3-Bromoaniline: A Comprehensive Look into Science, Safety, and Industry
Historical Development
Organic chemistry has never been a static science. The use of halogenated anilines like 3-bromoaniline dates back over a century. Early on, chemists understood the power that small tweaks in a molecule deliver in pharmaceuticals and dyes. Synthetic routes became more refined through the twentieth century as researchers demanded cleaner reactions and better yields. Each breakthrough shaped the fabric of major chemical industries. From the first time 3-bromoaniline came off a bench-top, chemists recognized its unmistakable smell and the opportunities it unlocked — especially once the chemical age matured and organic synthesis began to scale.
Product Overview
3-Bromoaniline, a halogenated aromatic amine, draws significant attention in both applied and basic research. The simple act of placing a bromine atom on the meta position of aniline alters its entire reactivity profile. Its popularity stems from this unique chemistry. As someone familiar with bench work and process design, I’ve seen this compound come up in countless reaction screens — always providing a different reactivity compared to its ortho or para cousins.
Physical & Chemical Properties
This white to pale yellow crystalline solid has a faint amine aroma that sticks in memory after time spent handling pure compounds. Its melting point falls between 30°C and 33°C, and it begins to boil at around 220°C under atmospheric pressure. The density hovers just above that of water, about 1.6 g/cm³. Poorly soluble in water, it dissolves easily in most organic solvents, which often determines work-up protocols in the lab. The presence of both a strongly electron-donating amino group and an electron-withdrawing bromine ring tweaks its acid-base and nucleophilic behavior—directing synthesis in subtle, meaningful ways.
Technical Specifications & Labeling
Suppliers deliver 3-bromoaniline by purity grade. For small-scale synthesis, 97% or better suits most pharmaceutical and fine chemical applications. Product labels show the CAS number 591-19-5, full molecular formula C6H6BrN, and batch-specific data such as melting point and water content by Karl Fischer titration. Certificate of analysis sheets frequently include GC or HPLC traces to confirm not just purity but also absence of harmful isomers or residual starting material. Those details make a difference in regulatory affairs and ensure specifications match downstream requirements in industrial or academic research.
Preparation Method
Synthesis of 3-bromoaniline typically begins with a nitration-bromination-reduction sequence. Most chemists introduce the bromine into a protected aniline or use direct bromination of nitrobenzene derivatives, knowing regioselectivity favors the meta position under many conditions. Catalytic hydrogenation or metal-acid reductions then cleanly convert nitro intermediates into the amine. The big challenge is avoiding other brominated isomers. Careful control of stoichiometry and temperature does the trick. Process engineering groups go further by recycling spent reagents and optimizing yields, as waste minimization is crucial in cost-sensitive or environmentally regulated industries.
Chemical Reactions & Modifications
This compound acts as both a building block and a linchpin in organic synthesis, where cross-coupling reigns supreme. The bromine enables Suzuki, Buchwald-Hartwig, and Ullmann reactions, opening doors to biaryl scaffolds and advanced intermediates. Modifying the amino group offers access to diazonium salts, acyl derivatives, or sulfonamides. I’ve used 3-bromoaniline myself as a jump-off point for heterocycle formation and medicinal chemistry projects, and the options grow every year as new catalysis technologies emerge. Each pathway benefits from the balance between activation by the nitrogen and deactivation by the bromine, giving synthetic chemists a unique reactivity palette.
Synonyms & Product Names
Beyond “3-bromoaniline,” this compound has several names: meta-bromoaniline, m-bromoaniline, and 3-bromobenzenamine. Older IUPAC texts and catalogs sometimes list it by its registry numbers or as 1-amino-3-bromobenzene, depending on region and industry. In my own experience searching for suppliers or cross-checking publications, recognizing all synonyms can save time and confusion, especially when data curation or procurement is on the line.
Safety & Operational Standards
Working with aromatic amines calls for vigilance. Skin and eye irritation crop up quickly if PPE slips, due to both the amine base and halogen atom. Inhalation risk also shouldn’t be downplayed, since long-term exposure to anilines links to methemoglobinemia and other effects. Regulations require clear hazard labeling (GHS07/08), strict storage standards, and fume-hood operations. Professional labs also monitor air quality and maintain spill kits at stations where operators transfer or process this chemical. Adhering to published exposure limits and environmental controls goes beyond regulatory compliance; it protects every person who handles or disposes of these materials. Creating a safe work environment starts with training and constant vigilance.
Application Area
Fine chemicals, dyes, pharmaceutical ingredients, and agrochemical syntheses draw on 3-bromoaniline’s reactivity. Medicinal chemists use it to build kinase inhibitors, anti-infectives, and custom ligands as its intermediate properties streamline lead optimization. Dye manufacturers have long benefited from its brominated core, shifting the hue and permanence of azo and anthraquinone pigments. Functional materials scientists also value the halogen for tweaking electronic properties in conductive polymers, light-emitting diodes, and semiconducting films. Each sector drives constant demand for cleaner, purer, and more sustainable production routes — and as the world pivots toward greener technologies, user industries increasingly report on the sourcing and lifecycle impact of these specialty building blocks.
Research & Development
At the R&D frontier, researchers continually explore new cross-coupling protocols leveraging 3-bromoaniline as a partner in metal-catalyzed transformations. Focus sharpens on milder, more selective conditions using earth-abundant catalysts. Academia and industry share a commitment to improving atom-economy and process safety. Computational chemists model the electronic structure to guide synthesis, while analytical scientists refine techniques for high-throughput screening of new derivatives. In my previous collaborations, success often hinged on creative derivatization or overcoming scale-up bottlenecks — both areas where 3-bromoaniline routinely serves as an accessible, cost-effective scaffold.
Toxicity Research
Toxicologists have studied both acute and chronic effects of 3-bromoaniline. Animal testing points to moderate toxicity, with doses above several hundred milligrams per kilogram producing signs of central nervous system depression, hemolysis, and even death. Repeated exposure builds up risk for blue skin coloration (methemoglobinemia) and liver enzyme changes. In vitro, the compound interferes with cellular respiration, so facilities keep close control over waste and exposure, especially where vulnerable personnel might come into contact. Disposal must follow chemical safety guidelines, as the substance is hazardous to aquatic life and can persist in the environment.
Future Prospects
As global priorities shift toward environmental sustainability and stricter regulation of hazardous chemicals, the production and use of 3-bromoaniline faces new scrutiny. Firms invest in greener bromination techniques and look for bio-based routes with lower carbon footprints. Creative catalysis will reshape its application in pharmaceuticals, as regulatory pressures rise to limit genotoxic impurities and guarantee traceability from raw material to tablet. Researchers scan for non-toxic substitutes without trading away the powerful reactivity of anilines. Having watched the industry move over the past few decades, future demand for cleaner, safer, and more traceable intermediates will drive both the science and business of 3-bromoaniline.
What is the chemical formula of 3-Bromoaniline?
Getting to Know 3-Bromoaniline
3-Bromoaniline shows up in the labs of anyone working with dyes, pharmaceuticals, or certain polymers. Its name hints at its makeup: a benzene ring, an amino group, and a bromine atom attached somewhere on that ring. Experience in organic synthesis brings a lot of familiarity with these types of molecules, and 3-Bromoaniline’s structure follows the typical aromatic system—one of the backbones of organic chemistry.
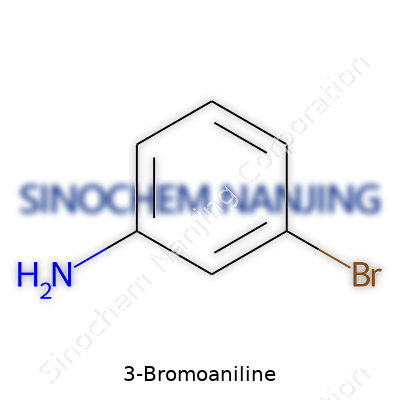
The Formula Itself: C6H6BrN
Anyone looking at the raw data will see that 3-Bromoaniline’s chemical formula is C6H6BrN. That tells a story with just a few letters and numbers: six carbons, six hydrogens, one bromine, and one nitrogen. Working with these chemicals usually means double-checking the formula before sending in an order or planning an experiment. Confusion often creeps in due to the many positions where substituents on benzene can land. The “3-” in 3-Bromoaniline means the bromine sits one carbon away from the amino group around the ring.
Why Positions Matter in Chemistry
Placement makes a real difference. In the aniline family, swapping positions around that benzene ring leads to new substances with different properties. For example, 2-Bromoaniline, 3-Bromoaniline, and 4-Bromoaniline each react in their own way because of simple changes in atom location. That seems small, but in drug development, those differences shape how safe or effective a compound might be. I once spent days chasing a synthetic route down the wrong path because I mixed up para- and meta- isomers of a similar compound, which didn’t behave the same during a reaction.
Uses You’ll See in the Real World
The value of 3-Bromoaniline isn’t limited to textbook diagrams. Chemists working in dye manufacture rely on this compound as a building block. 3-Bromoaniline stands up to various reactions—nucleophilic substitution, coupling, reduction—opening doors to new molecules that might become part of a pigment or drug. Research into cancer treatments, for example, occasionally draws on aniline derivatives, including those with halogenated rings.
Safety and Handling Concerns
Despite the usefulness of compounds like 3-Bromoaniline, safety can’t fall by the wayside. The presence of both an amino group and a bromine atom means this molecule doesn’t mix well with eyes or skin. It has the sort of toxicity you’d expect from most aromatic amines, so ventilation and gloves are part of everyday handling. In the past, I’ve seen minor lapses—like skipping goggles for a “quick” weighing—result in serious eye irritation. Lab culture stresses respect for personal protective equipment, and with good reason.
Improving the Flow from Idea to Substance
Anyone working with chemical synthesis will see room for smoother pipelines. Information about compounds like 3-Bromoaniline feels scattered: academic papers in one place, supplier specifications in another. A shared database with up-to-date safety notes, synthesis strategies, and regulatory status could unclog the search for details. Chemists used to keep notebooks for every scramble, but modern databases promise a real boost in accuracy and speed. The right formula, in the right place, makes every part of the process—from the benchtop to the marketplace—simpler and safer.
What are the common applications of 3-Bromoaniline?
Organic Synthesis in the Lab and Industry
3-Bromoaniline shows up a lot when chemists search the toolkit for making new molecules. This compound often helps researchers build larger, more complex chemical structures. Scientists use it to put together pharmaceuticals, agrochemicals, and advanced materials. Its distinctive setup — an aniline group with a bromine atom on the third position — makes it easy to manipulate through many organic reactions. You see it in Suzuki coupling, Buchwald-Hartwig amination, and other well-known organic chemistry methods. The bromine atom gives chemists a handle for swapping it out for other groups, expanding the list of molecules they can make. Speaking from personal experience in the lab, using 3-Bromoaniline cuts down the number of reaction steps to reach key intermediates. It saves time and narrows down the pathway when you need to make something specific.
Pharmaceutical Development
Medicinal chemists often rely on 3-Bromoaniline to build new drug candidates. Many pharmaceutical compounds take shape from aniline derivatives. Adding a bromine at the third spot makes all sorts of modifications possible, which boosts a scientist’s chance of tuning a molecule’s biological activity. Several antihistamines, fungicides, or anticancer agents sprouted from this building block. Some research teams I’ve worked with tweak 3-Bromoaniline to search for molecules with improved selectivity or better safety margins. It’s easy to underestimate how a single substituted aniline opens new doors in drug discovery, but the ripple effect in medicinal chemistry is real. Besides offering a foundation, it speeds up optimization — yielding promising candidates for pre-clinical testing faster than traditional approaches.
Agrochemicals and Dyes
Farmers rely on chemicals to protect crops and boost productivity. Many herbicide and fungicide molecules originate from the 3-Bromoaniline structure. Agrochemical companies turn to it when developing new products to address resistant pests or weeds. The unique arrangement of atoms lets researchers test small changes, sometimes leading to breakthroughs where older chemicals stopped working. Beyond agriculture, dye manufacturers use 3-Bromoaniline to create vivid pigments for textiles, ink, paints, and plastics. The brominated aniline structure brings reliable color-fastness and a broad palette. From a practical point of view, being able to fine-tune how a dye looks or how long it lasts can give products a real-world edge.
Material Science and Research
Researchers exploring polymers and advanced materials draw on 3-Bromoaniline to shape new compounds with special electrical or physical features. In electronics, it takes part in forming organic semiconductors for applications like solar cells and sensors. Scientists mix it into reaction networks, using its reactivity to install side chains or other chemical “modules” that tweak how a plastic or film works. Having access to a small molecule like this gives inventors a starting point for creating materials that stand up to demanding tests or harsh conditions.
Addressing Health and Environmental Concerns
3-Bromoaniline carries risks, especially in large-scale use. Handlers deal with potential toxicity, and environmental releases can raise red flags. Labs work with strict controls to avoid exposure through skin or inhalation. Factories design containment and waste management systems that handle residues responsibly. The chemical sector continues to improve purification processes and explore less hazardous alternatives, echoing growing regulatory concerns. Some researchers push for “greener” routes to these building blocks, using catalytic or enzymatic methods that cut down on harmful byproducts. Looking at how quickly science adapts, there’s real hope that these improvements stick and become widespread. Fact is, working safely with 3-Bromoaniline matters as much as what gets made from it.
What safety precautions should be taken when handling 3-Bromoaniline?
Personal Experience With 3-Bromoaniline
I remember my first real encounter with 3-Bromoaniline during an undergraduate organic synthesis. My instructor told us about the dangers before we reached for the bottle. A sharp, almost irritating scent filled the air, even though the container seemed perfectly sealed. That moment hammered home why we do not cut corners with chemical safety.
Understanding the Risks
3-Bromoaniline comes with hazards you can’t ignore. It has an irritating effect on the skin, eyes, and respiratory tract. Swallowing it or getting it on your skin can trigger more than a mild rash; it sometimes causes lasting irritations or sensitization. Chronic exposure affects the liver and kidneys, so it deserves a level of respect. Manufacturers and regulatory bodies like OSHA and the European Chemicals Agency classify it as harmful, with good reason. Even small spills produce vapors and noxious dust that linger in poorly ventilated spaces.
Building A Respectful Routine
Most mishaps can be traced to routine slip-ups. No one plans to spill chemical solutions or remove their goggles before the end of the task, but that’s often how accidents happen. Working with 3-Bromoaniline means protecting your skin and lungs every time. Always choose lab coats, nitrile gloves, and wrap-around eye protection; for prolonged work or large quantities, use a face shield and chemically resistant apron. More than once, I saw how a forgotten wrist or the skin above the glove cuff turned red within minutes after a splash. Full coverage matters, especially when fatigue leads to careless moments.
Good ventilation is not a luxury; it’s a basic need. This chemical gives off vapors that regular HVAC won’t handle. Any time I worked with gram-scale amounts, I turned to a fume hood with a strong sash and reliable airflow. The hood keeps those fumes and dust away from lungs and eyes. If you use labs without reliable ventilation, move operations to a space with a working hood, no excuses. Shortcuts here leave people with headaches or worse.
Storage and Spill Response
3-Bromoaniline stays stable if kept cool and dry, away from oxidizers or acids. Ordinary cabinets don’t always offer enough protection. Secondary containment—plastic tubs with fitted lids—catches leaks nobody expects. Assigning responsibility for regular cabinet checks prevents surprises from unnoticed drips or broken containers.
Spills need quick, calm responses. A minor drop on a benchtop calls for inert absorbent—kitty litter or commercial spill pads work really well, scooped up with gloves. The debris doesn’t go in regular trash; labeled waste containers keep the janitorial team and the rest of the lab safe from secondary exposure.
Training and Culture Matter Most
No safety gear substitutes for hands-on training. In my best labs, new students practiced how to remove gloves, handle simulated spills, and read safety data sheets before touching any chemical. Oversight—someone who checks and reinforces good habits—turns rules into real habits.
Finally, it’s hard to overstate the value of a strong safety culture. I worked once in a space where someone reported even minor near-misses openly. Normalizing this transparency helped more than any sign or checklist. Speaking up if someone skips goggles or sprays something outside the hood protects everyone—no heroics, just a team looking out for each other.
Working with 3-Bromoaniline doesn’t need to feel intimidating. Clear rules, complete protection, and open communication keep accidents rare and confidence high.
What is the molecular weight of 3-Bromoaniline?
The Context Behind the Calculation
After years of handling chemicals during late-night undergraduate labs and later in industrial settings, I’ve learned labs rarely focus much on the real-world weight of tiny molecules. Yet, in moments where a synthesis hinges on just getting a few numbers right, the value can’t be ignored. 3-Bromoaniline, with the formula C6H6BrN, is one of those slightly quirky yet important compounds popping up in dyes, medicines, and organic synthesis. Forget for a moment what it’s used for, let’s just look at what the numbers say.
Getting Real: The Numbers That Matter
Molecular weight calculations used to feel like busywork at the start of a project. But here’s the calculation, without the boredom: carbon’s atomic mass comes in at about 12.01 g/mol, hydrogen at 1.01, nitrogen at 14.01, bromine much heavier at 79.90. The arithmetic rolls like this:
- Six carbons: 12.01 x 6 = 72.06
- Six hydrogens: 1.01 x 6 = 6.06
- One nitrogen: 14.01
- One bromine: 79.90
Add these up, you get 172.03 g/mol. It’s not just about plugging numbers into a formula. Chemical engineers, researchers, and quality-control specialists use this number every day, double-checking that their process is on track. Inconsistent weights betray impurities or poor labeling, risking experiments and even safety.
Why It Matters in Practice
Years ago in a dusty college storeroom, I grabbed a bottle labeled 3-Bromoaniline. By habit, I double-checked the label—but the displayed molecular weight didn't match the number in my lab manual. A manufacturing slip or a typo could foul up every calculation for reactant ratios. That 172.03 number isn’t just trivia; it affects every step that follows. For every chemist who’s measured out powders, a tiny mistake balloons into lost hours, wasted materials, or, far worse, a blown experiment.
Quality, Accuracy, and Trust
Researchers rely on accurate data both for compliance and for discovery. Documentation must tie each batch of 3-Bromoaniline to its correct molecular identity, or regulatory fines and lost credibility pile up. In pharmaceuticals, stakes rise even higher; dosing errors can turn from technical glitches to health threats. This pushes teams to cross-check, not just trust a single reference.
The Role of Fact-Based Practice
Good practice starts with looking up values in trusted sources—textbooks, peer-reviewed papers, and chemical catalogs from established suppliers. I’ve caught mismatches in old data tables, and I now double-check at least one reputable database before using a molecular weight in a synthesis or equation. This matters for audits, patent filings, and keeping projects reproducible when teams collaborate across borders.
Pursuing Solutions
Teams get further by building redundancy into data management. Digital lab notebooks flag entries with mismatched molecular weights, while inventory management software pulls updated chemical info before release. This habit might look fussy but saves time and money. Training staff to catch these discrepancies becomes part of onboarding in any forward-thinking lab.
Final Thoughts on Data-Driven Chemistry
3-Bromoaniline, sitting at 172.03 g/mol, represents the chemical industry’s larger relationship with trust and transparency. Getting that number right strengthens every experiment, every analysis, and every result. For anyone working with chemicals, never treat molecular weight as a background detail. The integrity of research depends on these everyday numbers.
What are the storage conditions for 3-Bromoaniline?
What Makes 3-Bromoaniline Different?
3-Bromoaniline carries more risks than some other lab chemicals. This compound goes into dyes, pharmaceuticals, and other specialty materials, but it also brings real hazards. The smell alone will remind anyone in a lab about the need for proper precautions. If someone stores this chemical incorrectly, it loses stability and turns into a safety concern. Its unique structure—an aromatic ring with both bromine and amine groups—can be reactive, especially if exposed to moisture, air, or high temperatures.
Temperature, Moisture, and Air: Crucial Factors
Keeping 3-Bromoaniline in a cool, dry spot isn’t just a suggestion—it’s the difference between predictable results and a nasty surprise. At room temperature, sealed up tight and away from sunlight, the compound remains stable and doesn’t break down. Humidity or condensation turns a harmless-looking bottle into a contaminated mess. I remember once walking into a university storage room that reeked of spoiled chemicals partly because someone left bottles open on a humid day. Closed cabinets with good seals saved the rest of the stock.
Oxygen also tends to stir up trouble. Even though this material doesn’t ignite as easily as some solvents, it does have a habit of reacting if left in the open. Keeping jars closed, using containers made of glass or approved plastic, and labeling them with the date prevents confusion and unwanted exposure. An old habit from organic chemistry lab class: check seals, check dates, then check again. Simple routines like this stop headaches before they start.
Avoiding Cross-Contamination and Spills
Some labs get sloppy after a few months. A bottle of 3-Bromoaniline on the wrong shelf next to acids or bases isn’t just an oversight—it’s a ticking time bomb. I saw a student once store a halogenated aniline beside a strong oxidizer. The chemical started to discolor within a week. One spill, or one accidental knock, could lead to toxic fumes or fire. Good practice means separation: dedicate a clear, isolated section of the storage cabinet. Post clear labels; avoid makeshift signage or paper scraps taped to the bottle.
Safety goggles and gloves aren’t optional—the compound burns skin and eyes quickly. A spill on the bench needs prompt cleanup, not a note left for the morning crew. Labs that stay disciplined pick up on these risks early, setting routines so nobody forgets. This isn’t just red tape; it’s respect for your own health.
Improving Storage: Simple Steps That Work
Major issues fade when teams invest in airtight, chemical-resistant containers. The upfront expense pays for itself by slashing waste and preventing ruined experiments. A simple purchase like a temperature monitor stops most surprises. Recording the date when opening bottles helps track age—a forgotten bottle always warrants a double check.
Labs that run regular training get fewer accidents. New staff pick up smart habits and recognize red flags before trouble starts. Some teams post quick-reference sheets outlining what belongs on each shelf. Ready access to spill kits, good ventilation, and weekly audits all cut down on risk.
Building a Culture of Accountability
Chemicals like 3-Bromoaniline demand respect in all stages—right from delivery until safe disposal. Students and techs who take pride in a well-run storage room rarely face big emergencies. By keeping containers sealed, checking labels, and storing away from incompatible substances, labs keep their people safer and their research on track. In a world full of shortcuts, seeing that yellow safety cabinet organized tells you a lot about how seriously a team takes its work.
| Names | |
| Preferred IUPAC name | 3-bromobenzenamine |
| Other names |
m-Bromoaniline 3-Aminobromobenzene 3-Bromo-1-aminobenzene m-Bromophenylamine Benzene, 1-amino-3-bromo- |
| Pronunciation | /ˈθriː ˈbroʊ.moʊ.əˌnɪl.iːn/ |
| Identifiers | |
| CAS Number | 591-19-5 |
| Beilstein Reference | 1209221 |
| ChEBI | CHEBI:22234 |
| ChEMBL | CHEMBL23343 |
| ChemSpider | 5738 |
| DrugBank | DB04280 |
| ECHA InfoCard | InChIKey=ATFUPGXHZJVOHJ-UHFFFAOYSA-N |
| EC Number | 611-202-5 |
| Gmelin Reference | 82211 |
| KEGG | C00609 |
| MeSH | D000773 |
| PubChem CID | 72204 |
| RTECS number | BW6650000 |
| UNII | 9Q3U18T66B |
| UN number | UN prototyping: "UN2811 |
| Properties | |
| Chemical formula | C6H6BrN |
| Molar mass | 186.04 g/mol |
| Appearance | Brown to yellow liquid |
| Odor | ammonia-like |
| Density | 1.610 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.96 |
| Vapor pressure | 0.0146 mmHg (25°C) |
| Acidity (pKa) | 4.17 |
| Basicity (pKb) | 9.64 |
| Magnetic susceptibility (χ) | -66.0e-6 cm³/mol |
| Refractive index (nD) | 1.641 |
| Viscosity | 1.19 mPa·s (at 25 °C) |
| Dipole moment | 2.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 93.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 51.9 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3112.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D07AC12 |
| Hazards | |
| Main hazards | Harmful if swallowed, in contact with skin or if inhaled; causes skin irritation; causes serious eye irritation; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H317, H411 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-1 |
| Flash point | 92 °C |
| Autoignition temperature | 615°C |
| Lethal dose or concentration | LD50 oral rat 2300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 1570 mg/kg |
| NIOSH | KW6825000 |
| PEL (Permissible) | PEL: 2 ppm |
| REL (Recommended) | 40 hours |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Aniline 2-Bromoaniline 4-Bromoaniline 3-Iodoaniline 3-Chloroaniline 3-Nitroaniline 3-Aminophenol |

