3-Bromotoluene: Uses, Synthesis, and Trends
Historical Development
Chemists in the late nineteenth century were constantly looking for new ways to modify simple aromatic compounds, hoping to create versatile building blocks for dyes, medicines, and growing industrial needs. The journey of 3-bromotoluene, also known as meta-bromotoluene, reflects this era of experimentation. Early synthetic chemists learned that swapping out hydrogen for a bromine atom on the aromatic ring could open up new pathways for further modification. This halogenated toluene quickly found a place in organic labs and industry settings, offering more than a simple curiosity on a shelf. As the decades passed, brominated aromatics like this became staples for research and manufacturing, not just in academic settings, but also in any place looking to build something new out of something simple.
Product Overview
3-Bromotoluene stands out as an aromatic compound sporting both a methyl group and a bromine atom attached to a benzene ring at the meta (3) position. Its molecular formula, C7H7Br, shows a neat arrangement that lets it serve as a platform for a surprising number of reactions. It shows up often as a colorless to pale yellow liquid, catching the eye and the interest of chemists for its straightforward structure and willingness to participate in a variety of chemical transformations. Suppliers typically stock this chemical in bulk and research quantities, helping academic and industrial labs tackle problems from the design of drugs to the manufacturing of agrochemicals or specialty polymers.
Physical & Chemical Properties
Curiosity often takes you from a bench with a small vial. 3-Bromotoluene’s boiling point sits around 184–186°C, making it easier to distill than heavier halogenated aromatics. Density hovers close to 1.39 g/cm3 at room temperature—a bit heavier than most hydrocarbons, thanks to the added bromine. It shows low solubility in water, a common trait among aromatic halides, but blends well with most organic solvents such as diethyl ether, chloroform, and benzene, making it a practical partner for many synthetic steps. Its refractive index of nearly 1.57 marks it as unmistakably aromatic. Volatile enough to catch your attention with its faint, sweet, and pungent odor, it never lets you forget you’re working with a real chemical, not a benign lab salt.
Technical Specifications & Labeling
Each bottle of 3-bromotoluene tells a careful story on its label. Purity levels, typically not less than 98%, matter greatly for reproducibility in research or strict process controls in industry. Suppliers print batch numbers and storage guidance, spelling out clear instructions to keep the chemical in tightly sealed containers, away from heat and open flames. Labels flag it as harmful—contact can irritate skin and eyes, so gloves and eye protection are a must. Transport codes and signal words (like “Warning” or “Caution”) leave no doubt about the need for care. For those running precise reactions, gas chromatography and proton NMR data offer reassurance that the liquid inside holds up to its promised quality, which bolsters confidence in the results that follow.
Preparation Method
Making 3-bromotoluene is all about smart chemistry with reliable starting points. The classic method starts with toluene, using controlled bromination to nudge the bromine atom to the right spot. In practice, achieving the meta (3) position takes more than just mixing reagents. Direct bromination of toluene usually favors the ortho and para positions because of the methyl group’s activating effect, so chemists take a workaround: first, introduce a nitro group at the meta position, then reduce it and swap it for bromine using a Sandmeyer reaction. This pathway lets chemists sidestep the issue of uncontrolled substitution and gives a clean product, ready for further transformations. Newer techniques, such as palladium-catalyzed cross-coupling, also crop up in some research or large-scale applications, especially when higher selectivity or step economy is needed.
Chemical Reactions & Modifications
Anyone who has spent hours at the bench knows the real value of 3-bromotoluene comes from its reactivity. The bromine atom acts like a handle, making this molecule a perfect platform for Suzuki or Heck coupling reactions, as well as nucleophilic aromatic substitution. That reactivity turns it into a stepping stone toward a range of products: attach boronic acids to the aromatic ring, transform it into amines or nitriles, and build complex multi-functional molecules for pharmaceuticals, liquid crystals, or organic semiconductors. When it comes to changing the methyl group, oxidation to benzoic acid or side-chain functionalization opens even more routes. Each modification offers new pathways that a creative chemist can take, and the reliability of these transformations secures 3-bromotoluene’s place in any well-stocked synthetic arsenal.
Synonyms & Product Names
Despite the focus on “3-bromotoluene,” this compound has earned a range of aliases in catalogs and textbooks. Some know it as m-bromotoluene, reflecting the meta designation, while others point out the IUPAC name: 1-bromo-3-methylbenzene. Names like 3-methyltoluene or simply bromotoluene crop up in some reference works, but chemical structure drawings always clarify any ambiguity. CAS number 591-17-3 acts as the final authority in ordering or regulatory paperwork, bypassing potential confusion from varied naming conventions.
Safety & Operational Standards
Whether working at the bench or in an industrial plant, safety rules guide every interaction with 3-bromotoluene. This compound, like many halogenated aromatics, can cause irritation to skin, eyes, or respiratory tract. Inhalation of its vapors raises concerns, so effective ventilation and fume hoods are standard. Proper gloves—nitrile or neoprene—offer basic protection. Spills prompt quick containment and disposal in line with hazardous waste protocols. Storage calls for well-sealed containers in a cool, dry spot. Good training and clear communication, backed by up-to-date Safety Data Sheets (SDS), keep things running smoothly even when working with large volumes. Watching colleagues and trainees develop careful habits never gets old, because a moment’s carelessness can ruin weeks of work.
Application Area
3-Bromotoluene’s fingerprints turn up all over industrial and research settings. It appears as a crucial intermediate for manufacturing dyes, pharmaceuticals, agrochemicals, and fine chemicals. Medicinal chemists lean on it as a starting point for crafting new drug scaffolds, especially when they need that meta substitution pattern. It often finds use in the creation of specialty polymers, with the bromine facilitating strong cross-linking or post-polymerization modifications. Research labs exploit its versatility for making ligand libraries and screening chemical space far beyond what unsubstituted toluene would allow. The electronics industry has also taken notice—materials scientists tweak the aromatic ring to tune plastic semiconductors and liquid crystals for displays, sensors, and other advanced devices.
Research & Development
Ongoing research constantly draws fresh potential from 3-bromotoluene. Chemists continue to hunt for greener, safer bromination routes using milder conditions and less hazardous reagents. Alternatives like flow chemistry or photochemical approaches look promising, both for better yields and improved scalability. Efforts also focus on making cross-coupling reactions more tolerant of functional groups, letting researchers assemble more complex molecules without tedious protecting group steps. Computational modeling now accelerates development, predicting how changes to bromotoluene’s structure might shape pharmaceutical activity or electronic usefulness. Real progress shows up in new molecules patent filings and peer-reviewed studies, proving the compound’s importance isn’t fading.
Toxicity Research
Current data suggest that, like other aromatic bromides, 3-bromotoluene raises moderate concern for acute toxicity if inhaled, ingested, or absorbed through the skin. Animal studies point to irritating effects at high doses, with longer-term exposure increasing risk for organ effects if basic safety steps aren’t followed. Environmental studies flag the persistence of halogenated organics in water and soil, and work continues to understand breakdown pathways and bioaccumulation risks. In my time working with this compound, I’ve seen firsthand the need for extra care in waste disposal—labs that respect disposal rules help protect local waterways and air quality far more than they realize. Ongoing research into less toxic manufacturing steps and biodegradable derivatives stands to benefit health and the environment.
Future Prospects
Looking ahead, 3-bromotoluene will only deepen its influence as a platform for new chemistry. Green synthetic routes, better automation for large-scale reactions, and increasingly sophisticated computational tools will shape how labs and industries use this straightforward molecule. Its role in the next generation of drugs, materials, and agricultural chemicals remains secure, partly because it’s easy to modify and adapt for changing needs. Linking this compound’s history, practicality, and versatility gives chemists the tools to solve problems from carbon capture to electronic innovation. Watching how research evolves with fresh ideas and new demands shows the importance of keeping reliable intermediates like 3-bromotoluene close at hand.
What is the chemical structure of 3-Bromotoluene?
Straightforward Chemistry: Understanding 3-Bromotoluene
Anyone who’s spent time in a chemistry lab knows the sight and smell of substituted toluenes. 3-Bromotoluene looks simple on paper, but behind that simplicity lies a structure that says a lot about reactivity and utility in organic synthesis.
Breaking Down the Structure
Start with the benzene ring. Every organic chemist met this six-carbon hexagon early on. Toluene bolts a methyl group (–CH3) to the ring. With 3-bromotoluene, one hydrogen on that ring gets swapped for a bromine atom (Br). The “3” points to the bromine sitting on the third carbon away from the methyl group, following standard ring numbering.
The proper name for this compound uses the IUPAC system: 1-bromo-3-methylbenzene. Its formula reads C7H7Br. Visualize the benzene ring, place a methyl group at position 1, then put a bromine at position 3. Both groups shape how the molecule reacts and interacts with other chemicals.
Why It Matters in Research and Industry
Some might ask what difference a small change like this makes—swapping a single hydrogen for a bromine. In practice, it matters a lot. Bromine atoms on benzene rings serve as functional handles. Chemists can use them to stitch together new molecules with precision, especially in coupling reactions. Think Suzuki or Heck coupling.
In my experience, working with substituted aromatics teaches a person to value stability and selectivity. For example, 3-bromotoluene stands up well to both air and light on a shelf—qualities that make storage and handling easier than dealing with some more reactive organics.
Pharmaceutical development uses molecules like this all the time. Medicinal chemists tweak rings with groups like methyl and bromine to shift activity, solubility, and toxicity. Agrochemical design follows similar logic. It’s not just about what sits on the benzene ring—it’s also about getting one specific isomer, like the “3-bromo” variety, because each isomer interacts differently inside a biological system.
Safety and Handling Facts
Nobody should pour 3-bromotoluene down a drain or handle it bare-handed. Like most brominated organics, it can cause skin irritation and brings environmental risk if spilled. Fume hoods keep vapors out of your face. Wearing gloves, goggles, and lab coats is basic laboratory discipline.
From experience, using a brominated aromatic demands attention to safety. In one teaching lab, careless use left a lasting chemical odor in the workspace for days. Good ventilation and proper disposal keep the lab safe for the next project.
Looking at Solutions for Safer Use
Best practice means training everyone to understand what’s in those brown bottles. Substance-specific labels, clear safety data sheets, and routine waste pickup stop mistakes before they become lab incidents. Universities and companies could keep investing in greener alternatives when possible, but as it stands, nothing replaces the utility of halogenated intermediates in synthesis. Using them with care and respect is part of responsible science.
What are the main uses or applications of 3-Bromotoluene?
Breaking Down 3-Bromotoluene's Place in Chemistry
Plenty of everyday items start out in beakers and flasks in a lab, and 3-bromotoluene fits into that picture. This chemical may not make headlines the way big pharma pills or the latest electronics do, but its mark shows up quietly in crucial manufacturing steps.
Connecting to Pharmaceuticals and Crop Protection
In the world of drug chemistry, 3-bromotoluene stands out as a cornerstone for building blocks. Medicinal chemists count on it as a starting point, kind of like how carpenters use two-by-fours. Through simple tweaks—mostly swaps of a bromine here or a methyl group there—it forms the backbone for making key ingredients in active pharmaceutical compounds. Chemists aiming for anti-inflammatory drugs, antihistamines, or even cancer therapies will turn to derivatives based on this aromatic compound.
Similar chemistry happens in agriculture industries. Many crop protection products trace their roots back to small, versatile chemicals. 3-bromotoluene provides a simple entry point for making herbicides and pesticides. I’ve talked with agronomists who recognize this link even if they never handle it themselves. Their focus lands on cost and reliability, and upstream chemical choices like this one keep their costs predictable.
Driving Synthesis in Dyes and Specialty Chemicals
If you look around at your clothes or packaging, dyes and pigments fill out most of the color. Chemical plants use 3-bromotoluene to produce azo dyes and other colorants. It adds value precisely because it lets chemists plug plain benzene rings with extra atoms—tailoring colors in ways that weren’t possible fifty years ago. The fact that it’s stable but still reactive promotes flexible synthesis, which supports creative new products everywhere, from textile inks to advanced paints.
Material Science: Beyond the Lab Bench
Many new materials get their start with aromatic compounds like this one. Silicon chips and LED lighting rely on high-purity chemicals for doping and etching. In these electronics fields, purity matters as much as performance. I once visited a semiconductor facility where technicians suited up head to toe, tracking the tiniest trace elements. Precursors like 3-bromotoluene feed into those production lines. Small impurities can throw off million-dollar batches, so the chemical’s repeatability and purity are worth their weight in gold.
Challenges and Safer Practice
Some challenges needle the industry. Toxicity and safe handling top the list; this isn’t something you want floating around outside of tightly controlled settings. Companies investing in better labeling, worker training, and robust supply chains go a long way toward limiting risks. Upgraded air filtration and better personal protective equipment cut accidental exposure.
Chemists themselves can help by designing new processes that use less hazardous starting materials or produce fewer byproducts. Green chemistry marches forward as companies notice the pressure from regulators and consumers. In my own projects, shifting to less hazardous reagents not only reduced risks but trimmed costs tied to waste disposal and regulatory paperwork.
Finding Smarter Paths Forward
New research circles around finding alternatives with smaller environmental footprints. Enzymatic methods and catalyzed reactions edge closer to being affordable, even outside of the lab. Collaborative work between suppliers, academics, and manufacturers speeds up this progress. The drive to improve not only reflects competitive industry—it’s about providing the world with safer products from the ground up.
What are the storage and handling recommendations for 3-Bromotoluene?
Why 3-Bromotoluene Demands Respect in Storage
Anyone working with chemicals quickly learns that ignoring basic safety principles can lead to trouble, no matter how routine the compound. 3-Bromotoluene might not sound like the nastiest substance out there, but the real risks surface through daily use. In my years helping labs put smarter protocols in place, ignoring details with aromatic bromides like this one invites setbacks—spills, ruined reagents, even fire scares.
Let’s start with flammability. You won’t see 3-Bromotoluene ignite just from careless handling, but vapor build-up in poorly ventilated spaces puts everyone on edge. I still remember a research group losing weeks of samples after vapors interacted with static discharge. So, smart storage means cool, dry spots, away from direct sunlight or heat sources like radiators. Flammable cabinets rated for organics remain a best bet.
Direct Environmental Risks
I’ve watched as even seasoned chemists make the mistake of uncapping aromatic solvents near open drains. One drop eventually finds its way into local water supplies, and the environmental headaches multiply. 3-Bromotoluene doesn’t dissolve completely in water, but run-off risks pose headaches for wastewater treatment plants. Local guidelines urge tight secondary containment —trays or tubbing to catch leaks. It’s more than a regulatory box to tick; this curbs small errors before they become multi-agency messes.
Personal Safety Routines—A Lesson from the Field
One problem: the tendency to treat aromatic bromides like routine hydrocarbon solvents. They’re not. Gloves degrade fast with repeated contact, so nitrile or neoprene outlast thin latex every time. Eye shields matter. In my own experience, even cautious colleagues have gotten careless pipetting, only to end up rinsing out burning eyes. Even low vapor pressure doesn’t excuse skipping the chemical hood; fumes hang in still air, building up unnoticed over a few hours. I’ve learned that if you’d hesitate to sniff it, it’s better kept behind glass with the blower running.
Fire Hazards and Chemical Compatibility
3-Bromotoluene plays rough with oxidizing agents—think peroxides or concentrated nitric acid. I’ve investigated warehouse fires where someone shelved halogenated aromatics beside oxidizers, one bottle leaked, and then all bets were off. Simple fix: separate storage by chemical class, using clearly labeled shelves and fresh inventory logs. It’s about daily running, not just the poster on the wall.
Solving Practical Problems—Easy Solutions, Big Payoff
Decent room airflow, sealed bottles with intact O-rings, and refrigeration for opened containers—these habits matter more than just manufacturer brochures. Before moving, shaking, or blending, check seals. If labels look faded or mangled, re-label clearly and log the inspection date. That ounce of prevention has saved multiple labs I’ve worked with from expensive disposal bills or safety audits.
When training new techs or students, simple walk-throughs work better than slide decks. Show every risk, from slippery floors after a small spill to the gradual decay of stored product if exposed to light. Stress personal accountability—each user owns the consequences of careless storage.
Bottom Line on 3-Bromotoluene Handling
Every bottle becomes a test of attention. From solid PPE—right gloves, splash goggles, proper lab coats—to keeping careful logs and tightly managing contact with air and moisture, each small habit protects not only the present lab, but the next project down the hall. Good stewardship makes sure today’s experiments don’t turn into tomorrow’s clean-up stories or safety reports.
What safety precautions should be taken when using 3-Bromotoluene?
3-Bromotoluene Isn’t Your Everyday Chemical
Anyone who has logged hours in a chemistry lab knows some chemicals demand extra respect. 3-Bromotoluene, a clear liquid used for organic synthesis and pharmaceuticals, sits firmly in that territory. Safety measures are not optional—they are what keeps accidents off the table.
PPE Does More Than Satisfy Regulations
Gloves, goggles, and a good lab coat form the basic armor. It sounds like a broken record, but skin contact with 3-Bromotoluene can cause irritation. Splashing it in the eye? That turns a regular afternoon into a visit to an eye wash station. Beyond the basics, splash-resistant aprons and face shields matter when handling larger volumes or pouring from clumsy containers.
What’s in the Air Matters
3-Bromotoluene gives off vapors, and those can irritate lungs and nasal passages. Fume hoods aren't lab furniture—they are serious safety gear. Airflow keeps vapor levels low and cuts down risks of accidental inhalation. Each time a hood fan turns on, it delivers peace of mind as well as protection. I once worked a late shift where the hood died midway through a distillation—I had a sore throat for a day and learned quickly to check for a working airflow sensor before starting anything.
Ventilation and the Workspace
Stuffing chemicals in a tiny closet or a windowless back room spells trouble. Decent ventilation spreads out any fumes, reduces exposure, and makes life easier for everyone working nearby. The Occupational Safety and Health Administration (OSHA) points out that limiting exposure is the simplest step to prevent health problems down the road. Effective fume removal always trumps a cracked window.
Storage: Out of Sight, Not Out of Mind
3-Bromotoluene doesn’t play well with oxidizing agents or strong bases. Store it in a cool, dry place, away from incompatible chemicals. If you’ve seen cabinets labeled “flammables only” or noticed those funny-looking yellow safety lockers—that’s where these sorts of solvents should live. Keep containers tightly sealed, and label everything clearly. No one should need a guessing game when it’s time to grab a reagent.
Spill Response Shows Preparedness
Eventually, someone knocks over a flask or a bottle. It pays to know the drill: evacuate the area, use spill kits built for solvents, and never use a shop broom or home mop. My own worst lab day came from fumbling a half-full bottle onto the benchtop; the clean-up waste stayed with our hazardous materials handler until the next disposal run. Soap and water aren’t enough against organic solvents, so make sure those spill kits hold the right absorbents.
Training Beats Overconfidence
No one masters lab safety just by skimming the safety data sheet. Real knowledge comes from hands-on training, by seeing what works and learning from close calls. Workshops, refreshers, and on-the-job walkthroughs help everyone handle chemicals like 3-Bromotoluene the right way. Walking into a lab where folks trust their gear and their team always feels different from a place where people cut corners.
Disposal Isn’t Just About Pouring Down the Drain
Spent 3-Bromotoluene doesn’t get tossed down the sink. Solvent waste builds up fast, and proper disposal protects more than just the immediate staff. Labeled waste containers, set pickup schedules, and tracking usage keeps things legal and keeps regulators satisfied. Mislabeling or mixing incompatible wastes leads to fines and real danger—something no lab can afford.
Respect Earned Through Habit
Lab safety relies on habits, not luck. PPE, fume hoods, good storage, smart spill response—these steps make an average workday routine instead of risky. Treating 3-Bromotoluene with respect keeps work productive and trouble-free for everyone at the bench.
Is 3-Bromotoluene available in various purities or grades?
Why Purity Levels Matter
Having worked in a laboratory where quality control made or broke results, I know firsthand how the smallest trace of impurity in a chemical can throw off a whole synthesis. One compound that comes up regularly in pharmaceutical and material science research is 3-bromotoluene. It’s always in demand for coupling reactions, but people sometimes overlook the fact that not all 3-bromotoluene is created equal. The purity and grade of this compound can change how it behaves in a lab or factory setting.
Different Grades Serve Different Needs
Researchers in fine chemical synthesis usually go for high-purity or analytical-grade 3-bromotoluene. In my own projects, skipping out on the purer version just to save a bit of money often led to head-scratching chromatography data or unwanted side-products. High-purity batches, sometimes reaching 99% or even higher, help keep reactions predictable. These grades often come with a certificate of analysis, which provides a breakdown of what possible contaminants could lurk in the bottle.
Not every process needs the shiniest bottle, though. Some industrial manufacturers use technical-grade 3-bromotoluene for intermediate steps where they fully expect to filter, distill, or otherwise clean up the product downstream. That’s common in scenarios where end-use isn’t tightly regulated, or small amounts of impurity won’t disrupt performance. For example, a batch destined for bulk dye production may tolerate more impurity than pharmaceutical research, where strict regulatory oversight steps in.
Quality, Regulation, and Human Safety
Neglecting purity doesn’t just waste time and money, it can have real safety risks. Contaminants can create toxic byproducts, corrode equipment, or put workers at risk. Pharmaceutical companies face extra scrutiny because their compounds have to meet not just lab standards, but the tough rules from agencies like the FDA or EMA. Labs that use subpar quality for early testing sometimes run into trouble when they move to scale up, realizing too late that impurities quietly complicate analysis or approval processes.
A few years back, I worked with a team that jumped at a “cheaper” supplier for aromatic intermediates. They found residue from leftover halogenated byproducts, which caused serious reactivity issues and prompted a costly recall of half a year’s worth of pilot-scale runs. Cleanup and paperwork were just part of the fallout—the project’s timeline took a hit, and trust with downstream partners frayed.
Supporting Reliable Sourcing and Transparency
Suppliers who put transparency up front stand out. Reputable producers include batch-specific documentation, third-party certifications, and offer customer support for regulatory documentation. Traceability lowers the chance of mishaps due to mysterious contaminants. Customers benefit from knowing exactly what's in their flask or reactor.
Improving industry standards comes down to knowledge sharing and communication between buyers, chemists, and suppliers. Asking smart questions about a chemical’s source, impurity profile, and production methods builds confidence and keeps surprises to a minimum. A healthy market supports both high-purity research and scalable industrial use, with clear choices for each kind of application.
Better Outcomes Through Smart Choices
Pick the grade of 3-bromotoluene that lines up with the need at hand. Regulatory compliance, reaction reliability, and cost-efficiency all hinge on the right level of quality. That lesson has paid off many times in my own work and in the industry at large—choose wisely, and the chemistry takes care of itself.
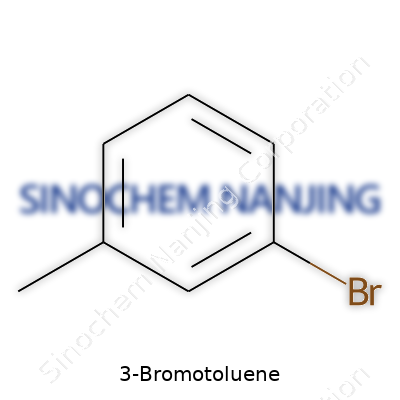
| Names | |
| Preferred IUPAC name | 1-Bromo-3-methylbenzene |
| Other names |
Bromotoluene m-Bromotoluene 3-Methylbromobenzene 1-Bromo-3-methylbenzene |
| Pronunciation | /ˈθriː ˈbroʊ.moʊ.tɒl.juːiːn/ |
| Identifiers | |
| CAS Number | 591-17-3 |
| 3D model (JSmol) | `JSmol.loadInline("data/model/pdb/3-Bromotoluene.pdb");` |
| Beilstein Reference | 1209226 |
| ChEBI | CHEBI:84277 |
| ChEMBL | CHEMBL15404 |
| ChemSpider | 7412 |
| DrugBank | DB04115 |
| ECHA InfoCard | 100.006.829 |
| EC Number | 209-207-0 |
| Gmelin Reference | 5662 |
| KEGG | C14350 |
| MeSH | D017935 |
| PubChem CID | 7237 |
| RTECS number | XS9625000 |
| UNII | 48A5M73I5V |
| UN number | NA1993 |
| Properties | |
| Chemical formula | C7H7Br |
| Molar mass | 185.04 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Aromatic |
| Density | 1.395 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 3.9 |
| Vapor pressure | 0.45 mmHg (25 °C) |
| Acidity (pKa) | Acidity (pKa) of 3-Bromotoluene: "41 |
| Basicity (pKb) | 6.2 |
| Magnetic susceptibility (χ) | -73.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.539 |
| Viscosity | 0.85 mPa·s (20 °C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 129.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 49.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –4490.7 kJ/mol |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 74 °C |
| Autoignition temperature | 571°C |
| Lethal dose or concentration | LD50 Oral Rat 3000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 4,400 mg/kg |
| NIOSH | B0145 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 250 mg |
| Related compounds | |
| Related compounds |
Bromobenzene Bromotoluenes 2-Bromotoluene 4-Bromotoluene Toluene |

