Exploring 3-Bromo-1,2-Dimethylbenzene: A Chemical Perspective
Historical Development
Chemical synthesis has always thrived on the backbone of aromatic halides. 3-Bromo-1,2-Dimethylbenzene traces its roots to organic chemistry labs of the mid-20th century, as researchers searched for reliable building blocks for drug-like molecules and advanced materials. In those days, scientists leaned on halogenation reactions to probe reactivity in methylated benzenes. This compound, in particular, taught chemists valuable lessons about direct halogen substitution and the nuanced control needed in working with methyl-substituted aromatic rings. Chemists who tinkered with toluene derivatives in search of new pharmacophores found this compound’s non-trivial preparation a helpful challenge that shaped methods still used today.
Product Overview
3-Bromo-1,2-Dimethylbenzene—also known by its synonym, 3-Bromo-o-xylene—remains a staple in the synthesis of fine chemicals. You will encounter this compound in the intermediate step for specialty dyes, fragrances, liquid crystals, and pharmaceutical building block production. Companies supplying advanced organic compounds often list it under catalog numbers like NSC 90420 and 1-Bromo-3,4-dimethylbenzene. The clear, colorless to pale yellow liquid sports a sharp, aromatic odor typical of xylenes, signaling both its chemical structure and need for proper ventilation during use.
Physical & Chemical Properties
Physically, 3-Bromo-1,2-Dimethylbenzene brings certain consistencies. It boasts a boiling point in the range of 200 to 204°C and a melting point well below room temperature, making it a liquid under standard lab conditions. Its molecular weight comes out to 185.06 g/mol, and the density sits around 1.32 g/cm³ at 20°C. The compound displays moderate solubility in alcohols and ethers but stays stubbornly immiscible in water. The bromine makes the ring more reactive toward certain transformations compared to methylbenzenes lacking halogens, a fact that many synthetic chemists appreciate when seeking functional diversity.
Technical Specifications & Labeling
In most laboratories, labels on bottles mention the chemical formula as C8H9Br, with purity often exceeding 98%. Suppliers regularly note hazardous classifications, referencing the dangers of flammability and skin or respiratory irritation. Containers use UN identification numbers for shipping, typically falling under dangerous goods regulations. The label may feature hazard pictograms and statements pulled straight from GHS guidelines, not as scary warnings, but as reminders: respect the compound’s potential risks and you minimize trouble.
Preparation Method
The typical synthesis path works by directed bromination of o-xylene. Pouring bromine into a cooled solution of o-xylene, with iron (III) bromide as a catalyst, instructs the halogen to land mostly in the meta position, away from competing methyl groups. A careful addition rate, plenty of stirring, and controlled temperature hold the selectivity. In my experience, running the process too warm or dumping bromine all at once triggers polysubstitution, reducing yields. After reaction, washing with sodium bisulfite removes unreacted bromine, a crucial step to keep the workup clean. Extraction and distillation follow, rewarding the patient chemist with a pure product.
Chemical Reactions & Modifications
Reactivity of 3-Bromo-1,2-Dimethylbenzene makes it valuable in coupling procedures, like Suzuki or Heck reactions. You can swap that bromine for a methyl, vinyl, or aryl group, opening pathways to unique biaryl scaffolds or heteroaryl compounds. Metal-halogen exchange with lithium reagents unlocks further functionalization right on the aromatic ring, letting researchers build complex targets piece by piece. The methyl groups aid regioselectivity, discouraging over-reaction. Nucleophilic aromatic substitution, despite being less common on benzene rings, picks up pace in the hands of expert chemists working with electron-withdrawing partners.
Synonyms & Product Names
Alongside its main title, 3-Bromo-1,2-Dimethylbenzene pops up as m-Bromo-o-xylene, NSC90420, and 1-Bromo-3,4-dimethylbenzene. These names surface in purchase orders, chemical inventories, and material safety data sheets. For research ordering and cataloguing, having the CAS number 576-23-8 at hand speeds up the process and avoids confusion. Keeping track of multiple synonyms proves essential for supply chain management and reliable sourcing, since product names change company to company and across countries.
Safety & Operational Standards
From a safety standpoint, 3-Bromo-1,2-Dimethylbenzene demands respect. Liquid-phase aromatic bromides can trigger irritation of the eyes, skin, and respiratory tract. Well-ventilated hoods, face shields, and sturdy gloves form the first defense. Fire risk warrants storage in cool, properly labeled cabinets. Regulatory agencies treat this compound as hazardous, so labs track storage and disposal in line with EPA and OSHA guidelines. Spills should not soak into soil or drains, especially since brominated organics accumulate in ecosystems. Fresh air, chemical-resistant gloves, and education on the latest safety posters make a daily difference in preventing accidents.
Application Area
Applications cover pharmaceutical intermediates, especially as a precursor for more elaborate aromatic molecules. Agrochemical and pigment manufacturers also turn to 3-Bromo-1,2-Dimethylbenzene during the construction of active or colorant scaffolds. Advanced materials groups use the compound to introduce tailored properties into polymers and coatings, particularly where electronic or optical features demand precise structural motifs. I’ve seen colleagues in chemical start-ups use it as a launching point for sensor materials and new functional chromophores. This versatility draws in researchers from synthetic, medicinal, and industrial chemistry alike.
Research & Development
Research efforts keep pushing what’s possible with aromatic bromides. Innovations in green chemistry seek to swap out toxic solvents, reduce waste, and improve selectivity in bromination procedures. IAC, Suzuki, or Buchwald-Hartwig couplings receive periodic upgrades, with ligands and catalysts tweaked for greater speed and less expense. Academic projects still publish new applications for this compound, including the assembly of fluorescent tags and drugs aimed at hard-to-treat illnesses. The ability to introduce bromo groups early on, then modify the product downstream, appeals to scientists who value modular synthetic strategies.
Toxicity Research
Questions of toxicity have cropped up in both workplace and environmental discussions. Research points to moderate acute toxicity by ingestion or skin contact, with delayed effects most likely tied to bioaccumulation and organ system exposure. Animal studies indicate central nervous system depression at high doses, while longer-term effects remain less understood without substantial data. Experiments show that breakdown in soil moves slowly, highlighting environmental persistence. Waste management protocols, including incineration and chemical neutralization, step in to address these issues. Chemists trained in handling organohalide waste help avoid environmental harm by scrupulous adherence to regulatory standards.
Future Prospects
Looking ahead, demand for 3-Bromo-1,2-Dimethylbenzene seems firmly rooted in the growing field of molecule construction. Drug discovery, materials science, and pigment technology all benefit from access to finely tuned intermediates. More sustainable production methods—maybe biocatalytic or using recyclable reagents—could make this compound’s preparation greener, reducing the use of hazardous materials. Diversification in downstream chemistry, including new catalytic cycles and in situ modifications, continues to expand what this old but reliable intermediate can achieve. Eyes in both industry and academia remain locked on improvements in safety, efficiency, and ecological impact.
What is the chemical formula of 3-Bromo-1,2-Dimethylbenzene?
Getting to Know This Compound
Chemistry isn’t always about what goes on in a lab with bubbling beakers. The names and formulas of organic compounds might look intimidating, but there’s a real logic to the way they’re structured. Take 3-Bromo-1,2-Dimethylbenzene. This name looks long, but each part points toward a very specific molecular recipe. Anyone familiar with aromatic rings knows that benzene carries six carbon atoms in a loop, linked by alternating double bonds. Attach two methyl groups and a bromine atom in the right spots and you get our topic for today.
Drawing the Structure from the Name
In every introduction to organic chemistry, understanding how to name and draw molecules forms the backbone of real progress. Here, "1,2-dimethyl" shows that two methyl groups (–CH3) cling to the first and second carbon atoms of the benzene ring. The “3-bromo” part signals a bromine atom hooked to the third carbon. Counting around the ring and keeping those groups as close together as possible, you sketch the skeleton. Put it all together and you see a benzene ring with a methyl at carbon 1, another at carbon 2, and a bromine at carbon 3. You end up with a formula: C8H9Br.
Why the Formula Matters
Details matter. Getting the formula right isn’t just about solving a puzzle. A small error in chemistry can cascade through to real-world results, whether in pharmaceuticals, plastics, or the next research breakthrough. Name and formula alignments keep safety high in labs and let different teams communicate without getting wires crossed. From experience, even a small confusion between a methyl and an ethyl group can send a project down the wrong road, cost time, or impact product purity.
Link to Everyday Reality
Learning to read these formulas and structures teaches precision. In college, struggling over aromatic ring isomers made me realize patience could pay off. As straightforward as C8H9Br looks, drawing every single hydrogen in helped me avoid simple slip-ups. It's easy to skip a detail if you haven’t deeply understood the system, which holds true far outside the classroom.
Relevant Data and Scientific Context
The chemical industry runs on accurate naming and formula conventions. According to the International Union of Pure and Applied Chemistry (IUPAC), clear nomenclature ensures everyone speaks the same language globally. In pharmaceutical manufacturing, regulatory authorities like the US Food and Drug Administration routinely audit compound designations to avoid expensive recalls or mislabeling accidents. Even in everyday items—paints, solvents, agricultural products—small mistakes in molecular formulas can result in toxins slipping into consumer hands.
Seeking Solutions
Consistency and education keep the system on track. Hands-on models and digital tools now flag errors before they snowball. Chemistry teachers who bring structure-drawing games into their lessons stick with students much longer than lectures full of abstract tables. I have seen open-access resources and chemistry databases improve accuracy and speed, especially for new students and small-scale labs without access to expensive software.
Moving Forward
Getting the chemical formula right—C8H9Br for 3-Bromo-1,2-Dimethylbenzene—provides a strong foundation in both education and lab work. It gives confidence not just to specialists but anyone who deals with chemicals, from students to engineers to health professionals. The clearer and more reliable the system, the safer and more effective real-world science can be.
What are the main uses or applications of 3-Bromo-1,2-Dimethylbenzene?
Chemical Foundations Everyone Benefits From
Every time someone hears about another chemical compound, the big question is simple: What’s it for? 3-Bromo-1,2-dimethylbenzene stands as one of those behind-the-scenes agents in the world of modern chemistry. With its two methyl groups and a bromine stuck to a benzene ring, this compound gives chemists a handy tool for many projects—most of which we never see, even though the results might wind up on pharmacy shelves or in advanced materials all around us.
Making Medicines Better and Faster
Medications rarely get produced from a single reaction. Most of the steps call for compounds that are chemically flexible and easy to manipulate. That’s where this bromo-methylbenzene comes in. Its structure lets it work like a building block during the synthesis of specialized drugs. For example, researchers use this compound to tack on different groups through Suzuki, Stille, or Heck reactions. It doesn’t take a stretch of imagination to see why this matters—each step toward the active ingredient in a medicine usually means stringing together chains in a super-specific way, and brominated aromatic compounds like this offer both precision and reliability.
From personal experience in a pharmaceutical lab, research projects regularly slow down when a key intermediate proves tough to handle. Easy-to-react compounds like 3-bromo-1,2-dimethylbenzene speed up development, meaning life-changing treatments could reach people a little quicker. With the right reliability, companies don’t waste months trying to troubleshoot dead-end reactions.
Fueling Everyday Materials
Polymers fill homes, workplaces, and even hospitals with everything from packaging to medical gear. This particular compound shows up as a starting material for high-value resins and polymers, especially those needing specialty performance, like resistance to chemicals or high temperatures. When manufacturers want to create custom plastic parts or advanced film coatings, these small aromatic building blocks let them tweak the end product so it holds up under stress and heat.
Stepping Up in Agrochemistry
Modern farms need targeted solutions to keep pests and disease off crops. That usually means inventing new pesticides and herbicides every year. Chemists lean on compounds such as 3-bromo-1,2-dimethylbenzene for developing crop protection agents. Its reliability and versatility let teams explore new molecules that farmers will actually use. By fine-tuning a structure starting with a well-understood intermediate, scientists can predict how a new compound might break down or interact with the environment, helping to minimize unwanted runoff.
Solutions and Looking Ahead
Chemicals like this do open up some tough questions. Handling and disposal require real care. For every upside, there’s the risk of exposure, pollution, or the fallout from poor end-of-life practices. Industry teams are investing in safer and more sustainable synthesis methods—greener solvents, milder conditions, recycling of side-products.
Modern science lives by collaboration and transparency. Published safety data, open-access protocols, and tightly regulated supply chains help everyone handle these powerful tools with respect. By sharing what works and what fails, researchers and companies keep the most important advances moving forward without putting health or the planet second.
What are the physical and chemical properties of 3-Bromo-1,2-Dimethylbenzene?
Physical Profile Up Close
3-Bromo-1,2-Dimethylbenzene stands out as a clear liquid at room temperature. Its molecular formula, C8H9Br, gives it a weight that’s heavier than plain xylenes, mainly because of the bromine atom. The liquid sports a fairly high refractive index and doesn’t show much color, making it easy to spot impurities just by looking for haze or tints.
Most folks working in a basic chemistry lab will notice a sharp, sometimes sweet chemical smell when opening a vial. This isn’t surprising, considering its structure echoes many aromatic compounds. Don’t let its clear look trick you—this liquid will stain lab coats and fingers if spilled, thanks to the sticky nature of halogenated aromatics.
The boiling point lands at around 217–220°C, much higher than toluene or xylene. Those who have tried distilling aromatic bromides notice you need more energy and decent ventilation. At the same time, that boiling point helps make the substance more stable on the bench, since it won’t evaporate quickly during regular use. The flash point—roughly 108°C—still demands attention from safety officers. No need for dramatics, but don’t leave bottles next to open flames.
Chemical Personality
Chemically, 3-Bromo-1,2-Dimethylbenzene scores high points for versatility. The methyl groups on positions 1 and 2 sit next to the bromine at position 3. This arrangement gives the molecule both electron-donating and electron-withdrawing influences, making it useful for selective reactions. The bromine atom invites classic cross-coupling approaches, including Suzuki and Heck reactions. Synthetic chemists use it as a starting block for building up more complex molecules.
In my experience, the methyl groups protect certain positions on the benzene ring, so electrophiles don’t just wander all over the molecule. This helps steer reactions to the empty spots, which cuts down on unwanted byproducts. Taking aromatic halides through nucleophilic aromatic substitutions gets tricky, but the methyl groups help slow down wild side-reactions, which is a relief during organic labs.
Water doesn’t mix with this liquid; try shaking up a test tube and watch the stubborn separation happen. For dissolving, you’ll have more luck with organic solvents like ether, acetone, or chloroform. I’ve seen labs store it in basic glassware—brominated aromatics don’t corrode glass or react with regular storage materials, so there’s less risk than with acids or strongly oxidizing agents.
Why Properties Count in a Working Lab
Anyone doing organic synthesis knows physical and chemical features guide how you handle and use chemicals day-to-day. A non-volatile, stable liquid like this won’t fill the lab with fumes, but there’s always that risk of absorption through skin, especially since it blends so well with oils. Gloves, goggles, and fume hoods aren’t just for show.
Its high boiling point and bromine content push waste disposal into special categories. You can’t pour leftovers down the drain or mix them with usual lab solvents without following disposal protocols. Employers watch out for these halogenated aromatics in the waste stream since they carry environmental risks.
Green chemistry movements look for ways to replace halogenated solvents and reagents, but sometimes the chemistry just works best with brominated compounds. Exploring recyclable reagents and better waste streams could be the answer moving forward. Many labs have started reclaiming used halogenated materials in-house, cutting costs and waste. These steps take the sting out of using useful but potentially problematic chemicals like 3-Bromo-1,2-Dimethylbenzene.
What safety precautions should be taken when handling 3-Bromo-1,2-Dimethylbenzene?
Straight Talk on Chemical Safety
People often see chemical names like 3-Bromo-1,2-Dimethylbenzene and tune out, but even so-called “routine” chemicals bring risks worth knowing. Over the past decade working in chemical labs, I’ve seen corners cut and people regret it. This compound packs more punch than its bland name suggests. Meaning: one splash, one fume cloud, and you’re dealing with a hazard.
Understanding the Risks
The structure—a benzene ring with bromine and methyl groups—signals possible skin and respiratory irritation. The brominated side introduces extra volatility compared to non-halogenated cousins. What matters: long exposure adds up fast, especially without gear or if a spill hits your skin.
Gear Up Before You Touch the Bottle
You don’t need to guess. Safety data sheets (SDS) stress gloves, but not just any kind—nitrile stands up better than latex against organics. Always wear goggles or safety glasses: one tiny drop can mess up your eyes for good. Go for a lab coat that covers your arms and closes snugly. For those transferring or weighing larger volumes, a chemical-resistant apron helps when a splash gets wild.
Air and Ventilation
3-Bromo-1,2-Dimethylbenzene vapors don’t have much mercy on lungs. Even if it doesn’t smell strong, inhaling fumes can sting your nose and throat, and repeated hits damage airways over time. You gain a lot by working in a certified chemical fume hood—never out on the benchtop. If your hood isn’t pulling air as it should, don’t work until it’s fixed. People trust that “quick tasks” outside a hood are safe, but the risks are the same.
Storage and Fire Hazards
Don't stash this chemical just anywhere. A cool, dry place, away from sunlight, slows down degradation and fume release. Keep it away from oxidizers and bases—chemical fires start this way, and most labs aren’t ready to fight them. Flammable liquid cabinets aren’t just for show—use them.
What to Do When Things Go Wrong
Spills feel scary, but fast action limits harm. Mop up small spills with absorbent pads, not paper towels, and never touch the liquid without gloves. Ventilate the area, and don’t hesitate to call for help in case of large spills. For skin contact, rinse with cold water for 15 minutes—don’t wait for a burn or rash.
Long-Term Health and Training—Not Optional
You remember the first time you work with a new chemical, but one month later, it can feel routine. That’s why regular safety refreshers make a difference. OSHA doesn’t make these rules for nothing—long-term benzene exposure links to headaches, skin issues, and sometimes worse. Calling in a safety officer for a walk-through doubles as a reminder for everyone. Easy-to-read signage and checklists help cut down on mistakes, especially for new team members.
An Ounce of Prevention
Most of my colleagues would tell you the same: Treat every hazardous chemical with respect, invest in good PPE, keep up with training, and don’t let shortcuts become the norm. Experience shows that the right habits, gear, and plans make all the difference in keeping people safe around 3-Bromo-1,2-Dimethylbenzene and chemicals just like it.
How should 3-Bromo-1,2-Dimethylbenzene be stored and disposed of properly?
Respect the Chemistry
Handling 3-Bromo-1,2-Dimethylbenzene isn’t just about following a checklist—every chemist and lab worker knows that spills, improper storage, and lazy disposal have real consequences. Anyone who’s spent time in a university teaching lab will probably remember walking in on a scene where an unlabeled bottle with a sharp, strange smell sat on an unventilated shelf, left by someone hoping to let it become “someone else’s problem.” The risks stick with you: skin contact, fire hazards, environmental hazards. Taking shortcuts can put people and the environment at risk, including neighbors and animals who never agreed to join your experiment.
Proper Storage Habits
Most aromatic bromides need a cool, dry place and sealed containers made of glass or high-quality plastic. Years ago, during a summer internship in a research lab, I saw a leaky cap turn a storage area into a clean-up project and a breathing hazard. Only tightly closed containers, clearly labeled, will do. We always kept compounds like this in ventilated chemical cabinets marked for organics and halogenated aromatics. No storing near oxidizers, acids, or open flames—this isn’t the bottle you squirrel away on a forgotten shelf. We checked inventory every week and rotated stock to spot cracked containers before they became a bigger problem. The Occupational Safety and Health Administration (OSHA) makes these recommendations for a reason—you don’t want a violation, but nobody wants an unnecessary hospital visit, either.
Ventilation counts. I’ve seen poorly ventilated store rooms cloud up after bottles broke, and I’ve seen fire marshals turn red pointing out chemical clutter. Invest in a flammable storage cabinet with real ventilation. This step isn’t just compliance; it’s common sense, keeping chemicals away from heat and static.
Smart Disposal Matters
Most municipalities treat substances like this as hazardous waste. Tossing it down the drain or in the trash can wreck local water or soil. Once, a local stream near our town showed traces of aromatic solvents—other labs had dumped waste, and the local wildlife paid the price. Those lessons stick. Reputable labs train staff to separate halogenated organics for licensed chemical disposal services. A manifest tracks every step, from storage to pickup. This record may sound excessive, but if somebody gets sick or the environment is damaged, tracing the waste becomes critical. Local, state, and federal laws all demand this level of care, with the Resource Conservation and Recovery Act providing the legal backbone in the US.
Sometimes there’s pressure to save money or time—pouring waste into an “all-purpose” solvent bin, for instance—but a fine or accident will cost much more. Chemistry staff who take part in regular hazardous-waste training understand not just how to dispose of compounds, but also why that effort matters. Experience in a busy laboratory environment taught me to always double-check the waste stream and ensure each container matched regulations. Chemical safety is a shared responsibility that keeps everyone safe, including the people who never enter a lab.
Protecting People, Protecting Place
Treating chemicals like 3-Bromo-1,2-Dimethylbenzene responsibly isn’t an option. It’s an ongoing habit that takes preparation and some respect for the rules. From the first day in the lab, working side by side with others, the need for vigilance becomes clear. Clear labeling, secure storage, and strict disposal guidelines never become outdated, and they protect lives and landscapes long after a project ends.
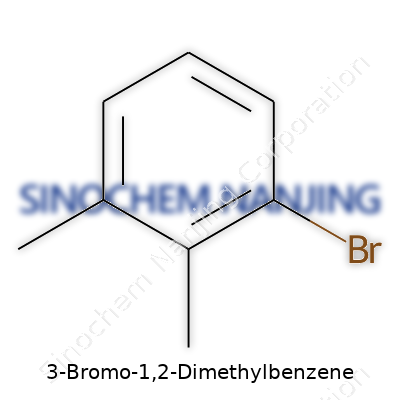
| Names | |
| Preferred IUPAC name | 1-bromo-3,4-dimethylbenzene |
| Other names |
3-Bromo-o-xylene 3-Bromo-1,2-xylene 1,2-Dimethyl-3-bromobenzene |
| Pronunciation | /θriː-ˈbroʊ-moʊ waɪn tuː daɪˈmɛθɪlˈbɛnˌziːn/ |
| Identifiers | |
| CAS Number | 591-17-3 |
| 3D model (JSmol) | `CC1=C(C)C=CC=C1Br` |
| Beilstein Reference | 1908562 |
| ChEBI | CHEBI:51304 |
| ChEMBL | CHEMBL3811312 |
| ChemSpider | 68212 |
| DrugBank | DB04125 |
| ECHA InfoCard | 01a204e6-5b8a-46db-92d7-1c7016d2e04f |
| EC Number | 202193-67-1 |
| Gmelin Reference | 77830 |
| KEGG | C21152 |
| MeSH | C08.945.996.419 |
| PubChem CID | 12053732 |
| RTECS number | DC3325000 |
| UNII | 3T8J1K74T1 |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C8H9Br |
| Molar mass | 183.07 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Aromatic |
| Density | 1.34 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.9 |
| Vapor pressure | 0.4 mmHg (25°C) |
| Acidity (pKa) | 35.00 |
| Basicity (pKb) | 3-Bromo-1,2-Dimethylbenzene does not have a relevant pKb value, as it is not a base. |
| Magnetic susceptibility (χ) | -84.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.552 |
| Viscosity | 0.886 cP (25°C) |
| Dipole moment | 1.47 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 87.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4687.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P305+P351+P338, P312, P337+P313, P362+P364 |
| Flash point | > 74 °C |
| Autoignition temperature | Autoignition temperature: 500°C (932°F) |
| NIOSH | DI2275000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Bromo-1,2-Dimethylbenzene is not specifically established by OSHA. |
| REL (Recommended) | REL: Not established |
| Related compounds | |
| Related compounds |
1,2-Dimethylbenzene 3-Iodo-1,2-dimethylbenzene 3-Chloro-1,2-dimethylbenzene 3-Nitro-1,2-dimethylbenzene 1,2,3-Trimethylbenzene |

