3-Bromopropionic Acid: A Real-World Look at a Niche Chemical
Historical Development
3-Bromopropionic acid came out of early 20th-century organic chemistry as researchers explored new ways to add functionality to basic carbon chains. Historical records show it emerged from work aimed at understanding carboxylic acids and their halogenated derivatives. As laboratories around the world honed synthetic methods, handling halogenated acids grew easier and safer. Academic curiosity and industrial need drove demand for selective alkyl chain halogenation, opening the door for tailored building blocks like this one. Its clinical spotlight flickered brighter in the 1990s, when scientists investigated glycolytic inhibitors for cancer therapy and sought alternatives to more toxic agents.
Product Overview
3-Bromopropionic acid usually appears as a colorless to pale yellow crystalline solid or thick liquid, with a strong, irritating smell reminiscent of pungent acetic acid mixed with sweetness. Chemists and technical staff know it under several names and recognize its potential as a small, reactive tool for introducing bromine and carboxy groups into organic molecules. It enters supply chains in laboratory-grade bottles ranging from small vials for academic investigation to larger quantities for specialty synthesis in pilot plants or pharma research.
Physical & Chemical Properties
Anyone handling this acid quickly notices how well it dissolves in polar solvents—especially water, ethanol, or acetone—making it a flexible intermediate for many synthesis strategies. The solid melts near 35 °C, but under standard lab temperatures, it typically softens and forms an oily layer that clings to glassware. Its boiling point sits around 204-206 °C, but strong hydrogen bonding and protic character mean it doesn’t evaporate eagerly below that. As a monobromo carboxylic acid, it readily donates protons in basic conditions and offers a handy electrophilic site on the bromo-carbon, setting up a playground for substitution and elimination reactions. Chemists appreciate the distinct IR and NMR signatures, which confirm both purity and substitution pattern during routine quality control.
Technical Specifications & Labeling
Bottles carry hazard diamonds and R/S codes that immediately warn chemists about its irritant properties and the dangers of possible inhalation or skin exposure. Purity varies but generally stays above 98% for research or specialty chemical purposes. Trace impurities often include hydrolyzed products or the starting materials left from its synthesis process. Labels list chemical abstract numbers, batch data, synthesis and shelf-life timelines, and proper disposal instructions. Shelf stability depends on moisture control and light protection, underscoring the need for careful inventory management to prevent decomposition or unwanted side reactions.
Preparation Method
Most of the world’s 3-bromopropionic acid supply comes from controlled halogenation of propionic acid or its derivatives. In the lab, the propionic acid undergoes substitution using phosphorus tribromide or hydrobromic acid, often with temperature and solvent carefully calibrated. Some processes favor bromination of the methyl group on propionic acid under radical conditions, followed by rapid purification using distillation or crystallization. Manufacturers tweak these methods to limit byproducts, like dibromo or polymerized materials, and to improve yield. Reaction vessels and fume hoods remain essential, as the process releases acidic vapors and can rapidly escalate if moisture control slips.
Chemical Reactions & Modifications
This acid stands out as a flexible starting material in both academic and applied organic synthesis. Its bromo group activates the adjacent carbon for nucleophilic attack—making it possible to introduce new amines, thiols, or alcohols in one-pot conversions. Synthetic chemists use it to create carboxylic acid derivatives, turning it into esters, amides, or salts depending on the downstream need. It reacts readily with bases, but the bromo position still allows for substitution or elimination, so the same molecule delivers a wide toolkit in a single flask. In medicinal chemistry, modifying the acid tail with diverse nucleophiles leads to new compounds for further testing, serving as a launch point for lead optimization in drug discovery.
Synonyms & Product Names
You’ll find 3-bromopropionic acid listed under plenty of synonyms: β-bromopropionic acid, 3-BPA, and even propionic acid, 3-bromo-, especially in regulatory and patent literature. These alternate names matter when ordering, as catalog numbers and supplier databases often store them under each alias. In the research and regulation world, it also crosses paths with its sodium salt (3-bromopropionate sodium), which often shows up in biological assay work or metabolic studies.
Safety & Operational Standards
After years in laboratories, one learns to respect halogenated acids like this. Safety documentation makes the risks clear: 3-bromopropionic acid can burn skin and eyes, create breathing hazards as an aerosol, and pollute groundwater if spills aren’t handled properly. Labs enforce gloves, face shields, and strict ventilation. Waste must go offsite for specialist disposal—neutralizing acidic material before discharge remains a top priority. Spills prompt immediate use of spill kits, and many teams keep calcium carbonate or sodium bicarbonate on hand for neutralization. Occupational safety guidelines demand meticulous record-keeping, exposure monitoring, and emergency protocols, since insurance and lab audits rely on this evidence.
Application Area
The molecule sees its most active use as a specialty intermediate in pharmaceuticals and biomedical research. Its chemical reactivity suits enzyme inhibition and metabolic pathway studies. In disease research, 3-bromopropionic acid turns up as a glycolytic inhibitor in cellular models of cancer, with particular focus on targeting tumor metabolism. This application’s gained traction as the search for targeted cancer therapies intensifies. Medicinal chemists synthesize analogs for further pharmaceutical screening, tweak the acid for improved delivery, and use it as a scaffold for more complex drug candidates. Technical teams in chemical manufacturing rely on it for custom syntheses, producing small-molecule building blocks or labeled compounds for analytical calibration.
Research & Development
Inside the R&D world, teams push 3-bromopropionic acid’s reactivity to expand chemical libraries, find novel cancer inhibitors, and create reference standards for high-precision measurement. Chemical biologists modify ring systems and side-chains using this building block. Investigators at research hospitals and universities study cell metabolism in detail, using this acid’s glycolysis inhibition properties to dissect biochemical pathways. Companies invested in synthetic methodology treat it as a case study for green synthesis improvements, testing out new reaction conditions that minimize toxic byproducts or maximize atom economy.
Toxicity Research
Glycolytic inhibition underpins both the promise and peril of 3-bromopropionic acid. Animal and cell studies demonstrate strong cytotoxicity against cancer cells—but with off-target risks in healthy tissue, especially in organs sensitive to energy disruption like the liver and brain. Clinical research flags dose-dependent toxic effects, and regulatory review stresses careful monitoring of exposure times, delivery methods, and downstream metabolites. Reliable toxicological data anchor all decisions about workplace limits, environmental release, and therapeutic candidates, so scientific transparency and open publication matter. Long-term ecological effects remain under evaluation, especially as more labs test similar molecules for broad applications.
Future Prospects
Looking ahead, demand for new cancer metabolism therapeutics and advanced chemical synthesis will keep 3-bromopropionic acid in circulation. Researchers explore analog molecules to reduce toxicity while maintaining potency, and chemical supply firms develop cleaner, less hazardous manufacturing routes. Green chemistry innovations around bromination and carboxylation methods offer hope for safer, more sustainable production. Increasing regulatory scrutiny on toxic intermediates drives investment in closed systems and digital traceability for supply chains. A tightening focus on both efficacy and occupational safety presses academic and industrial communities to share best practices, improve standards, and find safer alternatives for hazardous synthons. As focus on precision medicine grows, compounds that let scientists probe or tweak biochemistry at a granular level—like 3-bromopropionic acid—will stay valuable, so long as health, safety, and stewardship keep pace.
What is 3-Bromopropionic acid used for?
How 3-Bromopropionic Acid Steps Into the Spotlight
3-Bromopropionic acid remains a name many people outside laboratories barely recognize, yet scientists keep coming back to it. I’ve spent years talking to folks in research labs and the pharmaceutical sector, where people always hunt for something that can tip the balance in tough fights, like cancer. That’s where this compound starts to get interesting.
Connecting Science to Real Life: What’s the Point?
Researchers keep asking themselves how to slow down or kill cancer cells. Turns out, these cells eat up a huge amount of sugar to keep thriving. They run on a process called glycolysis. 3-Bromopropionic acid messes with this sugar-eating pathway. So rather than attacking healthy cells, it goes after the tumor’s weak spot — energy production. That feels like a big deal, because targeting cancer metabolism could sidestep the toxic side effects of many traditional chemotherapy drugs.
Studies in cells and animals show that 3-bromopropionic acid knocks out enzymes involved in glycolysis, especially hexokinase II. Scientists find this promising for both aggressive brain tumors and other fast-growing cancers. Researchers from Johns Hopkins, among others, highlight survival gains in laboratory models, suggesting far-reaching potential.
Medical Promise Raises Real-World Questions
Even with these breakthroughs, the clinical side still faces roadblocks. Doctors worry about safety. 3-bromopropionic acid can hurt healthy tissue if not handled properly. Pharmaceutical teams wrestle with dosing and delivery methods. That’s been a huge hurdle with cancer drugs forever — striking tumors without damaging the body. Some research groups believe that wrapping this compound in nanoparticles or using targeted delivery might shield healthy cells. This technology feels like science fiction, but trials are ongoing.
Beyond Cancer: More Doors Open
Beyond oncology, there’s movement to explore antifungal uses. The same way it pauses cancer metabolism, it disrupts energy systems in certain pathogens. For fungal infections that resist common medications, this could open lifesaving options. On the chemistry and biotech front, it shows value as a building block for more complicated molecules. Scientists sometimes use it to make other drugs or specialty chemicals that end up in agriculture, lab research, or niche industries.
What Keeps Progress Moving?
Regulators keep a close watch on any new drug, especially those that tinker with basic cell processes. That’s probably a good thing, because patient safety trumps speed. Progress depends on transparent research and doctors sharing raw trial data. The National Institutes of Health and various global agencies call for open data sharing so patients know the real risks and science doesn’t get cut corners. Teams in Europe, Asia, and the U.S. push for clinical trials, but also for fair access if something eventually works out.
Paths Forward
Today, hope often rides on new directions in cancer therapy and infection control. With 3-bromopropionic acid, the journey from laboratory finding to safe medication still stretches out in front of us. If researchers crack the challenge of targeting sick cells while protecting healthy tissue, millions worldwide could benefit. Open, honest science and strong oversight drive those odds forward, and make the difference between a chemical curiosity and a lifesaver.
What are the safety precautions when handling 3-Bromopropionic acid?
Why 3-Bromopropionic Acid Demands Caution
3-Bromopropionic acid falls into a class of chemicals where respect for personal safety isn’t optional. This stuff brings both corrosive and toxic elements to the table, and mishandling it can have serious consequences. I’ve spent years in research labs, and I can spot trouble before it shows up as an emergency room visit. No one should underestimate a compound just because it looks like a simple white powder.
Direct Contact Calls for Immediate Attention
One splash, and your skin can burn or turn red fast. The acid eats through gloves that aren't built for chemical warfare. Eyes feel the pain almost instantly, causing long-term injury. I once saw a colleague brush against a drop with their bare wrist, thinking only a rinse would solve it. That mindset sent them to get medical attention. Chemical exposure isn’t a learning opportunity—it’s a risk that often doesn’t come with warnings.
Breathing It In: More Than a Bad Smell
Goggles and gloves only go so far, especially if 3-Bromopropionic acid becomes airborne. A hurried transfer or clumsy pour can send a sharp, nose-burning vapor into the air. It irritates airways and can make you dizzy. I’ve learned that good ventilation isn’t a luxury, but a requirement. Fume hoods weren’t designed for aesthetics. Proper airflow keeps invisible threats from settling in your lungs.
Smart Storage Prevents Unwanted Surprises
Keeping this acid in sealed, chemically resistant containers is the kind of boring routine that saves fingers, eyes, and careers. Store it away from bases and strong oxidizers. The wrong shelf mate can spark a reaction that does more than mess up your experiment. Storing chemicals neatly, labeled in secondary containment, keeps reactions where you want them: inside the flask, not across the bench or floor.
Preparation Beats Panic
I always check the safety data sheet before cracking open a new bottle, even when I think I know the drill. Regulations around PPE—such as wearing chemical-resistant gloves (nitrile, not latex), goggles that seal, and long sleeves—are more than rules; they’re lines of defense. Once, during a hot summer, I considered ditching the lab coat. A reminder from that earlier accident changed my mind, and it’s served me well. Sweat is easier to wash than chemical burns.
Waste Management Keeps Hazards Under Control
After using 3-Bromopropionic acid, waste should not go down any ordinary drain. Collecting it in a properly labeled container, as instructed by local hazardous waste procedures, eliminates headaches for colleagues and for the folks handling disposal later on. One lab I worked in had a scare from poor labeling that led to a spill; it taught everyone real fast how much trouble sloppy habits can bring.
Training Makes All the Difference
Nobody is born knowing how to handle risky chemicals. Regular safety drills, reviewing protocol, and keeping first aid kits within reach save lives. I found that hands-on training sticks in your mind more than the longest manual. Practicing spill response, proper disposal, and emergency eyewash procedures builds muscle memory that shows its value in a crisis.
Culture of Accountability Keeps Us Safe
3-Bromopropionic acid reminds every chemist I know that confidence can’t outsmart carelessness. Pulling up the SDS, suiting up with the right gear, and handling waste properly don’t just keep one person safe—they protect everyone in the lab. I make it a habit to remind colleagues about the basics, because safety slips the moment it’s treated like a chore.
What is the chemical structure and molecular weight of 3-Bromopropionic acid?
Understanding the Skeleton: Chemical Structure
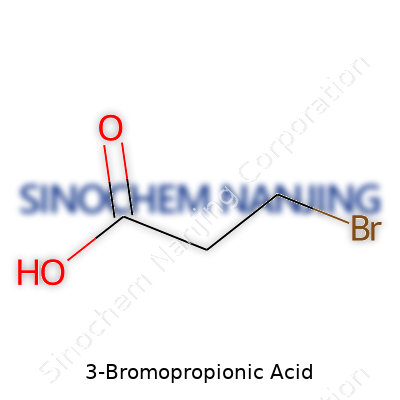
Looking at 3-bromopropionic acid, it becomes clear how a few atoms make all the difference. At its core, this molecule holds a three-carbon chain, topped at one end with a carboxylic acid group and at the other, a bromine atom tags along. Chemists label it as C3H5BrO2. Visually, you lay out the backbone with two carbons in between, one linking the acid group (–COOH) and the other pointing to bromine (–Br). The structural formula reads: Br–CH2–CH2–COOH.
Molecular Weight: More Than Just a Number
For research, molecular weight shapes dosing, measurement, and safety. 3-bromopropionic acid weighs in at about 167.98 g/mol. Most scale calibrations and handling calculations rely on this single value. Standard chemistry classes teach students that this number comes from tallying atomic weights: carbon (12.01 × 3), hydrogen (1.01 × 5), bromine (79.90), and the two oxygens (16.00 × 2), leading to precision during synthesis and application.
Context Shapes Importance
Anyone working in a synthetic chemistry lab meets these molecules not on paper, but in flasks and beakers. 3-bromopropionic acid attracts attention because it acts as a building block, finding uses in research as a metabolic inhibitor or an intermediate for pharmaceuticals. Its structure paves the way for various reactions, from simple acid-base to nucleophilic substitutions, because the bromine serves as a reactive spot. Nothing beats knowing what you’re holding, especially with hazardous digits like bromine attached—both for safety and for yield.
Supporting Facts: Handling, Health, and Ethics
Chemical safety data sheets point out that 3-bromopropionic acid needs careful handling. The presence of bromine increases reactivity, demanding solid protocols: gloves, proper labeling, and good ventilation stand as minimums. Exposure can cause irritation or worse, so even a small miscalculation from ignoring its chemical weight or structure might lead to overexposure or contamination. Studies in cancer research once spotlighted this acid as a potential glycolytic inhibitor, underlining that knowing a molecular weight isn’t just homework—it can steer drug development.
Responsible Practices Encourage Safer Labs
Relying on trusted sources—textbooks, peer-reviewed journals, MSDS certificates—backs each step of chemical work with credibility. Transparency and accuracy matter, no matter how routine a procedure seems. Training new chemists using real case studies drives home how much accuracy shields against mistakes. Personal experience confirms: the best chemistry teachers double-check weights and labels, staying humble because one error can throw off results or risk safety.
Next Steps: Solutions Rooted in Science
Both students and professionals need more than a list of formulas. Reliable training, sharp safety habits, and routine checks on chemical inventories reduce risk. Moving lab classes toward evidence-based protocols, emphasizing the why behind ingredients, grows respect for these details. Fact-checking every label and calculation helps avoid costly, sometimes dangerous, errors. At the end of the day, science demands both precision and a little humility.
How should 3-Bromopropionic acid be stored?
Understanding the Risks
3-Bromopropionic acid, a clear or slightly yellow liquid, smells a bit like acetic acid, but you probably notice it more for its punch in the lab. Its main danger comes from its corrosive nature—it can burn skin and eyes, and isn’t something anyone wants in their lungs. My first real introduction to this compound happened in a university lab, when a fellow student tried transferring it outside of the fume hood. That mistake cost him a week of classes. This acid messes with tissue on contact, and a little spill goes a long way.
Why Moisture and Heat Spell Trouble
Moisture doesn’t just dilute 3-bromopropionic acid—it can lead to unwanted reactions. Leaving a bottle open can shorten shelf life and damage labeling, too. Excess heat increases vapor pressure, so vapors leak out and put everyone at risk. Years ago, an improperly sealed sample sat near a radiator over a university break and filled the air with acrid fumes. The memory lingers, both in scent and in policy—now, acids live far from heat or direct sunlight.
The Right Containers Really Matter
Glass bottles or sturdy plastic containers with well-fitted lids offer strong barriers. Any weak seal or damaged bottle cap turns minor leaks into major emergencies. Each supplier recommends the original bottle for a reason: the material stands up to corrosion and doesn’t let the acid degrade over time. I’ve seen improvising go wrong—an old soda bottle gave out and ruined a small cabinet.
Keep It Out of Reach and in the Dark
No matter how experienced someone is, accidents happen with chemicals left in high-traffic spots. A dedicated chemical storage cabinet, with lock and ventilation, cuts down the chance of accidental exposure. Storing 3-bromopropionic acid with strong oxidizers or bases turns a hazard into a potential disaster. I once watched a senior researcher double check the shelf labels before closing up for the weekend—a simple habit that pays off every time.
Label Everything, Log Everything
A clear, legible label prevents confusion and saves time in an emergency. Dates matter—older acids can change and get riskier with time. Up-to-date logs show who last handled each bottle and when, so nothing sits ignored until it’s too late. It’s a lesson I picked up after a forgotten vial nearly threw off a research project. Keeping simple records on hand means surprise incidents get traced fast.
Handling Spills and Exposures the Right Way
Gloves, goggles, and lab coats stand between skin and dangerous acids. A spill is no time to cut corners—you need a proper spill kit handy, and quick access to running water or an eyewash station makes all the difference. One professor taught us to rehearse responses, not “just in case,” but because close calls don’t give warning. Poster guides near storage areas keep everyone sharp, even after long workdays.
Better Training, Fewer Accidents
Most mistakes with hazardous chemicals trace back to missing knowledge or shortcuts. Refresher safety sessions, honest conversations after mistakes, and clear checklists for storage routines go further than any rulebook. I’ve found that open, pressure-free talks create smarter, safer lab habits. Ensuring staff and students understand not just how, but why proper storage matters, builds respect for the risks and helps everyone walk away in one piece.
Is 3-Bromopropionic acid available in different purities or grades?
Ask anyone in a chemistry lab about 3-bromopropionic acid, and you’ll hear all sorts of stories about how much difference a small impurity makes. This chemical isn’t something you grab by the barrel without a thought. The way it gets used in research, pharmaceuticals, and industrial processing puts the spotlight on purity every time, and not just for the sake of ticking boxes. Safety, accuracy, and cost hang in the balance.
Why Purity Levels Matter in Practice
Take a synthetic organic chemist working on a new compound. A batch of 3-bromopropionic acid with just a few percent impurity can throw the whole outcome into chaos. Perhaps it’s a trace of water or leftover starting material from the manufacturer’s process. The reaction may not work at all or turns out extra byproducts, wasting time and money. Purity directly shapes the success of months of planning in the lab.
Drug-makers face even higher stakes. Regulators like the US FDA and EMA don’t leave much room for guessing when lives depend on quality. Pharmaceutical-grade ingredients often get tested down to parts per million for certain contaminants. Even a small batch of acid that doesn’t meet strict guidelines can land a factory in regulatory hot water, or worse, hurt patients relying on those medicines.
The Different Grades You’ll Find
3-bromopropionic acid rolls out in different grades to meet these broad demands. For casual lab use or industrial synthesis, technical grade is pretty common. This version comes with reasonable purity, suitable for situations where a bit of extra residue doesn’t risk failure. A bottle labeled “technical” might do fine for cleaning glassware or testing some early ideas.
Step up to analytical grade, and it gets a lot cleaner. Suppliers invest in extra purification and testing so scientists can trust their measurements down to the smallest decimal. No one wants a jumbled chromatogram just because a supplier got sloppy. This grade finds its way into quality control labs and research settings where precision is the difference between discovery and dead ends.
Pharmaceutical grade sits at the top. Producers need to use strict protocols, rigorous documentation, and validation processes. Auditors can walk in and ask for records tracing every bottle back to the starting materials. Healthcare depends on this reliability, but so does any regulated industry that prizes repeatability and traceability.
What Purity Means Beyond the Label
I’ve chatted with chemists who get by with technical grade for bulk reactions, saving money where they can. Still, I know more than a few researchers who lost weeks of progress due to an unexpected impurity, sometimes from a batch they’d trusted for years. Trust gets built not just on certificates of analysis, but on real-life results and the willingness of suppliers to answer questions—or provide extra details when rolls of the dice aren’t in the budget.
The price gap between grades can look steep, but headaches from failed experiments or rejected batches aren’t cheap either. Good suppliers publish full test results, clearly state what contaminants might be present, and update their customers when anything in the process changes. This transparency arms researchers and manufacturers with the knowledge to pick the right product every time.
Building Better Trust in Chemical Supply
There’s plenty of room for improvement. Sharing detailed impurity profiles, confirming batch consistency, and supporting questions before and after the sale all make a difference. Buyers get more confident, projects run smoother, and mistakes take fewer casualties. More open conversations between users and suppliers pushes the whole field toward safer, higher-quality choices. That benefits everyone downstream, from university students in the lab to patients counting on safe, effective medicine.
| Names | |
| Preferred IUPAC name | 3-Bromopropanoic acid |
| Other names |
3-BPA Propanoic acid, 3-bromo- 3-Bromopropanoic acid β-Bromopropionic acid |
| Pronunciation | /ˌθriːˌbroʊmoʊproʊˈpɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 590-92-1 |
| Beilstein Reference | 1206914 |
| ChEBI | CHEBI:63915 |
| ChEMBL | CHEMBL109188 |
| ChemSpider | 71475 |
| DrugBank | DB07715 |
| ECHA InfoCard | 100.012.686 |
| EC Number | 211-678-5 |
| Gmelin Reference | 8115 |
| KEGG | C01027 |
| MeSH | D001997 |
| PubChem CID | 6578 |
| RTECS number | TY8400000 |
| UNII | W9N3XPP4YY |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | UFCQGCVYYJFCSM-UHFFFAOYSA-N |
| Properties | |
| Chemical formula | C3H5BrO2 |
| Molar mass | 167.02 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | pungent |
| Density | 1.636 g/cm3 |
| Solubility in water | Soluble in water |
| log P | 0.5 |
| Vapor pressure | 0.00909 mmHg at 25°C |
| Acidity (pKa) | 4.50 |
| Basicity (pKb) | 1.84 |
| Magnetic susceptibility (χ) | -52.3e-6 cm³/mol |
| Refractive index (nD) | 1.463 |
| Viscosity | 2.2 mPa·s (20 °C) |
| Dipole moment | 2.1734 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 120.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -504.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1083.3 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314 |
| Precautionary statements | P260, P264, P270, P271, P273, P301+P312, P302+P352, P304+P340, P305+P351+P338, P311, P321, P330, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | **3-1-2-W** |
| Flash point | Flash point: 113 °C |
| Autoignition temperature | 220°C |
| Lethal dose or concentration | LD50 oral rat 703 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 177 mg/kg |
| NIOSH | Inconclusive |
| PEL (Permissible) | Not established |
| REL (Recommended) | 20 – 25°C |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
2-Bromopropionic acid 3-Chloropropionic acid Propionic acid 3-Bromopropionitrile 3-Bromopropan-1-ol |

