3,5-Dichloroaniline: Yesterday’s Chemistry and Tomorrow’s Questions
History Carried Forward
3,5-Dichloroaniline has roots that stretch back to the early 20th century, showing up in the chemical records just as new industries began to trust science for color, medicine, and agriculture. My own curiosity about these compounds really began during later college years, stumbling on case studies of anilines in legacy dye plants. Anilines turned textiles vibrant and played a controversial role in the evolution of herbicides, and 3,5-Dichloroaniline stepped in, thanks to its nifty chlorine substitutions that drew interest for selective reactivity. Those old patent archives and chemical catalogs tell a story of discovery, incremental tweaks, and the race to either scale up cost-effective routes or skirt patent claims with molecular creativity.
The Compound at Hand
This molecule brings two chlorine atoms onto the benzene ring—a picture that organic chemists spot quickly. With these modifications, it stands out from its relatives. The powder or crystalline form shows a yellow-white color, and if you ever get near a small sample (as I did during graduate research), there’s a sharp, pungent odor. Unlike plain aniline, those extra chlorines make it handle less smoothly—insoluble in water, easily soluble in common organics. Handling it really woke me up to just how much a tiny change in atom placement can change everything: stability, solubility, and application fit.
Technical Details and Labels
On the technical side, you’ll see it sold with clear hazard tags: irritant, toxic upon higher exposures, and not something to mishandle. Labels in the lab demand respect. Melting points sit around 70-75 degrees Celsius. The chemical community often codes it under CAS No. 626-43-7. Synonym lists pop up across research papers: 3,5-Dichlorobenzenamine, m-Dichloroaniline, and others that reflect the naming conventions of different eras and countries.
Getting It Made: The Preparation Pathways
Synthesizing 3,5-Dichloroaniline can start from 3,5-dichloronitrobenzene, which gets reduced using iron filings and hydrochloric acid, or other modern hydrogenation roads. Back in my early research days watching seasoned chemists run these reductions, the takeaway was always about tradeoffs: iron acid reduces cost but demands careful waste management, catalysts give cleaner results but raise budgets. Environmental compliance also shifted the balance, especially as stricter rules narrowed what we did with leftover iron sludge and acidic brines.
Tinkering with Structure: Reactions and Modifications
This compound isn’t just a static building block—its amine group and those chlorines invite further chemistry. Researchers have extended its legacy with acylation, diazotization, or even as a launchpad for making sulfonamides or phenylurea herbicides. In industry meetings and conferences, I often witnessed sharp debates about the value of modifying deployment at the ring versus the functional group, with priorities driven by target performance, not theoretical elegance.
What’s in a Name?
Across suppliers, you get different names thrown around, but the critical identifiers come from chemical abstracts and IUPAC. Overzealous renaming sometimes stumped purchasing agents in my experience—especially overseas—so double-checking against a structure or CAS number always saved time.
Staying Safe with Dichloroanilines
Exposure stories never stay theoretical for long. While handling this in the lab, I’ve seen colleagues experience mild irritations after accidental skin contact. Standard practice relies on gloves and fume hoods, with strict disposal routes for any contaminated material. The tougher question is worker safety across the industrial chain, especially where personal protective equipment gets less priority. Globally, regulatory agencies like OSHA and REACH have tightened both exposure limits and disposal standards, but enforcement varies, especially outside well-funded labs.
Where It Ends Up: Applications
In the real-world market, 3,5-Dichloroaniline’s reputation grew with agricultural science. The molecule gets transformed into herbicides or fungicides, and pigment industries love it as a precursor for yellow dyes and specialty colorants. While I never worked in a colorant plant, major advances in fastness and shade customization owe a debt to dichloro derivatives like this one. Analytical chemists also deploy it in developing sensors, but that always felt like a niche compared to field-scale agriculture or mass-market pigments.
On the Research Front
Academic attention remains significant. Studies explore reaction mechanisms or push into greener, milder synthesis. Scientists experiment with enzyme catalysis to dodge toxic byproducts, and recycling spent catalysts has come up in several collaborations I joined. The gap between university blue-sky thinking and industry constraints is obvious, but grant money and the right partnership can turn small-scale research into the next benchmark process. Still, a lot of promising routes get shelved if they can’t make economic sense by the metric ton.
What About Toxicity?
Long-term concern never went far from dichloroanilines. Toxicology labs find that acute exposure can damage kidneys and liver, hitting rodents the hardest in lab tests. Regulatory data points to carcinogenicity suspicions, although strong evidence in humans remains debated. Legacy contamination at old manufacturing sites sparks real-world anxiety for communities, not just theory. Getting the word out and building trust with affected residents takes more than regulatory filings—it means open data, straight talk, and, in my experience, a willingness to talk face-to-face at public forums.
Looking Beyond
The future of 3,5-Dichloroaniline splits between stricter controls and smarter chemistry. Digitization of safety data helps spot risks sooner, but moving toward safer alternatives or greener synthesis matters more for real-world impact. There’s room for biotech and catalytic breakthroughs—smaller environmental footprints, lower health costs, and products that do the job with less collateral damage. As the industry adapts, the human story underneath still asks for vigilance, open science, and honest exchange between policymakers, companies, and the public. If chemistry’s history teaches anything, it’s that outcomes improve most when technical brilliance pairs with humility about what we still don’t know.
What is 3,5-Dichloroaniline used for?
Climbing Into the Story Behind this Chemical
Ask anyone who’s spent a little time poking around the world of chemistry and they’ll recognize certain chemicals not just by their technical names, but by the roles they play in real life. 3,5-Dichloroaniline stands out as one of those. Labs, plants, and farmers have made use of it for decades, but its story stretches far beyond technical reports.
A Backbone in Pesticide Production
Walk into a field in the Midwest with wheat, or down in southern China where rice grains wave in the wind, and one thing ties many farms together: weed control. 3,5-Dichloroaniline forms a big part of this process. It serves as a starting material for manufacturing the herbicide diuron, a chemical that’s helped keep farm fields cleaner for nearly 70 years. Diuron knocks out pesky weeds with some real efficiency, so farmers saw their yields hold steady and the fight against unwanted plants got a little easier. By volume, much of the world’s 3,5-Dichloroaniline finds its way into these chains of production.
Dye Factories Count on It
I remember spending years researching industrial dyes, stumbling across 3,5-Dichloroaniline again and again. It isn’t just about coloring sweaters and jeans. This chemical helps create pigments for plastics, inks, and paints. So, every time you spot a vibrant shade in an old book or on a playground slide, this compound probably sat somewhere along the line. The compound gives certain hues their sticking power, brightness, and even staying power under tough sun.
Other Places the Chemical Ends Up
Factories put it to work as a building block in manufacturing drugs, specialty chemicals, and even in substances made for treating water. The pharmaceutical world has tinkered with it in the past to produce medicines and diagnostic agents. While it’s not the star player in most pharmacy cabinets today, its involvement in synthesis steps can’t be denied.
Concerns and Risks Hanging Over Its Use
Living in the era of persistent chemicals, I learned that not every miracle compound comes without baggage. 3,5-Dichloroaniline breaks down slowly in the environment. Runoff from agricultural land and waste from dye factories can dump it into streams and rivers. Studies from the EPA warn of toxicity to aquatic life and nervous system effects in animals. Workers exposed at plants handling the chemical have reported skin irritation and breathing problems. Public health agencies keep an eye on it because people don’t want more persistent organic pollutants drifting into their daily water.
Balancing Need and Responsibility
Companies have options. Using closed manufacturing systems prevents leaks. Stronger personal protective equipment shields workers. Wastewater treatment plants can add advanced filtration to snag contaminated runoff. Farmers can measure out pesticides with more precision to reduce spread. At my old research lab, we set up workshops to track chemical inventories and monitor air quality, making everyone part of the safety solution. Pressure from communities and environmental watchdogs keeps the industry alert.
At the end of the day, chemicals like 3,5-Dichloroaniline don’t exist in isolation. Their legacy rests in hands—farmers, factory workers, regulators—who decide how to keep food growing and colors vivid, without ignoring those who live next to factories, streams, or fields.
What are the safety precautions when handling 3,5-Dichloroaniline?
The Realities of Working with 3,5-Dichloroaniline
I’ve seen plenty of situations where a few routine shortcuts with chemicals turn into a real mess, fast. 3,5-Dichloroaniline comes with a set of hazards most can’t smell or see. It floats around as a white to light brown solid, and a couple whiffs of dust or accidental splashes cause much more than a headache. Once, during a plant inspection, I watched a coworker spill some powder while measuring, and soon after started coughing—with his eyes getting redder by the minute. It brought home for me how quickly things get out of hand.
The Real Dangers Behind the Name
There’s no sugarcoating what happens if you slip up with 3,5-Dichloroaniline. Skin rashes, eye irritation, and trouble breathing turn up after only brief exposure. The compound doesn’t take long to soak through gloves if you’re not using the right kind, making hand protection a serious business. The bigger concern usually sits in the lungs—fine dust or vapors quietly settle, hitting you harder the longer you go without a respirator.
Building Habits that Make a Difference
I keep coming back to basic habits: don’t work alone and take the time to check protective gear before opening a container. No one wants to spend time tracking labels or hunting for the right gloves, but ignoring the rules turns problems from minor to major. The best labs and shops train every worker to know the difference between rubber gloves and nitrile, and to double-check for leaks before each use. Chemical-resistant aprons, safety goggles, and the habit of changing gloves before touching your face or eating—these steps save more than just skin.
Good Ventilation, Good Health
A strong exhaust fan seems simple, but that kind of engineering control stops dust clouds in their tracks. If I had to name one upgrade that always pays off, it’s adding or improving local fume hoods. During cleanup or mixing, opening a window won’t cut it. Only a real fume hood, checked and maintained each month, keeps the room from getting thick with invisible danger. Routine checks and logged airflow measurements help spot trouble before it snowballs.
What Emergency Plans Mean in Practice
At a place where 3,5-Dichloroaniline gets used, chemical showers and eyewash stations need to work, not just look ready. Walk through an average shop, and you’ll see eye-wash stations blocked by boxes or chemical drums. In an emergency, time gets measured in seconds. Every worker should know the layout—ideally, through regular drills—not just by memory from orientation. After a spill or splash, quick decisions matter most: stripping off soiled clothing, rinsing longer than seems necessary, seeking fresh air even if you "just feel a little off." The difference between an incident and a trip to the ER often lies in how quickly these responses come together.
Tackling Waste Management
Unlabeled waste bottles set up confusion, and guessing which bin to use can lead to bigger problems during disposal. Having clear steps for labeling, storing, and removing 3,5-Dichloroaniline waste keeps both garbage haulers and coworkers safe. My experience says, encourage reporting of any unclear containers—or better yet, build a buddy system to double-check every drop of unused material. Map out emergency contacts, and keep up-to-date safety data sheets posted where they’re visible, not buried in a drawer.
Why Respect Becomes the Best Safety Tool
No one’s immune from mistakes. Even smart, experienced folks skip gear or rush through cleanup. But respect for chemicals like 3,5-Dichloroaniline—earned from seeing what happens when things go wrong— pushes everyone to slow down and do things right. If you work with it or know someone who does, a little extra effort today means everyone gets to go home healthy.
What is the molecular formula of 3,5-Dichloroaniline?
Digging Deeper Into 3,5-Dichloroaniline
Walk into any chemistry lab, and you’ll notice a particular attention to details like molecular formulas. Seemingly simple numbers and letters pack a real punch—especially for compounds used in research and industry, such as 3,5-Dichloroaniline. This compound carries the molecular formula C6H5Cl2N. A clear understanding of this formula doesn’t just help on paper; it’s integral for anyone handling it, whether in pharmaceuticals, agriculture, or academic research.
Why This Formula Deserves a Closer Look
Anyone who’s ever spent time working with chemicals knows small changes can transform a harmless substance into something risky or powerfully effective. With 3,5-Dichloroaniline, each part of the formula tells a piece of its story. The benzene ring (C6H5) serves as its foundation, while two chlorine atoms (Cl2) rest at the 3 and 5 positions. Nitrogen (N) seals the identity as an aniline derivative. Their combination shapes how the molecule reacts, how it’s handled, and even how it’s regulated.
The value here comes from more than just structural basics. Chemists depend on such information to predict solubility, toxicity, and reactivity. The presence of two chlorine atoms impacts not only biological activity but also environmental persistence. Years spent in the lab taught me that every project risks derailing over a mistaken formula or an overlooked element. At this level, accuracy protects people, projects, and reputations.
Building Trust Through Clarity and Safety
Clarity starts with sound education. Strong knowledge of formulas like C6H5Cl2N forms the backbone of safe lab practices. Over the years, I’ve seen beginner chemists reach for reference books or pull out their phones to double-check formulas before moving forward. Mistakes have a cost. Chlorinated anilines sometimes display unexpected reactivity, so even a minor lapse can affect safety or the outcome of an entire synthesis.
Chemical manufacturers also bear significant responsibility. Standardized product data sheets and correct labeling support everyone down the line—from warehouse workers to academic researchers. Consistent, transparent information helps the entire supply chain dodge unnecessary hazards. Letting a manufacturing mistake slip through to the user would create a domino effect of safety problems.
Moving Toward Stronger Practices
Accurate formulas save time and potential headaches, but there’s room to tighten up practices further. Teaching should focus on the “why” behind every structure, not just memorization. Open-access resources and better quality digital databases keep errors from creeping into the lab. I’ve found that regular hands-on review sessions in the classroom, especially those that focus on real-world case studies, drive the point home better than rote lectures.
For more advanced users, spectroscopic analysis and other technologies offer a direct way to verify compound identity. Refusing to take formulas at face value encourages critical thinking—a priceless habit in both academia and industry.
Final Thoughts
Understanding the molecular formula of a compound like 3,5-Dichloroaniline drives safer and more reliable chemistry. C6H5Cl2N is more than a string of characters; it’s a key to working responsibly in science and industry. By prioritizing accuracy and transparency, everyone using or studying this compound benefits—from entry-level lab techs to seasoned researchers.
Is 3,5-Dichloroaniline hazardous to human health?
What is 3,5-Dichloroaniline?
3,5-Dichloroaniline pops up in a lot of chemical manufacturing. Factories use it to make dyes, pesticides, and drugs. It's nothing fancy—just an industrial chemical with two chlorine atoms hooked to an aniline ring. Because of its widespread industrial use, it has ended up in places it probably shouldn't: workplaces, wastewater, even some soil and water sources.
Health Risks Linked to Exposure
Once you dig into the toxicology, it gets clear—3,5-Dichloroaniline doesn’t belong near people who aren’t wearing gloves and respirators. The International Agency for Research on Cancer lists related anilines as potential carcinogens. If you run across this compound in a factory, inhaling dust or vapors opens the door to headache, nausea, skin irritation, and higher risk of liver or kidney problems. Workers exposed over months can develop hemolytic anemia, where red blood cells break down faster than the body can make them.
Animal studies ring alarm bells. Rats fed 3,5-Dichloroaniline developed organ damage and cancerous changes in certain tissues. These findings shouldn’t be dismissed. What happens in the lab doesn’t always line up with what happens to real people, but most folks agree—regular exposure is playing with fire.
Short-term exposure doesn’t get a pass either. If you splash this stuff on your skin, it soaks in quickly. Burns, rashes, and blisters often follow. Breathing in the compound often leads to nose and throat irritation, asthma-like symptoms, and feeling lightheaded.
Why Ordinary People Should Care
Industrial chemical spills and poor waste management have already spread anilines far beyond the factory fences. Environmental studies find them in streams and soil samples near old chemical plants. Fish and wildlife can suffer, and human health—especially children’s health—hangs in the balance when these contaminants move up the food chain.
If you have a vegetable garden near an old industrial site, or spend time fishing in polluted rivers, those chemicals might be coming home with you. Municipal water treatment does a decent job but doesn’t always catch everything. I’ve seen news stories where small towns have wrestled with lingering old chemical waste in their water. The lesson is simple. Trust matters, but double-checking local water quality reports or contacting environmental agencies is just common sense if you’re anywhere near a chemical plant.
Taking Real Steps Toward Safety
Factories can reduce risks by using closed systems, ventilation, and regular air monitoring. Workers should have up-to-date protective gear, no shortcuts. Government standards limit workplace exposure, but oversight can always improve.
Most importantly, safe disposal of industrial waste needs strict enforcement. Accidental spills wipe out years of clean-up in just days. Managers who look the other way or skip maintenance do everyone a disservice. Supporting strict environmental standards and tighter reporting requirements should be a community responsibility, not just a burden for inspectors.
Doctors need to know which symptoms point to chemical exposure. When I talk to healthcare professionals, they admit—cases of rare anemia or odd rashes sometimes slip through the cracks unless someone asks about workplace hazards.
Building Informed Communities
It boils down to information and action. Workers should know the risks, neighbors should be able to push for transparency, and businesses must put this knowledge to use in every tank and valve. If you suspect contamination nearby, use the tools available—local environmental hotlines, water tests, and if needed, legal resources.
How should 3,5-Dichloroaniline be stored?
Seeing Chemical Hazards Up Close
I remember stepping into a small industrial storeroom for the first time. Chemical smells hung heavy in the air, and old dented barrels lined the walls. You don't forget the gritty realization that careless storage can mean real danger. 3,5-Dichloroaniline might look harmless as a white to light-brown solid, but that idea vanishes fast when you learn how little it takes for toxic fumes to spread or skin to burn.
The Real Risk Behind the Name
This compound plays a role in making dyes, pharmaceuticals, and agrochemicals. But its byproducts and dust can cause real trouble if inhaled or if they linger on surfaces where people work. Research over the years links exposure to acute irritation of the eyes, skin, and lungs. Lab studies have shown repeated or long-term contact raises the risk of organ damage and may cause severe allergic reactions. That’s not a story anybody wants to live out.
Temperature and Ventilation Matter More Than You Think
Picture a concrete storeroom during July with no airflow—temperature rises, containers sweat, and vapor starts to build. 3,5-Dichloroaniline does not catch fire easily, but its vapors do spread more when the air stagnates. Storing this chemical in a dry, cool area below 25°C slows down its breakdown and helps prevent dangerous fume build-up. Good airflow—think well-designed exhaust fans, not just open windows—keeps problems from stacking up in neglected corners.
Spill Containment and Shelf Sense
Chemical barrels balancing on weak wooden shelves led to one of the worst spills I’ve seen. Those mistakes get expensive and dangerous fast. Putting 3,5-Dichloroaniline in rigid, sealed containers and storing them on sturdy, chemically resistant shelving stops accidents before they start. Floor-level secondary containment, like plastic spill trays lined with absorbent pads, catches drips before they spread across concrete. Mixing up the storage of strong acids, oxidizers or flammable solids alongside this compound often makes things riskier, so keep them separate.
Label Everything—Precision Saves Lives
I worked with a supervisor who could spot a missing or faded label at fifty paces. Clear, permanent labeling saves time and, sometimes, lives. Mark each container with the chemical name, hazard symbols, date received, and the contact for the person responsible. Regular checks help catch mistakes before they become problems. Updates from workplace chemical safety audits are gold—ignore them, and sooner or later, someone pays the price.
PPE and Easy Access to Emergency Gear
Think about grabbing a bottle and realizing too late that gloves or goggles sit in another room. Working with 3,5-Dichloroaniline needs gear on hand: nitrile gloves, goggles, and chemical-resistant clothing. Keep eye wash stations and safety showers nearby—every second counts during an accident. Workers deserve regular training, not just a one-time orientation. People remember drills more than lectures.
Guidelines Offer Structure, But People Make Safety Happen
The National Institute for Occupational Safety and Health (NIOSH) and OSHA both lay out clear expectations for toxic chemical storage. Real safety springs from workers who do not cut corners, who look out for each other and call out shortcuts before they create harm. Kicking the can down the road as “someone else’s problem” solves nothing. I've lost count of stories about good people hurt by routine neglect. These chemicals reward respect, not carelessness.
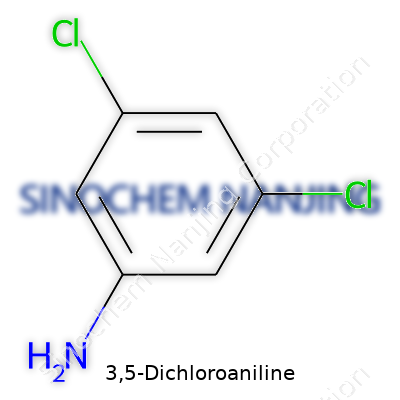
| Names | |
| Preferred IUPAC name | 3,5-dichlorobenzenamine |
| Other names |
3,5-Dichlorobenzenamine m-Dichloroaniline 3,5-Dichlorophenylamine |
| Pronunciation | /ˌθriːˌfaɪv.daɪˌklɔːroʊ.əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 626-43-7 |
| Beilstein Reference | 1209242 |
| ChEBI | CHEBI:34644 |
| ChEMBL | CHEMBL18978 |
| ChemSpider | 14221 |
| DrugBank | DB14089 |
| ECHA InfoCard | 100.008.204 |
| EC Number | 202-094-6 |
| Gmelin Reference | 5958 |
| KEGG | C06582 |
| MeSH | D003658 |
| PubChem CID | 6927 |
| RTECS number | KR1400000 |
| UNII | UNII2G9TA7E16Y |
| UN number | 2811 |
| Properties | |
| Chemical formula | C6H5Cl2N |
| Molar mass | 162.04 g/mol |
| Appearance | White to gray crystalline solid |
| Odor | amine-like |
| Density | 1.44 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.57 |
| Vapor pressure | 0.002 mmHg (25°C) |
| Acidity (pKa) | 3.55 |
| Basicity (pKb) | pKb = 9.09 |
| Magnetic susceptibility (χ) | -69.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.618 |
| Viscosity | 1.32 cP (25°C) |
| Dipole moment | 3.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 142.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -73.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2945 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, in contact with skin or if inhaled. Causes serious eye irritation. Suspected of causing cancer. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H317, H319, H332, H410 |
| Precautionary statements | P261, P280, P302+P352, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-2-1 |
| Flash point | 150 °C |
| Autoignition temperature | 470°C |
| Lethal dose or concentration | LD50 (oral, rat): 650 mg/kg |
| LD50 (median dose) | LD50 (median dose): 650 mg/kg (oral, rat) |
| NIOSH | SN9800000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Aniline 2,4-Dichloroaniline 3,4-Dichloroaniline 3,5-Dichlorobenzoic acid 3,5-Dichlorotoluene |

