3-Aminophenylarsonic Acid: Tracing the Past, Examining the Present, and Looking Ahead
Historical Threads in Arsenic Chemistry
Chemists have chased the secrets of arsenic for centuries, but the story of 3-aminophenylarsonic acid came about as people looked to pair the versatility of the aniline ring with the unique, sometimes unsettling, effects of arsenic. Early on, the agricultural industry hoped organic arsenicals could protect crops and livestock from parasites. In the 1900s, before regulatory oversight took hold, 3-aminophenylarsonic acid found its way into animal feed as a growth promoter and anti-parasitic agent, its popularity fueled by the hope for healthier flocks and herds. Over time, mounting evidence of environmental and health risks overshadowed the initial optimism. Faster-growing chickens meant more food on tables, but long-term research revealed the trade-offs: arsenic residues in soil, water, and even meat products. When regulatory agencies clamped down, whether in the EU, US, or Asia, it wasn’t out of caution alone—it was the result of painstaking toxicology and stubborn public health data.
Getting Acquainted: Chemical Identity and Features
Some chemicals demand attention by their very structure. 3-aminophenylarsonic acid isn’t flashy to the naked eye—just a pale, sometimes crystalline powder—but its chemistry packs a punch. With a benzene ring anchoring an amine on one carbon and an arsonic acid group on another, it stands out. This arrangement gives the compound both water solubility and the ability to partner with a range of other chemicals. The molecular formula, C6H8AsNO3, means it walks a tightrope between stability and reactivity. I’ve handled compounds like this in the lab; they seem benign at a glance, but a proper respect for their toxicity matters from the first scoop.
Packing Details: Technical Specifications and Proper Labeling
Strict standards shape the packaging and labeling of substances like 3-aminophenylarsonic acid. Chemists and plant workers look for clear labeling with standardized hazard pictograms, batch numbers, and purity metrics. From my own years working with specialty chemicals, clear labels and tamper-evident containers cut down on mistakes that can put entire projects, or even lives, at risk. Industry standards for storage—cool, dry spaces, away from acids and oxidizers—aren’t just box-ticking exercises. They guard against degradation and dangerous reactions. Mishandling even traces can cause regulatory headaches and unnecessary injuries.
Where Science Meets Synthesis
Most often, people synthesize 3-aminophenylarsonic acid by treating aniline derivatives with arsenic acid or sodium arsenite under controlled conditions. The reactions require careful temperature regulation and precise measurement of reactants, skills drilled into chemists from day one. Yields, purity, and byproduct profiles depend on tiny tweaks—the pH, the cooling times, the stoichiometry. Even minor mistakes affect not just the bottom line, but also the safety profile of the resulting batch. Scaling from bench-top glassware to industrial vats compounds these risks. Best practices call for fume hoods, gloves, goggles, and airtight planning.
Reactivity and Ways to Push the Molecule Further
3-Aminophenylarsonic acid doesn’t just sit idly in storage. Chemists push it into condensation reactions, link it to new aromatic partners, or use its amine group for diazotization and coupling. In a world always seeking novel drugs or new agrochemicals, tweaks to this core structure beckon as a route to fresh bioactivity. With arsenic's reputation, most chemists steer clear of frivolous tinkerings. In my own projects, every new derivative required deep research, structural confirmation, and extensive review before earning a trial run outside the glovebox. As safety rules tighten, fewer labs—and fewer industrial sites—take on such chemistry. Those that do must stay sharp, respecting the delicate balance between creativity and risk.
What’s in a Name?
Like many chemicals, 3-aminophenylarsonic acid wears several hats. Sometimes it appears as “meta-aminophenylarsonic acid,” or under various registry numbers. Historical literature and trade reports sometimes jumble the synonyms, stirring confusion for today’s scientists trying to trace a sample's life story. My colleagues and I often needed to double- or triple-check inventory records just to make sure what we ordered matched what arrived. The best defense: maintaining tidy records and cross-referencing every synonym and identifier.
Staying Safe: Procedures and Standards
Working with arsenic compounds means memorizing safety protocols. The glovebox becomes a familiar friend. Fume hoods hum daily. Double-chambered waste containers line benches, ready for every milligram of spent reagent or contaminated glove. Even routine clean-up takes on extra gravity. Industry standards, set by organizations like OSHA or REACH, exist to keep exposure low and discourage unsafe shortcuts. Accidents with arsenic are often irreversible; symptoms creep up slowly. A mentor once told me you know a good chemist by how seriously they take their PPE—dry facts, but true enough to save lives. Rigorous air monitoring, medical surveillance, and thorough training set apart responsible labs from those courting disaster.
Putting 3-Aminophenylarsonic Acid to Work
Initially, people turned to this compound as a way to treat poultry and swine, convinced it would stymie parasites and spur growth. For some decades, these applications dominated industry and underpinned a sizeable chunk of the animal ag sector’s profits. Over time, attention turned toward environmental and human health fallout. Arsenic residues began piling up in the soils around intensive farming operations, drifting into water tables and making their way into nearby ecosystems. Researchers found that the original promise clashed with social responsibilities; this led farmers and governments to roll back or ban its use. These days, any legal application involves close oversight and a constant search for alternatives.
Chasing Answers: Toxicity Research and Its Ramifications
Scientists have tested arsenicals for decades, searching for toxic thresholds, accumulation patterns, and metabolic breakdown products. 3-Aminophenylarsonic acid, like other organic arsenicals, doesn’t break down easily in soils. Long-term studies, including some that made headlines during my early career, showed that the compound—both directly and through its metabolites—builds up in food chains. Regulators stepped in after repeated studies connected residues in edible meat with chronic health effects. Arsenic compounds can stress kidneys and liver, shift gene expression, and cause neurological effects with sustained exposure. Today, more labs focus on detection and remediation—heavy investment in sensitive analytical chemistry and bioremediation strategies—to counteract a chemical legacy stretching back generations.
Pushing the Boundaries: Where Research and Regulation Intersect
Even as traditional uses fell out of favor, research kept probing the molecule’s possibilities. Analytical chemists develop fresh detection methods—trying to pull out arsenic traces from complicated environmental samples. Environmental engineers chase cost-effective cleanup, studying everything from microbial arsenic reducers to new filter materials. Pharma researchers scan arsenicals for anti-cancer properties; after all, one of the oldest cancer treatments, arsenic trioxide, came from the same basic element. Each research proposal weighs two sides: potential breakthroughs and the burden of responsibility. These trade-offs fuel spirited debates in research ethics panels and journals. Funding only follows those who can prove both innovation and care.
The Road Ahead: Future Prospects and Closing Thoughts
Looking forward, fewer chemists and companies try to push 3-aminophenylarsonic acid into new products, but its influence persists. The environmental footprint can’t be erased quickly, and cleaning up legacy sites—with contaminated soil and groundwater—will anchor research budgets for years. Analytical testing capacity grows, promising detection of ever-smaller residues and enforcing stricter compliance. On the R&D front, some researchers chase the holy grail of safe, effective, non-toxic organoarsenicals, though with every passing regulation, this portfolio shrinks. The real hope lies in safe alternatives that farmers, doctors, and the environment can live with. A generation after its heyday, 3-aminophenylarsonic acid stands less as a chemical for the future than a reminder: scientific progress must balance ambition with awareness of consequences.
What is 3-Aminophenylarsonic Acid used for?
A Look at Where 3-Aminophenylarsonic Acid Fits In
3-Aminophenylarsonic acid, known to some as an organoarsenic compound, found its spot in the agricultural world decades ago. Back in the days of my family’s run-down farm in the Midwest, folks leaned on any tool that promised strong crops and healthy livestock. This item once showed up in livestock feed, especially for poultry and swine, aiming to keep disease at bay and push animals to gain weight faster. Corporate and small farm operations often favored it for efficiency. They wanted robust animals and a stable bottom line.
This compound’s reputation grew because it could knock down certain bacterial infections and help animals use nutrients better. That mattered for a few reasons. Producers chasing bigger yields didn’t like sickness eating their profits, and safe, consistent feed fell into their routine. By the ‘70s and ‘80s, a whole class of arsenical feed additives, this compound included, turned into household names for many farmers hoping to keep up with market changes and growing demand for animal protein.
Risks No One Could Ignore
With all this use, someone had to ask whether it made the food supply and the environment riskier. Research turned up evidence that arsenic from feed made its way into meat, manure, and water. Studies at universities such as Johns Hopkins and UC Berkeley highlighted how small amounts in soil and food could build up in the human body over time, leading to serious health concerns. Farmworkers and scientists both caught on to the dangers, and families started to push for changes. It stood out to me that things in animal feed could end up in our water or vegetables downstream.
Regulators in countries like the United States took note. Agencies such as the FDA examined the risks and eventually pulled approval for most uses of this compound around 2013. By then, health experts argued that safer alternatives existed, and feeding this chemical to animals just didn’t make sense anymore. Many countries followed, with Europe already having a stricter stance years earlier. I remember seeing this change on local farms as labels started to change and more natural feeding policies popped up in co-op meetings.
Where Do We Go Next?
The story of this chemical points to bigger issues in food safety, environmental health, and animal welfare. Remaining true to the facts, 3-aminophenylarsonic acid doesn’t see much use today, but historical contamination stuck around in certain soils and water sources. Cleaning up old waste sites and monitoring food supplies takes continued effort. Community awareness helped spark these changes, showing how families and farmers together can nudge companies and officials to clean up their act.
As new feed additives come along, lessons from the past should shape policy and practice. Everyone in the chain—growers, scientists, consumers—shares a responsibility to question the safety of what goes onto farms and what ends up on our tables. Support for transparency and science-based testing helps prevent another risky chemical from slipping through the cracks. Open access to research and plain talk between experts and neighbors can spark more ethical, healthier decisions for animals, people, and the land around us.
What are the safety precautions when handling 3-Aminophenylarsonic Acid?
Understanding What You're Handling
3-Aminophenylarsonic acid carries risks that can’t be shrugged off. It's an organoarsenic compound, and folks who've worked in research or on the industrial side know arsenic grabs headlines for a reason. The dangers go beyond stories from textbooks. This compound can cause health problems if it gets on your skin, in your eyes, or if you breathe it in. My own time working in a university lab made me realize real danger sits in everyday tasks—reaching for a bottle or weighing out a powder without thinking twice.
Sticking to the Basics of Protection
Direct contact is trouble. Think about everything your hand touches: door handles, pens, smartphones. Before handling, grab gloves—nitrile or latex, they do the trick against chemical splashes. Eyes are often ignored until the sting sets in. Always slip on goggles, even for what looks like a quick task. One peer at my old job got some dust near her eye just once; she won’t forget how quickly pain taught respect. Simple choices like a cotton lab coat or a disposable apron stop the compound from clinging to your clothes and making an unwanted trip home.
Air Quality and Ventilation Matter
Many folks underestimate powders and dust. Breathing them in feels like nothing until coughing or worse shows up well after exposure. Working inside a certified fume hood stops inhalation at the source. Never skip the fume hood and never trust a DIY shortcut. Proper exhaust fans do their part, but fume hoods are king for personal safety. If your workspace lacks these basics, ask your workplace to fix it. Your lungs aren’t worth the risk.
Storage: Out of Sight, Out of Harm’s Way
Don’t leave containers on benches or stashed in open drawers. Seal them tightly after every use. Label everything clearly, so nobody’s left guessing. I’ve seen accidents start because someone thought they’d grabbed ordinary reagent instead of something much worse. Keep 3-aminophenylarsonic acid in a cool, dry, locked chemical cabinet, far from anything you’d want near food or drink.
Spill Clean-Up and Waste Disposal
If a spill happens, don’t use bare hands or ordinary paper towels. Sweep up solid spill residue with a plastic scoop or a small broom and pan, then transfer it all to a designated hazardous waste container. Every piece of trash—used gloves, wipes, containers—should go straight into labeled, sealed hazardous waste bags. Liquid spills need absorbent pads and extra patience. Always let your supervisor know immediately, even for small messes.
Knowing What to Do If You’re Exposed
Quick thinking saves more than embarrassment. If the compound touches your skin, rinse under running water for at least 15 minutes. Splashing in your eyes? Hold eyelids open in a gentle stream of water, no rush, just steady washing. If dust or fumes were inhaled, move to fresh air and alert your health officer right away. Never hesitate to seek medical help. Early attention makes recovery easier and way less stressful.
Continuous Training and Respect for Chemicals
Getting comfortable or cutting corners turns small jobs dangerous. Training means more than just signing forms. Ask questions. Watch out for your coworkers. In my experience, the buddy system and good habits—like always running checklists before you start—help everyone leave the lab with no stories to tell except good ones. Good safety makes work smoother, and nobody regrets staying on the safe side. If you see a better way to keep your team safe, speak up. Feedback matters more than bravado when dealing with chemicals like this.
Handle 3-aminophenylarsonic acid with care and never underestimate its risks.What is the chemical structure of 3-Aminophenylarsonic Acid?
A Closer Look at the Molecule
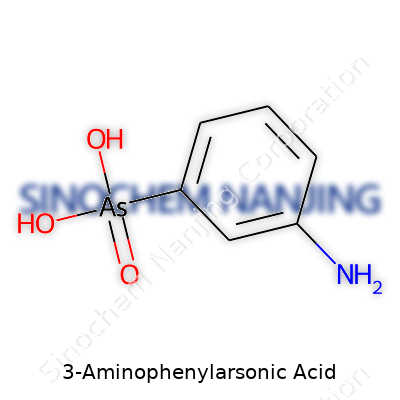
3-Aminophenylarsonic acid stands out in the world of organic chemistry due to its unique combination of an aromatic ring, an amino group, and an arsonic acid function. Its structure relies on a benzene ring that anchors two groups: a –NH2 (amino) at the meta position and an –AsO(OH)2 (arsonic acid) at the para position relative to each other. Here, the word "meta" marks the third carbon away from the arsonic acid group on the ring.
Looking closer, the molecule carries the formula C6H8AsNO3. The backbone—six carbon atoms arranged in a hexagonal pattern—serves as a scaffold. One corner of this ring sports the -NH2 group, bringing basicity and the ability to form hydrogen bonds. The arsonic acid group attaches through an arsenic atom double-bonded to one oxygen and singly bonded to two hydroxyl (-OH) groups. This specific arrangement links organic chemistry with elements of inorganic chemistry, thanks to arsenic’s central role.
The Relationship between Structure and Use
Folk working in veterinary science or agriculture might remember 3-aminophenylarsonic acid from its legacy as a feed additive, used in the past to boost growth and prevent disease in poultry and swine. Structure influences everything: the placement of the amino group makes the compound more water-soluble, which means it spreads through body tissues more efficiently compared to similar compounds. The arsonic acid group, with its affinity for both oxygen and hydrogen, binds tightly in biological environments.
As an undergraduate, I first saw the skeletal structure on an exam sheet. There’s something almost artistic about the way biology and chemistry meet on that ring. Its use as a feed additive drew fire after people learned more about arsenic accumulation in meat and soil. The molecule’s stability allows it to persist in animal systems and slip into manure, which then runs off into water or fields. This feature, rooted in its chemistry, explains continued scrutiny from regulators and researchers.
The Conversation Around Safety and Alternatives
Scientists tie the effect of 3-aminophenylarsonic acid to both its structure and how organisms process it. The arsenic part doesn’t just disappear after ingestion. Over time, transformation into inorganic forms increases risks for environmental and public health. Studies point out that traces still linger around farming sites, long after governments block its use.
Addressing the fallout, some researchers have worked on breaking down residues in the environment, using bacteria that grab arsenic and turn it into less harmful forms. Others push for complete alternatives, aiming for additives that skip the challenges created by the arsenic atom altogether. One promising avenue includes plant-based growth enhancers or probiotics, shaped to mirror the performance seen from arsonic acids but with far less baggage.
The structure of 3-aminophenylarsonic acid calls for responsibility in its application. A deeper look at its chemistry explains not just its utility but also its lasting impact. Openness about its makeup supports better regulation and safer innovation, offering a lesson for other molecules still on the market.
How should 3-Aminophenylarsonic Acid be stored?
Why Storage Even Matters
Every compound brings its own headaches. Take 3-Aminophenylarsonic acid, for example. This stuff isn’t baking soda. It lands on the shelf with a whole set of risks, including both toxicity and reactivity. I remember my first week in a chemistry lab: the supervisor told me, “This isn’t just about following rules. It’s about protecting your lungs, your neighbors, and the place you work.” Turns out, he was right. Keeping chemicals like this away from moisture, from incorrect temperatures, and from curious hands matters way more than any rulebook says.
Not All Shelves Are Equal
It’s too easy to think any cool, dark spot is enough. For 3-Aminophenylarsonic acid, that’s wishful thinking. Slight heat or excess light can play havoc, sometimes setting off decomposition. A dry environment keeps this powder stable, so desiccators and tight-sealing containers come into play fast. Glass works better than plastic; the latter sometimes reacts or leaches over time. The compound’s toxicity also rules out open access. It really belongs in a locked cabinet marked with hazard labels—no bland warnings, but proper GHS labels showing the risks.
In places I’ve worked, the best setups use silica packs to fight moisture and bypass those temptation-prone bottom drawers. The fewer hands that handle toxic organoarsenic compounds, the safer everyone feels, and there’s less chance of environmental spills.
Temperature: Not Just a Number
Room temperature usually lands around 20-25°C, but “room” can mean a lot, especially during a heatwave or power outage. This acid keeps best below a firm 25°C. Warm rooms cause volatile stuff at the cap to vaporize a little faster, and those vapors usually mean extra risk. Refrigerators sometimes help, though only if they’re cleared for storing hazardous materials. Never put food nearby. At my old lab job, one slip during a blackout meant toxic chemicals met the ice cream sandwiches in the break room fridge. That turned into an expensive clean-up and a few strained apologies to the team.
Getting Real About Safety
Some people trust their memory for safety steps, but signs and checklists win every time. An up-to-date log—one that actually gets updated—means nothing sneaks up. I’ve seen bright red tapes used to close off cabinets, and written instructions posted above the shelf to keep new interns from guessing at what’s inside. Chemical companies know regulators like OSHA and the EPA expect storage protocols and reporting: a misplaced jar could become a regulatory and legal problem overnight.
Better Practices, Fewer Problems
Mistakes fall harder in small spaces. Double containment—putting the primary container into a second, resistant box—often keeps spills contained. Disposal plans should sit right next to storage policies: if the stuff expires, the path from shelf to sink doesn’t exist. Special vendors help with pick-up, and safety data sheets are more than paperwork—they act as roadmaps for safe storage, handling, and accident response.
3-Aminophenylarsonic acid reminds us that handling real chemicals needs real attention. With the right habits and clear policies, these risks shrink a whole lot. The experts who’ve seen accidents know that smart storage never happens by chance.
What are the potential health hazards of 3-Aminophenylarsonic Acid?
Understanding the Chemical
3-Aminophenylarsonic Acid, often used in animal feed and as a research chemical, draws attention for more than its scientific curiosity. This substance, linked to the production of veterinary drugs in the past, brings up real health concerns for both workers and everyday folks.
What’s in the Air We Breathe?
People working near this chemical run the risk of inhaling dust or particles. Breathing it can irritate the nose, throat, and lungs. Simple symptoms like coughing or sneezing might not seem serious, but they point to a deeper problem. Over time, breathing in arsonic acids like this can damage lung tissue. A friend who worked in a feed processing plant spoke about the persistent nosebleeds and dry coughs he and some colleagues developed after handling animal supplements containing these compounds. These symptoms don’t always clear up just by stepping outside for fresh air—repeated exposure raises the risk of more lasting lung issues.
Direct Contact Risks
Spilled powder doesn’t just vanish. Skin contact can spark rashes, redness, or lingering irritation. Eyes are especially sensitive; dust or splashes sting and lead to redness and watering. I remember a college lab partner who brushed his face after a spill. It took a trip to the campus clinic and a full day before his eyes felt normal again. Lab safety rules might seem overcautious, but experiences like that make a lasting impression.
What Goes Into the Body Stays
Eating or swallowing any compound with arsenic opens the door to chronic health problems. Studies show arsenic-linked chemicals, once inside, accumulate in organs. Over months or years, low-level intake ties to nerve disorders, hand and foot tingling, skin splotching, and even a greater risk for certain cancers. The World Health Organization singles out inorganic arsenic as a known carcinogen, and organic forms like 3-Aminophenylarsonic Acid don't always stay “harmless.” Bacteria in the digestive tract can convert so-called safer compounds into more toxic forms.
Wider Environmental and Food Risks
Animal feed used to contain this chemical more often, especially in poultry production. That decision doesn’t just affect animals. Traces of arsenic from manure can run off fields and enter local water supplies. Years ago, some small-town water tested far above normal arsenic levels thanks to runoff from nearby farms. Long-term exposure through drinking water links directly to cancer, heart problems, and delayed childhood development. The U.S. Food and Drug Administration stepped in and banned most of these uses, but leftover soil and water contamination still worries parents and health workers in farming towns.
Looking for Solutions and Protections
Regulation cut back the biggest risks, but workers and researchers still handle these chemicals. Proper training, masks, gloves, and sealed containers really make a difference. Routine air quality checks inside feed mills and labs can catch problems before they snowball. For communities, soil and water testing offer the best clues about lingering pollution. Switching to genuinely safer animal supplements and encouraging transparency about chemical use in agriculture also matter. Speaking up, sharing stories, and holding companies accountable carry just as much weight as legislation.
| Names | |
| Preferred IUPAC name | (3-aminophenyl)arsonic acid |
| Other names |
3-Aminophenylarsonic acid meta-Aminophenylarsonic acid 3-APAA 4-Arsanilic acid meta-Arsanilic acid m-Aminophenylarsonic acid |
| Pronunciation | /ˈθriː əˌmiːnoʊ ˌfɛnɪl ɑːrˈsɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 98-50-0 |
| 3D model (JSmol) | `3D model (JSmol)` string for **3-Aminophenylarsonic Acid**: ``` NC1=CC=CC(=C1)As(=O)(O)O ``` |
| Beilstein Reference | 1573322 |
| ChEBI | CHEBI:28660 |
| ChEMBL | CHEMBL1238 |
| ChemSpider | 14217 |
| DrugBank | DB14096 |
| ECHA InfoCard | ECHA InfoCard: 100.003.498 |
| EC Number | 213-669-5 |
| Gmelin Reference | 53937 |
| KEGG | C14354 |
| MeSH | D000653 |
| PubChem CID | 6923 |
| RTECS number | CQ9625000 |
| UNII | V6Q4E3H29Z |
| UN number | UN1547 |
| Properties | |
| Chemical formula | C6H8AsNO3 |
| Molar mass | 233.07 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.73 g/cm3 |
| Solubility in water | Soluble |
| log P | -2.0 |
| Acidity (pKa) | 4.20 |
| Basicity (pKb) | pKb = 7.86 |
| Magnetic susceptibility (χ) | -62.0×10⁻⁶ cm³/mol |
| Dipole moment | 2.20 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -607.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1188 kJ/mol |
| Pharmacology | |
| ATC code | QH460 |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes skin irritation. Causes serious eye irritation. Suspected of causing cancer. |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | InChI=1S/C6H8AsNO3/c7-8(10,11)6-3-1-2-5(4-6)9/h1-4H,9H2,(H2,7,10,11) |
| Signal word | Danger |
| Hazard statements | Hazard statements: "H302, H319, H332 |
| Precautionary statements | Precautionary statements: P261, P264, P270, P271, P272, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P330, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 3-1-0 Health:3 Flammability:1 Instability:0 |
| Lethal dose or concentration | LD50 (oral, rat): 2800 mg/kg |
| LD50 (median dose) | LD50 (median dose): 800 mg/kg (rat, oral) |
| NIOSH | NA |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-Aminophenylarsonic Acid: 0.01 mg/m³ as As |
| REL (Recommended) | 0.02 mg/m3 |
| Related compounds | |
| Related compounds |
Arsanilic acid Nitarsone Roxarsone 4-Aminophenylarsonic acid Phenylarsine oxide |

