3,6-Dihydroxyphthalonitrile: Chemistry’s Curious Building Block
Tracing the Roots of 3,6-Dihydroxyphthalonitrile
Chemists have pushed boundaries for more than a century, and the history behind 3,6-dihydroxyphthalonitrile feels like a reflection of their relentless tinkering. Early organonitrile research in the mid-20th century brought a new world of aromatic compounds into sharp focus, and phthalonitrile derivatives quickly became hot property for researchers in dyes and advanced materials. 3,6-dihydroxyphthalonitrile comes from that legacy—a compound built for versatility, often landing in chemical schemes where both reactivity and precise modification matter. Lab notebooks from the 1950s scattered across Europe and the US show early mentions, as research teams shaped phthalonitrile structures for more complex applications. These small steps led to the robust toolbox we see today, with 3,6-dihydroxyphthalonitrile acting like a springboard for wider chemical innovation.
Getting to Know the Compound
3,6-Dihydroxyphthalonitrile shows up as an off-white to pale-yellow crystalline solid. It carries two reactive hydroxy groups at the 3 and 6 positions of a benzene ring, bracketed by a pair of cyano groups. This pattern puts it in a sweet spot for organic synthesis: The hydroxy groups welcome further reactions, while the nitrile groups tie the compound to broader phthalocyanine chemistry. Those nitrile handles remain crucial, since they can be transformed into amides, carboxylic acids, or even more exotic fragments. What really makes this molecule stand out is its dual functionalization, setting it apart from plainer phthalonitrile relatives.
Properties That Matter in the Lab
From the scientist’s perspective, the performance of 3,6-dihydroxyphthalonitrile is a blend of stability and usability. Its melting point lands well above room temperature, so it won’t melt away under standard conditions. In the fume hood, it dissolves best in polar solvents including dimethyl sulfoxide or DMF, but shows little love for water. The molecule’s aromatic ring keeps it relatively stable, resisting quick oxidation or hydrolysis—an advantage for long-term storage. Under strong bases or acids, those hydroxy groups can step into reactions, often firing off esters or ethers, while the nitriles only react with determined, targeted approaches. Its robustness means shipping, storage, and the odd bottle forgotten on a shelf don’t cause rapid decay or unpredictability.
The Science of Making 3,6-Dihydroxyphthalonitrile
For anyone actually working at the bench, producing 3,6-dihydroxyphthalonitrile can feel like threading a needle. A favorite approach starts with 3,6-dibromophthalonitrile; a nucleophilic substitution with potassium or sodium hydroxide swaps out bromines for those all-important hydroxy groups. Handling requires skill—the mixture heats up, the air fills with potent scents, and careful cleanup follows. Post-reaction steps call for acidification, precipitation, then multiple rounds of filtration and crystallization, since impure batches can stubbornly persist. Yields hinge on clean glassware and unwavering patience. For decades, chemists have nudged every parameter—from base strength to temperature and reaction time—to shave off side products and boost returns, leading to decent, reliable syntheses.
How the Molecule Changes and Grows
In the lab, few compounds offer as clean a starting point for modifications as this one. Chemists can protect the hydroxy groups with silyl or acetyl functions to channel the nitriles into reactions without interference. Conversely, those cyano arms morph smoothly into amides or carboxylates using hydrolysis, and advanced work may turn them into complex rings by cyclotetramerization—a key step for building phthalocyanines. The phenolic groups let the molecule form esters, ethers, or even serve as anchor points for polymer backbones. Instrumental methods, especially NMR and IR, allow anyone to watch every transformation step by step. Supported by strong chromatography work, this rich chemistry underpins both academic curiosity and real-world synthesis efforts.
Keeping Work Safe and Reliable
Working with nitriles always nudges chemists to respect safe practice. Gloves, goggles, lab coats, and discouraging open flames are daily routine. Lab ventilation does more than protect against fumes—it prevents buildup of cyanide and phenolic residues. Waste solutions head straight to dedicated disposal tanks, keeping bench tops free of both hazardous and persistent byproducts. Lessons from past accidents highlight the consequences of relaxed attitudes: Even one batch escaping or one broken bottle can contaminate a space for weeks. Training sessions stress proper labeling and storage, emphasizing that even routine tasks demand focus. Researchers share anecdotes about exposure-induced headaches or mild skin irritation as reminders not to take shortcuts.
Why 3,6-Dihydroxyphthalonitrile Finds Its Place in Industry and Research
Alone, the molecule rarely wins headlines, but as a chemical building block its value skyrockets. Dyes, pigments, and optical materials all benefit from the structures that arise from this compound. Research teams use it to forge phthalocyanines, molecules that color everything from blue-green ink to advanced solar cells. These precursors also spur development of organic semiconductors, fostering innovation in flexible electronics. In medicine, researchers anchor diagnostic agents to the aromatic core, pushing the boundaries of imaging and therapeutics. Patent literature tells the real story, ranking 3,6-dihydroxyphthalonitrile among foundational bricks in hundreds of chemical strategies meant to make colorants, sensors, and advanced coatings more effective.
Digging Into the Science: Research, Toxicity, and the Path Forward
Scientists do not take unknowns lightly, and safety studies for aromatic nitriles often precede large-scale work. 3,6-Dihydroxyphthalonitrile, like its chemical siblings, can irritate skin or sensitive tissues after direct contact. Long-term toxicity data for the compound is thinner compared to more common chemicals, but studies flag possible hazards from both inhalation and absorption. Laboratories tend to err on the side of caution, limiting exposure and documenting any odd reactions. Recent environmental work examines how such molecules break down in wastewater and soil, with hopes that proper containment and disposal limits risk outside the lab. As more data comes in, chemists will need to adapt their procedures for both human safety and environmental protection.
Looking to the Future
It’s easy to gloss over a compound tucked away on a shelf, but 3,6-dihydroxyphthalonitrile shows up in the footnotes of many breakthroughs. New demand for advanced pigments, responsive materials, and medical diagnostics keeps the compound in the sights of chemical manufacturers and R&D outfits alike. Industry trends push for greener techniques, so teams now look for ways to scale up production with less waste and safer solvents. Advances in catalysis, flow chemistry, and purification give real hope for cheaper, cleaner routes to the molecule. In emerging fields—think flexible electronics and bio-inspired imaging—researchers circle back to compounds like this, eager to build from its core structure. Investing in deeper safety assessments, faster synthesis, and broader application surveys will let manufacturers and scientists tap its full promise—without losing sight of the lessons that history and hands-on work have taught the field.
What is 3,6-Dihydroxyphthalonitrile used for?
Looking Deeper at a Common Lab Ingredient
Ask anyone familiar with organic chemistry and synthesis about 3,6-Dihydroxyphthalonitrile and you’ll get a nod. This compound—built around a phthalonitrile core with two hydroxyl groups—shows up in labs with serious potential. But what pushes researchers to keep it around? Why do chemists chase it, test it, and build new things on its backbone?
Push to Create High-Performance Materials
Phthalocyanines don't make headlines like AI or quantum computing, but plenty of modern devices rely on materials that owe their existence to intermediates like 3,6-Dihydroxyphthalonitrile. Chemists reach for this compound as a stepping stone. The two nitrile groups and the twin hydroxy functions make it easier to build large, complex molecules that hold up under heat, resist corrosion, and show surprising color properties. Synthetic dyes, OLED displays, and organic solar cells often have a bit of this molecule’s influence in their chemical history.
Color, Light, and Electronics
Phthalocyanines—crafted using 3,6-Dihydroxyphthalonitrile—land in printing inks, coatings, and sensors. These pigments don't just give a bold blue or green; they show stability against sunlight, moisture, and heat. That matters for art conservation, industrial packaging, and medical imaging equipment. The backbone built with this intermediate has allowed companies to deliver displays with sharper colors, longer lives, and friendlier manufacturing processes.
What’s more, research teams every year push for better solar panels and batteries. Phthalocyanine derivatives show up because their structure works well in organic semiconductors. In a world that hungers for affordable and sustainable energy, every little efficiency gain counts. That push for efficiency comes back to reliable starting materials—it’s not glamorous, but it’s real progress you can see on a power bill.
Medicine and Imaging
My experience working with diagnostic imaging researchers taught me that the right pigment can change more than just a photograph; it can open new pathways for seeing inside the body. Compounds related to 3,6-Dihydroxyphthalonitrile lead to agents used in MRI scans and photodynamic therapies for tumors. The purity and yield of the early-stage chemical shape the safety and accuracy of the final medical product. If anything goes wrong at the start, it costs both time and patient trust. This is not hypothetical—bad batches have caused major recalls in the past, which nobody in healthcare wants to revisit.
The Issue: Safety and Sourcing
Every upside comes with challenges. Manufacturing practices for compounds like 3,6-Dihydroxyphthalonitrile still generate hazardous waste. Disposal and ventilation take serious planning; mistakes harm both local workers and surrounding communities. Companies must also validate every shipment for purity. Impurities in the base chemical can cause entire product lines to fail, wasting resources and cash. I've seen projects go sideways over a contaminated drum sourced from a new supplier trying to cut costs.
Responsible Paths Forward
Better safety protocols, closed-loop solvent systems, and third-party ingredient verification offer practical steps. Some companies invest in green chemistry alternatives or cleaner synthesis routes. Others require strict supplier audits and full traceability documents on every link in the supply chain. Research into biocatalysts or less toxic starting materials continues as a long-term hope. Transparency improves customer trust, and safer processes protect everyone from workers in the lab to families who rely on medical tech.
Innovation may grab the headlines, but small choices in chemistry—like using 3,6-Dihydroxyphthalonitrile responsibly—help shape results that last.
What is the chemical formula of 3,6-Dihydroxyphthalonitrile?
Examining the Formula: C8H4N2O2
3,6-Dihydroxyphthalonitrile, a chemical with the formula C8H4N2O2, deserves attention beyond textbook definitions. This compound features a benzene ring as its backbone, adorned at the third and sixth positions with hydroxyl groups, along with nitrile groups at the first and second positions. For those who have handled precursors for specialty dyes or advanced organic semiconductors, the structure rings a bell. It's specific, with each atom in the formula speaking to its place in synthesis and application.
Why Structure Matters: Chemistry in Practice
Two hydroxyl groups transform what would be a simple phthalonitrile. Those -OH attachments set up key hydrogen bonding opportunities. In my time experimenting with similar compounds, this means more solubility in water, but also more reactivity—both valuable and risky, depending on the context. Chemists taking on organic synthesis routines use the precise layout of functional groups to guide what reactions might work. For anyone building phthalocyanine dyes or high-performance pigments, these tweaks determine final color and durability.
Application and Real-World Importance
Laboratories use 3,6-dihydroxyphthalonitrile as a stepping stone. The two hydroxyl groups make it more reactive than the base phthalonitrile. This pair of functional groups lets researchers aim for specific substitutions and open avenues for more complex ring systems—critical in pigment design, especially for the electronics industry. My own experience prepping advanced pigment precursors taught me that small modifications to these base units impact purity and yield. Even a minor contaminant can clobber the electrical properties of a final organic semiconductor.
Health, Handling, and Environmental Considerations
The presence of nitrile groups often raises questions of safety. Inhalation or skin exposure to nitriles requires solid lab protocols. I’ve always relied on personal protective equipment and rigorous ventilation standards, especially for chemicals destined for electronics or medical diagnostics. Accidents in the lab—thankfully rare—highlight the importance of understanding chemical behavior and proper storage. Environmental impacts extend past the bench. Nitrile-containing intermediates demand controlled disposal or careful recycling routines, so local regulations matter.
Challenges and Opportunities in Synthesis
Synthetic routes to 3,6-dihydroxyphthalonitrile tend to involve multi-step reactions. Efficient production can mean fewer side products, less solvent waste, and smaller footprints. The challenge lands squarely on the ability to control reaction temperatures, timing, and purification steps. In my own lab work, even the grade of starting material plays into the overall success. Tools like high-performance liquid chromatography and spectroscopy become non-negotiable.
Looking Ahead: Sustainable Chemistry
Attention now turns to sustainability in chemicals production. Reducing waste and choosing greener solvents isn’t just a trend—it shapes the future of how we make and use building blocks like 3,6-dihydroxyphthalonitrile. Industry shifts towards renewable feedstocks and closed-loop syntheses signal a direction that's long overdue. As someone who has watched the field evolve, I see potential for updated methodologies that cut environmental cost without sacrificing the precision that molecules like C8H4N2O2 demand.
What are the safety precautions when handling 3,6-Dihydroxyphthalonitrile?
Getting Real About Chemical Risk
A lot of people spend their days dealing with chemicals, whether at a lab bench or on a factory floor. If you’ve ever handled 3,6-Dihydroxyphthalonitrile, you know the kind of respect it demands. Safety isn’t a checklist you gloss over—it’s the biggest difference between finishing your shift safely and ending up in the ER. Even handling something that sounds tame on paper brings risk. If you ignore those risks, your health might not bounce back.
Know the Material
3,6-Dihydroxyphthalonitrile looks plain but can irritate the skin and eyes. Breathing in its dust doesn’t do your lungs any favors. Years in research labs taught me that the worst accidents start small: absentminded rubbing of your eye, tossing dirty gloves in the wrong place, or letting dust build up in a spot someone doesn’t expect.
Protection Shouldn’t Wait
Anybody handling this chemical should wear solid chemical-resistant gloves—not the thin, cheap ones from the grocery store. Splash-proof goggles save your eyes from a painful mistake. A proper lab coat or apron keeps your clothes and skin safe. The fact is, even a tiny skin contact can cause redness or rashes. Clean shoes matter too. I’ve watched colleagues track chemical dust out of the work zone and into shared spaces. That’s how a solo risk becomes a team problem.
Work Spaces Need Respect
Working in a well-ventilated area does more than tick a box on a form. I used to stick my nose too close to open reactions—bad habit. You want an exhaust hood or active ventilation. Never eat or drink while working here. Open food in a lab picks up dust and becomes a carrier. A messy work bench multiplies risk. I learned to clean as I go: spilled powder, open containers, loose gloves—get rid of them.
Clean-Up Counts
Washing hands isn’t just a ritual. After glove removal, run your hands under water and use soap. If the powder gets on clothes, change as soon as possible, and wash those garments separately. It’s embarrassing to bring chemical dust home, but worse if kids or pets come into contact.
If Something Goes Wrong
Eye splash? Rinse for a good fifteen minutes and head to a medical professional. Skin contact? Wash right away, use soap, and don’t let the exposure linger. Accidents grow when people freeze or panic. Know where to find eyewash stations and showers before things get hectic.
Training Matters
No shortcut matches good training. Watching someone with years of chemical work behind them, you see careful, steady routines—glasses checked, gloves inspected, labels double-read. The best workplaces run drills, make time for refreshers, and encourage a culture where nobody rolls their eyes at another worker’s concern.
Storage Solutions
Keep 3,6-Dihydroxyphthalonitrile sealed in clearly marked containers, away from food, acids, and other incompatible substances. Store it in a place with solid ventilation, out of reach from water sources and sunlight. I once saw a badly labeled jar cause a scare—labels matter, every single time.
The Bottom Line
Safe handling of chemicals isn’t about luck. My own mistakes taught me it’s steady habits, solid equipment, and an honest attitude about risk that protect people. Respect the material, look out for each other, and act fast when something slips. That’s how you cut down incidents and get everyone home safe.
How should 3,6-Dihydroxyphthalonitrile be stored?
Understanding the Real Risks
Storing chemicals like 3,6-Dihydroxyphthalonitrile should never turn into a guessing game. Years of handling similar aromatic nitriles taught me that the fine print matters just as much as the broad strokes. Lab accidents often happen not because of what people know, but because of what gets ignored. This compound is no exception. It comes as a pale or off-white powder, sensitive to warmth, light, moisture, and rough handling.
What Happens Without Proper Storage
One faulty jar seal or a little extra heat can turn a stable solid into a significant hazard. Most people underestimate how quickly air or moisture seeps in. Once, I watched a small batch clump and brown overnight in a sweaty storeroom. Chemical degradation doesn’t always announce itself loudly—but even minor breakdown products can introduce fresh risks, like toxic fumes or unexpected reactivity.
Practical Storage Methods
Nobody wants to handle a compromised reagent. Clean, dry containers make all the difference. Glass jars with airtight PTFE-lined lids block moisture and oxygen better than old screw caps ever could. Metal will sometimes react, so plastic works as long as it doesn’t leach or deform. My own preference comes down to high-density polyethylene jars. If the lab budget allows, double-container storage in a desiccator goes a long way to protect your supply.
Key Environmental Controls
Dusty, poorly ventilated rooms turn basic chemical storage into a gamble. Storing 3,6-Dihydroxyphthalonitrile calls for a dedicated chemical cabinet—preferably one that stays below 25°C and away from sunlight. Temperature swings speed up degradation and boost the chance of condensation inside the bottle. If you’re in a humid part of the world, a small, powered desiccator may quickly pay for itself.
Labeling and Segregation
Store this compound away from acids, bases, and strong oxidizers, since it doesn’t play nicely with those materials. Some labs lump all “organics” together on big shelves. In practice, this leads to contamination and, sometimes, runaway reactions. I always mark containers with clear dates and hazard symbols. Updated safety data sheets hang where anyone can grab them.
Emergency Precautions
Human error happens. I recall a late-night researcher who popped open an old bottle and nearly fainted when a musty, acrid odor poured out. He learned—too late—about the dangers of poor ventilation and forgotten expiration dates. Labs dealing with 3,6-Dihydroxyphthalonitrile need good airflow, a spill kit, and easy access to goggles and gloves. If an accident occurs, someone must know where to find the nearest eyewash station and emergency exit.
The Role of Training and Oversight
No storage solution works without responsible people and regular checks. I never trust memory alone; written logs and rotational inspections catch problems early. Just last year, a colleague avoided disaster by noticing powder caking on a rarely used jar. Active oversight, paired with a deep respect for chemical unpredictability, saves more than time and money; it saves lives.
What is the purity and grade available for 3,6-Dihydroxyphthalonitrile?
Why Purity Isn’t Just a Number
Every chemist who has handled 3,6-dihydroxyphthalonitrile knows purity isn’t just about lab reports. Your reactions depend on it. A batch that’s a percentage point off can throw months of research off track. The standard on the market usually sits at 98% or higher for research-grade material. For demanding applications such as dye synthesis, high-performance coatings, and specialty electronic materials, even trace impurities will ruin your yield or compromise end product quality.
Available Grades: A Buyer’s Minefield
Most commercial suppliers offer 3,6-dihydroxyphthalonitrile as a fine powder, coming in the so-called “analytical” or “reagent” grades. What this usually means is that the purity checks in at or above 98%. Anything below, and the product gets relegated to “technical grade.” From experience, this technical material doesn’t hold up in sensitive applications. You see odd colors in the solution, unexpected byproducts, or worse—your spectra show garbage peaks that are tough to track down.
Who Needs What Grade?
In my own lab life, we always hunt for above 99% purity for any work heading toward publication or real-world use. High-purity batches have documentation: NMR data, HPLC traces, and sometimes GC-mass spec. Without those, a responsible chemist stays alert for contamination from metals, residual solvents, or incompletely reacted precursors.
Industrially, some users accept a little less purity if the application is robust, but there’s still a big difference between 95% and 98%. Small changes invite problems—clogged filters, lower yields, missed deadlines. For scale-up, that’s money lost. Nobody in industry wants that surprise weeks before shipping product.
What Really Matters: The Impurities
Not all impurities are created equal. Even at 98% purity, the last two percent might contain phthalic acid, hydroquinone, or leftover solvent. Some of these impurities interfere with downstream chemistry, especially in catalytic cycles or material science projects. Last year, I saw a whole lot ruined because extra hydroquinone enjoyed oxidizing like wild, and the end polymer changed color. So the documentation that comes with a premium grade isn’t just paperwork—it’s a map to what could go wrong.
The Business Side: Sourcing and Solutions
Reliable suppliers, often those based in North America, Japan, and Germany, build reputations on documentation and repeatable supply. If a catalog looks sketchy or won’t provide certificates of analysis, engineers and scientists go elsewhere. To manage cost, a lot of smaller facilities purify in-house using recrystallization or column chromatography, but that adds time and risk. Outsourcing supply to a trusted partner saves hassle, and for critical projects, having two suppliers is a must. Science isn’t forgiving to one-source dependency when shipments get held at customs or quality drifts.
Navigating the Future of Purity
With countries tightening regulation on import and chemical purity, the bar keeps rising. Digital tracking and transparency about impurities help labs and companies avoid costly mistakes down the line. In my experience, it always pays to ask hard questions of your supplier before the invoice is signed.
Summary Table
| Grade | Purity (%) | Best Use Cases |
|---|---|---|
| Analytical/Reagent | 98%+ | R&D, publications, electronics, medical research |
| Technical | 95-97% | Early process screening, non-critical manufacturing |
| Names | |
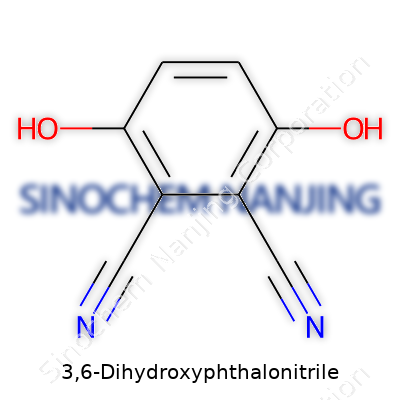
| Preferred IUPAC name | 3,6-dihydroxybenzene-1,2-dicarbonitrile |
| Other names |
3,6-Pyridinedicarbonitrile, 1,4-dihydroxy- 3,6-Dicyanoresorcinol Phthalonitrile, 3,6-dihydroxy- |
| Pronunciation | /ˈθriː.sɪks daɪˈhaɪ.drɒk.si ˌθæ.ləˈnaɪ.trɪl/ |
| Identifiers | |
| CAS Number | 40024-42-8 |
| 3D model (JSmol) | `load =C1=C(C=C(C(=C1O)C#N)O)C#N` |
| Beilstein Reference | 163873 |
| ChEBI | CHEBI:28148 |
| ChEMBL | CHEMBL3759709 |
| ChemSpider | 11440494 |
| DrugBank | DB08347 |
| ECHA InfoCard | 100.051.044 |
| EC Number | 609-085-6 |
| Gmelin Reference | 80850 |
| KEGG | C14368 |
| MeSH | D017708 |
| PubChem CID | 118145 |
| RTECS number | UR4375000 |
| UNII | L063OK7N3N |
| UN number | Not regulated |
| CompTox Dashboard (EPA) | DTXSID8070349 |
| Properties | |
| Chemical formula | C8H4N2O2 |
| Molar mass | 164.12 g/mol |
| Appearance | White to light brown solid |
| Odor | Odorless |
| Density | 1.682 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.23 |
| Vapor pressure | 0.0076 mmHg (25 °C) |
| Acidity (pKa) | 4.60 |
| Basicity (pKb) | 1.17 |
| Magnetic susceptibility (χ) | -0.78×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.617 |
| Dipole moment | 3.9 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 142.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -267 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -927 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P305+P351+P338, P321, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 246°C |
| Lethal dose or concentration | LD50 (oral, rat): >5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 1340 mg/kg |
| NIOSH | UR2975000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
3,4-Dihydroxyphthalonitrile 3,5-Dihydroxyphthalonitrile 3,6-Dimethoxyphthalonitrile 3,6-Diaminophthalonitrile Phthalonitrile 3-Hydroxyphthalonitrile 6-Hydroxyphthalonitrile |

