3,5-Dimethoxycinnamic Acid: A Deep Dive
Historical Development
Research into cinnamic acid derivatives has roots stretching back to the 19th century, when European chemists explored plant extracts looking for bioactive compounds. 3,5-Dimethoxycinnamic Acid emerged from this wave of discovery, noted early on for its unique placement of methoxy groups on the phenyl ring. As interest in natural product synthesis and modification grew, so did attention to this molecule, which mirrored components in the flavor and fragrance sector, and later found roles in chemical biology labs. Over decades, incremental improvements in synthetic methodology have made this compound more accessible to researchers probing the limits of molecular modification and pharmaceutical discovery.
Product Overview
3,5-Dimethoxycinnamic Acid falls under the family of substituted cinnamic acids, standing out with two methoxy groups at the 3 and 5 positions of its aromatic ring. Structurally, it brings together the phenyl group and the acrylic acid moiety, giving it reactivity that makes it a genuine platform for further chemical invention. Laboratories and small-scale manufacturers alike see it as a stock building block for making new aromatic carboxylic compounds, pushing research into new materials and drug precursors. Its consistency in structure and reliable reactivity keeps it in steady demand among chemists aiming for predictable results.
Physical & Chemical Properties
This compound shows up as a white to off-white crystalline powder, stable under standard conditions. It has a molecular weight of 208.21 g/mol. The melting point sits around 151–154°C, which speaks to reasonable thermal stability. The compound dissolves in organic solvents like ethanol, methanol, and dimethyl sulfoxide, but shows limited solubility in water. The presence of the electron-donating methoxy groups on the aromatic ring changes both its polarity and its chemical behavior, making it react more readily in nucleophilic substitution and electrophilic aromatic substitution than unsubstituted cinnamic acid.
Technical Specifications & Labeling
Bulk producers and laboratory suppliers list assay values generally above 98%, ensuring minimal contaminant content for research needs. Packaging relies on light-resistant materials, given some sensitivity to prolonged UV exposure. Labels highlight the batch number, purity, storage recommendations (preferably cool, dry, and away from direct sunlight), and chemical hazard symbols as per GHS requirements. Many suppliers add COA (certificate of analysis) documentation, giving researchers confidence in reproducibility and traceability. The labeling reflects current regulatory standards, aligning with chemical inventory regulations in North America, the EU, and Asia.
Preparation Method
The most widely used synthesis for 3,5-Dimethoxycinnamic Acid involves the Perkin reaction, a time-tested approach that couples 3,5-dimethoxybenzaldehyde with acetic anhydride in the presence of an alkali salt, typically sodium acetate. The process yields the corresponding cinnamic acid derivative after hydrolysis and acidification steps. Improvements in solvent selection and temperature control over the years have pushed yields higher, and workup steps have become more efficient, cutting down on waste and energy use. Some labs switch to the Knoevenagel condensation for small-scale runs, taking advantage of milder conditions and fewer by-products. Attention to purification is crucial, and best results come from careful crystallization or column chromatography.
Chemical Reactions & Modifications
Chemists appreciate the reactivity of 3,5-dimethoxycinnamic acid for several reasons. The carboxylic acid function is ripe for standard esterification, amidation, and reduction, generating esters, amides, and alcohols that each find use in probing structure-activity relationships or building molecular scaffolds. The methoxy groups steer substitution chemistry, often serving as handles for demethylation, halogenation, or oxidative modifications. The double bond invites reduction to the corresponding saturated acid or can be used as a site for Michael addition. In my experience, combining these handles opens the door to a huge family of derivatives, each with subtle shifts in chemical and biological behavior. Researchers find that a small tweak in the aromatic domain can lead to big changes in downstream reactivity.
Synonyms & Product Names
3,5-Dimethoxycinnamic Acid is referenced in literature under several names, depending on the context and supplier. Common synonyms include 3,5-Dimethoxy-trans-cinnamic acid, trans-3,5-Dimethoxycinnamic acid, and (E)-3-(3,5-dimethoxyphenyl)acrylic acid. Product numbers vary, but chemical catalogs keep CAS number 1163-37-9 as a unique identifier, sidestepping the confusion created by variant trade names. This uniformity in cataloging matters, especially for regulatory compliance and academic reference.
Safety & Operational Standards
Handling demands personal protective equipment and proper ventilation. Users should avoid direct contact with skin and eyes, since low-level irritant effects are common across cinnamic acid derivatives. Inhalation risks are low for small-scale lab use, though dust management keeps exposure at a minimum. Disposal follows hazardous organic waste streams under most institutional guidelines. Good Laboratory Practice (GLP) protocols ensure consistent safety, from the weighing station to the reaction flask. Inventory and labeling standards prevent mix-ups and signal emergency responders if spills or accidental exposures occur.
Application Area
The main users of 3,5-Dimethoxycinnamic Acid are pharmaceutical researchers, synthetic organic chemists, and specialists in material science. In medicinal chemistry, this compound serves as both a pharmacophore and a lead for analog synthesis, as the methoxy group arrangement often shows promise in modulating biological activity. Some labs explore its antioxidant or antimicrobial activities, seeking alternatives to standard aryl carboxylic acids. Light-absorbing properties, owed to the conjugated double bond and aromatic substitutions, pull in research from the dye and additive sectors. Experience in collaboration projects shows this compound functions as a reliable base-shelf material for test reactions and custom syntheses, giving teams a consistent starting point for elaboration.
Research & Development
Recent years have produced dozens of papers turning over new stones in the chemistry of this molecule. Teams have published on tailored modifications of the acid function, the exploration of bioisosteres based on its framework, and the search for optimized derivatives with better pharmacokinetics. Advances in green chemistry push for solvent systems and catalysts that lower the environmental impact of synthesis. Computational chemistry aids in rational design, predicting where adding or shifting substituents could favor new biological targets. Research momentum holds steady, building off a solid foundation of synthetic experience and incremental methodology tweaks.
Toxicity Research
Available studies point to moderate toxicity, not far off from related cinnamic acid derivatives. Acute oral and dermal exposure in animal models shows low absorption and rapid excretion, with limited accumulation. Genotoxicity and carcinogenicity results so far trend negative at low concentrations, but long-term exposure datasets remain thin. Environmental toxicology calls out some persistence in soil and water, common for phenyl derivatives, so responsible disposal becomes part of daily practice. In lab work, users lean on standard precautions rather than working in fear of unexpected hazards. Good documentation of all incidents keeps the toxicity profile transparent and up to date.
Future Prospects
Since many current drug discovery campaigns now prioritize greener processes and structural novelty, compounds like 3,5-Dimethoxycinnamic Acid fit right into project portfolios. Growing access to enzymatic and flow chemistry techniques hopes to scale up its functionalization and make downstream processes more sustainable. Machine learning algorithms, fed by decades of published reaction data, promise more efficient pathway design starting from this base. The biotechnological angle, engineering microbes to biosynthesize cinnamic acid derivatives, is picking up, too, responding to demand for non-petroleum sources. For industry outsiders, it may look like just another specialty chemical, but for researchers pushing boundaries, it unlocks new directions in molecular design, targeted therapy, and advanced materials. As funding expands for safer chemicals and faster drug leads, this molecule squares up as a workhorse and a springboard.
What is 3,5-Dimethoxycinnamic Acid used for?
Digging Into Daily Science
Looking at the label on a lab bottle, “3,5-Dimethoxycinnamic acid” might read as a dry chemical name, but this compound actually ends up woven into real challenges and breakthroughs. If you ask chemists or pharmaceutical researchers about it, you’ll get stories about long hours spent coaxing complex molecules from simple sources. I remember during a summer in grad school, working alongside a postdoc mixing up synthetic pathways, this acid acted as a cornerstone for reactions we could never have solved otherwise.
Supporting New Treatments
Its role in medicine stands out right away. Medicinal chemists use 3,5-Dimethoxycinnamic acid as a scaffold to build other molecules. Think of it almost like a frame in a workshop that gets shaped into painkillers or anti-inflammatory agents. It’s not that the acid itself ends up in your medicine cabinet, but drug discovery teams latch onto its structure to create compounds with better activity against everything from joint pain to cancer cell lines. This step seems small, but sometimes a single methoxy group is the difference between a useful drug and a useless one. Published research backs this up, showing how these derivatives show anti-tumor and antimicrobial activity in carefully controlled studies.
Food Science Isn’t Left Out
If you have ever wondered how flavor chemists keep coming up with new taste notes for everything from candy to energy drinks, basic building blocks like 3,5-Dimethoxycinnamic acid help drive these innovations. With its roots in the larger group called cinnamic acids, it can spawn natural-sounding flavors or hint at “spicy” and “vanilla-like” profiles. In my own kitchen experiments, working with natural extracts for homemade syrups, related acids changed the way certain spices pop in a soda or liqueur.
Plant Science and Natural Research
3,5-Dimethoxycinnamic acid falls right in line with metabolic research carried out on plants, especially when looking for natural antioxidants. Extracting and analyzing these compounds from roots or herbs gives agricultural engineers new leads for boosting disease resistance or even improving shelf life for fresh produce. Studies point to its presence in medicinal plants like Scopolia tangutica, where it builds up as part of the plant’s natural defenses.
Facing Safety and Environmental Responsibility
Handling chemicals in the lab teaches everyone that you need to balance excitement with caution. 3,5-Dimethoxycinnamic acid doesn’t set off alarm bells on its own, but scale matters. Large-scale synthesis leads to byproducts and waste. Green chemistry points the way forward. Newer labs push for cleaner production methods, smarter solvent choices, and recycling leftover materials. As a result, these improvements help both student researchers and global manufacturers cut pollution and reduce risks down the line.
Looking Forward: What’s Next?
The uses for 3,5-Dimethoxycinnamic acid stretch beyond textbooks or ingredient lists. Open-minded research keeps uncovering new uses in polymers, coatings, or even as additives for plastics, aiming to give these everyday materials better weather resistance or stability. As research teams swap discoveries and refine their techniques, you watch curiosity build into something that can change how we approach both health and technology. At every step, this molecule offers another tool for anyone ready to explore deeper.
What is the chemical formula of 3,5-Dimethoxycinnamic Acid?
Getting Down to the Basics of 3,5-Dimethoxycinnamic Acid
Take a closer look at 3,5-dimethoxycinnamic acid and you’ll see a small detail that sets the tone for all its chemical behavior: C11H12O4. This formula isn’t just a string to memorize for exams or trivia. It spells out exactly how this molecule operates in the real world – in a lab, in nature, or even as a potential building block for new medicines.
Understanding Its Structure and Why It Matters
Matching the formula to its actual structure gives clues about its personality. There’s a benzene ring, spiced up with two methoxy groups sitting at the 3 and 5 positions. Down that same backbone, a double bond connects the ring to a carboxylic acid group, making it part of the cinnamic acid family. Those two methoxy groups do more than look pretty. They help nudge how the molecule behaves – changing how it dissolves, how it interacts with other chemicals, and giving it a unique flavor in the crowded world of organic acids.
The Importance Beyond the Chemistry Textbook
People often overlook compounds like this, thinking they’re only interesting for someone wearing a lab coat. In some of my college lab days, I came across related compounds in natural products – in spices, wildflowers, and even some everyday foods. These molecules deserve attention because they end up influencing antioxidant activity, plant defense mechanisms, and even serve as blueprints for drug development.
A lot of medicinal chemists, for example, use the backbone of cinnamic acids as a launching pad for designing drugs. By tweaking those methoxy groups, or swapping the acid chain, researchers try to boost desired benefits in anti-inflammatory agents, or to fight bacteria more effectively. The hard, numerical language of chemical formulas opens the door to an entire world of possibilities and application.
Why Details Like These Aren’t Just Trivial
Most people don’t think about how something as simple as the formula C11H12O4 might change the way certain products behave or how safe they might be. Regulatory bodies—think the FDA or international equivalents—pay close attention to every atom in that string when approving everything from preservatives to synthetic flavorings. Winemakers or those working in food chemistry can stumble into legal gray zones without realizing small changes in chemistry may create new molecules that require different safety checks.
Building Trust in Science and Everyday Life
It’s easy to brush past names like 3,5-dimethoxycinnamic acid, but knowing even a little about its structure helps unpack why scientists care so much about formulas and labels. Clarity and traceability stem from full chemical formulas. When trust in science wobbles, open information helps steady the conversation.
If more folks made a habit of checking the details or asking what names and formulas mean, there’d be less confusion and sharper public input on science policy, nutrition, and consumer safety. Putting some effort into chemical literacy pays off, even if you’re not running titration experiments or working at a regulatory agency.
What are the storage conditions for 3,5-Dimethoxycinnamic Acid?
A Practical View on Storage
Anyone who has ever handled specialty chemicals, even outside a lab, knows storage isn’t just about “putting substances away.” It’s about keeping the product usable, pure, and most importantly, safe. 3,5-Dimethoxycinnamic acid, a fine white or off-white powder often found in research labs and sometimes in fragrance chemistry, fits into this picture.
Keeping It Cool and Dry
A common headache comes from moisture. With powders like 3,5-Dimethoxycinnamic acid, water vapor in the air can trigger clumping or even start unwanted chemical changes. Based on chemical data and years of storage practice, keeping this compound in a dry, well-ventilated area makes a difference. Ambient humidity has caused many a ruined batch or forced unnecessary replacements. Silica gel packs or desiccators help, but companies that pay attention to basic climate control see fewer material losses.
Light and Heat: The Quiet Threats
Direct sunlight hurts more than just your eyes or your skin. Organic powders break down from exposure to UV rays, slowly turning from white to yellow or brown, sometimes changing their chemical profile. Heat can speed up decomposition too, sometimes making chemicals less effective or even hazardous. Having seen expired reagents stored near sunny windows, I’ve witnessed firsthand how even a slight temperature rise messes with stability and purity.
The lesson is clear: place chemicals like this in dark, temperature-stable rooms. Refrigeration (between 2°C and 8°C) extends the shelf life, though for short-term work, a cool, shaded shelf usually suffices. The constant up-and-down temperature swings cause condensation, so any cold storage should stay dry, not just cold.
Container Matters
Choosing the right container plays a bigger role than most realize. Glass vials or tightly sealed HDPE bottles block air, glare, and stray chemicals from mixing in. Loose lids, gasket-free caps, or old, scratched containers lead to contamination. Years of working in research showed me that careless storage—the “just screw the lid back on” habit—leads to impure samples and odd lab results.
Labeling, Security, and Regular Checks
Every label should show the chemical name, concentration, and expiry date. That sounds obvious, but I’ve found mystery bottles in many a shared chemical cabinet. Mistakes in labeling result in wasted time, extra expense, and occasionally dangerous mix-ups.
Storage cabinets for 3,5-Dimethoxycinnamic acid keep users safe if they limit access to trained personnel. Lockable chemical cabinets and open logs become a double protection—against accidents and carelessness. Institutions following the best lab practices include regular inventory audits, where expired or decomposing material gets removed long before issues arise.
Protecting Both People and Product
Safe storage isn’t just about ticking boxes for regulations. It’s about respecting the lab and everyone in it. Reliable storage of 3,5-Dimethoxycinnamic acid means separating it from strong oxidizers and acids—a piece of wisdom that’s saved more than one shelf in my experience. Spills get much less likely, reactions less frequent, and you prevent fires by keeping incompatible substances apart.
Well-controlled storage conditions stand as the simplest way to protect both the purity of research chemicals and the well-being of everyone who handles them. Proper chemical stewardship supports both progress and safety, making a small investment in containers and climate control worth every cent.
Is 3,5-Dimethoxycinnamic Acid available in different purities?
Seeing the Bigger Picture in Chemical Purity
I’ve seen more lab benches covered in reagent bottles than most people see coffee mugs in a break room. Flipping through catalogs, it’s clear that chemical purity isn’t for show—especially not with compounds like 3,5-Dimethoxycinnamic Acid. This isn’t obscure chemistry. Some use it in pharmaceuticals, others in plant studies or material science. What catches my eye is that most suppliers organize their listings by purity, and folks have real reasons for caring about those little numbers next to “% Pure.”
Why Purity Isn’t an Afterthought
Step into any lab or production floor, and you’ll hear stories of failures that trace back to dirty chemicals. Subtle impurities can jam up a reaction, ruin an assay, or skew a promising result. I remember hunting down a contaminant in a supposedly “high grade” reagent that clouded our whole experiment. So, when scanning the options for 3,5-Dimethoxycinnamic Acid, I look for those three main types: technical, laboratory, and high purity grades.
Suppliers often grade this acid by its intended use. Technical grade works for non-critical applications, where traces of byproducts or leftover reactants don’t throw anyone off. Laboratories demand more, so purity climbs above 98%, and QC labs often spring for even finer. If an active pharmaceutical ingredient needs this compound, then the story shifts—all eyes go to the certificate of analysis. That means checking not just the percentage, but the absence of specific trace elements, heavy metals, or organic residues.
How Purity Shapes Real-World Outcomes
Ever tried scaling a reaction with a low-purity batch? It’s like baking a cake with flour mixed with sand. Some research teams catch a lucky break, but most find the reproducibility crashes. I’ve seen it lead to wasted materials, broken timelines, and unnecessary troubleshooting. Pharmaceutical production can’t afford these gambles. Even a fraction of a percent off in purity sometimes means that a drug batch won’t pass regulatory checks.
Different grades show up because one-size-fits-all never fits science. Certain syntheses get by with fewer purity constraints. I’ve heard from lab friends working on early-phase material discovery; they use technical grade to save money and time. Shift to preclinical or clinical testing, and no one touches a sample below 99%. These settings call for hplc, gc, and nmr data—no guesses or gray areas.
Chasing Solutions and Raising the Standard
It’s not enough to choose a shiny high-purity chemical off the shelf. I suggest labs demand transparency: ask for sourcing, batch numbers, and traceability. Some suppliers cut corners on documentation, so I encourage working with those who back up purity with real data, full spectra, and impurity profiles. Comparing costs sometimes feels tempting, especially on limited grant funds, but sacrificing reliability leads to bigger problems.
For those wrestling with purity bottlenecks, collaboration with quality vendors pays off. Look for those that deliver consistent results over time—not just a headline number. Push back if the certificate lacks detail. Greater awareness and higher demands from buyers indirectly improve the standards across the industry. The days of “just good enough” reagents are giving way to a more careful approach, because lost time and flawed outcomes cost much more down the line.
What safety precautions should be taken when handling 3,5-Dimethoxycinnamic Acid?
Why Safety Matters with 3,5-Dimethoxycinnamic Acid
I spent years sharing lab benches with folks who saw no difference between a harmless salt and a fine organic powder. That attitude racks up casualties pretty quickly. Even chemicals that sound fancy—like 3,5-Dimethoxycinnamic Acid—deserve respect for what they can do to skin or lungs under the wrong circumstances. This compound finds its way into research, sometimes crop science, and even the materials side of pharmaceuticals. Anytime powders fly or solvents stir, there’s always a risk to people and the workspace.
Protect Your Skin and Lungs from Exposure
This compound doesn’t leap out of the bottle, but trust me: fine crystals get onto hands, faces, sleeves, and everything you touch—especially if you keep lids off. I learned fast that regular gloves don’t always cut it. You want gloves of nitrile or neoprene, never powdered latex. A good lab coat saves shirts from stains and skin from long-lasting irritation. Eye protection ends up a daily savior; chemical splashes are rare, but they happen before you even blink.
Breathing in small particles brings bigger problems. Any compound with aromatic rings deserves a fume hood. Standard practice in my lab called for weighing inside an enclosure so dust didn’t hang in the air. Face masks or respirators come out when ventilation isn’t enough, and every decent lab keeps these at the ready.
Dealing with Spills and Clean-Up
Spills breed chaos. Cluttered benches multiply the risk. I learned this as a rookie, knocking over a sample that painted the bench and the floor. Grab a spill kit, make sure it has absorbent powder, gloves, and a disposable scraper. Sweep up slowly, starting from the outside. Avoid dry sweeping that creates more dust. Wipe down the area with damp towels. Double-bag everything before tossing it in the chemical waste bin.
Storage Making Life Simpler
Sealed containers win every time. Heat, sunlight, and open air break down sensitive compounds in ways that endanger future work or cause unexpected reactions. I always kept such chemicals labeled clearly, noting opening date and any changes like color or texture. Don’t let acids sit on bottom shelves; even small bottles deserve space at chest height, away from incompatible reactants or food and drink.
Training and Common Sense Always Win
No one does their best work alone, especially with specialty chemicals. Experienced researchers and students benefit from walking through a safety review, not just signing off on a checklist. Good teamwork means alerting others when something seems off in storage or behavior. I never regretted speaking up when a container looked odd or when someone ignored the fume hood rule. Mistakes get fixed, lessons get learned, and dangerous habits fade with honest conversations.
Building a Respectful Lab Culture
People trust labs that protect them, and that starts with simple rules everyone follows. Good habits, real communication, and access to the right gear turn odd-sounding acids into just another part of the day. That’s how research stays productive, safe, and welcoming to newcomers eager to learn.
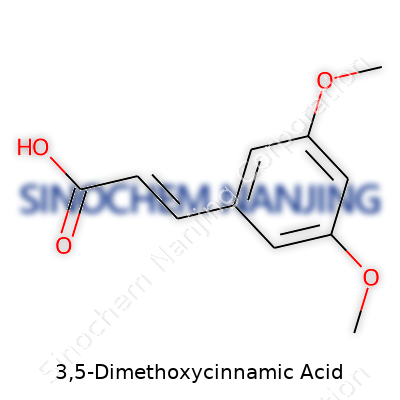
| Names | |
| Preferred IUPAC name | (E)-3-(3,5-dimethoxyphenyl)prop-2-enoic acid |
| Other names |
3,5-Dimethoxy-cinnamic acid 3,5-Dimethoxyphenylacrylic acid Cinnamic acid, 3,5-dimethoxy- 3,5-Dimethoxycinnamate |
| Pronunciation | /ˈθriːˌfaɪv daɪˌmiːˈθɒksi sɪˈnæmɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | [4499-55-0] |
| 3D model (JSmol) | `3D structure; JSmol='C1=CC(=CC(=C1OC)OC)C=CC(=O)O'` |
| Beilstein Reference | 1207586 |
| ChEBI | CHEBI:24866 |
| ChEMBL | CHEMBL1200873 |
| ChemSpider | 14638273 |
| DrugBank | DB08309 |
| ECHA InfoCard | 17e3bd0c-6f4e-40cc-bf8d-8944147e75f4 |
| EC Number | 228-236-7 |
| Gmelin Reference | 82133 |
| KEGG | C09662 |
| MeSH | D000069412 |
| PubChem CID | 154909 |
| RTECS number | GV2808000 |
| UNII | L4RQZ6TA9F |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | urn:lsid:epa.gov:CompToxDashboard:DTXSID3068818 |
| Properties | |
| Chemical formula | C11H12O4 |
| Molar mass | 208.22 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.239 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 1.90 |
| Vapor pressure | 0.0000112 mmHg at 25°C |
| Acidity (pKa) | 4.06 |
| Basicity (pKb) | 13.25 |
| Magnetic susceptibility (χ) | -60.9 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5660 |
| Viscosity | Viscous solid |
| Dipole moment | 3.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 312.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -407.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1346.8 kJ/mol |
| Hazards | |
| Main hazards | Causes skin and eye irritation. |
| GHS labelling | GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P362+P364, P501 |
| Flash point | 162.1 °C |
| NIOSH | RN1006 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 mg |
| Related compounds | |
| Related compounds |
Cinnamic acid Ferulic acid 3,4-Dimethoxycinnamic acid 4-Methoxycinnamic acid 3,5-Dimethoxybenzoic acid |

