3-Bromophenol: Chemistry, Development, and Prospects
Historical Development
Chemists have worked with phenolic compounds for more than a century, searching out new ways to modify their structure and harness new functionalities. 3-Bromophenol started gaining traction in the late 19th and early 20th century, especially after the commercial scale production of bromine made aromatic bromination practical. Back in those days, advances came fast as researchers learned to control reactions more precisely—making products like this in a more predictable and reproducible way. Scientific journals tracked updates as synthetic strategies improved, giving labs and manufacturers reliable access to pure 3-bromophenol for the first time. Over decades, demand tracked closely with surges in dye chemistry, new pharmaceutical candidates, and the needs of pesticide research.
Product Overview
3-Bromophenol shows up in labs and industry as a versatile intermediate. Chemists value the bromine atom sitting on the meta position of the phenolic ring, since it opens up unique pathways for further modification. Its presence helps introduce the functionality necessary for more complex molecules, or for tuning the reactivity of a reaction—vital for applications in organic synthesis, biochemical testing, and in a variety of fine chemical products. A key selling point revolves around its purity, since impurities can derail sensitive transformations further down the line. Labs routinely find it in powder or crystalline form, with a strong, unpleasant, often pharmaceutical-like odor.
Physical & Chemical Properties
3-Bromophenol exists as a white to light tan powder or flaky solid, melting between 32°C and 36°C. Its molecular formula is C6H5BrO, and it weighs in at about 173 grams per mole. Solubility stands out as an important feature: it dissolves well in common organic solvents, like ethanol, diethyl ether, and chloroform, but much less so in water. The substance emits distinct, piercing fumes—many will remember the sharp smell from early lab work. As a weak acid, its phenolic hydroxyl group reacts with bases, while the bromine provides a ready leaving group for classic substitution or coupling reactions. The compound should always be stored away from heat and light to prevent degradation or unwanted side reactions.
Technical Specifications & Labeling
3-Bromophenol arrives with detailed labeling for purity, hazard classification, and handling instructions. Most sources guarantee at least 98% purity, and packages display batch analysis, melting point range, and critical impurity data. Modern labels also carry signal words and GHS pictograms, marking it as hazardous to both health and the environment. Specific warnings address inhalation, skin, and eye risks, along with emergency first aid steps. Barcodes or QR codes often link to digital safety data sheets, offering immediate access to in-depth handling and storage advice for professional settings.
Preparation Method
The go-to synthesis route relies on electrophilic aromatic substitution. Starting from phenol, bromination in the presence of a controlled amount of bromine and water yields mono-brominated products, with temperature and reagent ratio dictating which isomer forms. Skilled chemists guide the reaction to favor the 3-position, blending temperature control, mild oxidizing agents, and buffering to minimize unwanted dibromo or tribromo byproducts. Afterward, purification calls for recrystallization or column chromatography to isolate pure 3-bromophenol. In industry, the emphasis falls on efficiency—recovering and recycling solvents, precise reaction monitoring, and always aiming to minimize emissions or waste.
Chemical Reactions & Modifications
In practical synthesis, 3-bromophenol serves as a foundation for Suzuki couplings, where the bromine atom helps link aromatic rings using palladium catalysts and boronic acids. This method has proven indispensable in drug development, letting chemists build more complicated molecules from accessible building blocks. Nucleophilic substitution reactions replace the bromine with other groups, changing properties without complicated multi-step syntheses. The phenolic group—thanks to its reactivity—can be protected, alkylated, or acylated as needed. This chemical agility makes 3-bromophenol an essential structure in custom synthesis, material science studies, and analytical method development.
Synonyms & Product Names
3-Bromophenol wears several alternative names, especially as product catalogs draw customers from different regions. Common synonyms include meta-bromophenol, m-bromophenol, and 3-hydroxybromobenzene. International trade identifies the compound through CAS number 591-20-8, while safety or shipping documentation might also reference EINECS or other registry numbers. No matter the name, reputable suppliers maintain traceability back to original certificates of analysis, so chemists feel confident sourcing the right substance for mission-critical work.
Safety & Operational Standards
People who work closely with 3-bromophenol quickly learn the importance of robust lab safety procedures. Direct skin or eye contact brings risk of irritation, and vapor inhalation causes headaches or respiratory discomfort. Gloves, goggles, and chemical fume hoods serve as essentials. Given the environmental toxicity, especially toward aquatic organisms, strict disposal rules apply. Labs follow regulatory frameworks such as OSHA, REACH, and relevant ISO guidelines, keeping handling and storage well within safe limits. Emergency plans, air monitoring, and regular safety audits reinforce best practices—especially as labs increasingly focus on occupational health and environmental stewardship.
Application Area
3-Bromophenol’s reach stretches into pharmaceuticals, polymers, and biochemistry. Early-stage drug discovery programs value it for scaffold diversification, helping teams generate libraries of candidate molecules. In materials science, custom polymers emerge from structures built around the phenol core, with the bromine atom playing a critical role in crosslinking or introducing new functions. Environmental chemists have also picked it up for analytical methods: as an internal standard, tracer, or calibration chemical in complex testing scenarios. In industrial settings, its unique reactivity supports the creation of specialty dyes, and some cases show promise in agrochemical discovery paths—though increased regulatory hurdles add hurdles to environmental testing there.
Research & Development
Modern R&D teams study 3-bromophenol not just for its direct applications, but as a point of access to new chemical space. Coupling reactions have become more reliable, with better catalysts driving up yield and selectivity. Analytical chemists check its behavior in liquid chromatography, searching for impurities in high-value batches. Academic groups often use it to model fundamental reaction mechanisms, teaching the next generation of synthetic chemists the ins and outs of substitution and directed ortho metalation. As green chemistry principles spread, researchers chase ways to cut hazardous byproducts, swap out troublesome reagents, and recycle solvents more effectively—all with the aim of making every step safer, more cost-effective, and sustainable over time.
Toxicity Research
Toxicology profiles show that 3-bromophenol can pose real risks if mishandled. Acute exposure irritates tissues; high concentrations bring concerns for central nervous system depression and potential liver or kidney distress. Chronic studies on rodents suggest low-level hazards, especially with repeated dermal exposure. Environmental research points to aquatic toxicity—small doses can impact fish and invertebrates, and breakdown products may persist in waterways longer than some would expect. Responsible labs take these findings seriously, implementing containment, closed-system synthesis, and regular training aimed at minimizing accidental spills or airborne exposure. Monitoring and risk assessments remain front-line tools for any research team handling sizable quantities.
Future Prospects
Looking ahead, 3-bromophenol occupies a space shaped by ongoing innovation and mounting safety expectations. As synthetic techniques sharpen, new routes promise to reduce waste, increase selectivity, and offer greener alternatives to traditional bromination. Demand in pharmaceuticals and materials research looks set to rise, with cutting-edge projects searching out fresh uses for phenolic building blocks. With regulatory landscapes changing, manufacturers and academic labs need to stay proactive—refining safety practices, prioritizing environmental responsibility, and sharing findings across industries. As I see it, the core challenge lies in translating technical chemistry advances into sustainable industrial realities, balancing practical need with occupational and public health safeguards for future generations.
What is the chemical structure of 3-Bromophenol?
A Close Look at 3-Bromophenol’s Structure
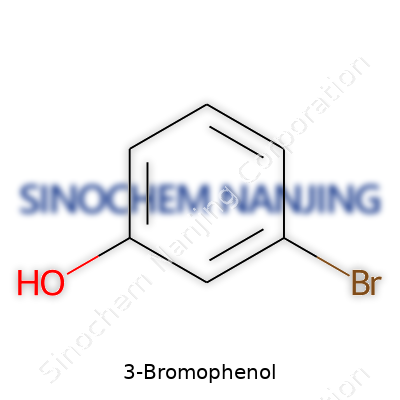
3-Bromophenol drops you right into the world of organic chemistry. It’s not just another compound on a list. Its backbone—one six-carbon benzene ring—runs with two groups that make all the difference. Sitting at the third carbon, you’ll find a bromine atom. On the very first carbon, an -OH group hangs on. They’re just two small groups, but together, they set up a chain of effects through that aromatic ring.
Anyone learning about aromatic compounds spots a pattern: the benzene ring brings stability, but every group you bolt on changes its character. In this case, the -OH group swings things toward increased polarity, absolute necessity for many lab reactions. The bromine packs in more unpredictability; it’s heavy and electronegative, throwing electron distribution out of balance and impacting how 3-Bromophenol reacts.
Why 3-Bromophenol Matters
You might not bump into 3-Bromophenol at the local store, but pharmacists, industrial chemists, and research scientists all know its value. You’ll find it is used as an intermediate for bigger projects. For example, the pharmaceutical industry leans on compounds like this to build drugs and develop new treatments. Brominated phenols often find their way into antiseptics, dye production, and sometimes in the world of pesticides.
Stepping back, chemistry students and researchers rely on 3-Bromophenol’s structure when exploring substitution reactions and teaching resonance concepts. The ortho, meta, and para terms—familiar to anyone with a semester of organic chemistry—come into focus here. Because the bromine group sits at position 3 (meta to the -OH group), the way these two groups push or pull electrons changes possible reaction routes compared to its isomers, 2-bromophenol or 4-bromophenol.
Health, Safety, and Environmental Notes
As with most halogenated benzenes, careful handling beats regrets. The bromine atom gives 3-Bromophenol extra firepower as far as toxicity and potential irritant properties go. Anyone in the lab uses gloves, goggles, and works inside a fume hood, since even small amounts can trigger skin or respiratory reactions.
From an environmental perspective, brominated compounds—this one included—can linger long after a reaction’s over. They often break down slowly and sometimes accumulate in water supplies. Disposing of waste properly can’t just be a good intention. It’s necessary. Regulatory authorities worldwide lay down clear rules for disposal because these residues threaten both animal and human health if allowed to spread.
The Pursuit of Safer Chemistry
Green chemistry pushes teams to replace halogens like bromine in new compounds whenever possible, to dodge health and persistence issues. For academic labs and industry R&D, switching to more sustainable phenol derivatives is growing in popularity. Some firms invest in new catalysts and methods that skip brominated intermediates and focus on biodegradable options. Surrounding communities and ecosystems stand to benefit from this shift, even though it means extra cost and effort up front.
If you ask around among chemists, most would say learning about compounds like 3-Bromophenol builds a foundation. Its structure bridges core organic chemistry concepts and the very real impact laboratory decisions make, whether in medicine, manufacturing, or sustainability.
What are the main uses of 3-Bromophenol?
Understanding the Chemistry Behind It
3-Bromophenol sounds like a niche term, but mention it to anyone who’s spent time in a synth-lab or who tracks chemical ingredient lists, and it feels immediately familiar. With a molecular formula that puts a bromine atom onto a simple phenol ring, this compound lands with a sharp, distinctive odor. Its presence signals more than a quirky smell, though: it shapes the backbone of more products and processes than most folks realize.
Key Ingredient in Making Pharmaceuticals
Drug discovery often needs molecules with a “hook”—a reactive site that chemists can grab onto and transform. 3-Bromophenol provides exactly that with its bromine, making it perfect for a chemistry process called cross-coupling. This step bridges different molecular pieces, helping researchers craft new active compounds. The pharmaceutical world leans on it for building blocks of antibacterials, antifungals, and early-stage candidates for cancer treatments. Industry reports highlight a steady increase in demand for brominated aromatics, especially in regions driving generics manufacturing. In my own time working alongside chemists crafting active drug ingredients, few intermediates appeared as frequently as simple brominated phenols.
Pesticides and Crop Care
On the agriculture side, pests don’t stand a chance when chemists pull from phenol derivatives. 3-Bromophenol’s reactive position lets it act as a core part of fungicides and pesticides. Agrochemical producers look for molecular scaffolds that break down at a predictable rate in the field—this compound fits that bill and brings the chemical “handle” needed for making effective products. Plenty of patent literature lists it as a step in making certain crop protection chemicals. Farmers rarely see the name, but its fingerprint shows up in the compounds they rely on.
Flavors and Fragrances: A Curious Role
People sometimes catch a whiff of 3-Bromophenol in seafood or even certain wines, thanks to natural processes and microbes. At trace levels, it influences “iodoform-like” or “medicinal” notes. Some food chemists even study how it appears in smoked fish or aged spirits. Chemists sometimes use it as a starting point for synthetic flavors that need a touch of smokiness. Getting these aroma chemicals right takes real skill: too much ruins a blend, but just enough carves out distinctive, memorable notes. Food safety regulations put strict controls around using such compounds, and expert panels often weigh in on safe exposure levels.
Research, Testing, and Specialized Manufacturing
Academic chemists love reliable, well-behaved molecules for experiments. 3-Bromophenol serves as a reference standard in analytical chemistry—think calibration mixes or purity checks for lab instruments. Some technical literature points out its usefulness in dye manufacturing and as a building block for certain plastics. Almost every specialty chemicals supplier lists it, counting on steady small-batch sales to university labs and R&D outfits.
Concerns and Responsible Handling
Even a compound with so many benefits can’t escape scrutiny. Researchers know 3-Bromophenol raises environmental concerns if handled carelessly, because halogenated organic materials resist breaking down in nature. Regulatory agencies set workplace exposure limits and require proper disposal to avoid contamination. I once watched a team develop a new waste-treatment process specifically aimed at clearing phenolic and brominated residues from a plant’s output. Solutions involve cleaner catalysis, better recycling in production, and onsite scrubbing technology. Chemical safety stays front and center as companies try to balance innovation with responsibility.
Practical Value and Future Outlook
From synthesizing the pills in a medicine cabinet to shaping the taste profile of a seafood dish, the reach of 3-Bromophenol keeps growing. The challenge lies in using its power wisely: supporting agriculture, helping science move forward, and keeping an eye on health and environmental impacts at every step. These topics show up time and again in real conversations between researchers, product developers, and regulators who want chemistry to serve—and not harm—people’s lives.
Is 3-Bromophenol hazardous to health or the environment?
What 3-Bromophenol Does in Industry
Plenty of labs keep 3-bromophenol stocked. Chemists rely on it for building pharmaceuticals, dyes, and even some flavor or fragrance compounds. With its pungent smell, you won’t forget it in a hurry. If you work anywhere near a bench, you know that phenols often come up during organic synthesis. But any substance that hangs around in research environments invites a practical question: is it safe?
Reactions With the Body
While 3-bromophenol isn’t as familiar as some old-school toxins, exposure concerns aren’t just theoretical. Phenols tend to irritate skin and eyes. A splash on your hand can leave a nasty rash or worse—most phenolic compounds penetrate skin and reach the bloodstream. 3-bromophenol, thanks to its halogen (bromine) group, might hang around longer in tissue than plain phenol. That increases the risk message you get from the safety data sheets, which advise protective gloves and goggles, even for quick handling.
Breathing 3-bromophenol fumes won’t do your lungs any favors, either. The sharp odor isn’t just unpleasant; inhaling significant amounts could irritate airways. I learned early to trust my nose—a whiff usually means the bottle doesn’t belong open any longer than absolutely necessary. Symptoms like coughing or headaches start popping up with poor ventilation or spills.
Long-term studies on humans are rare, but repeat exposure to similar compounds shows up as liver and kidney damage in animal testing. Some halogenated phenols have also raised red flags in mutation studies. Treating it like a “use and remove” compound, not something to keep hanging around, keeps risks in check.
Impact After Disposal
Letting 3-bromophenol go down the drain isn’t just a bad habit; it has consequences. Phenols, especially the halogenated ones, stick around in water and soil long after you’ve moved on. In wastewater, standard treatment struggles to fully break these molecules apart. If the chemical drifts into rivers or groundwater, fish and other aquatic life get hit first. Even low concentrations stunt growth, harm reproduction, and disrupt normal behavior in small organisms.
Halogens in chemicals can make biodegradation much slower than nature’s pace. You don’t have to look far to find evidence of persistent phenols in surface water worldwide, often stemming from improper disposal in labs or factories. Once in the ecosystem, the problems echo through the food chain, affecting both wildlife and people.
Making Safer Choices
Facts show the need for extra care. National guidelines—including right-to-know sheets and waste disposal laws—aren’t just red tape. They exist because these risks are real. Your actions in the lab matter. Wearing the right layers saves a trip to the ER. Using fume hoods keeps the stink and toxins out of your lungs. Signing the hazardous waste log ensures chemicals don’t sneak into public water.
Some places search for greener alternatives, swapping out halogenated compounds where possible. Even small changes in purchasing—buying less, tracking inventory better—helps limit what needs disposal. Chemistry’s toolbox is vast; using thoughtful risk assessments lets teams pick safer options with similar performance.
No one should ignore the hazards tied to 3-bromophenol. Mistakes and shortcuts with phenols can ripple far beyond your own bench or factory floor. It’s possible to use and respect this compound without inviting danger—if we treat it as more than just another bottle on the shelf.
How should 3-Bromophenol be stored and handled?
Respecting Chemical Hazards
Anyone who’s spent time around a chemical lab knows 3-Bromophenol leaves a strong, distinctive smell in the air. That sharp odor speaks for itself. It usually indicates you’re working with something that can’t be shrugged off as harmless. Over the years, I’ve learned that safety steps aren’t there as background noise. They make the difference between a clean operation and a dangerous mistake.
Why Attention Matters
Ignoring safe storage for 3-Bromophenol increases the risk of fires, leaks, and toxic exposure. Inhalation can trigger headaches and nausea, and even skin contact should be avoided. This is more than written guidelines—these are real risks, backed by years of toxicological studies. The chemical’s volatility and toxicity turn a careless accident into a health emergency fast.
Storing with Precision
Many people new to lab work think a simple closed cabinet will do the job. That’s wishful thinking. 3-Bromophenol belongs in a tightly-sealed glass container, always labeled, and far away from any heat source. Flammable lockers work well. Never settle for metal containers, as this compound reacts with certain metals, corroding the container, causing leaks, and creating hazards nobody wants to clean up.
Environmental Factors
Heat, light, and humidity speed up chemical breakdown. Every time I’ve seen a colleague overlook storage temperature, problems followed—volatile vapors, ruined samples, or worst of all, pressure build-up. Regular room temperatures usually work, but always keep the space visibly dry and out of direct sunlight. Dampness encourages container corrosion.
Handling Practices in Action
Handling 3-Bromophenol means relying on good habits: gloves, safety goggles, and a working fume hood are day-to-day essentials. Rushing means spills and accidental inhalation. In my own practice, I learned the hard way that skipping a quick check of the hood fan before pouring can fill a room with vapors that linger for an hour. Proper ventilation saves a lot of regret and effort.
Planning for Spills
Accidents still happen. Absorbent pads, neutralizing agents, and clear clean-up instructions help prevent small mistakes from spiraling. Teams should rehearse spill protocols—knowing exactly what to do takes the panic out of the moment. Treat every near-miss as a lesson, tightening up personal routines and workplace checklists.
Supporting Safety Culture
Safe storage and handling thrive in environments where people speak up, replacing shortcuts with best practices. Supervisors have a responsibility to keep training fresh, holding regular refreshers to update everyone on the latest guidelines. Open communication allows issues to surface early, from faulty equipment to mislabeled jars. I’ve seen places where speaking out is the norm, and they run smoother operations and avoid the long-term headaches that corners cut bring.
Simple Solutions Make the Difference
Quality chemical storage units, sturdy personal protective equipment, and regular habit checks form the backbone of a safe lab. Reviewing chemical inventory for expired or damaged containers helps catch problems before they grow. By embedding these small steps into daily routine, everyone in the lab gets a fair shot at a safe and productive day.
What is the molecular weight and CAS number of 3-Bromophenol?
Science in the Lab: Details Matter
Working with chemicals like 3-Bromophenol isn’t just about mixing things and hoping for a reaction. Small details matter. Take its molecular weight—173.01 g/mol. For a chemist, this number pops up almost every day. Measuring out the right mass of a compound is not something anyone can eyeball, especially with fine powders or crystalline solids. It doesn’t matter if you’re fresh out of college or have twenty years behind a fume hood; accurate molecular weight means accurate calculations. This is what keeps experiments reproducible.
In the past, I’ve seen what happens when the numbers get sloppy. A missed decimal means a reaction yields nothing, or worse—spills over, makes a mess, wastes time and money. With substances like 3-Bromophenol, every milligram makes a difference. You can’t prepare standard solutions without this value. If you’re testing for trace contaminants or running HPLC, your calibration only holds up if your math does.
3-Bromophenol: Pinpointing a Substance in a World Full of Compounds
The CAS number—591-20-8 for 3-Bromophenol—serves like a unique fingerprint. Many compounds share similar names, but chemicals aren’t always labeled consistently across the globe. Having a CAS number kills the confusion. It lets researchers, suppliers, and regulators speak the same language. When I order from chemical catalogs, I never rely solely on a name. It avoids errors—costly shipments, and sometimes serious safety issues.
On a few occasions, looking up a structure only by its name led to headaches. Is it the ortho, meta, or para version someone’s talking about? Someone says “bromophenol”; they might mean 2-Bromophenol, 4-Bromophenol, or our guy here—3-Bromophenol. The CAS number cuts through that fog.
Real-World Importance: Accuracy, Safety, and Efficiency
A single compound’s details ripple out way further than the beaker. Researchers building new medicines or designing plastics start with pure, well-identified ingredients. Let’s say a team is studying the breakdown products of flame retardants, where 3-Bromophenol sometimes turns up. Regulators tracking chemical hazards in factory waste use those same bold numbers: molecular weight for monitoring, CAS for reporting and tracking.
Bench chemists and regulatory bodies share a common challenge: trust in the data. Accurate labeling is the backbone here. I’ve seen enforcement agencies reject entire shipments because documentation got sloppy. Using a CAS number like 591-20-8 avoids months of costly delays. Industrial hygiene reports, environmental fate studies, and analytical chemistry methods rely on unambiguous identification.
Solutions for Better Practices
The old days of scribbling names without numbers on vials are long gone. We need to keep pushing for digital inventories, shared chemical libraries, and barcoding that link straight to CAS details and molecular data. Simple steps go a long way: every sample container in the lab gets labeled with both name and CAS number. In supply chains, requiring the CAS number on every invoice speeds up customs checks and docking. In the classroom, teaching students to treat these identifiers as non-negotiable standards sets them up for responsible science.
3-Bromophenol might seem like just another reagent, but it’s a good reminder that getting the basics right turns the gears of safe, efficient science. Take every number seriously—makes the work smoother and safer for everyone.
| Names | |
| Preferred IUPAC name | 3-Bromophenol |
| Other names |
m-Bromophenol 3-Hydroxybromobenzene m-Hydroxybromobenzene |
| Pronunciation | /ˈθriːˈbroʊməˌfiːnɒl/ |
| Identifiers | |
| CAS Number | 591-20-8 |
| Beilstein Reference | 1209221 |
| ChEBI | CHEBI:27814 |
| ChEMBL | CHEMBL1369 |
| ChemSpider | 10256 |
| DrugBank | DB03267 |
| ECHA InfoCard | ECHA InfoCard: 100.005.223 |
| EC Number | 202-164-7 |
| Gmelin Reference | 63266 |
| KEGG | C01717 |
| MeSH | D000386 |
| PubChem CID | 69477 |
| RTECS number | BZ9625000 |
| UNII | FF28E8J5V4 |
| UN number | UN2837 |
| Properties | |
| Chemical formula | C6H5BrO |
| Molar mass | 173.01 g/mol |
| Appearance | Colorless to pale yellow solid |
| Odor | Phenolic |
| Density | 1.49 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.96 |
| Vapor pressure | 0.023 mmHg (25 °C) |
| Acidity (pKa) | 9.3 |
| Basicity (pKb) | 9.3 |
| Magnetic susceptibility (χ) | -74.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.573 |
| Viscosity | 1.881 mPa·s (20 °C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 93.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −8.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3330.9 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. Harmful to aquatic life with long lasting effects. |
| Precautionary statements | Precautionary statements: P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 100 °C |
| Autoignition temperature | The autoignition temperature of 3-Bromophenol is **605°C**. |
| Lethal dose or concentration | LD50 (oral, rat): 293 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 3-Bromophenol: "1400 mg/kg (oral, rat) |
| NIOSH | BR2450000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 |
| Related compounds | |
| Related compounds |
Phenol 2-Bromophenol 4-Bromophenol |

