3-Chloro-4-Methoxyaniline: More Than a Chemical Reagent
Looking Back: How 3-Chloro-4-Methoxyaniline Has Shaped the Lab
The story behind 3-Chloro-4-Methoxyaniline stretches back to the mid-century boom in organic chemistry, when researchers scrambled to produce new building blocks for dyes, drugs, and plastics. Back then, aromatic amines grabbed attention because of their flexibility during chemical transformations. The specific pairing of a chlorine atom with a methoxy group on an aniline core didn’t happen overnight. Chemists built up to it, step by step, testing out which substituents created stable yet reactive molecules. In this environment, 3-Chloro-4-Methoxyaniline earned its spot for offering a unique balance of reactivity and selectivity. Lab heads valued every opportunity to spin a new derivative, and this compound offered plenty of directions to take.
Stepping Into the Lab: What Makes It Stand Out
Three substituents jump off its benzene ring—an amino group, a chloro atom, and a methoxy group. Simple at first glance, but that trio packs an impressive punch for chemists looking to control a reaction. Its melting point often registers under 60°C, pointing to moderate thermal stability but not enough to survive industrial heat. The pale color and needle-like crystallinity speak to its purity after careful recrystallization. Solubility in organic solvents like ethanol and ether helps with extractions, making workups in the lab that much smoother. Handling the solid powder, even when packed in a brown-glass bottle and labeled with familiar hazards, drives home that organic chemistry asks for vigilance as much as curiosity.
Technical Specs Beyond the Label
3-Chloro-4-Methoxyaniline carries more than just a chemical formula: C7H8ClNO. In practice, molar mass usually reads as 157.6 g/mol, with density falling in line with most small organic molecules. Labs demand accurate labeling—proper identifiers, batch purities, shelf life, and hazard warnings. Safety sheets stress the importance of eye protection and fume hoods, not just for this compound but for the off-gassing that some impurities can provoke. Analytical teams double-check batches by running NMR and IR spectra, confirming both substitution patterns and absence of unforgiving contaminants. For the folks preparing calibration standards or scaling up reactions, skipping these details would wreck any consistent workflow.
The Method Behind the Molecule: How Chemists Prepare It
Making 3-Chloro-4-Methoxyaniline isn’t as simple as blending edge-of-the-shelf reagents. The favored route often starts with 3-chloro-4-methoxynitrobenzene. Catalytic hydrogenation—sometimes Pd/C, sometimes iron and acid—pulls that nitro group down to an amine. Careful control over pressure, temperature, and timing guards against over-reduction, which can trigger side reactions. The product doesn’t always come out pure, so experienced chemists deploy salt formation or solvent extraction before reaching for that purist’s technique—recrystallization. At scale, control over exothermic steps, cooling rates, and filtration become make-or-break factors, with yield maximization and safety running on parallel tracks.
What Happens Next: Reactivity and Modification Channels
Benzene rings love to host new substituents, and the mix of electron-withdrawing chlorine and electron-donating methoxy groups tunes the reactivity of 3-Chloro-4-Methoxyaniline in clever ways. Nucleophilic substitutions tend to favor the positions dictated by these groups’ directing effects. The amino group opens new doors, accepting acetylation, diazotization, or coupling reactions to craft more complex molecules—useful for dye intermediates or pharmaceuticals in early-stage development. This interplay between the groups often reminds me of setting up a chessboard: every piece matters, and one move sets up the next.
Familiar Names and Identity in the Chemical World
In catalogs or journal articles, this compound slips under several aliases. You’ll find it called 3-Chloro-p-anisidine, 4-Methoxy-3-chloroaniline, or just “aniline derivative” depending on the application or researcher’s preference. CAS numbers and molecular identifiers show up in procurement paperwork, but in practice, a quick glance at the label and the structure brings the chemist right back to the reactivity profile and lab safety protocol they’ve learned to trust.
Not Just a Label: Keeping it Safe in the Workplace
Safety considerations come right to the front. Aromatic amines warrant respect, not just because of their skin and eye irritant properties, but because of potential long-term risks flagged by toxicity studies on related compounds. Labs treat spills and dust with the seriousness they deserve—gloves, goggles, and proper ventilation. Disposal never takes shortcuts as regulations treat these chemicals with justified caution. Comprehensive training avoids surprises, protecting both workers and the environment. Many labs lean heavily on established best practices, and with compounds like 3-Chloro-4-Methoxyaniline, there’s no room for improvisation.
Where Science Meets Something Practical: Application Areas
In the hands of a skilled chemist, 3-Chloro-4-Methoxyaniline turns into much more than a research sample. The dye and pigment industry draws on its reactivity to make the colors that line store shelves, especially in textiles and plastics. Pharmaceutical research teams use it as a scaffold for synthesizing candidate molecules, sometimes adjusting substitution to tweak pharmacological properties. Crop protection chemists have explored its potential in fine-tuning herbicides or fungicides. The appeal lies in the balance this molecule strikes—reactive enough for practical transformations, but manageable for controlled experiments, making it popular for research and product development alike.
Curiosity on the Bench: Trends in R&D and Exploratory Chemistry
Some of the most exciting developments tied to this compound emerge from the intersection of synthetic organic chemistry and green chemistry. Teams strive to reduce hazardous waste and energy use, running reactions on smaller scales or swapping traditional solvents for water or ionic liquids. Newer methodologies search for single-step transformations and environmentally friendlier catalysts. Published papers track the journey from small-scale syntheses to more efficient pathways that could suit scale-up. I’ve watched research groups pivot from classic batch chemistry to continuous flow systems, learning from every iteration how a classic molecule like this can still spark new discoveries.
The Other Side of the Story: Digging Deeper into Toxicity Concerns
Anyone who’s handled or examined the broader health profiles of substituted anilines knows they often bring unwelcome baggage. Toxicity studies highlight potential for adverse effects, with a focus on skin sensitization, organ toxicity, and, with repeated exposure, possible carcinogenicity. Regulatory agencies look for detailed reports on handling, exposure levels, and waste streams, especially if large batches move beyond the bench. Researchers monitor environmental persistence because breakdown products sometimes show up in wastewater or soil science studies. Colleagues share experience with both risk assessment protocols and personal protective strategies—they know toxicology often moves slower than synthetic innovation, so caution stays central.
Peering Ahead: What’s Next for 3-Chloro-4-Methoxyaniline?
As labs expand green chemistry and digital tracking systems, 3-Chloro-4-Methoxyaniline sits at a crossroads. Researchers weigh the classic reactivity profile against changing safety standards and rising demand for greener processes. Fine-tuning synthesis pathways by using recyclable catalysts, switching over to smart, low-impact solvents, or building synthesis into continuous rather than batch processes—these aren’t just trends, but real opportunities in the field. Future directions look set to balance inventive science with real-world application, shining a light on how even well-studied compounds still play a central role as both a challenge and an inspiration in chemical innovation.
What is 3-Chloro-4-Methoxyaniline used for?
The Role in Chemical Manufacturing
3-Chloro-4-Methoxyaniline shows up most often in the world of chemical manufacturing. This compound isn’t something found in a kitchen cabinet. Instead, you’re likely to see it inside a laboratory, where chemists use it as a key starting material. They appreciate the way its structure invites further reactions, especially when creating more complex molecules.
Pharmaceutical researchers put 3-Chloro-4-Methoxyaniline to work when they make small-molecule drug candidates. Many drug molecules begin life as simple aromatic amines like this one, known to tweak the underlying chemical properties that companies hunt for—better solubility, improved targeting in the body, or longer shelf life. Monash University’s medicinal chemistry teams mention how methoxy and chloro groups open up pathways to new anti-inflammatory and anticancer agents. For each step on that journey, materials like 3-Chloro-4-Methoxyaniline shape what’s possible in the test tube, showing why understanding every intermediate matters.
Dyes, Pigments, and Everyday Color
Art supplies and printed fabrics don’t usually make headlines, but the colors come from painstaking chemistry. This aniline derivative slips into dye and pigment manufacturing. The presence of chlorine and methoxy groups makes for deep, lasting color that resists fading. Textile makers trust this class of molecules when they want to bring out reds, blues, and violets that stay bright after washing.
Synthetic dyes sometimes pose risks for health or the environment. Lab safety data sheets record how certain aromatic amines need careful handling because they might irritate skin or have toxic effects. Companies in Europe and North America follow tight safety rules. They treat these chemicals with respect, investing in closed systems and improved personal protection to limit accidental contact.
Agrochemicals and Crop Protection
Walk through any large-scale farm, and the chemistry beneath your feet plays a huge part in boosting crop yields. The structure of 3-Chloro-4-Methoxyaniline, with its ability to bind and release additional atoms, comes in handy for synthesizing herbicides or fungicides. These products don’t just keep weeds or mold at bay; they keep harvests predictable so more people get fed.
Farmers today face tight rules on which chemicals reach plants and soil. Innovations in agrochemicals build on compounds like this one to design molecules that break down faster after they’ve done their work—less environmental residue, fewer worries downstream. Agrochemical developers know public trust depends on tuning old recipes with safety and transparency at the core.
Broader Impact and Responsibility
People far from a research bench feel the ripple effect of chemicals like 3-Chloro-4-Methoxyaniline. Tracing a piece of printed packaging or washing a bright T-shirt brings the question of where these chemicals came from, whether rules for health and environment had teeth, and whether the manufacturing left a light footprint.
Chemists, factory workers, and end consumers all benefit when companies dig into supply chain transparency. Product documentation, third-party safety audits, and responsible sourcing matter—not just to pass inspections but to support real-world safety. Experiences from friends working in industrial firms taught me that the best companies go beyond compliance. They run pilot projects for greener chemistry or offer retraining to keep people safe as rules shift.
What Comes Next
Every new industrial era sees older chemicals reinvented for fresh, safer uses. Research into biodegradable dyes, non-toxic intermediates, and wearable sensors hints at the legacy 3-Chloro-4-Methoxyaniline leaves. The next generation of chemists might use what they learn from it to build smarter, cleaner technology, closing the gap between innovation, practicality, and environmental care.
What is the molecular formula of 3-Chloro-4-Methoxyaniline?
Diving into the Structure
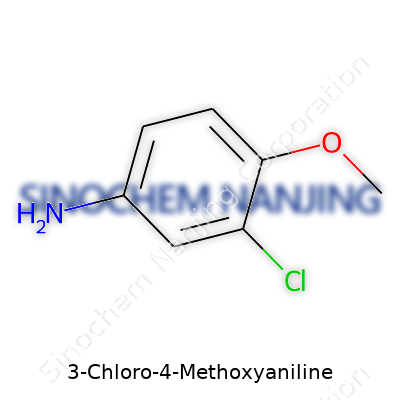
Sometimes chemistry looks like a puzzle that flips logic on its head. 3-Chloro-4-Methoxyaniline isn’t some random jumble of letters. Its structure matters, especially in labs where real people study, test, and try to make sense of molecules. If you pull up the skeleton of this compound, you’ll find a benzene ring as its backbone. At the 3-position, a chlorine atom stands out. At the 4-position, a methoxy group sweeps in. An amino group settles in at the 1-position. Each of these groups changes how the molecule acts and how people can use it in the real world.
Counting Atoms: The Formula
Chemists don’t settle for simple names, so they line up every atom, every bond. For 3-Chloro-4-Methoxyaniline, that count ends up as C7H8ClNO. Break it down: seven carbons form the ring and side chains; eight hydrogens dot the ring and attach to the methoxy and amino groups; one chlorine is tucked onto the ring; one nitrogen forms the core of the amine; one oxygen floats in from the methoxy group.
This formula is not plucked from thin air. Textbooks, online chemical databases like PubChem, and chemists in pharmaceutical or agrochemical fields recognize it. It shows up in safety data sheets, helping keep people safe, and guides analysts checking for impurities in manufacturing. C7H8ClNO isn’t just a piece of trivia—it’s a checkpoint so that mistakes don’t slip into research or industry pipelines.
Why Accuracy Counts
I remember how easy it felt to mess up early in chemistry. Miscount one atom and suddenly a substance can become toxic or lose the property you want. In real lab practices, mistakes can spill over into faulty data or even safety incidents. Misreading a molecular formula means the wrong chemical winds up in a reaction, or the waste disposal steps miss hazardous elements. With something like 3-Chloro-4-Methoxyaniline, accuracy runs the show, not just for legal compliance but for trust among workers, researchers, and end users.
Good science comes from getting the small things right. If people are careless, the wrong molecule can slip through—maybe causing drug side effects, or letting a harmful impurity into a material. Respect for the tiny details keeps many people safer and drives breakthroughs.
Better Solutions for Safer Chemistry
Access to trusted chemical information should feel as basic as having a clean glassware cabinet. Digital databases like ChemSpider and regulatory sources regularly review and update molecular formulas and properties. Making these tools part of daily routines in research and manufacturing shops stops guesswork. Handbooks and electronic databases help double-check work. Investment in clear labeling, standardized training, and continuing education makes it easier for people to get things right from day one.
If a question about a formula pops up, pausing and checking directly with primary sources beats relying on memory or word of mouth. Labs that put quality control front and center don’t just follow rules—they grow a culture where double-checking becomes a habit, not a chore. That approach saves headaches, budgets, and sometimes lives. People who spend time getting to know the molecules they use don’t just solve puzzles—they help build a better, safer chemistry world.
What are the storage conditions for 3-Chloro-4-Methoxyaniline?
Common-Sense Chemistry Doesn’t Stop on the Lab Bench
A lot of lab chemicals look unremarkable, but every bottle can carry risks unless you treat it with respect. 3-Chloro-4-Methoxyaniline doesn’t draw much attention—still, turning your back on safe storage can backfire. Years working alongside chemists and lab managers have taught me this isn’t just about regulators or protocols. It’s about staying healthy, preventing sneaky contamination, and protecting resources. Mistakes get expensive fast.
What 3-Chloro-4-Methoxyaniline Needs for Storage
The basics start with dryness, darkness, and a tight seal. This compound doesn’t mix well with moisture or light. Exposure to air can also spoil its stability over time. Shelf life drops, and those small changes you don’t notice can throw off experiments. Glass jars with screw caps feel old-school, but they’ve stood the test because plastics can let vapors through or degrade unexpectedly.
Direct sunlight might not seem like a big deal, but chemical degradation can sneak up fast. Ultraviolet rays trigger reactions you can’t spot until your yields drop or your results turn strange. A cool, dark cabinet or chemical refrigerator blocks that risk. Most lab refrigerators hold at 2-8°C and aren’t shared with food—they give the extra security this compound deserves.
What a Chemical Label Means Beyond Words
The Safety Data Sheet lists health hazards—3-Chloro-4-Methoxyaniline causes irritation, and longer-term exposure can increase health risks. Accidental spills in storage areas cause problems not just for the person on shift, but for everyone who enters later. I’ve watched colleagues recover from rashes and worse after underestimating something as minor as residue on a doorknob. If the workspace smells odd or you see signs of discoloration, it’s time to check the container for leaks or damage.
Smart Placement Cuts Down on Emergency Calls
A lesson I learned early: don’t store incompatible chemicals next to one another “for convenience.” Aniline derivatives can react with oxidizers, acids, and certain bases. A forgotten bottle set next to a bleach container turns small negligence into a big incident. Always double-check the shelf plan and make sure spill trays or secondary containers are in use before going home.
Crowded shelves often mean containers get bumped or swapped by mistake. Consider separate cabinets or marked areas for toxic organics, and avoid storing bottles above eye level. Gravity moves faster than you when a bottle falls; cleanup becomes much less stressful if everything has a designated place within easy reach.
Solutions for Storage Challenges
Proper ventilation keeps fumes in check. Even though you might not smell anything strong, vapors build up over time, making headaches and chronic exposure cases more likely. Fume hoods or vented storage keep air clear and staff in better shape. Fire safety cabinets rated for flammable or toxic compounds add another layer—strong metal, tight seals, and ground-level placement all matter.
I keep an emergency cleanup kit close by, stocked with spill pads, gloves, and goggles. Label containers with readable dates, and rotate old stock forward so nothing expires at the back of a cabinet. Forgetting to check the expiration date can mean ruined syntheses or risky decomposition products. In my experience, double-checking storage saves more headaches—and more lab funds—than any quick shortcut ever could.
Is 3-Chloro-4-Methoxyaniline hazardous or toxic?
What We Know About 3-Chloro-4-Methoxyaniline
3-Chloro-4-Methoxyaniline comes up in factories that make dyes, drugs, and other specialty chemicals. This compound, on the surface, blends in with other aniline derivatives, but the chloro and methoxy groups set it apart from basic aniline. It’s more than just a lab curiosity; it heads straight onto the floor of industries that rely on molecular customization.
Health and Workplace Impact
I’ve seen chemical handling routines at manufacturing plants. Personal protective equipment always hangs right by the door, not just for show. Workers know what direct skin contact or inhalation risks look like. The problem with 3-Chloro-4-Methoxyaniline is its toxic profile, echoing lessons from aromatics and substituted anilines. Similar molecules have caused headaches, dizziness, methemoglobinemia, and allergic skin reactions in those handling or even just breathing unfiltered dust or vapors.
3-Chloro-4-Methoxyaniline carries toxic risk because of its structure. The body struggles to break down these substitutions safely. That means, if you inhale it or it touches your skin, you might face not only irritation but more serious conditions like blood and organ toxicity. That’s just not a gamble worth taking for a wide population of workers, especially in settings with limited training or subpar ventilation.
Environmental and Societal Worry
Runoff from chemical plants rarely stays on-site. Once a chemical like this leaks into soil or streams, local communities deal with it firsthand. Years ago, I covered a story on riverbank contamination from synthetic dye factories. The clean-up effort required proper hazardous waste facilities, and plenty of testing showed how fast aromatic amines showed up in sediments and wells. The added chlorine and methoxy groups here often slow natural breakdown, which means groundwater takes longer to recover. Wildlife and downstream agriculture sometimes suffer long after the plant moves on. It’s a slow-moving problem, one seldom seen on headlines, but it hits home for anyone living near a chemical complex.
What Can Change in Handling Practices?
Clear labeling works; no one should reach for this chemical without knowing they need gloves, goggles, and a face mask. Too often, companies skip basics due to budget pressure or sheer routine. Those working with 3-Chloro-4-Methoxyaniline need solid training. I’ve seen factories improving their fume hoods and better spill control gear, and not just for show. Routine air testing gets real results, quickly alerting supervisors if limits creep beyond safety standards. Waste should travel directly to hazardous treatment plants, not public landfills.
At a broader level, agencies must keep updating exposure limits as new toxicology research appears. Community monitoring programs let residents catch trouble before it grows. Sharing data—good, bad, or ugly—helps everyone see the stakes. Weighing the economic benefit of making a specialty chemical against the health cost to workers and neighbors should not just fall to companies. Local governments ought to have a seat at the table too.
Finding a Safer Way Forward
No factory will toss every hazardous raw material in a day, but improvement can move faster than it does now. Engineers test alternative synthetic routes or substitutes that cut out chlorine or methoxy groups entirely. Pressure from public awareness and worker advocacy matters. Questions about toxicity do not disappear simply because a material arrives in a drum stamped “industrial use.” The reality remains: strong safety culture beats wishful thinking every time people and chemicals share a workspace.
What is the purity specification of 3-Chloro-4-Methoxyaniline?
Looking Past the Numbers
For anyone who works in fine chemicals, the details around 3-Chloro-4-Methoxyaniline hit differently than generic purity statistics. Every chemist knows the Cas number: 6375-47-9. But the work rarely stays that sterile. In practice, purity isn’t just about pride, or ticking off analytical milestones. It shapes the quality chain all the way from a bench in an R&D lab to the moment a client’s HPLC spits out the expected peak.
The Numbers Backed by Needs
Suppliers selling 3-Chloro-4-Methoxyaniline commonly offer material at 98% purity, and some batches run to 99% or more. Analytical reports typically spell things out using HPLC or GC. This seems simple: pick the highest spec available, sign off, move forward. Yet real-world experiences tell everyone why this type of accuracy counts. Tiny impurity levels—often measured below 1%—can spark major headaches in scale-up synthesis or registration dossiers. If you’re targeting an API intermediate, a catalyst poison, or even an unexpected trace of heavy metal can set back a project by weeks.
Where the Stakes Get Personal
My time in lab coats brought home what high purity really means. At lower specs, I dealt with byproducts that fouled reactions, leaving sticky residues and ghost peaks on NMR. Running with 98% or higher almost always brought fewer interruptions, cleaner filtrations, and better reproducibility. One misjudged batch meant chasing phantom side products for hours—even days—which ate up grant funds faster than anyone liked to admit.
Health and Environmental Angles
Analytical rigor isn’t about bureaucracy. Personal safety and environmental management both hinge on proper documentation. Even one percent of a persistent chlorinated byproduct changes how a facility must handle waste. Reach, GHS, and related guidelines press for less ambiguity and more disclosure, especially as European and U.S. regulatory frameworks bite harder. If the data sheet excludes key impurities, operators find out the hard way from alarms or unplanned shutdowns.
Supplier Transparency and Documentation
Not every producer approaches documentation the same way. Some offer a single certificate, with basic data on melting point and loss on drying. Others deliver full NMR, IR, and HPLC overlays. The value lies in clear reporting. I’ve learned to chase suppliers who walk through impurity profiles, outline solvent residues, and justify their heavy metal data. Without this, project managers add risk in places they least want to see it. The best partners pick up the phone, explain peaks, and own up to batch variations.
Moving Toward Smarter Sourcing
Scientists and buyers can’t afford to look only at the price per kilogram. They must push for even tighter documentation, regularly review supplier consistency, and dig into supply chain transparency. Digital supply platforms help, but old-fashioned communication remains irreplaceable. Those hard-won lessons from failed syntheses carry weight: always verify with third-party testing if stakes run high.
References:- Sigma-Aldrich Certificate of Analysis, 3-Chloro-4-Methoxyaniline
- European Chemicals Agency: Substance Infocard, 3-Chloro-4-Methoxyaniline
- Personal experience as synthetic organic chemist, 2010–2020
| Names | |
| Preferred IUPAC name | 4-methoxy-3-chloroaniline |
| Other names |
4-Amino-1-chloro-2-methoxybenzene 3-Chloro-p-anisidine m-Chloro-p-anisidine 3-Chloro-4-methoxybenzenamine |
| Pronunciation | /ˈθriː-klɔːrəʊ-fɔːr-mɛθˈɒksi-əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 10256-03-8 |
| 3D model (JSmol) | `3d:CC1=CC(=C(C=C1Cl)N)OC` |
| Beilstein Reference | 1718655 |
| ChEBI | CHEBI:18908 |
| ChEMBL | CHEMBL301998 |
| ChemSpider | 14204 |
| DrugBank | DB08370 |
| ECHA InfoCard | 03d5c4b7-7d4d-48af-8dbc-7c1dbfa0ad67 |
| EC Number | 202-738-6 |
| Gmelin Reference | 108059 |
| KEGG | C14370 |
| MeSH | D036673 |
| PubChem CID | 68199 |
| RTECS number | BY6300000 |
| UNII | 34U72QTG8N |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C7H8ClNO |
| Molar mass | 157.6 g/mol |
| Appearance | Light yellow to brown solid |
| Odor | Amine odor |
| Density | 1.25 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 1.79 |
| Vapor pressure | 0.00422 mmHg at 25°C |
| Acidity (pKa) | 6.05 |
| Basicity (pKb) | 10.04 |
| Magnetic susceptibility (χ) | -59.27·10^-6 cm^3/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | 300 cP (25°C) |
| Dipole moment | 2.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 181.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −62.2 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3738 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, in contact with skin or if inhaled; causes skin irritation; causes serious eye irritation; may cause an allergic skin reaction; may cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H317, H334, H341, H351 |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P337+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-1 |
| Flash point | 104°C |
| Autoignition temperature | Autoignition temperature: 540°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1990 mg/kg |
| NIOSH | SN9866000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m³ |
| Related compounds | |
| Related compounds |
3-Chloroaniline 4-Methoxyaniline 4-Chloroaniline 3-Methoxyaniline 3-Chloro-4-nitroaniline 3-Chloro-4-fluoroaniline 4-Chloro-3-methoxyaniline 2-Chloro-4-methoxyaniline |

