Looking Deeper into 3,5-Dinitroaniline—An Editorial Reflection
Historical Development
3,5-Dinitroaniline has roots that date back to the formative days of organic chemistry. Chemists first isolated aniline-based compounds in the nineteenth century, opening floodgates for nitroaromatic research. A need for stronger dyes and more potent intermediates led to deeper study of aniline derivatives. Researchers, chasing better color fastness and stability in textiles and paints, found ways to introduce nitro groups to aromatic rings. Through those experiments, they discovered both the promise and the risk—higher reactivity comes with higher hazards. Textbooks tell the story of breakthroughs that came from trial and error, hard work in basic laboratories, and plenty of chemical burns. Life in the lab didn’t revolve around automation or sophisticated instrumentation then. Instead, tired hands and sharp noses picked up subtle differences in product and byproduct. It’s not just old stories—these lessons on effort, error, and perseverance lay a foundation for all research into potent intermediates like 3,5-Dinitroaniline today.
Product Overview
Ask any chemist about aromatic nitro compounds, and 3,5-Dinitroaniline won’t be far down the list. This compound stands out because of its dual nitro substitution pattern, making it both a useful intermediate and a challenging substance to handle. Its stature grew alongside the use of synthetic dyes, explosion-resistant materials, and agricultural chemicals. Engineers and researchers keep returning to it for those same reasons: predictable reactivity, high yields in certain reactions, and a link to more complex molecules. On a lab shelf, it’s not the flashiest bottle, but beneath the label hides a key ingredient for everything from colorants to advanced polymers.
Physical & Chemical Properties
3,5-Dinitroaniline forms bright yellow crystals, a signature trait for many nitroaromatics. Its melting point sits higher than many straight-chain organics but lower than highly conjugated systems. Warm hands will never melt it on a summer day, but a gentle heat in the lab brings it to a melt before it combusts. Odor can give hints about volatility, though this compound isn’t especially aromatic—at least not in the sense your nose registers. In water, its solubility falters, yet strong acids and bases treat it like a guest, coaxing it into solution for further modification. The presence of two electron-withdrawing nitro groups lets it serve as both a challenge and an asset in reactions. In my own time at the bench, small changes in pH, solvent mix, or reaction time produced noticeably different outcomes, underlining how much care each handling step needs.
Technical Specifications & Labeling
Labeling of chemicals in real-world storage matters more than just following rules. Clear Signal Words, prominent hazard codes, and bold NFPA diamonds protect workers from serious harm. When you see bottles of 3,5-Dinitroaniline in a storeroom, every inch of tape, every sticker matters. Warnings about explosive risk and toxic dust do more than meet regulatory standards—they save lives. The specification sheets go beyond listing purity; they describe how trace contaminants can change performance or safety. In research or manufacturing, handling small quantities under a fume hood—with personal protective equipment—turns from recommendation to necessity once you’ve watched nitrated powders ignite or cause burns. This isn’t just chemistry formality—your safety depends on respect for the material and the clarity of every label you see.
Preparation Method
Early routes to 3,5-Dinitroaniline involved direct nitration of aniline itself, pushing the limits of classical organic synthesis. Today, most chemists still rely on stepwise introduction of nitro groups, using mixtures like concentrated nitric and sulfuric acid. The control of temperature, rate of addition, and acidity levels guides the process toward the target substitution pattern. Outside the pristine world of academic research, practical challenges pile up. Uncontrolled exotherms, runaway reactions, and toxic fume releases keep operators on their toes. The waste produced—a mix of acidic water, unreacted starting material, and dangerous side products—demands careful disposal. Responsible labs focus on minimizing hazards, counting every drop and weighing risks at each stage. As regulations around waste and emissions grow tighter, chemists adapt, using milder reagents or closed reactor systems to keep both operators and the environment safe.
Chemical Reactions & Modifications
Reactivity, in the context of 3,5-Dinitroaniline, offers a double-edged sword. On one hand, the nitro groups activate specific positions for further modification—from reductions to radical substitutions. On the other hand, the same groups make the molecule less stable in energetic environments. I remember the challenge of carrying out reduction reactions: too little control, and you generate tar or worse, hazardous exotherms; too much solvent, and yields evaporate along with your patience. Still, selective reductions transform these nitro groups into valuable amines or hydroxylamines, enabling development of dyes, pigments, and even pharmaceutical scaffolds. The aromatic core gives a sturdy backbone, resisting unwanted rearrangement or polymerization under most conditions. Students sometimes underestimate the risks in post-modification, especially during scale-up, but a few hours spent cleaning up a spill or rebuilding a setup can refocus anyone’s attention on safety.
Synonyms & Product Names
Looking at catalogs from chemical suppliers, 3,5-Dinitroaniline hides behind plenty of names—m-Xylidine dinitrate, 1-amino-3,5-dinitrobenzene, sometimes Dinitroaniline. Each of these speaks to a slice of chemical history and application. The parade of synonyms can create confusion in the literature and in stockrooms, risking mistakes in ordering or handling. For newcomers and veterans alike, recognizing alternative names prevents costly errors. Clarity in naming helps bridge research, manufacturing, and regulatory conversations, so teams speak the same language—no matter their position or department.
Safety & Operational Standards
In a world that too often takes shortcuts, safety protocols around 3,5-Dinitroaniline draw clear boundaries. Meeting regulatory minimums won’t suffice where toxic dust and explosive potential linger. Gloves, respirators, and adequate ventilation shift a workspace from hazardous zone to controlled lab. Training doesn’t stop at annual refreshers; it emerges every day in habits, reminders, and debriefs after accidents or near-misses. I’ve seen teams freeze after a minor spill, the silence punctuated only by the hiss of fume extractors. Those moments shake up complacency and reinforce the lessons on containment, prompt cleanup, and incident reporting. Adherence to guidelines—whether OSHA, REACH, or GHS—forms the backbone of modern laboratory and plant management. Only by respecting the properties of these chemicals does the research community continue forward with confidence.
Application Area
Beyond the laboratory, 3,5-Dinitroaniline finds roles in dye manufacture, agricultural chemicals, and as an intermediate for specialty energetic compounds. Textile industries rely on its reactivity for chromophore synthesis, creating colorfast dyes suited for plastics, fibers, and inks. In agriculture, derivatives act as weed control agents, targeting specific pathways in plant metabolism. Specialty explosives and propellants sometimes spring from further chemical manipulation of its nitroaromatic skeleton. The versatility here shouldn’t eclipse the challenges—such as environmental persistence or human toxicity—which prompt ongoing research and tighter controls. Effective applications spring from targeted synthesis, meaning not every sector needs broad access to raw 3,5-Dinitroaniline. Restricting usage to trained, equipped professionals is one meaningful way to manage risk while still harvesting chemical value.
Research & Development
Innovation keeps the story of 3,5-Dinitroaniline alive. Universities, corporate labs, and startups all chase greener, safer production methods. Catalytic processes offer hope for lowering waste and energy consumption, but industry inertia—old equipment, legacy procedures—slows adoption. Computer modeling now predicts reactivity and hazard before a single flask fills with acid. Data from these simulations, coupled with in-situ monitoring, have allowed several groups to sharply reduce both waste and risk, fostering a new generation of sustainable chemistry. In my own work, collaboration between synthetic and environmental chemists revealed how downstream metabolites behave, influencing both product design and regulatory approaches. Funding constraints sometimes pressure teams to cut corners, but rising public expectations about the environment push budgets toward long-term safety and efficiency instead of quick wins.
Toxicity Research
Concerns about nitroaromatic toxicity go back decades. Work in the 1970s and 80s peeled back layers of mystery around DNA binding, carcinogenicity, and environmental mobility. 3,5-Dinitroaniline, like its siblings, poses risks to liver and kidney over repeated exposure, with laboratory animals highlighting vulnerabilities. Waste handling grows urgent: effluents containing traces of this compound linger in water tables for years, threatening aquatic life and, by extension, drinking water safety. Researchers continue unraveling its metabolic fate, developing rapid detection assays and improved sorbents for wastewater cleanup. Practical solutions have emerged from pilot studies—advanced oxidation processes, biofiltration, and membrane technologies all help lower risk. Full elimination proves elusive, but reductions in emission and exposure levels show real progress. For anyone working with this compound, keeping up with peer-reviewed toxicity data is as important as tracking supplier inventory.
Future Prospects
Looking at tomorrow, the future of 3,5-Dinitroaniline will likely revolve around safety, sustainability, and specialty applications. Economic constraints and green chemistry movements nudge industry toward lower-impact processes, cleaner downstream profiles, and engineered biodegradation. Advances in robotics, in-line monitoring, and microreactor design point toward safer, smaller-scale synthesis. Researchers still bet on nitroaromatics for performance in dyes and targeted pharmaceuticals, leveraging selectivity honed by generations of chemists. If regulators harmonize rules across borders and the scientific community shares best practices, the story will veer toward safer workspaces and leaner waste streams. In my own lab circle, we talk less about banning chemicals outright and more about how to coax safer, smarter value from powerful reagents. For 3,5-Dinitroaniline, that’s a challenge worth the effort and the vigilance.
What is 3,5-Dinitroaniline used for?
Not Just Another Chemical Compound
3,5-Dinitroaniline doesn’t show up on kitchen shelves or in any bottle under the bathroom sink. Its story plays out across labs, factories, and some pretty gritty industry floors. Most people probably haven’t heard much about it outside chemistry circles, yet it powers processes behind products that show up in unexpected places.
The Drive for Color
For me, learning about the path from raw chemical to recognizable product carries as much appeal as learning how bread rises in an oven. 3,5-Dinitroaniline gets a spot on the list because dye manufacturers look for this compound to help them lay down brilliant colors. It works as a building block in the synthesis of azo dyes—the sort that bring intense reds, oranges, and yellows to textile fibers, plastics, inks, and even cosmetics. Textile workers, printers, and artists benefit from colorfastness and vibrancy made possible, in part, by this compound.
Linking Chemistry with Manufacturing
I’ve watched chemists liven up dull swatches of fabric with just small changes to a formula. 3,5-Dinitroaniline makes these feats a little more predictable. Because it feeds into the creation of dyes designed to stick, it helps clothing keep color after dozens of spins in the laundry. A chemical link forms, holding pigment to fiber, which matters even more to families watching clothing bills. Dyestuff companies lean on it for consistency in their batches.
Beyond the Color Wheel
The story doesn’t stop at dyes. 3,5-Dinitroaniline sees use in laboratories as an intermediate for specialty chemicals. Here, it appears in the recipes for various agricultural and pharmaceutical products. In crop protection, certain herbicides need specialized molecules. Chemists have used this particular compound to create weed killers that cut down broadleaf competition in fields. Farmers counting on those herbicides depend on reliability and safety, since both impact crop yields and consumer health.
Keeping Safety and Environment in Focus
Handling 3,5-Dinitroaniline isn’t risk-free. It earns hazard warnings—direct contact can irritate skin, eyes, and mucous membranes. Regular workers have protocols for protective equipment and safe disposal. Spills or careless dumping could move toxic substances into soil and water. This raises a red flag for environmental and occupational specialists, since mishandling impacts more than just factory staff. Safety regulations serve as more than a checklist. They trace back to stories of industry workers who saw long-term effects of chemical exposure. Addressing these risks, companies put money into training, air monitoring, and alternative processes, aiming to limit harm both inside the facility and out in the community.
What Comes Next?
Tech keeps marching on. Research groups look for greener ways to get the colors and chemicals we want, with less environmental baggage. Some teams experiment with enzymes and renewable feedstocks as alternatives, but familiar compounds like 3,5-Dinitroaniline won’t disappear overnight. As the conversation about chemical safety and sustainability grows, the challenge becomes how to support industries reliant on these chemicals, help workers stay safe, and nudge companies to try cleaner options. Every step counts—on factory floors, in regulatory meetings, and while shopping for anything from T-shirts to farm produce.
Is 3,5-Dinitroaniline hazardous to health?
What Is 3,5-Dinitroaniline?
This yellow crystalline powder crops up in different corners of chemical manufacturing, especially in making dyes and pesticides. Its structure packs two nitro groups onto an aniline backbone. Chemistry can sound abstract, but here we talk about a compound with real impacts in workplaces and communities.
Routes of Exposure
Jobs in dye, pigment, and pesticide production often bring people into direct contact with 3,5-Dinitroaniline. Inhalation dust, skin contact, and accidental ingestion rank as the primary ways someone could absorb it. Chemists and operators usually work with gloves, goggles, filtration systems, and strict controls to limit exposure. Sometimes, though, older facilities or gaps in training slip through the cracks. Airborne dust, spills, and improper storage can raise real risks, especially for those handling bulk quantities or cleaning contaminated equipment.
How Does It Affect the Body?
Studies done on related nitroanilines show these chemicals can irritate eyes, cause rashes, and affect the respiratory system. In severe or repeated high-level exposure, nitro compounds trigger methemoglobinemia—a disorder where your blood’s ability to carry oxygen drops. Symptoms like blue lips, rapid heart rate, dizziness, and even seizures may develop. Workers who noticed these effects sometimes needed hospital care for oxygen support or specific medical antidotes.
Animal tests have linked high exposures to liver and kidney injury, as well as blood changes. Long-term, persistent exposure raises bigger flags for cancer risk or birth defects. Solid proof for human cancer risk doesn’t exist yet, but many nitro compounds show up as mutagens in laboratory tests.
What Do Health Agencies Say?
Agencies like OSHA, NIOSH, and the European Chemicals Agency flag 3,5-Dinitroaniline as requiring real caution and controls. The US Environmental Protection Agency lists it as toxic, which means manufacturers need to track and report large-scale usage or accidental release. In Europe, it faces strict risk assessments under REACH regulations. Community members living near manufacturing sites sometimes worry about air or water contamination, raising pushback against unregulated releases.
Reducing Hazards—Realistic Steps
Practical safety means more than signs on the wall. Facility managers can put in strong local ventilation, maintain equipment, and run regular occupational health checkups for workers. Providing proper respirators, chemical-resistant gloves, and eye protection matters. Training helps people spot trouble, like leaks or dust buildup, before it grows into a bigger problem.
Outside of direct manufacturing, waste management and environmental controls keep the larger population safer. Regular monitoring in nearby waterways and soil, rapid cleanup after spills, and transparent community communication make a difference. Companies can substitute safer chemicals in their production lines where possible, which often proves better for everyone in the long run.
Looking Out for Each Other
Years spent around chemical plants or reading the stories of former workers has taught me respect for these risks. Small routine exposures show up in blood work, and regular health clinics for employees become not just policy, but reassurance. Community groups often push hardest for tighter enforcement, and their pressure has led to tougher rules.
Nobody can dodge every workplace risk, yet smart policy, transparent communication, and reliable protective measures help tip the odds in favor of health rather than harm.
What is the chemical structure of 3,5-Dinitroaniline?
The Nuts and Bolts of Its Structure
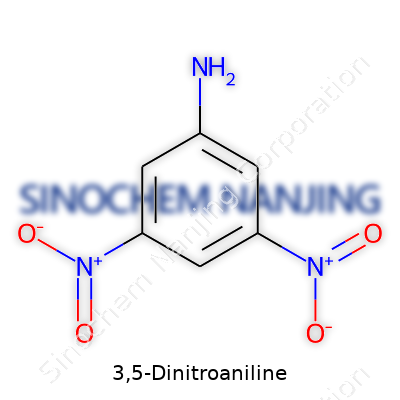
3,5-Dinitroaniline stands out as a unique molecule. You have a benzene ring forming the backbone, which is something most people recognize as that classic hexagon in chemistry class. Attached to that ring, at the third and fifth positions, sit two nitro groups (-NO2). At the first position, there’s an amine group (-NH2). Take a step back and that combination — one amine and two nitros on a benzene ring — gives the compound its distinctive character. Its full formula: C6H5N3O4.
Simplicity on paper, yet complexity in function. Visualizing it: the benzene ring acts like the base, amine sits at the top, and nitro groups hang out two carbons away on each side. This particular arrangement does more than just look good in a chemistry textbook. The positions of those nitro groups matter — placement changes the molecule’s behavior, sometimes in ways that give scientists a headache during synthesis or analysis.
An Everyday Life Connection
Chemicals like 3,5-dinitroaniline aren’t just obscure research topics. Farmers rely on related compounds as herbicides. In high school, I spent a summer working for a landscaping company. The owner kept talking about certain types of weed killers, their colors, and safety when mixing chemicals. Turns out, chemicals sharing the nitroaniline structure pop up in several herbicide formulations, especially those targeting weeds while sparing crops.
There’s a flip side as well — 3,5-dinitroaniline doesn’t dissolve in water very well. That influences how it behaves in soil and how long it hangs around after an application. The same chemical stability that makes it effective can also mean it sticks around longer in the environment than anyone wants.
Why Structure Matters in the Real World
Structure does more than dictate the basics of chemistry; it shapes everything from manufacturing to environmental safety. Those nitro groups make the molecule less reactive under normal conditions, which helps reduce accidental breakdown. On the other hand, the presence of the amine group still allows scientists to manipulate the compound to create new materials or tweak existing ones.
Many safety protocols trace back to these specific structural elements. The nitro groups bump up the potential for toxicity and environmental persistence. Handling guidelines, safe disposal requirements, and protective gear aren’t suggestions — they’re built around hard-won experience with compounds that don’t always behave as expected.
From an educational angle, the structure of 3,5-dinitroaniline helps students understand important organic chemistry concepts without wading through endless jargon. Once you see how positioning nitro groups affects a molecule’s water solubility or potential reactivity, practical chemistry starts to click.
What Needs Attention
Researchers know the risks that come with nitroaniline-based compounds. Monitoring what ends up in fields and waterways takes commitment, not shortcuts. Regular soil and water testing, investment in research for biodegradable alternatives, and tight controls during manufacturing all play a part in reducing negative impacts. Policy only goes so far. Farmers, scientists, and everyday consumers share responsibility — each decision adds up.
Plenty of unknowns still hover around these chemicals. More transparency from manufacturers would go a long way, alongside clear labeling and open reporting on latest health studies. Regular safety training in agriculture remains important, backed up by frequent and accessible public updates regarding any new findings or incidents.
Learning about the structure of 3,5-dinitroaniline isn’t just theory — it impacts how professionals handle, measure, and respect what’s in the bottle, underfoot, or flowing downstream.
How should 3,5-Dinitroaniline be stored?
Why Proper Storage Matters
Working in research and industrial labs for over a decade has taught me the hard way that risky chemicals like 3,5-dinitroaniline demand a lot of respect and a solid storage plan. This aromatic nitro compound doesn’t make headlines like some explosives do, yet incidents can still creep up when folks overlook small but critical details. Nasty odors, yellow dust on containers, hard-to-clean workspaces — signs like these suggest something’s not right behind the scene.
Nitroanilines show sensitivity to heat, friction, and bumping. Even if 3,5-dinitroaniline sits lower on the hazard scale compared to trinitrotoluene or picric acid, faulty handling or sloppy storage raises the potential for chemical decomposition. More than once, I’ve seen containers with caked-on residue stuck in corners of fume hoods because somebody skipped a step in the cleanup process.
Choosing the Storage Space
A consistent lesson: store 3,5-dinitroaniline in a cool, dry spot, with plenty of ventilation and no sunlight creeping in. I’ve walked into supply rooms in mid-July that felt like greenhouses, jars sweating more than the staff. Heat stress on these compounds accelerates breakdown, sometimes resulting in pressure buildup or unpredictable reactions if moisture finds its way inside.
Fire-resistant cabinets made for flammables and explosives have proven their value time and again. Doors close smoothly, shelves take the weight, and chemical spills can be contained. Fume hoods play a part during transfers or weighing, but they never serve for long-term storage — too much movement, temperature swings, and risk of contamination from other chemicals.
Container Choices: Not All Lids are Equal
Glass bottles with tight seals keep out humidity better than cheap plastic. Labels matter too, since handwritten notes tend to fade, and mystery jars never inspire confidence. Safety teams I’ve worked with always advocate for pressure relief caps or compatible venting systems. One slip with a cracked cap, and you end up with powder on your fingers and headaches from fumes.
Old inventory should never linger. I learned to keep an updated log and cycle through stock by expiration date, with monthly checks for crystal growth or leaks. That sort of diligence keeps you honest, and it keeps everyone safer.
Housekeeping Rules
Segregation runs the show. Keep 3,5-dinitroaniline away from strong acids, bases, reducing agents, and oxidizers. Even in shared labs, I push to see solid barriers or secondary containers. Using trays to catch spills means cleanup goes faster if someone drops a jar or misjudges a scoop.
Personnel training makes an enormous difference. Storage rules aren’t worth much if folks don’t understand why they exist. Talking through real incidents with new lab staff changes the culture. Simple tweaks — double-checking lids, jotting down observations, rotating stock — all lower the risk in everyday operations.
Addressing the Gaps
Regulations often lag behind best practices. Regular reviews, updated standard operating procedures, and clear communication between management and on-the-ground staff close the gap. Investing in better cabinets and ventilation may not win awards, but watching out for each other and having a voice in storage policies does more to protect both health and reputation.
Few things matter more in hazardous materials work than trust and vigilance. Chemicals such as 3,5-dinitroaniline force teams to make regular safety a shared habit, not just a rule in the manual.
What are the safety precautions when handling 3,5-Dinitroaniline?
The Reality of 3,5-Dinitroaniline Risks
People working in chemistry labs often meet chemicals that look harmless, yet carry a punch when it comes to toxicity. 3,5-Dinitroaniline fits that profile. Now, the yellow powder has its uses, mostly as a building block for dyes and pesticides, but it doesn’t like mistakes. Its dust irritates the nose and throat and if it settles on your hands, problems grow. Anyone who has accidentally rubbed their eyes after handling similar stuff knows the burn. Breathing its dust isn’t just unpleasant—it can tear up your airways and spark headaches. Safety talk isn’t scare tactics. The danger is real.
Simple Steps That Prevent Accidents
For some, gloves might feel awkward. They get sweaty. But bare hands make handling this powder a risky game. Nitrile or neoprene gloves make all the difference, stopping powder from reaching the skin. Protective glasses are not overkill either. Few chemical workers forget the sting when dust hits the eyes—cheaper to deal with steamy goggles than with permanent eye damage. I once watched a colleague spend weeks recovering after splashing a far milder chemical in his eyes. Now, he never skips his glasses, even for “just a minute.”
Lab coats serve more than appearances. They keep street clothes clean, but more than that, they form a barrier when chemicals spill. In a shared workspace, that single step stops folks from spreading powder into break rooms or public spaces. Closed-toed shoes sound like a fashion rule, but they keep chemical burns off the feet. Every worker who’s dropped a flask can tell you why sandals and lab chemicals just don’t mix.
Why Good Ventilation Isn’t Optional
Breathing fine dust can do harm over time. Fume hoods or well-designed extraction fans keep the air safer for everyone. In an old lab with poor ventilation, it only takes half a shift to realize headaches start popping up. After fixing the airflow, those sick days drop. The key is consistency. If the hood is there, use it. If it's not working, speak up. Turning a blind eye to poor air circulation never pays off.
Storage and Spills Demand Respect
Anyone storing 3,5-Dinitroaniline learns to read labels and keep containers sealed. This isn’t just neatness—humidity or accidental mishandling can lead to messy situations. Storing the chemical away from heat sources or sunlight keeps it from breaking down or reacting with other materials. At one job, chemicals stored above head height spelled disaster during an earthquake; ever since, I look for shelves that won’t let things topple.
Spills call for calm and the right tools. Small amounts can get swept up with damp paper towels, always while wearing gloves and glasses. Large spills? Time to alert everyone and use the spill kit. Never sweep dry powder—it throws more dust into the air.
Knowledge Lowers the Odds of Trouble
Training isn’t just bureaucracy. Each chemical behaves differently, and nothing replaces real-world practice mixed with sharp instructions. Material Safety Data Sheets don’t sit in drawers for nothing. Reading them before any new job keeps workers aware of symptoms to watch for and how to handle emergencies. One time, a quick glance saved me from mixing incompatible chemicals—just because it was the only time all week I had read the data sheet all the way through. That five-minute read easily avoided hours of cleanup and potential health problems.
Solutions Take a Team Effort
Culture at work makes a big difference. Leadership that rewards safety-conscious choices quickly reduces accidents. People speak up, report broken hoods, and stop each other before mistakes happen. Small tweaks—like setting out gloves and glasses near the door—nudge those who might skip steps. Supervisors who actually wear the gear they recommend show by example. Sharing stories (not just rules) about near-misses or lessons learned from spills creates a shared memory that guides newer folks. Little changes, repeated daily, build safer habits for everyone handling chemicals like 3,5-Dinitroaniline.
| Names | |
| Preferred IUPAC name | 3,5-dinitrobenzenamine |
| Other names |
1-amino-3,5-dinitrobenzene 3,5-dinitrobenzenamine |
| Pronunciation | /ˌθriːˌfaɪv.daɪˌnaɪtrəʊ.əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 618-87-1 |
| Beilstein Reference | 1638985 |
| ChEBI | CHEBI:27860 |
| ChEMBL | CHEMBL215358 |
| ChemSpider | 13644 |
| DrugBank | DB07715 |
| ECHA InfoCard | 03b574a2-8d59-40fd-bd32-3017c1b2b753 |
| EC Number | 202-877-8 |
| Gmelin Reference | 85790 |
| KEGG | C06605 |
| MeSH | D004082 |
| PubChem CID | 9869 |
| RTECS number | UA9625000 |
| UNII | 86C3V00U9A |
| UN number | UN1322 |
| Properties | |
| Chemical formula | C6H5N3O4 |
| Molar mass | 213.13 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.47 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.96 |
| Vapor pressure | 2.98E-5 mmHg at 25°C |
| Acidity (pKa) | 3.7 |
| Basicity (pKb) | pKb = 10.56 |
| Magnetic susceptibility (χ) | -56.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.670 |
| Dipole moment | 2.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 181.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −40.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1052.0 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed, causes serious eye irritation, may cause respiratory irritation, harmful to aquatic life. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-2-Expl |
| Flash point | 174 °C (345 °F; 447 K) |
| Autoignition temperature | 460 °C |
| Explosive limits | Explosive limits: "not known |
| Lethal dose or concentration | LD50 oral rat 590 mg/kg |
| LD50 (median dose) | Oral rat LD50: 590 mg/kg |
| NIOSH | DN 16800 |
| PEL (Permissible) | PEL: 1 mg/m3 |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 25 mg/m³ |
| Related compounds | |
| Related compounds |
2,4-Dinitroaniline 2,6-Dinitroaniline 3,5-Dinitroacetanilide 3,5-Dinitrobenzoic acid Aniline |

