Digging Deeper into 3,5-Dibromo-4-Hydroxybenzonitrile: More Than a Laboratory Curiosity
Historical Development
For anyone who’s walked the meandering road of organic chemistry, the name 3,5-Dibromo-4-Hydroxybenzonitrile might conjure a sinking feeling of effort. This compound, a product of careful halogenation and functional group management, has roots in the gradual expansion of synthetic methods for aromatic bromides and phenolic derivatives. Researchers in the last century looked for ways to push aromatic chemistry—they focused on activating and deactivating rings, managing substitutions, and optimizing reaction conditions. Somewhere in that quest, 3,5-Dibromo-4-Hydroxybenzonitrile stepped out from the shadows, an artifact of chemists’ growing control over regioselectivity.
Product Overview
3,5-Dibromo-4-Hydroxybenzonitrile isn’t found sprawled across store shelves, but it lives in countless reaction flasks, acting as a scaffold for more complex molecules. The molecule joins the ranks of halogenated phenols, a set of substances critical to both medicinal chemistry and materials science. Its elegance lies in its symmetry and functional distribution—a combination of bromines, a hydroxyl group perfectly placed, and a cyano group anchoring versatility. Behind its clinical name stands a bridge between basic research and practical innovation, proof that small, tailored molecules shape larger breakthroughs.
Physical & Chemical Properties
This white crystalline solid, wielding a faint chemical odor, doesn’t just catch the eye with its clean edges but draws attention during melting point analysis—often hovering in the expected range for simple aromatic bromides. Its moderate solubility in common organic solvents such as ethanol or acetone makes it manageable across synthesis procedures, even where water’s not a helpful companion. The molecule’s bromines confer weight; they push density higher than unsubstituted relatives. Those same bromine atoms bring reactivity, enabling routes for cross-coupling or further modification. Researchers can count on the stability of the nitrile group, which delivers resistance to many reagents but sits ready for transformation when demanded by a synthesis plan.
Technical Specifications & Labeling
Chemists expect precision and documentation when handling substances like this. Labeling usually emphasizes both hazards and handling instructions. If you frequent chemical stockrooms, you’ve likely seen clear hazard pictograms warning of potential health risks, sometimes paired with specifications like assay percentage, melting point, and storage advice. For 3,5-Dibromo-4-Hydroxybenzonitrile, specifications often note purity levels, appearance, and sometimes moisture content, since trace water can disrupt specific reactions. Test methods—for instance, NMR or HPLC—get a mention in most credible supply sheets, though bench chemists know these are only one thread in the much larger weave of chemical reliability.
Preparation Method
Aromatic nitriles like this one rarely appear without intention. Synthesis often begins with a suitably substituted hydroxybenzonitrile, then calls for careful bromination. The selectivity for the 3,5-positions emerges from the electron-withdrawing and activating tug-of-war between the hydroxy and cyano groups. N-bromosuccinimide (NBS) and elemental bromine remain go-to reagents, each with benefits and limitations. Good ventilation, pragmatic use of cold traps, and skilled timing matter a great deal—runaway bromination can turn progress into a setback. Purifying the resulting solid needs patience, with recrystallization playing a starring role for those aiming at high purity without structural compromise.
Chemical Reactions & Modifications
This molecule invites transformation. The bromines speak loudest, letting the compound join Suzuki or Heck couplings with palladium catalysts—platforms for crafting biaryl scaffolds in pharmaceutical or material research. The hydroxy group opens the door to etherification or acylation; it’s been a useful handle for modifying polarity or joining spacers during probe design. The nitrile brings scope for conversions too—hydrolysis, reduction, or even cyclization if driven by the right creative chemist. Each reactive node on this aromatic structure enables a new branch of exploration, supporting both method development and urgent application work.
Synonyms & Product Names
No chemist enjoys confusion in nomenclature, but some is unavoidable. 3,5-Dibromo-4-Hydroxybenzonitrile sometimes answers to alternates like 2,6-Dibromo-4-cyanophenol—underscoring the IUPAC/locant dance that happens in labs across continents. You also meet non-systematic lists in catalogues, ranging from basic descriptions (dibromohydroxybenzonitrile) to numeric abbreviations. Recognition across languages and industries matters for safety, sourcing, and sharing results, so experienced chemists triple-check before placing an order or reporting discoveries.
Safety & Operational Standards
Lab veterans never downplay safety with aromatic halides. Brominated species are no exception, often catching eyes for their moderate toxicity and unpredictable local irritation. Exposure can cause issues ranging from mild respiratory discomfort to more serious effects if absorbed or inhaled recklessly. Proper PPE—lab coats, gloves, goggles—makes a difference, and so does working inside functioning fume hoods. Containers demand labeling, and storage calls for exclusion from direct sunlight and incompatibles like strong bases or oxidizers. Disposal, guided by local regulations, must not cut corners, since halogenated organics often face extra scrutiny in waste streams. For those unfamiliar, understanding hazards before handling trumps all urges to rush a synthesis.
Application Area
Despite its unassuming name, this compound has found welcome into fields ranging from pharmaceutical research to materials science. In drug discovery, halogenated aromatics pave ways towards potent bioactives—either as direct scaffolds or synthetic intermediates. Researchers have leveraged 3,5-Dibromo-4-Hydroxybenzonitrile for diversification campaigns where rapid library expansion helps spot hits that bind to tricky biological targets. Outside pharma, the compound anchors polymer development and has attracted attention for dye and pigment research, where electron-rich and electron-poor groups help tune optical or conductive properties. These applications put it on the radar of both academic innovators and industrial formulation teams chasing next-generation products.
Research & Development
The molecule’s appeal in R&D boils down to its toolkit: it comes ready to explore new reactions and build new frameworks. Chemists chase pathways that join small, reactive molecules into larger, functionally rich architectures. The dibromo arrangement means researchers can try multiple coupling strategies, each direction offering unique access to complexity. Studies have used it to probe protein binding in biophysical chemistry and to create test materials meant to showcase conductivity or stability. Regular advances in organometallic catalysis have kept 3,5-Dibromo-4-Hydroxybenzonitrile in the loop for testing new palladium or nickel systems, sometimes serving as benchmarks for selectivity and efficiency.
Toxicity Research
Any time a compound holds promise in the lab, the environment and organismal safety question lurks close behind. Regulatory science often lags discovery, leaving researchers to probe toxicity with a mix of animal models and cell studies. Halogenated aromatics present risks—bromine atoms, in particular, get flagged for potential environmental persistence. Data on 3,5-Dibromo-4-Hydroxybenzonitrile remains patchy but suggests caution. Chronic or repeated skin contact shouldn’t fly under the radar, and inhalation of dust or vapors could stress sensitive bronchial passages. Wastewater studies warn against dumping—persistent organic pollutants have a knack for disrupting aquatic ecosystems at low concentrations. Conscientious labs commit to understanding toxicological impacts, not just to tick compliance boxes, but to protect colleagues and communities that live downstream from research.
Future Prospects
The story does not end on the page of a safety data sheet or in a single successful synthesis. Advancing synthetic methods keeps generating interest in molecules like 3,5-Dibromo-4-Hydroxybenzonitrile, especially as the call for sustainability, efficiency, and creativity grows louder in chemical industries. Its structure lends itself to modular approaches, and progress on greener halogenation—perhaps using less hazardous brominating agents or milder conditions—could make it even more viable in large-scale synthesis or automated flow chemistry setups. Regulation may push for alternative, less toxic halides, but for now, the compound stands as a key tool in the arsenal for forging complexity from simplicity. Those seeking new pharmaceuticals, advanced materials, or rapid screening platforms will likely keep this molecule close at hand, adapting its use as technology, safety standards, and scientific imagination keep moving forward.
What are the common applications of 3,5-Dibromo-4-Hydroxybenzonitrile?
Chemistry Shapes the Real World
3,5-Dibromo-4-hydroxybenzonitrile might sound like just another mouthful from a chemistry class, but those who work in environmental or pharma labs know it’s more than a string of syllables. I’ve spent afternoons looking into chemical structures, and you start to recognize which compounds show up often for a reason. This one pops up a lot, especially where people need targeted action and a predictable outcome.
Protection Beyond the Laboratory
Out in the real world, 3,5-Dibromo-4-hydroxybenzonitrile finds itself mixed into everyday solutions—especially as a biocide. If you’ve turned on a tap or dipped a toe in a swimming pool this year, some of the water might carry its touch, keeping harmful fungi and bacteria in check. Its action helps stop slime and algal growth, making it a quiet hero in maintaining clean water.
I grew up helping my uncle clean pools. We tested and treated water with more care than most people realize. Chemicals like this one punch above their weight, keeping recreational and drinking water safe. You’d be surprised how many agricultural irrigation lines rely on it for drip system hygiene.
Taking on Mold Where It Hides
This compound also defends wood, paint, and wallboard. Mold and bacteria can ruin building materials and trigger health problems. Rolling walls with paint or stain containing active agents like 3,5-Dibromo-4-hydroxybenzonitrile helps prevent disastrous outcomes—no sudden black spots on the ceiling or headaches from hidden spores. Anyone in construction or property management deals with the bills left behind when mildew takes over. The preventive value stacks up fast, both for homeowners and big real estate names.
Supporting Modern Medicine’s Building Blocks
Pharma research sometimes leans on this compound as a stepping stone. Medicinal chemists explore new drug candidates, and the brominated ring of this compound helps in tailoring molecules with precise properties. Labs synthesize derivatives for all sorts of clinical trials. If you’ve ever followed the journey of a potential medication from whiteboard to piloted use, you know every reagent matters. Safety and effectiveness often start with clean ingredients and smart selections at the very start of the process.
Wider Environmental Footprints
There’s always the challenge of keeping such chemicals out of trouble. Some biocides linger longer in soil and water than you’d guess. Research from environmental groups points to a need for more downstream monitoring. Efficient breakdown, smart application, and education all play a role in reducing unwanted residues. Regulatory agencies look for detectable traces in rivers, backing up real-time monitoring programs. Responsible use, strict tracking, and ongoing studies make a difference between protection and pollution.
Building Better Habits in Industry
Getting regular people involved in the conversation helps. Products with strong controls, clear labeling, and return or collection programs work best. A community that cares about both practical use and long-term safety stands a better chance at making each tool count without harming the places we live and work.
Every once in a while, a compound like 3,5-Dibromo-4-hydroxybenzonitrile shows how science can guard health, property, and even peace of mind. With honest evaluation, tight oversight, and open conversations, we can balance what we need today with what we want for tomorrow.
What is the chemical structure and molecular formula of 3,5-Dibromo-4-Hydroxybenzonitrile?
The Structure at a Glance
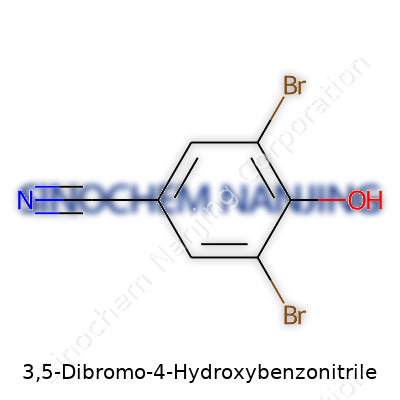
3,5-Dibromo-4-Hydroxybenzonitrile brings together a few recognizable chemical groups. The backbone is a benzene ring. At the heart of this molecule, two bromine atoms sit on the ring at the third and fifth positions. Right at the fourth spot, a hydroxy group (-OH) adds to the ring. Next to the first position, a nitrile group (-C≡N) attaches, shaping the rest of the molecule’s identity.
Chemists know this compound as C7H3Br2NO. Laying it out, the molecule’s structure runs like this: start with benzene, add a nitrile at position one, hydroxy at four, bromines at three and five. Each part matters, and the arrangement shifts how the molecule interacts with the world.
Why Structure Matters
Many folks look at molecular formulas and just see a string of letters and numbers. Behind every letter, though, there’s a story about how atoms bring out certain traits. Back in grad school, I ran into these brominated phenols during a project on antimicrobial compounds. The presence of bromine made a clear impact. Those two bromine atoms not only bump up the weight of the molecule, but they also change its reactivity. Even small tweaks, like shifting from a chlorine to a bromine, shake up the whole property set.
The hydroxy group gives this molecule some water-loving traits. It can form hydrogen bonds and helps the compound dissolve better in polar solvents compared to plain benzonitrile. Bromines bring heft and make the ring more stable—but they also make it less reactive toward some types of chemistry. The nitrile group adds its own layer, drawing electrons and pulling on the rest of the ring.
Uses and Potential
Most curious minds won’t find this compound lining pharmacy shelves. But it pops up in research work, particularly in the hunt for new pesticides and medicines. Benzonitrile derivatives, in general, have a reputation. Take a look at the agricultural world—hundreds of crop protection agents rely on little changes to rings like this one. Lab teams pick apart which tweaks block fungal growth or knock down insect populations. With public health, the structure offers a launching point for building new molecules that fight bacteria resistant to older drugs.
The trick with such chemicals is always balance. The structure makes the molecule stable enough to last in harsh environments, but researchers keep an eye on the buildup in soil and water. Safety rules at every stage—from the lab bench to manufacturing—protect workers, ecosystems, neighbors, and future generations. Regulations step in because compounds with halogens and nitriles don’t just vanish after use.
Moving Forward
Researchers aiming to work with such compounds lean on a mix of robust lab practices and green chemistry ideas. Swapping in less hazardous reagents, limiting unnecessary waste, and always keeping an eye on downstream effects help science move ahead safely. For those facing the challenge of making or using specialty chemicals like 3,5-Dibromo-4-Hydroxybenzonitrile, talking with experienced chemists and environmental specialists gives a practical edge. Science thrives when people share real experience, stay curious, and dig deep into every molecule’s story.
What are the storage and handling requirements for 3,5-Dibromo-4-Hydroxybenzonitrile?
Getting Practical with Lab Chemicals
Every scientist remembers their first introduction to the world of hazardous materials. My first close call started with an unlabeled bottle and a sharp, chemical smell. Even experienced chemists can get tripped up by inattention, so respect for compounds like 3,5-Dibromo-4-Hydroxybenzonitrile isn’t just about regulations—it’s about keeping yourself and your workspace safe.
Why Conditions Matter
This compound belongs to a class of brominated benzonitriles with strong oxidative character. It acts as a skin and respiratory irritant, and contact with water or sunlight may degrade the material. You won’t see vapor clouds rising from the bottle, but improper storage eventually leads to losses in purity or, worse, a dangerous incident. The research group at my former university learned this after discovering that unexplained “off” results traced back to old stock exposed to light and moisture.
Safe Storage Tips
The first rule is a cool, dark place with low humidity. I store these materials in tightly sealed amber glass containers. Some colleagues use metal cans, but for most research settings, glass with good closures is ideal. A silica desiccant inside a secondary container keeps things dry, especially in older buildings where humidity spikes with the seasons. Direct sunlight degrades this compound surprisingly quickly, so skip any shelf near a window.
Don’t store incompatible substances nearby. Brominated organics react with strong acids, bases, or oxidizers. I keep my benzonitrile derivatives away from nitric and sulfuric acids, as well as loose oxidizers like potassium permanganate. Flammables should remain apart, even though this compound doesn’t burn easily itself—mixing the wrong vapors leads to trouble.
Handling: Don’t Take Shortcuts
Disposable nitrile gloves and eye protection are a must. I avoid latex because certain solvents break the material down, and few things ruin your day faster than a glove that dissolves mid-transfer. Always work under a fume hood; even low-volatility materials produce low-level airborne particles. Static discharge poses a risk both for the handler and the product, so ground yourself before opening the bottle—especially during dry winters.
Spill control materials belong within arm’s reach. In my experience, spills rarely happen during measured work, but always during the rush of a deadline. Absorbent pads, baking soda for neutralization, and a clear path to the sink make all the difference. If a spill occurs, scrub with soap and water; alcohol-based cleaners work on the surface but double-check compatibility.
Best Practices Build a Safer Workplace
The chemistry community has seen injuries from ignoring guidance that seemed overcautious at the time. The most tragic part? Small lapses lead to big problems—a shared storage fridge that’s just a little too warm, a missing label, an open bottle left on the bench. Routine checks prevent accidents and keep your data clean by stopping contamination or decomposition before it begins.
Academic and industrial groups now lean on digital inventory management to track expiration dates and spot problems early. I always advocate for simple checklists and logbooks—old-school, but more reliable than memory during hectic periods. Working together and showing new colleagues proper handling sets a tone that supports both productivity and safety.
Approaching 3,5-Dibromo-4-Hydroxybenzonitrile with care comes down to respecting both the material and those who use it. Strict protocol isn’t red tape—it’s smart chemistry backed by hard-earned lessons from generations of lab workers.
Is 3,5-Dibromo-4-Hydroxybenzonitrile hazardous and are there any specific safety precautions?
Chemical Risks Aren’t Just Lab Concerns
3,5-Dibromo-4-hydroxybenzonitrile—no question about it, you need to take it seriously. Anyone who has ever spilled a chemical or caught a whiff of something sharp in a lab knows that mistakes don’t take long to cause problems. This compound, like many brominated phenols and nitriles, brings both reactivity and toxicity. It isn’t famous like cyanide, but it deserves respect, gloves, and goggles.
Toxicity Isn’t Just a Number
Research into halogenated benzonitriles highlights an uncomfortable truth: molecules with bromine and nitrile groups can irritate the skin, eyes, and lungs. They can cause lasting damage if misused. Small exposures might sting for a day but regular contact, especially without protection, could mean chronic dermatitis or worse. Some chemicals sneak into the body and show effects weeks or months later. Evidence links similar compounds to issues in aquatic life, raising concerns if any of this stuff gets rinsed down the drain.
Working With It Means Thinking Ahead
Science doesn’t just happen in test tubes—good habits start before you open a bottle. Each time I prepped for a synthetic reaction using halogenated organic compounds, I put on a lab coat, nitrile gloves, and safety glasses. That layer of defense made a difference. Ventilation counts, too: many organic dusts and vapors build up fast in small spaces. Fume hoods are not optional. I’ve seen colleagues shrug off hoods, only to suffer splitting headaches and itchy eyes after long hours with volatile substances.
Accidents Happen—Preparation Isn’t Paranoia
Working in education, I watched students forget about simple precautions, wiping their eyes with gloved hands or forgetting to tie back long hair. One time, a minor splash led to a week of pink, irritated skin. That taught everyone in the room how fast an irritating compound can ruin your day. Having spill kits ready, plus eye wash bottles clear of dust, isn’t a bureaucratic checklist—it keeps people safe. Equally crucial, the label and safety data sheet offer more than warnings; they give direct instructions for what to do in the heat of the moment.
Better Practices, Fewer Problems
Dry facts don’t always motivate better behavior, but seeing what exposure does makes the lesson stick. Using proper waste containers for brominated compounds protects not only the lab but the environment outside. Even in small academic labs, chemical waste gets collected and disposed of as hazardous material, never dumped or poured down sinks. Routine checks for leaks, clear labelling, and regular safety briefings work better than hope and luck.
Responsibility Extends Beyond the Lab
People using these chemicals outside traditional research settings—artists, hobbyists, small manufacturers—can cut corners easily. That’s where accidents pile up. Good judgment grows with experience, but sharing stories, enforcing rules, and making safety gear easy to use helps make safe habits routine. Whether working on a major synthesis or a quick test, the mindset stays the same: no shortcuts, no careless moments, no reason to risk your health for a shortcut.
What is the purity grade and available packaging sizes of 3,5-Dibromo-4-Hydroxybenzonitrile?
The Not-So-Glamorous Realities of Lab Chemicals
Anyone who spends time in a lab knows the frustration of tracking down specialty reagents like 3,5-Dibromo-4-Hydroxybenzonitrile. Most people outside of chemistry circles have never heard of this compound, but for researchers and formulators, attention to detail—especially purity and packaging—can decide whether your science moves forward, or hits a wall.
What Kind of Purity Are We Seeing?
Chemical purity is not just a badge of quality; it’s central to how reliable your experiments turn out. For 3,5-Dibromo-4-Hydroxybenzonitrile, the market expects purity grades above 97%. This level covers most synthetic and analytical needs. You might see some suppliers advertise technical grade for industrial R&D, but most academic and advanced commercial labs steer toward high-purity or analytical grade—sometimes listed at 98% or even 99% minimum. That means you’re not fighting through results tangled up by side impurities. As a researcher myself, I’ve scrapped weeks of work only to realize trace contaminants in a so-called “pure” standard were the real culprit. Over the years, I’ve come to insist on a detailed certificate of analysis and even ask vendors for batch-specific chromatography reports, since one company’s “high purity” might not match another’s definition. The bottom line: high purity cuts headache, makes data cleaner, and builds trust when you’re publishing results.
Packaging: More Than Just a Container
The size of the bottle doesn’t sound like an exciting detail until you find yourself paying for ten times what you actually need. Most suppliers offer 3,5-Dibromo-4-Hydroxybenzonitrile in laboratory-oriented sizes—my experience lines up with 1 gram and 5 gram amber glass bottles leading the pack, with options going up to 25 grams for scale-up or collaborative work. Some bulk suppliers can ship up to 100 grams, packed in sealed plastic or glass for industrial partners, but that price tag brings its own triple-checks for purity and safe delivery.I’ve faced situations where buying in excess led to degradation, especially because brominated compounds can be moisture-sensitive. Smaller containers cut down on waste and stop the heartbreak of opening a half-used jar, only to discover the contents have yellowed, clumped, or lost potency. If you need more, a reputable supplier should offer bulk packaging in double-sealed drums or HDPE containers, but don’t take shortcuts on your storage solutions just to save a little per gram on bulk orders.
The Takeaway: Details Matter
At first glance, purity grades and pack sizes seem like boring line items, far removed from the creativity and challenge of chemical synthesis. Yet every researcher I know has a story about a project derailed by a skipped detail in a spec sheet. The habit of demanding documentation, confirming storage compatibility, and choosing only what you need keeps budgets in check and data rock-solid.Most mistakes in chemistry don’t come from bad ideas—they start with bad materials. Next time you order a bottle of 3,5-Dibromo-4-Hydroxybenzonitrile, pay attention to these “little” details. It’s one way to show you’re serious about your science.
| Names | |
| Preferred IUPAC name | 3,5-dibromo-4-hydroxybenzenecarbonitrile |
| Other names |
Bromoxynil Brominal BZ Brominil Bromoxynil, analytical standard 3,5-Dibromo-4-hydroxybenzenecarbonitrile |
| Pronunciation | /ˈθriː,ˈfaɪv-daɪˈbroʊmoʊ-fɔːr-haɪˈdrɒksi-ˈbɛnzoʊˌnaɪtraɪl/ |
| Identifiers | |
| CAS Number | [33013-62-4] |
| 3D model (JSmol) | `3Dmol.js?model=CC1=C(C=C(C=C1Br)O)Br` |
| Beilstein Reference | 1002353 |
| ChEBI | CHEBI:134424 |
| ChEMBL | CHEMBL131040 |
| ChemSpider | 5339211 |
| DrugBank | DB07957 |
| ECHA InfoCard | 03f8e9e3-3ce9-4876-bbf2-2d2870d4c0b7 |
| EC Number | 20912-47-2 |
| Gmelin Reference | 82177 |
| KEGG | C14396 |
| MeSH | D017937 |
| PubChem CID | 71979 |
| RTECS number | BY5425000 |
| UNII | ND91Y96R5A |
| UN number | UN2671 |
| Properties | |
| Chemical formula | C7H3Br2NO |
| Molar mass | 295.90 g/mol |
| Appearance | White to Off-white solid |
| Odor | Odorless |
| Density | 1.98 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.9 |
| Vapor pressure | 4.7 x 10^-6 mmHg (25°C) |
| Acidity (pKa) | 7.57 |
| Basicity (pKb) | pKb ≈ 5.85 |
| Magnetic susceptibility (χ) | -63.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.694 |
| Dipole moment | 2.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -44.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AE26 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | > 270°C |
| Lethal dose or concentration | LD50 Oral Rat 3250 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 372 mg/kg |
| NIOSH | DT8925000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
2,6-Dibromo-4-hydroxybenzonitrile 3,5-Dibromosalicylonitrile 4-Hydroxybenzonitrile 3,5-Dibromobenzonitrile 3,5-Dibromophenol |

