Commentary on 3-Aminoquinoline: Science, Society, and the Future
Historical Development of 3-Aminoquinoline
Chemistry carries stories in its molecules. 3-Aminoquinoline stands as a prime example. Chemists first began exploring quinoline derivatives in the early 20th century, searching for new ways to fight diseases like malaria. Growing interest in aromatic heterocycles like quinoline led to the creation and detailed study of compounds such as 3-aminoquinoline. Early synthetic methods revolved around the Skraup synthesis, known for its rough conditions and variable yields. Over decades, newer synthetic strategies improved purity and output, making lab research more practical and economic. As labs tuned processes and researchers improved safety, the handling of these substances became less daunting over time. This shift shaped the possibilities for drug discovery, influencing generations of medicinal chemists and leading to a wider scope of application for such building blocks in pharmaceutical pipelines.
Product Overview: From Lab to Industry
Among the many molecules born in synthetic labs, 3-aminoquinoline has shown itself as a workhorse. Its structure—a quinoline backbone with an amino group attached at the third position—provides a foundation for various biological activity. Biologists and medicinal chemists prize this molecule for its potential in antiparasitic and antitumor research. Some compounds built on this scaffold have advanced into early pharmaceutical investigations targeting malaria and tuberculosis, and the broader range of nitrogen-containing heterocycles often appears in Hit-to-Lead stages of drug discovery. It's not just a research curiosity; practical applications stretch into agrochemistry, dye manufacturing, and even corrosion inhibitors.
Physical and Chemical Properties
Handling 3-aminoquinoline draws attention to its modest but notable properties. In solid form, it usually appears as a yellowish powder. This physical nature hints at aromatic stability, but the presence of the amino group brings enough polarity to play a role in solubility, influencing both reactivity and how labs consider storage. The melting point generally sits just above room temperature, making it easy enough to handle without risk of premature decomposition. In terms of chemistry, the balance of basicity and nucleophilicity in the amino group fosters a range of chemical transformations, from coupling reactions to reductive modifications. Solubility trends follow typical aromatic amines: limited water solubility but good interaction with polar organic solvents such as ethanol, DMSO, or acetone. Chemists seeing these properties quickly sort out the best conditions for running reactions or purifying targets.
Technical Specifications & Labeling
Reliable experiments start with well-characterized materials. Genuine samples of 3-aminoquinoline need careful specification. Purity should be established using standard methods: HPLC, NMR, or GC where appropriate. Trace solvents and byproducts from synthesis—like unreacted anilines or residual acids—undermine reproducibility. The labeling standards developed by experienced labs emphasize hazard classification, batch-specific data, and expiration dates. Each bottle of 3-aminoquinoline carries detailed identifiers not just for safety, but for accurate tracking and compliance with supply chain integrity, particularly where anti-counterfeiting measures matter. Results depend on trust in basic materials, so specs and labels aren’t just bureaucracy—they protect both science and safety.
Preparation Method: Practical Chemistry in Action
Real-world synthesis rarely mimics textbook routes. For 3-aminoquinoline, several main pathways exist. One time-honored approach starts from 3-chloroquinoline or 3-nitroquinoline. Nucleophilic aromatic substitution opens the door to install the amino group, often powered by ammonia under pressure or catalytic hydrogenation with supported metals. The choice of starting materials echoes the balance chemists strike between cost, scalability, and available equipment. On the research bench, small batches might use simple glassware and standard heating, while industrial settings shift to continuous flow or high-pressure reactors for bulk output. Each path writes its own story about the priorities—yield, purity, throughput—guiding process chemistry in labs worldwide.
Chemical Reactions & Modifications
After basic synthesis, innovation moves forward through modification. 3-Aminoquinoline stands as a flexible platform for further synthetic work: acylations and alkylations generate new derivatives by transforming the amino group. Electrophilic substitutions on the aromatic core give further depth, allowing the molecule to be tailored for specific biological targets or physical requirements in material science. Reductive methods can be used to fine-tune electronic properties or prepare precursors for polymerization. Each new reaction cracks open unexplored chemical space, feeding back into the cycle of research and discovery. Academic and industrial labs require this kind of flexibility to chase promising leads or adapt to emerging technical requirements.
Synonyms & Product Names
Names in chemistry reveal a lot about a compound’s journey. 3-Aminoquinoline has picked up multiple aliases in databases and catalogs: sometimes it’s 3-quinolinamine; sometimes researchers refer to it by short-hand indexes or trade identifications. These variations highlight both the spread and the specialty use cases—chemical suppliers, research papers, and safety data all may use different language depending on audience and context. Navigating these synonyms becomes essential for data mining and for avoiding duplicative research. It’s a fact of life for scientists and procurement teams alike: the vocabulary might shift, but the underlying compound stays the same.
Safety & Operational Standards
Lab safety often comes down to good habits, not just written guidelines. With 3-aminoquinoline, basic precautions matter. Standard PPE makes a difference: gloves keep skin clear, and working in a well-ventilated hood reduces inhalation risk. Accidental spills are cleaned up with absorbent materials designed for aromatic amines, not just everyday lab towels. Waste management needs attention too, since quinoline derivatives can linger in the environment and sometimes require specific treatment before disposal. Over the years, regulatory trends have demanded tighter tracking, whether for worker health or environmental diligence. Teams train new researchers with protocols based on lessons learned from actual mishaps, not just theoretical risks.
Application Area: Real-World Value
What drives continued interest in 3-aminoquinoline lies in its value across industries. Antimalarial research draws on this backbone because quinoline analogues block parasite growth at key life stages. In oncology, researchers test new derivatives for enzyme inhibition, especially in search of next-generation treatments. Chemical manufacturers value the compound as a precursor for dyes and advanced polymers—taking advantage of stability and tunable reactivity. Some labs in material science employ quinolines as ligands or chromophores, where electronic features turn out to be just as important as biological compatibility. These multisector uses send the compound on a journey from bench to bedside, and even into the world of advanced materials, far from its initial roots.
Research & Development Efforts
Investment in R&D for 3-aminoquinoline keeps resurfacing, especially as new screening technologies uncover unknown activities or properties. High-throughput platforms test hundreds of analogues for biological effects, feeding data back into machine learning models that prioritize which derivatives to pursue. Synthesis isn’t static—chemists continually refine conditions to minimize byproducts, trim down energy costs, or circumvent hazardous reagents. Collaborative efforts, sometimes crossing pharmaceutical, academic, and industrial boundaries, bring diverse expertise together under one roof. These partnerships generate acceleration: a promising anticancer analogue one year could transform into a licensed product ready for trials far sooner than in the past, especially with open-access data speeding up literature discovery.
Toxicity Research: Limits and Cautions
Examining toxicity shapes every discussion about chemical innovation. With 3-aminoquinoline, investigators must confront both acute and long-term exposure outcomes. Aromatic amines raise red flags—some compounds in this class can act as mutagens or irritants. As regulatory awareness grows about the risks posed by poorly understood lab chemicals, modern toxicology emphasizes careful profiling before moving from the lab to commercial use. Animal studies and in vitro data inform how processes adapt to protect workers, consumers, and the environment. Actual incidents serve as reminders: short-term exposure might not always cause immediate symptoms, but chronic contact ramps up risk. Better testing, combined with improved labeling and transparent communication, ensures chemical research doesn’t outpace safety.
Future Prospects
Looking forward, 3-aminoquinoline stands perched at a new era of chemical development. Next-generation synthesis methods—like photoredox catalysis or biocatalysis—promise cleaner, more sustainable preparation. The field of medicinal chemistry eyes this scaffold for its epidemiological relevance, especially with infectious diseases that adapt rapidly. Greater integration of computational modeling and AI could soon predict toxicity or activity profiles, narrowing down which modifications matter before labs commit valuable resources. Green chemistry advocates push for processes that generate fewer waste byproducts or use renewable feedstocks, changing the environmental footprint at scale. As the broader chemical industry steers toward responsible innovation, compounds like 3-aminoquinoline illustrate the balance between creative possibility and the responsibility to manage risks, deliver value, and protect communities linked by the invisible web of global chemistry.
What is 3-Aminoquinoline used for?
Stepping Into the World of 3-Aminoquinoline
Whenever I sit down to break down a chemical compound, I ask myself two things: what does it do, and why should anyone care? In the case of 3-aminoquinoline, both questions bring up some real excitement from the pharmaceutical world. Chemists rely on quinoline-based compounds for projects where only serious molecules will do, especially in tackling disease.
Why This Molecule Stands Out
3-Aminoquinoline isn’t the sort of chemical you see lining grocery store shelves or popping up in food products. Its home sits in research labs and the hard grind of drug development. Chemists working with this building block focus on its unique structure—a quinoline backbone with an amino group at a key spot. This basic change turns it into a versatile starting line for experiments and research that goes after some of the toughest health challenges we know.
Diving Into Malaria Treatments
Malaria remains relentless in many corners of the world. For decades, scientists have taken aim at this disease, trying to outsmart drug resistance and break the deadly cycle. 3-Aminoquinoline marks a central feature in a group of drugs called antimalarials. Chloroquine—widely used in the past—owes much of its power to a similar chemical foundation. Now that the parasite is growing resistant, the search is on for fresh molecules. Researchers pick apart compounds like 3-aminoquinoline, tweak their makeup, and test them to find new medicine candidates. Data from the World Health Organization points to the urgent need for these efforts since more than 600,000 people still die from malaria every year, mostly kids under five in Africa.
Cancer Research and Antiviral Potential
Big names in healthcare aren’t just looking at this compound for malaria. Scientists test quinoline derivatives for anti-cancer activity. The way 3-aminoquinoline interacts with DNA and other parts of tumor cells opens up routes for treatment ideas. Some research teams have even flagged it as useful in designing drugs to fight hepatitis and, in rare cases, viruses like HIV. These ideas remain at the research stage for now, but the early facts give experts reason to keep pushing.
Creating Tools for Chemical Synthesis
Every chemistry graduate student I know has spent late nights piecing together molecules. For them, 3-aminoquinoline offers a flexible foundation. Its ability to connect with different chemical groups means it shows up all over academic literature—chemists use it to make probes, dyes, and tools that help monitor biological changes in cells. This sort of work might not make headlines, but it matters every time a researcher sits down to “see” a disease process in a live cell for the first time.
Challenges and Responsible Innovation
Every big solution brings its own set of problems. Drug resistance rolls forward like a wave, wiping out one drug after another. Oversight is crucial. The more we use these compounds for treating malaria, infections, or even cancer, the more carefully researchers and healthcare workers have to track unexpected effects and emerging resistance. Investing in clear communication, funding basic research, and supporting local labs worldwide can balance the risks against the huge potential of compounds like 3-aminoquinoline.
What is the chemical formula of 3-Aminoquinoline?
Understanding the Core Structure
Anybody who’s worked in labs recognizes how certain chemical names carry weight. Mention 3-Aminoquinoline in a research setting, and chemists immediately picture a two-ring system, one nitrogen, and an amino group. The chemical formula, C9H8N2, packs in both history and potential. Quinoline compounds have figured prominently in malaria research and cancer drug development, so this molecule is hardly a background player.
Where 3-Aminoquinoline Makes a Difference
Medicinal chemists keep turning back to compounds like this. At the heart of many antimalarial drugs is the quinoline core, first discovered in ancient herbal remedies, modernized in the 1800s, and reinvented a dozen times since. 3-Aminoquinoline’s arrangement nudges the molecule into a position to interact with enzymes and DNA, making it a useful starting point for innovation. Chloroquine and amodiaquine—both rooted in quinoline chemistry—changed the fight against malaria. Scientists see C9H8N2 and think about where new treatments might start.
Why Naming and Formula Matter for Real Research
A formula written out on a page might not feel personal. Speaking from multi-year stints in organic labs, the tickets to progress are speed and accuracy. Misplacing an amino group turns a promising medicine into a dud—or worse. 3-Aminoquinoline is “3-amino” because the NH2 sits on the third carbon, and slight rearrangements can mean all the difference for safety and effectiveness. Some of the most frustrating wasted weeks in the lab come down to a misplaced group or misread structure, so clarity isn’t just paperwork. It’s about not setting research budgets on fire.
How Modern Tools Are Changing the Game
Computational chemists run simulations with C9H8N2 to spot new drug targets before anybody touches a flask. Big data and AI sort through thousands of derivatives in seconds. The right software catches errors that used to derail whole studies. Even so, none of these tools erase the basics: the chemical formula keeps everyone on the same page, whether a project is in Boston, Singapore, or Nairobi.
Diving Into the Safety Side
I remember the first safety seminar that made me look sideways at even simple compounds. 3-Aminoquinoline, like lots of nitrogen-containing organics, can irritate skin, eyes, and lungs if handled carelessly. The more labs move to automation, the fewer folks memorize MSDS sheets. Online chemistry forums fill with reminders: don’t skip gloves, work in a fume hood, label everything twice. The formula helps identify risk at a glance. Folks who cut corners usually have stories they’d rather forget.
Finding Solutions for Industry Needs
Pharmaceutical companies need reliable, clearly defined building blocks. Regulatory agencies demand unambiguous documentation—for researchers and for patient safety. Technology keeps improving routes to synthesize 3-Aminoquinoline efficiently. More green chemistry methods trim out harmful solvents; robust analytics ensure batches are pure. Industry partners push for quick integration of new data, linking every batch back to the original formula: C9H8N2.
Looking Ahead
Research never stands still. The next big leap in infectious disease or cancer might hinge on a minor tweak to a molecule like 3-Aminoquinoline. Getting the formula right might seem simple, but real breakthroughs often start with understanding the basics—before taking the risks and leaps that move science forward.
Is 3-Aminoquinoline hazardous or toxic?
Looking into Chemical Safety
Stepping into a lab, you get used to reading safety labels and wondering whether a chemical will sting, stain your skin, or cause something far worse. That feeling never fades, even for smaller chemicals like 3-Aminoquinoline, which might not ring bells for most but does show up across drug research and fluorescent dye work. The question keeps popping up: Is it hazardous, and what should people really know before opening a bottle?
What the Research Says
3-Aminoquinoline belongs to a group of compounds—quinolines—with complicated relationships to human health. Several quinolines show up on lists for possible toxicity, especially when handled regularly at research or industry scale. The U.S. National Library of Medicine points out that 3-Aminoquinoline causes irritation to skin, eyes, and the respiratory system. If inhaled or ingested, it can trigger symptoms including headaches, dizziness, and nausea.
There’s a reason chemical safety sheets shout for gloves and goggles. Accidental exposure often causes rashes, and a splash in the eye can lead to burning pain. Long-term or repeated contact raises bigger questions, with uncertainty over whether this might drive chronic effects. A review in the Journal of Chemical Health & Safety flagged the lack of deep toxicity trials, which tells me that researchers haven’t fully mapped how this compound acts in the human body if someone is exposed over months or years.
Potential for DNA Damage
It’s hard to avoid mention of quinolines without coming face-to-face with stories about mutations and DNA damage. Some close chemical relatives get listed by the International Agency for Research on Cancer as possible carcinogens. Direct data about whether 3-Aminoquinoline drives this kind of risk looks thin, but its structure and chemical cousins raise eyebrows.
At my own university, our safety training told stories of young researchers skipping gloves, thinking they’d save time. Months later, someone complained of chronic eczema and ended up tracing the problem to repeated splashes with aromatic amines—3-Aminoquinoline included. It doesn’t take much to set off a chain reaction with these compounds. Over-the-counter treatments can only go so far when you pick up a rash from chemicals.
Safe Handling Pays Off
Safety culture has changed for a reason. Fume hoods and gloves aren’t decorations; they really do reduce exposure and keep people healthy. Working in a lab that stored 3-Aminoquinoline, I always took the fume hood rule seriously. Even a quick weigh-out wafted a bitter smell, and the difference between a careful transfer in the hood and trying it on a crowded benchtop? No headaches, no cough, and no mess to clean off your hands.
Disposal stays important too. City wastewater plants rarely know how to treat specialty organics, and flushed leftovers may linger in the environment. Studies from environmental journals show some quinoline compounds end up in soil and water, shifting over time into new forms that may carry environmental risks we don’t measure enough.
Room for Solutions
Down the hall from my old workspace, someone once asked: “Why are we using this at all?” Sometimes there’s no perfect substitute for a specific chemical step, but process engineers and green chemistry teams keep hunting. Swapping in safer amines, cutting down on unnecessary use, and choosing closed systems for transfers all contribute to healthier labs.
Curiosity drives science forward, but respect for hazard regulations and sensible procedures keeps people in the field year after year. For any researcher new to 3-Aminoquinoline, it pays to read the label, remember that a compound’s calm appearance masks real risks, and look for less hazardous approaches whenever possible.
What are the storage requirements for 3-Aminoquinoline?
The Real Story Behind 3-Aminoquinoline Storage
Getting storage right for chemicals like 3-aminoquinoline isn’t about following textbook protocols. It’s about protecting lab workers, research, and the facility. Anyone who’s handled aromatic amines knows they can be finicky, sometimes even dangerous, if left to their own devices. I recall my first time in a research lab—no manual could prepare you for the smell or the anxiety of stacking brown glass bottles, hoping they wouldn’t react to a forgotten sunlight beam.
Risks of Careless Storage
3-Aminoquinoline carries risks. This isn’t about danger for its own sake. The chemical sees use in pharmaceutical research and dye manufacturing, making it a staple in plenty of labs. It comes as a yellow-brown powder, and its key threats stem from two properties: it’s potentially harmful on skin contact, and it may irritate airways if inhaled as dust. Storage errors lead to health problems or costly contamination. Mistakes cost more than cleanup—they can jeopardize entire research projects or the health of anyone nearby. In pharmaceutical research, contamination ruins bench work, months of progress, and even critical financial grants.
Practical Storage Requirements
No one in a bustling lab wants to double-check storage guidance every time a shipment arrives. Based on my years working in pharma R&D, these basic rules keep 3-aminoquinoline safe and stable. Keep it in tightly sealed containers—amber glass works best, especially in shared spaces. Ordinary polypropylene or polyethylene jars lose their integrity over time. Shelf stability drops off fast in sunlight or fluorescent light, as prolonged exposure degrades the powder. Stash it well away from heat sources. I’ve seen powder stick to stoppers and clump after accidental heat exposure, throwing off subsequent measurements and contaminating pipettes.
Dry conditions matter. Moisture finds its way into everything over time, and 3-aminoquinoline doesn’t handle damp air. It clumps, reacts with trace water, and can even break down, leaving impurities that sabotage research. Squeeze packets of silica gel into storage areas and keep the chemical off floors or windowsills, away from accidental splashes. Even a sudden downpour on an open window can ruin an entire bottle overnight.
Health and Regulatory Concerns
Safety data sheets remind researchers to keep 3-aminoquinoline locked away from acids and oxidizers. In one facility I worked at, a careless mix-up of bottle placement led to a slow-forming crust on the rim, which the postdoc only discovered during a late-night inventory. Inhaling even tiny amounts causes throat discomfort and coughing fits, so storing it near dusty bags or open shelving invites accidents. Gloves, lab coats, and well-ventilated storerooms aren’t optional—they’re enforced by responsible lab managers. Scientists know regulators inspect more than paperwork—they check shelves for proper labeling, secure lids, and even temperature logs. Failure means warnings, lost funding, or, worse, chemical spills ignored by oversight bodies. In my experience, teams with up-to-date labeling systems and regularly rotated chemical stocks keep both research and researchers safe.
Getting It Right—and Keeping It Right
There’s plenty written about best practices, but nothing beats direct responsibility. Yearly training refreshers help. Clear signage, locked cabinets, and a habit of quick visual checks go a long way. New students mess up—senior team members set the tone. Neatness, vigilance, and respect for the chemical turn guidelines into real safety. Labs built on these habits rarely face unplanned accidents or failed experiments caused by something as avoidable as improper storage. Getting storage right means more than compliance—it means protecting every person, experiment, and investment in the facility.
Can 3-Aminoquinoline be used in pharmaceutical research?
Digging into the Science
Chemists have long turned to heterocyclic compounds when looking for new molecules that can change the way we fight disease. 3-Aminoquinoline is one of those interesting structures. It pops up in scientific journals as a building block for a range of promising pharmaceuticals. This molecule carries a pair of nitrogen atoms tucked inside a fused ring, which gives it some unique chemistry. Medicinal chemists know that arrangements like this tend to interact well with enzymes or proteins that drive diseases.
Why the Structure Matters
The arrangement of atoms in 3-aminoquinoline matters because it’s not just decorative. It helps the molecule fit into the pockets of biological targets like a key in a lock. Researchers at universities and drug firms use it to create new compounds by swapping atoms around that nitrogen spot. Some even link it to other rings or chains to create hybrids, hoping to boost through blood barriers or stick to receptors that help block malaria or even some cancers.
What Are Scientists Using It For?
Exploring the medical literature, you’ll see 3-aminoquinoline showing up in projects aiming to treat malaria, bacterial infections, and auto-immune disorders. It’s in the family tree of some antimalarial drugs—chloroquine and amodiaquine have a similar backbone. Even after decades, researchers haven't run out of ways to tweak these molecules in search of lower toxicity or better activity against tricky strains.
I’ve spoken with researchers who look for re-invented drugs to solve resistance problems in malaria. For them, the ability to modify each position of the quinoline ring makes compounds like 3-aminoquinoline exciting to work with. Every small change can mean a big shift in activity or safety.
3-Aminoquinoline in Today’s Drug Labs
Drug discovery feels like panning for gold. Chemists keep exploring new derivatives of 3-aminoquinoline because each one could carry potential for something new. The search for new antibiotics and novel cancer therapies leads back to structures people have worked with for decades. According to PubMed and similar sources, researchers use this molecule in virtual screens, then synthesize and test the best hits against bacterial and cancer cells.
Some projects use 3-aminoquinoline to help tune selectivity—so a compound hits its intended target and leaves healthy cells alone. For companies and labs, quicker fails save money and time. A structure like this gives a lot of opportunities to make changes on the fly.
The Drawbacks and Risks
No molecule comes without baggage. Highly active compounds can carry side effects or trigger reactions in organs that have little to do with the disease. Chemists and toxicologists need to keep a close eye on new compounds built from 3-aminoquinoline, especially as researchers seek more potent versions. Early animal and cell studies act as a filter before sending anything down the lengthy pipe to clinical trials.
Drug resistance creeps up over time. History with antimalarials shows what happens if you lean too hard on one scaffold. Any new drug built on 3-aminoquinoline would need careful management, strong data sharing, and vigilance in the clinic to avoid meeting the same fate.
Ideas Moving Forward
Researchers are already feeding what they learn with 3-aminoquinoline into new platforms—like machine learning simulations—to sort through chemical combinations faster than before. Open data is helping bridge the gap between academic labs and pharma companies. Bringing more minds together increases the chances of finding something that treats disease without adding fresh risks. The chemistry around 3-aminoquinoline isn’t finished; it’s pushing forward with every experiment and every new theory.
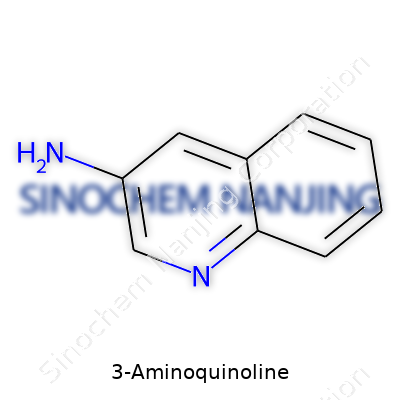
| Names | |
| Preferred IUPAC name | Quinolin-3-amine |
| Other names |
3-Quinolinamine 3-Quinolylamine |
| Pronunciation | /ˈæmɪnoʊ.kwɪnəˌliːn/ |
| Identifiers | |
| CAS Number | 86-17-3 |
| 3D model (JSmol) | `3DModel__JSmol__QN1c1ccc2nccc(N)c2c1` |
| Beilstein Reference | 84874 |
| ChEBI | CHEBI:38411 |
| ChEMBL | CHEMBL285866 |
| ChemSpider | 12071 |
| DrugBank | DB04837 |
| ECHA InfoCard | 03a0eaf4-b7b4-4f94-bcb5-aad1292a5d8c |
| EC Number | 213-035-3 |
| Gmelin Reference | 87589 |
| KEGG | C08678 |
| MeSH | D015352 |
| PubChem CID | 69414 |
| RTECS number | AT0705000 |
| UNII | U6M3PE2GDR |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C9H8N2 |
| Molar mass | 229.28 g/mol |
| Appearance | Light yellow to yellow crystalline powder |
| Odor | Odorless |
| Density | 1.23 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 1.23 |
| Vapor pressure | 0.0000346 mmHg at 25°C |
| Acidity (pKa) | 5.00 |
| Basicity (pKb) | pKb = 6.10 |
| Magnetic susceptibility (χ) | -57.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.719 |
| Viscosity | 1.254Pa.s (20°C) |
| Dipole moment | 2.52 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 163.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3234 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D04AA09 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P264, P270, P271, P301+P312, P304+P340, P311, P330, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 127°C |
| Autoignition temperature | 715°C |
| Lethal dose or concentration | LD50 (oral, rat): 750 mg/kg |
| LD50 (median dose) | LD50 (median dose): Mouse intravenous 114 mg/kg |
| NIOSH | BQ3500000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 100-500 mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Quinoline 2-Aminoquinoline 4-Aminoquinoline 8-Aminoquinoline Chloroquine |

