedures. In any space where this chemical moves, proper ventilation and explosion-proof motors need priority. Quality control folks should keep leak detectors handy, and everyone gets reminded that lingering odors aren’t a normal part of work.
Simple Steps Make Big Differences
Emergency plans go beyond something taped to the wall. Eyewash stations and safety showers should actually work, within ten seconds’ reach of anyone pouring or blending. Spill kits close by are nearly as important as the chemicals themselves. Local laws often call for written records and proper signage, but in real life, it’s those steady habits—storing with care, inspecting before use, handling with full PPE—that keep accidents at bay.
Sometimes, companies invest in ventilation upgrades or extra employee training after a close call. They realize quick fixes never match the value of a healthy team and avoided shutdowns. If every organization treats chemical handling as personal business, not just a rulebook exercise, fewer people end their day at the hospital and more go home proud of their safe record.
Is 3-Chlorobenzotrifluoride hazardous to health or the environment?
What is 3-Chlorobenzotrifluoride?
3-Chlorobenzotrifluoride may sound like something only chemists talk about. In reality, it shows up in paints, coatings, and some chemical manufacturing. With a crisp smell and low viscosity, manufacturers find it useful as a solvent and as a starting point in making other chemicals.
Health Impacts That Deserve Attention
Breathing in vapors of 3-Chlorobenzotrifluoride, even for a short while, sometimes leads to headaches, dizziness, or irritation in the nose and throat. Longer or repeated exposure increases the chance of skin problems or eye discomfort. On a few job sites I’ve visited, workers often mention a persistent irritation when handling this stuff, especially where ventilation isn’t great. It doesn't help that the compound evaporates easily at room temperature, filling the air with fumes quicker than many expect.
Peer-reviewed studies show its hazard level does not match that of the deadliest chemicals in industry, but people often overlook the cumulative effect. Direct skin contact dries out skin, and in high enough doses, lab animals showed damage to organs like the liver and kidneys. We don’t have piles of data on cancer risks for workers constantly exposed, but early toxicology suggests caution makes sense.
Environmental Concerns: More Than Meets the Eye
A chemical with “trifluoride” in its name rarely breaks down quickly in the wild. This one sticks around in soil and water, refusing to degrade as bacteria or sunlight try to do their work. One EPA summary flagged its persistence, which means traces linger for months or years. I’ve seen this story play out at factories next to rivers; runoff from a single year leads to chemicals turning up in sediment a decade later.
Fluorinated molecules like this resist treatments used in standard water plants. Fish exposed to chlorinated aromatics in studies sometimes show changes in behavior or reproduction, though researchers focus mostly on less stable cousins. Environmental scientists worry about trifluoride-based solvents joining the “forever chemical” pile, which concerns regulators everywhere.
Safer Handling and Practical Solutions
Good science points toward smart habits on the shop floor and in the lab. Workers who need to use 3-Chlorobenzotrifluoride do better with gloves that don’t wear out, goggles that seal well, and fume hoods running steady. Safety data sheets give clear warnings about spills, since the liquid spreads fast and seeps through cracks. Disposal takes planning—dumping down drains or in regular landfill creates long-term contamination. Incineration under strict controls works, but costs run higher than many companies want to pay.
Regulators in regions like the EU have begun asking for alternatives: solvents with lower volatility, biodegradable byproducts, and safer manufacturing routes. Switching isn’t always easy on the wallet, yet as public health costs grow—more respiratory issues, groundwater cleanups—more firms move toward safer chemicals.
Looking Forward
I’ve watched several industries adjust habits over time when faced with evidence of hidden risks. The road ahead for 3-Chlorobenzotrifluoride looks similar. As we learn more from real-world exposure and environmental monitoring, the drive for less hazardous materials will keep picking up. In the meantime, smart use and strong oversight make a difference in keeping both workers and communities safer from invisible hazards.
What is the shelf life and recommended storage temperature for 3-Chlorobenzotrifluoride?
What It Means to Handle Chemicals Safely
In the world of industrial chemicals, 3-Chlorobenzotrifluoride turns up often enough to make safe storage a real concern for labs, factories, and shipping staff. Practical storage habits not only preserve quality but protect people and the environment. I remember my early days in a lab, where labels and storage temperatures dictated whether we met safety codes—or scrambled to fix a leaky, ruined sample. Keeping quality high always starts with respecting guidelines.
The Real Shelf Life
Shelf life matters because a degraded solvent leads to unexpected reactions, loss of potency, and wasted money. I looked into the typical storage recommendations from suppliers recognized for reliability in the chemical trade. Most point to a shelf life of two years for unopened containers of 3-Chlorobenzotrifluoride. After two years, containers could see impurities form or the solvent edge toward instability, especially if humidity or high temps sneak in. Regular checks on expiration dates and condition go a long way. It is easy to forget what sits on a lower shelf and then reach for it during crunch time, only to lose a batch due to questionable solvent.
The two-year rule isn’t just a number penciled in for convenience. Manufacturers test for purity and degradation over time, and storage under the wrong conditions accelerates the process. In my own work, we kept a tight log of delivery dates and ran spot checks after a year for any chemical we knew could break down faster.
Keeping It Cool and Dry
Most chemical suppliers stay consistent: store 3-Chlorobenzotrifluoride between 2°C and 8°C, away from direct sunlight and moisture. At that temperature range (roughly 36°F to 46°F), the risk of unwanted side reactions plummets. Back in our lab, the dedicated flammable storage fridge became the unsung hero of day-to-day work, keeping sensitive compounds like this one fresh and reliable. Anyone trying to stretch shelf life beyond the guidance is playing with fire, both for results and safety.
Improper storage—think fluctuating temps, direct sunlight, or stacking near oxidizing agents—can mean the chemical breaks down or picks up moisture. That can translate into ruined experiments, hazardous spills, or compliance violations. I’ve seen teams forced to dispose of whole shipments because someone left solvents near a sunny window. Those sorts of mistakes cost money and undermine trust from leadership and inspectors alike.
Solutions for Safer Storage
Rely on temperature monitoring and logbooks for every storage area. Simple thermometers and humidity strips cost almost nothing compared to the expense of cleaning up a chemical spill. For any space with restricted airflow or no cooling system, regular spot checks are a smart investment. Avoid storing solvents in crowded rooms where a freezer door gets held open during inventory. Enough stories float around about freezer failures that no one needs another reminder about what spoiled chemicals can do to project timelines and staff morale.
Anyone using 3-Chlorobenzotrifluoride should also train new staff on chemical rotation practices. FIFO (first in, first out) works for solvents as well as onions, preventing forgotten bottles from lingering too long. That approach may sound old-school, but quality and safety both benefit.
Looking Ahead
Keeping 3-Chlorobenzotrifluoride effective means watching the calendar and the thermostat. In an industry built on safety and precision, it pays to do the basics right. Always check the label, consult the supplier documentation, and if in doubt, err on the side of caution. There’s no shortcut worth risking lab results, equipment, or, most importantly, the wellbeing of the people doing the work.
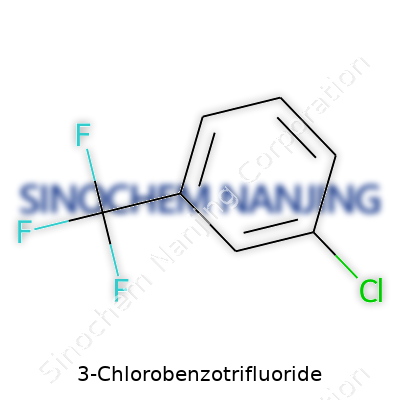
| Names | |
| Preferred IUPAC name | 1-Chloro-3-(trifluoromethyl)benzene |
| Other names |
m-Chlorobenzotrifluoride 3-Chlorobenzotrifluoride 1-Chloro-3-(trifluoromethyl)benzene 3-Trifluoromethylchlorobenzene m-Trifluoromethylchlorobenzene |
| Pronunciation | /ˌθriː-klɔːr.oʊˌbɛn.zəʊˈtraɪ.fljuː.ə.raɪd/ |
| Identifiers | |
| CAS Number | '98-16-8' |
| 3D model (JSmol) | `3D structure;JSmol;C1=CC(=CC(=C1)Cl)C(F)(F)F` |
| Beilstein Reference | 1307541 |
| ChEBI | CHEBI:84568 |
| ChEMBL | CHEMBL369218 |
| ChemSpider | 10619 |
| DrugBank | DB14096 |
| ECHA InfoCard | {'ECHA InfoCard': '100.010.481'} |
| EC Number | 209-384-8 |
| Gmelin Reference | Gmelin Reference: "2021867 |
| KEGG | C19136 |
| MeSH | D021217 |
| PubChem CID | 75648 |
| RTECS number | CB8925000 |
| UNII | K73CD9T18A |
| UN number | UN3334 |
| Properties | |
| Chemical formula | C7H4ClF3 |
| Molar mass | 186.57 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic odor |
| Density | 1.33 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.7 |
| Vapor pressure | 5.6 mmHg (20°C) |
| Acidity (pKa) | -0.23 |
| Basicity (pKb) | 12.85 |
| Magnetic susceptibility (χ) | -66.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.499 |
| Viscosity | Viscosity: 1.12 mPa·s (25 °C) |
| Dipole moment | 2.86 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -284.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1674 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 43 °C (closed cup) |
| Autoignition temperature | 605°C |
| Lethal dose or concentration | Lethal dose or concentration: LD50 oral (rat): 13,000 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 13,000 mg/kg |
| NIOSH | CN8575000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: 2.5 ppm (20 mg/m³) |
| Related compounds | |
| Related compounds |
3-Bromobenzotrifluoride 3-Fluorobenzotrifluoride 3-Iododenzotrifluoride 3-Methylbenzotrifluoride 3-Nitrobenzotrifluoride 4-Chlorobenzotrifluoride 2-Chlorobenzotrifluoride |

