3-Aminopyridine: A Closer Look at a Resilient Chemical
Historical Development
Aminopyridines have been quietly shaping industrial and scientific progress for over a century. Chemists first outlined the family of aminopyridines in the late 1800s, tracing their origins back to classic organic synthesis. These were the sort of compounds that gave old-school laboratories plenty of buzz. Pyridine itself stood out early on for its odd, fishy smell and basic character. Once researchers figured out how to bolt an amino group onto the aromatic ring, worlds opened up in both synthesis and applications. 3-Aminopyridine, with the amino group at the meta position, drew curiosity because of its unique reactivity and biological properties compared to its siblings. Over the years, demand grew as pharmaceuticals, dyes, and agrochemical sectors discovered the value of reliable pyridine derivatives.
Product Overview
3-Aminopyridine is not the sort of chemical most people will ever hear about, yet it keeps turning up in so many cutting-edge projects. Anyone who wears a lab coat knows its distinct, ammonia-tinged scent. Its widespread use in scientific research and industry speaks to its solid reputation for reliability and precision in reactions. 3-Aminopyridine serves as a building block, a reagent, and, at times, an active substance on its own. In the world of chemical synthesis, it’s respected for its versatility–it often finds its way into pharmaceuticals, crop protection agents, and more, demonstrating a remarkable ability to adapt to diverse technical needs.
Physical & Chemical Properties
Ask a chemist to describe 3-Aminopyridine and they’ll likely paint a clear picture: pale yellow or almost white crystalline powder, not too dense, with a melting point around 60-63°C. Its molecular formula, C5H6N2, carries the typical signature of a pyridine with an extra amino group lending it a touch of basicity beyond the parent compound. The compound dissolves fairly well in water and other polar solvents, which helps when mixing it into complex reactions. Thanks to its aromatic structure, 3-Aminopyridine doesn’t just play well with acids and bases; it also reacts in more specialized settings where electron-donating and -withdrawing effects matter. A faint, sharp odor reminds even the most experienced chemist not to underestimate its presence.
Technical Specifications & Labeling
Laboratories and manufacturers treat 3-Aminopyridine with a great deal of care because purity and stability are non-negotiable. Access to high-grade material–often at 98% purity or higher–matters for reproducible results, especially in pharmaceutical synthesis. Labels usually feature the chemical structure, international name, batch number, and purity. Exacting quality control limits moisture and impurities since even small deviations threaten downstream reactions, making robust testing protocols important throughout the supply chain. Contamination triggers waste and, in some cases, costly delays in research or manufacturing. So, good stewardship begins with clear labeling and a culture of thorough documentation.
Preparation Method
Preparation of 3-Aminopyridine usually follows two main paths in industrial contexts. Direct amination of pyridine derivatives, especially using classical Sandmeyer-type reactions starting from 3-chloropyridine, produces reliable yields. In certain setups, reduction of 3-nitropyridine delivers the compound with decent efficiency. Both routes require precise control of temperature, catalyst loading, and handling of intermediates, such as nitro or chloro compounds, which can misbehave under careless conditions. Green chemistry approaches now encourage milder conditions and fewer hazardous reagents, reflecting the industry’s move toward safer production and waste reduction. No matter the method, the goal remains the same: high yield, low impurities, and sustainable process design.
Chemical Reactions & Modifications
The amino group of 3-Aminopyridine gives chemists freedom to explore many reaction pathways. Acylation and sulfonation enable synthesis of new molecules for medicinal chemistry. Developers often convert the amine to derivatives such as amides, ureas, and hydrazides, which can enhance biological activity or tailor pharmacokinetics. Electrophilic aromatic substitution, while less pronounced because of the pyridine’s electron structure, still enables selective modifications in the ring system. Some researchers exploit this reactivity to produce more complex heterocycles, fueling the search for novel drugs or molecular probes. Reductive and oxidative coupling open even broader horizons, amplifying 3-Aminopyridine’s role in multi-step synthetic routes.
Synonyms & Product Names
3-Aminopyridine goes by several aliases in scientific literature and commerce. “Meta-aminopyridine” makes reference to its ring position, while “Pyridin-3-amine” crops up in IUPAC contexts. Catalogs sometimes list it as “3-Pyridinamine” or “3-AP,” reflecting shorthand for busy chemists. Whether it’s listed under these names or Cas No. 462-08-8, the chemical’s distinctive properties remain the same.
Safety & Operational Standards
Handling 3-Aminopyridine takes genuine caution born of respect for both its utility and its risks. Though not the most dangerous compound in the lab, it carries enough acute toxicity to warrant gloves, goggles, and fume hoods at all times. The solid or vapor can irritate skin, eyes, and respiratory passages. Chronic exposure, particularly over many months or years, can lead to more permanent health concerns, including nervous system effects. Anyone storing or transporting it must follow regulations on hazardous substances, keeping containers tightly closed and far from acids, oxidizers, or heat sources. Cleaning up spills calls for fast action and disposal of any contaminated materials according to local laws. No shortcut replaces a safety-minded culture grounded in training and diligence.
Application Area
3-Aminopyridine does the heavy lifting in several modern industries. In pharmaceuticals, it acts as a precursor for drugs targeting neurological disorders, including promising treatments for multiple sclerosis and Lambert-Eaton myasthenic syndrome. Some agrochemical producers sharpen their pesticide formulas using aminopyridine-based chemistry, optimizing effectiveness while watching for potential ecological impact. Materials scientists exploit its structure to produce new dyes and functional polymers. Custom synthesis firms value its building-block potential for developing innovative molecular scaffolds. It turns up behind the scenes and, often, ends up as the secret sauce in products that improve everyday life in subtle yet meaningful ways.
Research & Development
Research around 3-Aminopyridine never slows down, even as its basic chemistry seems well mapped. Innovation drives continuous improvement in synthesis, analytics, and application. Teams investigate analogs and derivatives to unlock better pharmacological profiles, more durable polymers, or sharper electronic properties. Green chemistry ideas float through most R&D labs as experts work to shrink solvent use, lower energy demands, and switch to renewable raw materials. Analytical chemists refine methods for trace detection and purity checks, serving both production and regulatory reporting. Better understanding of reactivity, bioavailability, and safety inspires product evolution, whether for medicine, electronics, or agriculture.
Toxicity Research
The study of 3-Aminopyridine’s toxicity attracts both clinical and environmental interest. Animal studies show that improper exposure produces nervous system aggression, convulsions, and in high doses, fatal outcomes. Medical uses always demand tight dosing regimens, patient monitoring, and careful risk-benefit evaluations. Researchers review metabolic pathways, excretion, and potential accumulation to set appropriate exposure limits for both patients and workers. On the environmental side, studies focus on breakdown rates in soil and water, examining how quickly and completely the substance degrades. Keeping toxicity in focus helps doctors, regulators, and industrial operators make informed decisions based on science, not just economic drivers.
Future Prospects
Looking ahead, 3-Aminopyridine’s story continues to unfold. Its proven past in drug research, materials science, and agrochemicals doesn’t stop others from looking for smarter, safer, or more sustainable alternatives. Synthetic chemists experiment with bio-based sources and cleaner process designs. Data from high-throughput screening and computational chemistry accelerates discovery of new derivatives. Regulatory pressure pushes for greener disposal, responsible sourcing, and tight control of health impacts. Some researchers hint that next-generation applications–perhaps in advanced energy storage or new nerve therapies–could push this venerable molecule into fresh territory once considered beyond reach. The pursuit of safer, more effective, and environmentally sound chemistry remains a strong current pulling 3-Aminopyridine forward into whatever new roles the future demands.
What is 3-Aminopyridine used for?
Understanding Its Role in Science and Medicine
3-Aminopyridine doesn’t pop up in everyday conversation, but step into a lab or a pharmaceutical manufacturing plant and this compound has a reputation. It’s a small organic molecule, shaped by a pyridine ring with an amino group, and turns a simple structure into a building block for many industries. In my years of studying chemical pathways and reading through stacks of pharmacology journals, this molecule stood out for a few reasons.
Specialty in Neurology
Doctors and researchers have explored 3-aminopyridine for its effects on nerve signaling. It blocks potassium channels in nerve cells. This blocking action allows nerves to send signals longer and more effectively, which comes in handy for conditions where these signals break down. For instance, researchers looked at its cousin, 4-aminopyridine, to improve movement in patients with multiple sclerosis. 3-Aminopyridine shares similar properties and has shown promise in studies focusing on neuromuscular diseases and spinal cord injuries. Some research teams believe it may help recover motor function after injury, which brings hope to many patients.
Value in Drug Synthesis
Beyond its direct medical uses, chemists like me have often reached for 3-aminopyridine when creating new pharmaceutical agents. Its structure serves as a core unit in the synthesis of antiviral drugs, cancer therapies, and anti-inflammatory medicines. It acts as a starting point—the scaffold—on which more complex medicine molecules can be built. Access to this chemical means researchers push forward in creating next-generation therapies. The ability to use it as a building block speeds up early-stage drug research and development.
Applications in Chemical Reactions
In industrial chemistry, 3-aminopyridine supports the manufacture of dyes, agrochemicals, and corrosion inhibitors. When I worked on a project aimed at developing improved crop treatments, derivatives from 3-aminopyridine allowed us to design molecules that protected plants from fungal diseases without toxic residue. This mattered, as safer crops yield better food security. Chemical manufacturers choose this compound precisely because it reacts efficiently and forms strong links with other molecules, driving innovation across fields.
Risks and Responsible Handling
Working with 3-aminopyridine means following strict safety measures. The compound has known toxic effects when handled improperly. Lab guidelines require gloves, proper ventilation, and eye protection every time handling or synthesis occurs. In my own laboratory experience, safety briefings around such chemicals focus on preventing exposure. Accidental ingestion or inhalation can harm the nervous system, reflect symptoms like muscle twitching or convulsions, and send staff straight to emergency care. These risks stress the need for ethics and responsibility in distribution and storage.
Looking Forward: Steps for Better Use
As new therapies push ahead and industrial innovation continues, open data sharing between labs and government regulators can reduce risks associated with chemical misuse or accidental exposure. Training sessions in research and university environments can reduce incidents, and investment in green chemistry could pave the way for safer derivatives with similar benefits. Collaboration ensures 3-aminopyridine continues to help, not harm, the communities and professionals relying on its unique properties.
What is the chemical structure of 3-Aminopyridine?
Understanding 3-Aminopyridine at a Glance
3-Aminopyridine belongs to a family of molecules with a pyridine ring, which is a six-membered structure holding five carbon atoms and one nitrogen atom. In 3-Aminopyridine, an amino group (-NH2) sits on the third carbon of that ring—pretty specific real estate in organic chemistry. The actual chemical formula is C5H6N2, painting a small but mighty picture. Scientists sketch the molecule as a hexagon, with a single nitrogen up top and an amino group pointing off that third carbon slot, something that signals big differences compared to other pyridine derivatives.
The Power and Pitfalls in How Molecules are Arranged
Small tweaks in these structures can turn a molecule from benign to a useful tool in the lab or hospital, or sometimes make it risky. The placement of that amino group on position three changes how this compound interacts with proteins and enzymes in the body. I’ve watched chemists puzzle over reactions just because a functional group switched places. The world of pharmaceuticals runs on these distinctions, as molecular structure often guides drug action. 3-Aminopyridine steps into this conversation mainly as a molecule with potential, particularly in nerve signaling studies.
Roles in Research and Medicine
Over the last decade, researchers explored 3-Aminopyridine’s ability to block certain potassium channels in nerve cells. Potassium channels shape the electrical activity in nerves, which drives muscle function and sensation. Shutting those channels off, even temporarily, may help restore function in damaged nerves, like what happens in certain types of paralysis or nerve injuries. Scientists tested related aminopyridines in clinical trials for multiple sclerosis, hoping to boost nerve signals in damaged pathways. Those drugs come from careful study of structure-activity relationships—and 3-Aminopyridine’s unique setup underlines just how crucial small shifts in a molecule’s architecture truly are.
Handling Safety and Trust
The backbone of trust in science and medicine rests on showing clear, evidence-based information. Reports indicate that 3-Aminopyridine itself can act as a toxin in higher doses, so people working with it take plenty of safety precautions. Public agencies like the National Center for Biotechnology Information (NCBI) keep this data accessible, listing hazards and recommended handling practices. It’s uncomfortable to talk about risks, but transparency about a compound’s profile always strengthens decision-making.
Pushing Forward Through Research
Cutting-edge research depends on having the right tools in the right hands. 3-Aminopyridine gives neuroscientists and clinicians a way to test nerve signal processes and potentially new treatments for nerve diseases. Keeping up with laboratory best practices and following regulatory guidance—such as those laid out by the U.S. National Institutes of Health—helps ensure progress carries less risk. Every new experiment adds a little more detail to the story, and all that traces back to how and where chemists place single atoms on a ring.
Looking Ahead for Safer and Smarter Solutions
The focus on 3-Aminopyridine’s structure isn’t just about chemical trivia—it stands as a reminder that precision in science saves lives. Continued research and open dialogue with safety experts, health professionals, and the public create a foundation for ethical discovery. When chemists and physicians keep their eyes on structural details and communicate findings plainly, everyone wins: researchers push boundaries, doctors make informed calls, and people get safer solutions for some of the toughest health challenges out there.
Is 3-Aminopyridine hazardous or toxic?
Understanding 3-Aminopyridine Beyond the Lab
3-Aminopyridine pops up in chemical supply catalogs, research labs, and industrial concoctions. Most people outside chemistry circles won’t recognize the name, but anyone working with pharmaceutical intermediates or specialty dyes has probably handled it. The thing about 3-aminopyridine: it’s not an everyday household chemical, but plenty of researchers, factory technicians, and students come into contact with it during daily work.
Direct Health Hazards: What Happens If You’re Exposed?
Breathing in dust or vapors from 3-aminopyridine brings on headaches, dizziness, nausea, and sometimes tremors. Touching it with bare skin can cause irritation, rashes, or worse if you don’t wash it off right away. Eyes burn and water when exposed to a splash. Swallowing even a small amount can send someone to the emergency room and spark dangerous nervous system reactions.
The U.S. National Institutes of Health lists 3-aminopyridine as a hazardous substance, with acute toxicity for humans and animals, and not just in monster doses. Ingestion or lengthy exposure can cause rapid heart rate, high blood pressure, restlessness, or seizures. The nervous system reacts quickly, with symptoms that don’t wait for slow build-up. There’s also evidence of toxic effects on the liver and kidneys after prolonged exposure.
Why the Risks Don’t Stop at Short-Term Symptoms
I spent some years in a university chemistry lab. We handled chemicals daily, following safety protocols that sometimes felt like overkill. But with chemicals like 3-aminopyridine, lax safety culture can’t be allowed. Chronic exposure, even at low doses, sees health issues compounding over time. Gloves and eye shields became a line of defense, not just a rule from a safety manual.
Spills and contamination aren’t rare during synthesis or transport. If a fume hood malfunctions or PPE slips, you could go home with more than a lingering smell on your clothes. Chemical burns and allergic reactions can spring up hours later. People working long hours around chemicals need a culture where double-checking protocols is automatic, not a chore.
The Need for Information and Better Handling
Finding clear safety data for some specialty chemicals gets tricky, and 3-aminopyridine is no exception. Everyone in the supply chain—from distribution staff to lab techs to waste disposal workers—should access information sheets that don’t just rattle off properties but explain practical risks. Too many labs and facilities lag on education, with underfunded training sessions or out-of-date manuals. That puts not just users, but anyone nearby, at risk.
Better Practices Make All the Difference
Moving toward safer workplaces means ventilation that’s properly maintained, training that covers not just emergency wash stations but also real-world accident scenarios, and clear labeling. If 3-aminopyridine can cause twitching, respiratory distress, and serious organ damage, then safe storage away from incompatible substances isn’t optional. Regular audits, routine health checks for at-risk workers, and clear incident reporting encourage accountability.
The structure of modern research and industry leaves no room for guessing or skipping steps when hazardous chemicals enter the picture. By treating every step—from understanding toxicity to controlling exposure—as a matter of worker health, organizations show respect for those whose hands do the actual work.
How should 3-Aminopyridine be stored?
Why Taking Care with 3-Aminopyridine Matters
Anyone who has worked in a lab probably knows that safety gets tested by the smallest details. 3-Aminopyridine stands out as one of those chemicals that looks pretty straightforward on its info sheet. It's a white-to-light yellow powder, it has that sharp, almost fishy odor—and it doesn't look half as intimidating as many others. That’s where people can lower their guard. This compound deserves respect, especially after you consider how it behaves over time and in certain conditions.
Getting Temperature and Air Right
The real test comes down to temperature and exposure. With 3-Aminopyridine, stable room temperatures feel right, usually around 20–25°C. Even in a storeroom without the hum of high-tech climate control, basic steps go a long way. I’ve seen chemicals left in mop closets or window sills soak up heat and humidity, ruining the purity and, sometimes, creating hazards researchers never expected. Sealing the container keeps out air and moisture, which drive up the risk of unwanted reactions, like slow oxidation. Every now and then, I’ve watched as humidity causes clumping and unexpected exothermic reactions—not something you want to explain to a safety officer.
Why the Container Isn't Just a Container
It’s easy to overlook, but not every plastic or glass jar is equal. In my time caring for chemical stockrooms, I’ve seen far too many cracked lids and odd substitutions. The right call comes down to using an airtight glass vessel, never metal, since metals and 3-Aminopyridine can interact in ways that ruin the batch and, rarely, spark corrosion. Glass also lets you spot early signs of trouble: moisture beading inside or color shifting. Always label everything, no matter how certain you feel now that you’ll remember what’s inside months later. It keeps the whole team on the same page and avoids cross-contamination.
Keep It in the Dark
Exposure to light can seem like a low-risk issue if you’re used to storing bulk powders. In reality, this compound doesn’t like sun or bright artificial bulbs. Even a few hours sitting in direct sunlight can kick off unwanted breakdown. Store it deep in a cabinet or a shaded shelf, way out of any sunbeams. Many labs pull blackout tape over doors as an extra guardrail, especially in rooms with big windows.
Remembering Security and Segregation
Experience taught me that it’s not just about how chemicals are packed away—what else sits nearby matters just as much. Strong oxidizers, acids, or reactive metals stored near 3-Aminopyridine ramp up the danger during spills or leaks. Keeping everything separated by hazard class saves lives and headaches. Lock chemical rooms and check stock every few weeks to make sure no leaks or damage sneak by.
Developing a Culture of Diligence
People talk about policies and SOPs, but in practice, it’s the small habits—double-checking seals, writing clear labels, confirming the storage temperature—that keep everyone safe. Don’t leave it up to chance or to memory. Facts back this up: OSHA and chemical safety boards have traced almost every lab accident to some lapse in those habits. The next time somebody new joins the team, show them these routines in action. If something seems off—a funny smell, a new stain or leak—report it right away. Respecting chemicals like 3-Aminopyridine isn’t just about storage; it’s about respect for everyone sharing the space.
What are the typical applications of 3-Aminopyridine in research and industry?
Chemistry Behind the Tools
3-Aminopyridine doesn't get the flashy press coverage that high-profile biotech breakthroughs attract, but step into any well-equipped lab and you'll find this compound working behind the scenes. Chemists pick up 3-Aminopyridine because of its small, nitrogen-rich structure. It's what researchers call a building block. In the real world, this means scientists use it as a starting piece to stitch together bigger, more complex molecules, sort of like using a good set of Lego bricks to create something brand new.
Going Beyond the Reaction Flask: Pharmaceuticals
Drug development eats up large parts of research budgets, and here's where 3-Aminopyridine sees some of its most important work. Companies use it to shape medications useful for treating neurological issues. Research papers often list 3-Aminopyridine among the lead components for synthesizing molecules targeting diseases like multiple sclerosis. In fact, the compound is a parent to 4-Aminopyridine, a drug helping MS patients with their walking. That connection might sound like one step removed, but in pharmacology, small changes in molecular structure mean life-changing results for real people.
Capturing Value in Agrochemicals
Growing up, I spent time on a family farm, and stories about crop losses haunted every harvest season. Synthetic chemistry offers some protection through better herbicides and fungicides. Many of these spray bottles owe their performance to chemical families that rely on pyridine rings, like the one found in 3-Aminopyridine. Agrochemical companies take this compound as a starting point, modifying it to build more effective crop protectants. These products land on fields worldwide, keeping food affordable and more secure.
Material Science: Surfaces and Sensors
You might not think small molecules like 3-Aminopyridine play a role in high-tech materials, but they show up in the details. In labs working on advanced coatings or organic electronics, scientists tweak compounds like this one to give surfaces new abilities. For example, adding nitrogen atoms (which this molecule has) changes electrical properties or helps new materials bind to metal or glass, making them perfect for sensors or specialty polymers. My time spent in a university lab saw several students experimenting with these derivatives in solar cell research and wearable devices.
Cleaner Reactions and Greener Choices
Regulations on pollution keep tightening, and waste coming from chemical manufacturing attracts attention from both governments and communities. 3-Aminopyridine supports greener chemistry because its structure allows straightforward reactions that don’t pile up waste byproducts. Research labs keep an eye on atom economy — a fancy term for how much raw ingredient ends up in the final product rather than the trash bin. With this compound, processes often run cleaner, making it a smart pick for scaling up greener, safer chemistry.
Pushing Forward with Safer Handling
Every chemical has its risks. 3-Aminopyridine has a bitter taste and can irritate skin. Training, protective gloves, and good fume hoods help keep everyone safe on the job. Looking ahead, finding ways to harness its reactivity while managing risks stands out for both scientists and workers in the manufacturing plants.
Shaping the Future
The reach of 3-Aminopyridine stretches wider each year. The molecules built from it show up in healthcare, agriculture, and next-generation technology. Investment in better synthesis methods and safer practices promises to keep this humble structure delivering value long after the headlines move on to the next big breakthrough.
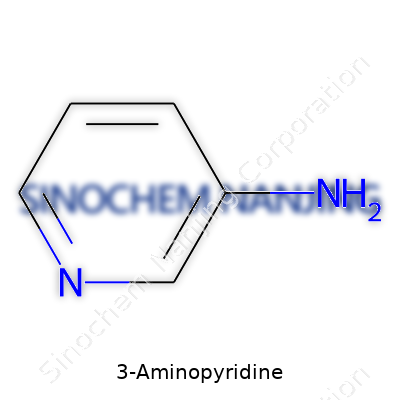
| Names | |
| Preferred IUPAC name | Pyridin-3-amine |
| Other names |
3-Pyridinamine 3-Pyridylamine Pyridine-3-amine meta-Aminopyridine |
| Pronunciation | /ˈæmɪnoʊ.pɪˈrɪdiːn/ |
| Identifiers | |
| CAS Number | 462-08-8 |
| Beilstein Reference | 527861 |
| ChEBI | CHEBI:18351 |
| ChEMBL | CHEMBL1126 |
| ChemSpider | 13980 |
| DrugBank | DB08285 |
| ECHA InfoCard | 100.011.433 |
| EC Number | 203-606-3 |
| Gmelin Reference | 80018 |
| KEGG | C02265 |
| MeSH | D016693 |
| PubChem CID | 1037 |
| RTECS number | UY8925000 |
| UNII | K6QN2EUYHV |
| UN number | UN2671 |
| Properties | |
| Chemical formula | C5H6N2 |
| Molar mass | 94.12 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | amine-like |
| Density | 1.119 g/mL at 25 °C(lit.) |
| Solubility in water | soluble |
| log P | 0.02 |
| Vapor pressure | 0.0147 mmHg (25°C) |
| Acidity (pKa) | 6.1 |
| Basicity (pKb) | 7.85 |
| Magnetic susceptibility (χ) | -43.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1. refractive index nD 1.583 |
| Viscosity | 1.014 cP (20°C) |
| Dipole moment | 2.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 165.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 96.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1383 kJ/mol |
| Pharmacology | |
| ATC code | N07XA03 |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H301: Toxic if swallowed. H311: Toxic in contact with skin. H331: Toxic if inhaled. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | Precautionary statements: P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0-Yes |
| Flash point | 86°C (186.8°F) |
| Autoignition temperature | 460°C |
| Explosive limits | Lower: 2.2%, Upper: 13.8% |
| Lethal dose or concentration | LD50 (oral, rat): 178 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 178 mg/kg |
| NIOSH | SN2988000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
2-Aminopyridine 4-Aminopyridine Pyridine Nicotinamide Isoniazid |

