Peeling Back the Layers of 3-Buten-2-One: An Editorial Commentary
Historical Development
People often consider the chemistry of the past a black box, but certain compounds–like 3-Buten-2-one–tell a richer story. Years ago, organic chemists spent tireless nights mapping out new molecules that could drive both industry and academic discovery. 3-Buten-2-one emerged during a wave of keen interest in unsaturated carbonyls. The laboratory synthesis and gradual industrial adoption carved a space for this molecule among the early crop of alpha, beta-unsaturated ketones, driven not only by curiosity but by real demands for efficient intermediates. The shift from small-scale reactions to larger operations mirrored the broader pattern of organic synthesis moving into real-world applications as chemical engineering matured.
Product Overview
Many folks outside the chemical field might overlook why manufacturers focus on 3-Buten-2-one, except that it bridges a gap between lab studies and day-to-day products. You can spot derivatives or reaction products from this simple molecule in the synthesis of flavors, fragrances, and pharmaceuticals. It rarely stays in its pure form for long–its reactivity means it gets snapped up in the next step of a reaction vessel quickly. That versatility has led research teams around the globe to use it for tasks from specialty coatings to drug synthesis. It is not flashy, but anyone in the know recognizes its punch.
Physical & Chemical Properties
The structure of 3-Buten-2-one sets up an intriguing interplay between a carbonyl group and an alkene. As a colorless to yellowish liquid, it carries a sharp, pungent odor that hints at its chemical vigor. The double bond adjacent to the carbonyl makes it something of a chemical crossroads, hosting nucleophiles on one side and electrophiles on the other. Under the right conditions, it boils at a moderate temperature–neither so volatile as to be uncontrollable nor so sluggish as to hinder work. Solubility in water and many organic solvents marks it as a flexible participant in multistep syntheses, which open the door to both academic and industrial workflows.
Technical Specifications & Labeling
In practice, those handling 3-Buten-2-one sort it by purity and batch quality. Laboratories push for minimal contamination to ensure reproducibility, while larger operations may relax standards to manage costs. Labeling usually puts high visibility on its volatility, flammability, and toxicity warnings, plus storage guidelines to minimize accidental exposure. All of this matters not for bureaucracy’s sake but because these details keep researchers and workers safer. That’s based on years in labs where a missing warning or a mismarked bottle leads to a world of trouble nobody wants to deal with.
Preparation Method
Chemists have come up with a list of ways to prepare 3-Buten-2-one, with routes often dictated by available starting materials and downstream requirements. The most direct path involves dehydrogenating butanone, though labs sometimes rely on dehydration of precursor alcohols or rearrangement reactions. Industrial setups lean on conditions that safely and efficiently scale up without blowing through safety limits or budgets. That process isn’t always glamorous–it can mean careful control of temperature, pressure, and catalyst purity. Each method reflects years of trial, error, and a few triumphs.
Chemical Reactions & Modifications
Think of 3-Buten-2-one as a crossroads in organic synthesis. The alpha, beta-unsaturation primes it for nucleophilic addition, making it a frequent star in Michael additions and related transformations. Chemists use it in aldol condensations, cyclizations, epoxidations, and reduction reactions, often to build more complex frameworks found in pharmaceuticals and specialty materials. Its conjugated system lets it feed directly into polymer chemistry, agrochemical development, and limited runs of rare flavors. Each application tests the limits of reaction conditions, with careful adjustments necessary to steer the outcome.
Synonyms & Product Names
Those working with chemicals long enough grow accustomed to a carousel of names for one compound, and 3-Buten-2-one is no different. Trade literature and academic papers use labels like methyl vinyl ketone, crotonone, and butenone. Alternate names sometimes spring from different naming conventions or from regional habits. This often causes confusion unless you know your way around chemical nomenclature – sometimes paperwork or material safety documentation can turn into a scavenger hunt.
Safety & Operational Standards
There’s no sugar-coating the hazards tied to this molecule. Even brief encounters with mist or vapor make your nose and eyes sting, and direct skin contact risks severe irritation. Over the years, workplace standards have gotten tighter on account of reported incidents and ongoing toxicology reviews. Fume hoods, gloves, and protective eyewear aren’t optional, but taken as given by anyone with a healthy respect for chemicals. In settings where large volumes come into play, rigorous ventilation, immediate spill containment, and regular training have become the backbone of safe handling.
Application Area
On the application front, 3-Buten-2-one threads through industries at a surprising number of points. In the realm of pharmaceuticals and fine chemicals, researchers value it for constructing complex, biologically active molecules. Makers of flavors and scents use its sharp profile for certain key notes, though strict limits follow because of toxicity. Modern polymer chemistry grabs this molecule for making high-performance plastics and coatings. Agrochemical companies see it as a springboard for herbicide and pesticide ingredients. Those ends all rely on the molecule’s ability to jump into further reactions, often unlocking properties that bulkier, less reactive precursors just can’t match.
Research & Development
In research, the goals stretch beyond finding one more use for 3-Buten-2-one. Modern laboratories chase greener synthesis routes to limit hazardous waste and energy consumption, knowing regulatory bodies pay closer attention each year. Scientists run studies to cut exposure and boost yields with recyclable catalysts, solvent-free reactions, or continuous flow systems that lower risk and cost. Universities pair with industry partners to test bio-based methods, tapping renewable carbon sources and redesigning processes that used to rely on heavy metals or fossil fuels. In fields like medicinal chemistry, this molecule stays relevant as a modular platform for quickly generating variants of drug candidates or screening libraries.
Toxicity Research
Experience reminds us that toxicity can define the fate of a molecule no matter how useful. For 3-Buten-2-one, the dangers loom from both short- and long-term exposure. Studies highlight its irritating and potentially carcinogenic nature, prompting extra caution and driving researchers to model exposure, set down clear acceptable limits, and adjust procedures in real time. Animal studies shed light on metabolic pathways and possible systemic effects, while epidemiological work watches for patterns in workers who spend years near even low doses. This side of the compound’s dossier keeps evolving, and responsible use tracks each new publication that comes out.
Future Prospects
Peering into tomorrow’s chemical landscape, 3-Buten-2-one won’t disappear anytime soon. Its reactivity, accessibility, and simple structure let it keep pace as chemists dream up new reactions and materials. Researchers hunt ways to tame its hazards without tossing aside its benefits, such as designing better protective equipment, smarter reactors, or even tweaking the molecule itself to dull its edge. Industrial demand for sustainable syntheses creates an opening for greener processes built around this compound. As data pours in, the best prospects combine smarter chemistry with a sharper eye for worker safety, community impact, and environmental footprint. Years in the lab taught me upstream choices matter just as much as downstream cleanups, and 3-Buten-2-one’s evolution probably offers a lesson in doing better–not just doing more.
What is 3-Buten-2-One used for?
Unpacking What’s Behind the Name
The name 3-buten-2-one doesn't roll off the tongue, but this chemical pops up in more places than most people realize. Chemists often call it methyl vinyl ketone. It looks simple on paper, built from just carbon, hydrogen, and oxygen, but its impact stretches far across several industries. Anyone working in labs has met the sharp, pungent scent—some describe it as something between vinegar and plastic. That smell signals how reactive and useful this molecule can be.
Big Player in Making Other Chemicals
Factories pump out 3-buten-2-one to build bigger, more complicated chemicals. It’s a workhorse for making things like pesticides, pharmaceuticals, and plastics. The double bond in its structure snaps into reactions, helping chemists stitch together long chains or add complex groups. This ability supports growing crops, mixing medicines, and shaping the body of cars or toys.
Pharmaceutical researchers lean on 3-buten-2-one for making certain pain medicines and antibiotics. It acts as a tool to snap in carbon atoms or tack on groups needed to tweak a drug’s behavior in the body. This might sound abstract, but these small chemical changes mean a pill dissolves just right or a pesticide targets the right bug without harming everything in the field.
Shows Up in Nature’s Toolbox
Some fruits and veggies, particularly when they ripen or get damaged, let off small amounts of 3-buten-2-one. Scientists picked up on this in ripening tomatoes and some cheeses. Its aroma helps define flavors and tells a story of freshness or spoilage. Years back, I worked sorting tomatoes on a farm. There was always a moment where a certain sharp tang settled over ripe piles. Turns out, this molecule helped send that cue.
The Safety Side of the Story
No one wants factory accidents or long-term exposures that harm communities or workers. Studies from groups like OSHA and the European Chemicals Agency point out that handling 3-buten-2-one needs respect—it can cause skin and eye irritation, and breathing in the fumes brings serious risks. Some animal studies raised cancer concerns. Good ventilation, careful storage, and strong regulations help contain the dangers. People shouldn’t fear the molecule itself, but nobody benefits from sloppy handling. Labs and plants using it set clear protective rules and use sensors to catch leaks fast.
Room for Safer Choices and Cleaner Outcomes
People working in greener chemistry circles see a challenge. Can we replace 3-buten-2-one in some uses, or design smarter ways to work with it? Processes that cut waste or swap out hazardous solvents have already made progress. Regulators nudge the industry to rethink old habits, and some companies now use renewable raw materials, so less pollution comes out the pipes. This isn’t about banning one ingredient—it’s about weighing costs, outcomes, and finding a better balance between science and health.
Why the Details Matter
Everyday life sits downstream from chemicals like 3-buten-2-one, even if most people never see them up close. From the freshness of breakfast to the supply of clean medications, controls and choices made far from public view touch families and neighborhoods. Knowing what goes into food, packaging, and drugs keeps everyone part of the discussion about safer workspaces and cleaner products. That level of transparency and shared responsibility is the kind of progress that keeps trust strong in science and industry.
Is 3-Buten-2-One hazardous or toxic?
Understanding 3-Buten-2-One
Ask a chemist about 3-Buten-2-one and you’ll likely hear terms like “methyl vinyl ketone.” Found in everything from industrial processes to cigarette smoke, it’s not a chemical many people bump into on purpose. Most uses involve making other chemicals, or crop protection. For anyone working or living near industrial zones, it shows up as part of air pollution concerns. Recognizing the risk means looking at both the science and real-world experiences.
Health Risks and Exposure
Breathing in air laced with 3-Buten-2-one tends to irritate the eyes, nose, and lungs pretty quickly. Toxicology data points toward this compound as both an irritant and a potential long-term health risk. Workers who deal with production and handling without proper protection risk chemical burns or lung damage. Reports from the National Institute for Occupational Safety and Health (NIOSH) set recommended exposure limits at levels where workers still smell it before it gets overwhelming. Exposure beyond those limits causes coughing, headache, and in rare cases, lung injury serious enough to send people to the hospital.
Lab studies back up those worries. Animals exposed to high levels suffer respiratory distress, liver strain, and effects on the central nervous system. Long-term exposures can mean cancer risk goes up—in fact, some international agencies flag it as a possible carcinogen. The Environmental Protection Agency (EPA) lists methyl vinyl ketone among high-priority substances for review, in part because it can be present in the air around factories and hazardous waste sites.
Environmental Impact
3-Buten-2-one doesn’t just disappear after release. It can move through the air, get into water sources, and stick around for a while unless broken down by sunlight or microbes. Plants and animals living near industrial sites sometimes face exposure through contaminated soil or water. That adds one more layer of concern for regulators and local communities. In drinking water testing, the compound rarely turns up at levels above what is considered safe, but accidental spills raise the stakes for local environments and people alike.
Keeping Workplaces and Communities Safer
Technology in workplaces dealing with this chemical helps keep the odds on the side of safety. Ventilation systems pull hazardous vapors away from workers, while chemical-resistant gloves and protective eye gear stop contact burns. Regular air monitoring lets safety managers act fast if levels creep up. No system works without proper training—everyone in the plant needs to know what to do if there’s a spill or a leak. This same approach goes for first responders and clean-up crews. Rules matter, but on-the-ground know-how saves more lives.
Moving Forward with Better Policy and Education
Regulation and education walk hand-in-hand with hazardous chemicals. Agencies like OSHA keep exposure thresholds under review, but staying safe often means looking out for local hot spots—especially where factories meet neighborhoods. Communities deserve accurate air monitoring reports and the right to know which chemicals sit close by. Companies practicing transparency build trust and offer an early warning if something goes sideways. Schools and public health organizations giving straight talk about chemical risks help us all make smarter choices about where we live and work.
Nobody wants surprise hazards in the air, water, or their workplace. Science spells out the risks; common sense and sound policy keep those risks from becoming reality.
What is the chemical formula and structure of 3-Buten-2-One?
Understanding 3-Buten-2-One
3-Buten-2-one remains one of those chemicals that pop up in both the lab and industry almost as often as questions about its properties do. The formula for this compound is C4H6O. Anyone who's ever cooked a meal using butter or fried onions has already been exposed to it, since 3-Buten-2-one, or methyl vinyl ketone, contributes those sharp, nutty aromas.
Chemical Formula and Structure
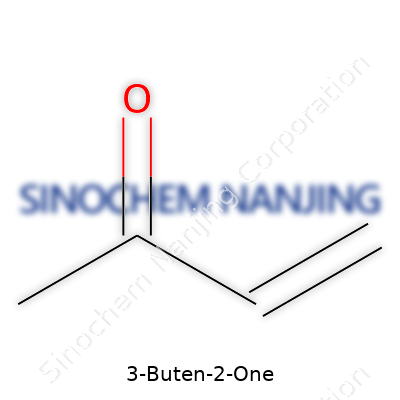
The backbone of this molecule tells a story of chemistry and function. 3-Buten-2-one uses four carbon atoms, arranged in a straight chain. The “buten” fragment means a four-carbon chain with a double bond, while “one” means a ketone group somewhere on the chain.
Draw it out and you get this: CH2=CH-CO-CH3. The double bond sits between the first and second carbons, while the ketone (C=O) is attached to the second carbon. This structure explains its high reactivity—a carbon-carbon double bond alongside a carbonyl group is like giving the molecule two sets of hands, eager to grab reactions from either side.
Why 3-Buten-2-One Matters
3-Buten-2-one takes a seat in more labs than textbooks let on. It works as a crucial building block in making certain pharmaceuticals, pesticides, and flavorings. In the chemical synthesis world, researchers value how easily it reacts. Adding stuff to a double bond or that sharp carbonyl means scientists can design more complex molecules, with fewer steps. That saves resources, time, and costs—three things that really matter, especially when research budgets stay tight.
3-Buten-2-one turns up outside the lab, too. This compound helps create the “cooked” flavor in foods, showing up in tiny amounts during Maillard reactions. Food chemists know that even a trace can change the way something tastes or smells. That's powerful. People concerned about food additives also track how much of this, and similar compounds, get added to processed foods.
Safety and Handling
Dealing with 3-Buten-2-one in pure form calls for major caution. Its double bond and carbonyl make it highly reactive. That's good news in a flask, but risky to skin and lungs. The vapors have a choking, irritating effect that you notice right away. Prolonged exposure can inflame mucous membranes and lungs. Safety data sheets urge anyone using it to work in a well-ventilated space with gloves and goggles.
Years in the lab teach the value of these warnings. Accidents happen quicker than people assume, especially with reactive ketones. Stories circulate of seasoned researchers coughing for days after one careless whiff. Strict attention to safety gear isn’t bureaucracy. It’s practical wisdom from chemists who learned things the hard way.
Solutions and Moving Forward
Scientists and manufacturers continue to search for greener ways to both make and handle chemicals like 3-Buten-2-one. Some look at catalysts to speed up reaction rates with fewer byproducts. Researchers also design better fume extraction systems and push for more robust personal protective equipment. Open communication between researchers, safety officers, and industry leaders leads to fewer injuries and better outcomes for the environment.
Tools and practices evolve, but the basics stick: Know what a molecule can do, stay aware of the risks, and keep asking how processes can be safer and less wasteful. 3-Buten-2-one brings utility and challenge. That mix drives innovation and sharper science on every bench where it gets used.
How should 3-Buten-2-One be stored and handled?
Handling Hazards: Respect What's in the Drum
3-Buten-2-One isn’t an everyday kitchen chemical. It's a harsh compound that can cause dangerous eye, skin, and respiratory irritation. I remember my early days in the lab, where the mere whiff of it meant you had to double-check your gloves and make sure your eye gear was in place. Folks sometimes underestimate vapors. Breathing them can be a real problem, so you have to keep your workspace well-ventilated. If you pour or transfer the chemical without a fume hood, you’re rolling the dice with your health.
Picking the Right Place: Where It Stays Matters
For years, I worked in facilities where careless labeling nearly caused accidents. 3-Buten-2-One needs a cool, dry spot far from sunlight and heat sources due to its flammability and tendency to degrade. Flammable cabinets save lives. Avoid metal shelves that might react with spills. Storage areas need clear signage, fire extinguishers nearby, and zero clutter. Anybody who's ever spilled a volatile liquid in a messy room knows you don’t want to be stumbling around for the cleanup supplies.
No Shortcuts: Use the Right Containers
I’ve seen old paint cans and soda bottles holding dangerous materials, and every time, I cringe. Only keep 3-Buten-2-One in chemically compatible containers, sealed tight. The right cap makes all the difference in keeping vapors contained and moisture out. Invest in containers rated for hazardous liquids; glass or special-grade plastics are best. Cheaping out on containers invites leaks, which risk both personal injury and fire.
Preparing for the Worst: Safety Tools and Training
People trust their luck far too much. I've watched coworkers handle potent chemicals with their bare hands, only to regret it hours later. Gloves, goggles, and lab coats aren’t just for show. Always have a spill kit within arm’s reach and train everyone on the floor to actually use it, not just admire it on the wall. Emergency showers and eyewash stations are worth every inch they take up. No one likes practicing safety drills, but skills lock in when you run through real scenarios.
Regulatory Reality: Know the Rules
OSHA and EPA rules aren't just bureaucratic paperwork – they’re based on grim past experience. Local fire codes often don’t allow unlimited quantities of flammable chemicals. Check material safety data sheets (MSDS), keep the right paperwork handy, and don’t cut corners to save time. Controlled access keeps unauthorized folks out. If you skip these steps, you risk more than fines—people get hurt.
Path to Safer Practice
People often ignore maintenance until something breaks. Regularly check container seals, inspect storage areas for leaks or corrosion, and keep track of how much 3-Buten-2-One you have. If a drum sits forgotten and unlabeled, danger multiplies. Label everything with dates and quantities. Clear handoff policies make sure no one inherits a mystery barrel.
Handling chemicals like 3-Buten-2-One safely isn’t about following procedures for appearances. It's about respect for what you’re dealing with, and for the people who work around you. Safety grows out of habits, education, and the courage to speak up if something looks risky. If the right tools, storage, and methods are in place, you won’t need luck to stay safe.
What are the safety precautions for using 3-Buten-2-One?
Understanding the Nature of 3-Buten-2-One
3-Buten-2-One, also called methyl vinyl ketone, doesn’t make headlines, but anyone who’s dealt with it in the lab knows it packs a punch. The clear liquid has a sharp, pungent smell, something like acrid fruit. It reacts fast—especially with skin and air. Safety isn’t just about ticking boxes here. It’s about keeping your own body out of harm’s way, as I learned the hard way in grad school after a whiff of the vapor left my eyes burning for hours.
Ventilation Is Everything
Nobody enjoys working in a stifling fume hood, but with 3-Buten-2-One, it’s nonnegotiable. The vapors shoot straight for your sinuses. OSHA’s classification places this chemical firmly in the “danger” zone for inhalation, blistering mucous membranes fast. I never risk it and neither should anyone else: if the fume hood’s broken, pack up until it’s fixed. Portable fans or open windows just don’t cut it. Local exhaust is the only real line of defense for your lungs.
Dress Like You Mean It
Cotton lab coats, not polyester blends, handle splash risks best since synthetic fibers can melt and stick to the skin. Long sleeves roll down for a reason, and nitrile gloves are a must—latexes won’t block this stuff, even for a quick pour. Don’t skip out on closed-toe shoes or impact-resistant goggles. I’ve seen a colleague’s regular glasses fail when a droplet managed to ricochet off the beaker. Eyes and lungs don’t get a second chance.
Keep Emergency Tools Within Reach
A working eyewash station sits right next to my bench. Time counts if 3-Buten-2-One finds its way to your face or hands; the ten seconds it takes to scramble for help could decide whether you lose a burn scar or your sight. Chemical spill kits matter, too: vermiculite and proper neutralizing agents, never just paper towels. I store the SDS sheet where anybody can grab it. Knowledge only pays off if you can get to it fast.
Storage and Handling: Cut No Corners
Heat and sunlight break 3-Buten-2-One down or make it explode, so I always tuck bottles in a metal safety cabinet far from heat sources. Metal cans beat plastic because the stuff reacts over time and weakens containers. Every label faces front, nothing sits unlabeled; mistakes with clear liquids lead to accidents. Disposal needs care, too: down the drain isn’t an option. Specialized chemical waste bins, checked weekly, keep surprises to a minimum.
Training and Practice Over Theory
Reading the manual doesn’t stick like the lesson you get from real drills. Safety training only works if it happens in the actual workspace, with equipment you know you’ll use. I’ve always pushed for regular hands-on reviews. Not every job gives that, but teammates tend to pay more attention after handling a simulated spill or a staged emergency. Muscle memory can save more than protocols ever do.
Creating a Culture, Not Just Following Rules
Nobody wants to land in the ER for ignoring a five-minute safety check. Sometimes one person breaking procedure creates problems for the whole lab. Speaking up feels awkward but ignoring unsafe shortcuts can cost someone a lot more. Collective watchfulness makes a place safer than any one expert can manage alone. If you want safety to stick, it takes habits, open conversations, and respect for the risks these chemicals bring every day.
| Names | |
| Preferred IUPAC name | but-3-en-2-one |
| Pronunciation | /ˈθriːˈbjuːtənˈtuːˌoʊn/ |
| Identifiers | |
| CAS Number | 78-94-4 |
| Beilstein Reference | 635246 |
| ChEBI | CHEBI:52741 |
| ChEMBL | CHEMBL13322 |
| ChemSpider | 12070 |
| DrugBank | DB04160 |
| ECHA InfoCard | 100.003.190 |
| EC Number | 211-146-5 |
| Gmelin Reference | 82852 |
| KEGG | C02855 |
| MeSH | D016529 |
| PubChem CID | 7907 |
| RTECS number | EL8575000 |
| UNII | TF3A5C631Y |
| UN number | UN 1224 |
| Properties | |
| Chemical formula | C4H6O |
| Molar mass | 70.09 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | strong, penetrating, unpleasant |
| Density | 0.846 g/cm³ |
| Solubility in water | soluble |
| log P | -0.4 |
| Vapor pressure | 3.7 kPa (at 20 °C) |
| Acidity (pKa) | 6.7 |
| Basicity (pKb) | 7.24 |
| Magnetic susceptibility (χ) | -42.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.421 |
| Viscosity | 0.43 mPa·s (25 °C) |
| Dipole moment | 2.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -116.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1432 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H301, H312, H315, H319, H332, H335 |
| Precautionary statements | P210, P261, P280, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-Buten-2-One NFPA 704: `"2-3-2"` |
| Flash point | 6°C |
| Autoignition temperature | 180°C |
| Explosive limits | Explosive limits: 2.6–15% |
| Lethal dose or concentration | LD50 oral rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 3-Buten-2-One: 140 mg/kg (rat, oral) |
| NIOSH | 201-159 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Buten-2-One: "2 ppm (6 mg/m3) (OSHA Ceiling) |
| REL (Recommended) | 0.03 ppm |
| IDLH (Immediate danger) | 400 ppm |

