3,4-Lutidine: Looking Beyond a Lab Reagent
Historical Development
Some compounds seem to spring up everywhere once you know their name. 3,4-Lutidine, or 3,4-dimethylpyridine, did not just appear on today’s chemical scene. Chemists started paying close attention to its unique aromatic structure well over a century ago when coal tar distillation opened the door to the pyridine family of compounds. Naming and clarity have been ongoing battles in chemical history, and this particular isomer stood out because those methyl groups on the 3 and 4 positions create a distinct blueprint. Research journals from the early 1900s already featured 3,4-lutidine as an ingredient for more complex molecules. At a time when most chemists had to rely on tedious separation and basic spectral techniques, recognizing its value showed an early appreciation for targeted molecule design and discovery. It’s a reminder that the evolution of chemistry grows on the shoulders of tedious, patient observations, and that progress often means re-examining simple molecules through new, sharper lenses.
Product Overview
For those who spend long hours with organic syntheses, 3,4-lutidine tends to show up as a lab staple—an oily, colorless liquid, not flashy but hard to replace. Chemists turn to it for its mild basicity, something that sets it apart from more aggressive bases that push reactions too far or too fast. Its gentle nature brings value in the preparation of pharmaceuticals and specialty chemicals, as it often gives much-needed control over reaction rates and selectivity. Its ease of purification and reliable physical properties provide practical advantages, especially when yields and reproducibility take priority. Chemical companies keep it in stock knowing it plays a background but ever-present role in fine-tuning outcomes. From my perspective, there’s a sort of working-class appeal to 3,4-lutidine—no fanfare, steady performance, and rarely the cause of headaches if handled with reasonable skill.
Physical & Chemical Properties
3,4-Lutidine’s profile starts with its aromatic ring and two methyl groups, which keep it from forming strong hydrogen bonds but do give it measurable bulk. Its boiling point typically sits just below 170°C, and its distinct amine odor hits you right away from an uncapped bottle. The density lands around 0.94 g/cm³—a bit lighter than water, easy to separate. You can see why it dissolves nicely in organic solvents but handles water less enthusiastically. Its pKa (about 6.7 for the conjugate acid) marks it as a manageable base, not strong enough to steal protons with reckless abandon. This measured basicity brings specificity to many catalytic cycles and substitution reactions. Small-scale operators and research labs find these characteristics translate into handling predictability, essential for reproducible research.
Technical Specifications & Labeling
Regulators and chemical suppliers demand clarity on labels for good reason, and 3,4-lutidine usually gets the works. Purity often runs above 98%, with tightly controlled moisture and acidity limits, especially where pharmaceutical or specialty grade batches head out the door. Most bottles come with hazard pictograms indicating flammability and a warning on inhalation risk. Clear synonym labeling—such as 3,4-dimethylpyridine and Cas No. 583-61-9—helps avoid confusion with other pyridine isomers, which can carry entirely different reactivity or toxicity profiles. From my own experience, mislabeling or sloppy handling in a busy lab creates a headache that nobody wants, making rigorous attention to specification sheets far more than bureaucratic red tape.
Preparation Method
Traditional synthesis of 3,4-lutidine involves the Skraup reaction or modifications tailored to pump up selectivity. Starting from simple precursors like formaldehyde, ammonia, and appropriate methylated derivatives, application of acids or oxidizing agents draws out the pyridine core. Over the years, catalytic innovations have dialed back on harsh reagents and improved atom economy. Green chemistry efforts encourage continuous flow techniques and microreactor setups to limit waste and exposure, reflecting a shift in priorities across the chemical sector. While bulk manufacturers optimize for ton-scale runs, custom synthesis in research focuses more on high purity and minor tweaks in reaction conditions. This balance between industrial practicality and lab innovation keeps 3,4-lutidine relevant and available despite growing regulatory pressure on nitrogen-containing organics and occupational exposure standards.
Chemical Reactions & Modifications
The methyl groups on the pyridine ring make 3,4-lutidine resistant to strong nucleophilic attack, allowing it to serve as a non-nucleophilic base—something every medicinal chemist appreciates when handling sensitive functional groups or fine-tuning regioselective alkylations. In addition, it tolerates a range of reagents: from alkyl halides to metallation agents. The electron-donating effect from the methyl groups pushes electron density into the ring and shifts reactivity away from the positions adjacent to nitrogen, helping to direct substituent additions for later transformations. Functionalization, like halogenation or oxidation, gives access to advanced intermediates for pharmaceuticals and agrochemicals. Researchers keep exploring how temperature, pressure, and catalyst tweaks shift yields or selectivity, a process shaped as much by patience as by flash-in-the-pan high-throughput screening.
Synonyms & Product Names
Anyone who’s dug through decades of chemical literature will recognize the importance of naming conventions. Besides “3,4-lutidine,” other terms like “3,4-dimethylpyridine” show up on safety data sheets and supplier catalogs. Some texts group all methylpyridines as “lutidines,” dividing them by the position of those methyl groups. This sounds straightforward, but mixing up 2,4-lutidine or 2,6-lutidine in a synthetic plan can derail a day’s work. While regulatory and safety standards keep databases in line now, legacy papers sometimes require careful cross-referencing. For anyone in chemical R&D or compliance, knowing these aliases minimizes mistakes and ensures accurate tracking for inventory and downstream environmental reporting.
Safety & Operational Standards
3,4-Lutidine demands respect in handling. Thanks to its volatility and strong ammonia-like odor, even a minor spill fills the room with an acrid scent. Prolonged exposure brings headaches and irritation; skin contact risks dermatitis; and, as with many pyridines, inhalation at high levels leads to systemic toxicity affecting the liver or nervous system. Fire is always in the back of the mind with solvents of its boiling range and flash point. Strict storage protocols—sealed containers, good ventilation, flame-proof cabinets—lower risks, with personal protective equipment and spill procedures part of everyday operation in responsible labs. Regulatory authorities like OSHA and REACH include 3,4-lutidine on their books, setting exposure limits and disposal routes. Over the years, stories of accidents become cautionary tales rather than daily reality, thanks to improved training and tighter operational controls.
Application Area
Chemists use 3,4-lutidine most often as a base in laboratory synthesis, but its role doesn’t end there. As an intermediate in pharmaceuticals, it offers value for building anti-infective agents, anti-inflammatory drugs, and crop protection chemicals. The selectivity from its steric environment allows complex molecule construction with fewer byproducts. Beyond the pharmaceutical sector, 3,4-lutidine has found a place in catalyst development, corrosion inhibition, and dye synthesis. Analytical chemists sometimes use it as a reference standard, given its sharp features in NMR and mass spectrometry. What stands out is the versatility arising from the combination of basicity and stability, making it useful in both exploratory research and scaled-up industrial campaigns. From university labs in North America to specialty chemical plants across Asia, its appeal comes from reliability and specificity—a prized combination for solving targeted synthesis challenges.
Research & Development
Research on 3,4-lutidine never really stands still. Each decade brings new ways to streamline its synthesis or adapt its backbone for modern needs. In the past, much attention focused on switching out the hazardous precursors or finding catalysts that would pop out the desired isomer instead of a messy mixture. These days, green chemistry drives projects to reduce waste, lower energy consumption, and swap out problematic solvents or metals. In pharmaceutical chemistry, the search for next-generation drugs draws on the lutidine scaffold as a launchpad for bioactive molecules. Environmental monitoring studies assess its fate in wastewater or air, pushing for treatment methods that break it down safely. Cross-disciplinary collaborations pull in physical chemists, toxicologists, and computational chemists, digging into every detail—how it interacts with biological systems, how it degrades, and how minute changes in the synthesis route can alter its toxicological footprint. Unlike some fleeting trends, research on 3,4-lutidine shows the value of deep, incremental improvement.
Toxicity Research
Toxicology data builds up, not in one dramatic study, but in layer after layer of long-term animal studies, case reports, and workplace monitoring. While 3,4-lutidine does not carry the acute lethality of more notorious aromatic amines, repeated exposure does reveal risks. Liver and kidney effects appear at higher doses, and inhalation leads to CNS symptoms in both animal models and poorly ventilated facilities. Recent research looks closely at metabolic byproducts once it enters the body—a reminder that the real risk sometimes comes from what the body does to a compound, not just the parent molecule’s properties. Environmental studies track persistence in soil and water, driving research into improved containment and remediation. Safety data continues to evolve as new tools, like high-throughput screening and metabolomics, shine light on chronic low-dose exposures. Toxicologists and chemical engineers compare findings, balancing industrial utility with workplace and environmental safeguards.
Future Prospects
3,4-Lutidine stands at a crossroads shared by many older chemical workhorses: keep up with environmental standards or fade out to make room for greener options. Researchers develop new catalysts and reactor designs aiming to improve selectivity and lower waste. In the drug development arena, lutidine-derived structures offer promising scaffolds for targeted therapies and diagnostic agents. Environmental scientists examine pathways for safe degradation, opening doors for more sustainable use. Regulations tighten as evidence accumulates about chronic exposure risks, prompting investment in containment, monitoring, and innovative green chemistry. Future use will depend on adaptability—both in industrial applications and in legal frameworks. The constant challenge is to leverage the familiar advantages of 3,4-lutidine while responding to the growing call for safe, sustainable, and health-conscious material choices.
What is 3,4-Lutidine used for?
The Role of 3,4-Lutidine in Chemical Synthesis
3,4-Lutidine, a dimethylpyridine, doesn’t usually feature in everyday conversation. For a chemist, though, it’s an old friend. This compound has just enough basicity and steric bulk to act as a mild, selective base—one that doesn’t overreact with everything in sight. Anyone who’s tried to control a tricky reaction knows the value of a gentle touch. Some bases cause trouble by reacting too fast or introducing side products. 3,4-Lutidine often keeps things simpler, making it useful for forming specific bonds, especially in pharmaceutical labs and crop science.
Industries That Rely on 3,4-Lutidine
Drug development teams often need to tweak the chemistry of a molecule without tearing down other parts of the structure. 3,4-Lutidine acts as a tool for just that job. It helps with acylation and alkylation reactions—meaning it supports the addition of new pieces, with fewer unwanted changes elsewhere. For folks making complex medicines or experimental compounds, this reliability matters. Most lab chemists value a reagent that reduces stray byproducts since every cleanup step takes time and erodes yield.
Out in the world of agrochemicals, 3,4-Lutidine also finds a place. Companies making herbicides or pesticides sometimes use it as an intermediate in their synthetic routes. Its specific geometry and reactivity mean it can help build molecules that wouldn’t come together as easily with other bases.
Why Safety and Experience Matter With 3,4-Lutidine
Anyone who’s smelled pyridine—the parent compound—remembers its biting, fishy odor and knows to respect these chemicals. 3,4-Lutidine shares those qualities. A strong-smelling liquid, it should be handled in a fume hood. Mistakes can cause headaches or worse. Working in a well-ventilated space, wearing gloves and goggles, stays standard practice. That’s not just company policy; ask any lab tech who’s ever had to clean up a spill.
Environmental Concerns
Pyridine derivatives have a reputation for sticking around in the environment if carelessly dumped. 3,4-Lutidine presents similar challenges. Because it resists breakdown, its release into waterways can harm aquatic life. Companies must manage their waste streams, using incineration or special chemical treatments—a step that adds cost but protects the world outside the lab. This point gets overlooked outside regulatory circles, but environmental safety starts with discipline at the source.
Surveys from the Environmental Protection Agency remind us that pyridine-based compounds can turn up in soil and groundwater when disposal practices slip. Companies and research institutes that don’t cut corners—who track their chemical handling—help keep these issues in check.
Looking for Alternatives
Some research groups test less toxic or more biodegradable compounds to replace traditional bases like 3,4-Lutidine. Progress takes time because not every substitute performs as well. Green chemistry teams work to find reagents with both the right chemical bite and a lighter environmental load. While there’s demand for safer options, nobody wants to give up results in the lab.
Final Thoughts on Responsible Use
3,4-Lutidine demonstrates how specialty chemicals walk a line between high utility and the need for respect. The time spent learning how to use it safely protects both people and the environment. Choosing the right base, managing exposure, and handling waste with care—these habits safeguard more than a chemical reaction. They shape the long-term impact of research and manufacturing, turning a useful tool into a responsible choice.
What are the physical and chemical properties of 3,4-Lutidine?
Getting to Know 3,4-Lutidine
3,4-Lutidine belongs to the pyridine family—a group of chemicals most chemistry folk cross paths with pretty early on. This compound carries two methyl groups attached to a pyridine ring, specifically at the third and fourth positions. That simple shift in structure changes a lot, not just on paper but in how this liquid behaves in the real world.
Physical Features That Speak Volumes
Pick up a bottle of 3,4-Lutidine and the first thing that stands out is its clear, colorless look. Don’t let the bland appearance fool you. Under your nose, you’ll notice a strong, fish-like smell. If a whiff reminds you of handling reagents in an undergraduate lab, you’re not alone. It has a density measuring around 0.978 grams per cubic centimeter, making it just a hair lighter than water. It’s a flammable liquid, catching fire at a flash point near 56 degrees Celsius. Anyone serious about lab safety keeps this stat in mind, especially in crowded or warm spaces.
If you pour some into water, the 3,4-Lutidine will mix, but it won’t disappear without a trace. Its moderate solubility gives people an easier time washing glassware, but it also means that accidental spills can travel further through drains, ending up where they shouldn’t.
Chemical Behavior and Why It Matters
Here’s where chemistry really makes its mark. The nitrogen atom on the ring acts as a magnet for protons. 3,4-Lutidine behaves as a weak base—nowhere near as aggressive as ammonia or sodium hydroxide, but definitely not passive either. Put it in a reaction, and it will snap up hydrogen ions or work as a catalyst. Organic chemists lean on it to control acidity when juggling sensitive molecules. This makes the compound valuable in pharmaceutical synthesis, agrochemical production, and dye manufacturing.
There’s a catch, though. That same chemical stubbornness makes 3,4-Lutidine hard to break down in the environment. Wastewater treatment plants struggle to process it if significant amounts sneak through. Small exposures cause headaches, throat irritation, or worse if mixed with other chemicals during accidents. That smell isn’t just an annoyance—it’s a warning sign to work in well-ventilated areas and to double-check storage protocols.
Respecting the Risks and Charting Solutions
Everybody working with 3,4-Lutidine has a duty to respect what this liquid can do. Over time, I’ve learned to keep a close eye on storage shelves, making sure flammable chemicals stay far from heat sources. Subbing in less hazardous bases works for simple tasks, saving the 3,4-Lutidine for the processes that truly need it. I’ve seen bigger labs employ closed systems and better scrubbers, limiting how much vapor escapes into the working space. Regular air checks keep a lid on long-term exposure risks.
Disposal shouldn’t happen down the drain or in regular trash bins. Specialized waste management companies know how to handle pyridines safely, preventing leaks into soil and groundwater. Team training sessions, even for experienced hands, go a long way—reminding people how to handle spills, use spill kits quickly, and spot early health effects.
Every Property Tells a Story
Digging into the details of 3,4-Lutidine uncovers more than numbers and facts on a page. These physical and chemical properties shape how this compound gets used, stored, and disposed of. Every chemist owes it to themselves and the world outside the lab to treat this liquid—and those like it—with care and respect. Facts and experience, taken together, help build safer and smarter habits, no short-cuts allowed.
Is 3,4-Lutidine hazardous or toxic?
What is 3,4-Lutidine?
3,4-Lutidine shows up in the chemistry world as a colorless liquid with a distinct smell. Scientists group it with pyridine derivatives, and it often gets mixed up in labs that work on organic syntheses or make special chemicals. The substance doesn’t get much attention outside those circles, but anyone using or near it might start to wonder if this compound poses any risks. That sense of curiosity about safety isn’t wasted, since small bottles of unusual chemicals always deserve extra scrutiny.
Toxicity – How Safe is It, Really?
3,4-Lutidine carries hazard labels for a reason. It causes irritation to eyes, skin, and the respiratory tract. Workers exposed to vapors without proper gear often end up coughing or complaining of watery eyes. Spills on the skin prompt an itchy, burning reaction, reminding anyone handling it that this is not something to take lightly. Not every organic solvent has the same punch, but pyridine cousins tend to hit with more noticeable effects than, say, household alcohols.
Long-term toxicity news gets mixed. Some animal studies have caught liver and kidney changes after repeated exposure, though scientific reports don’t point to major threats in small amounts. Many chemicals used in the lab trigger concern about possible carcinogenicity. There’s no clear stamp linking 3,4-Lutidine with cancer so far, but little long-term data shows what happens over decades. Detailed work from international chemical agencies slots it as harmful, but not the biggest villain in the bottle.
Flammability and Environmental Impact
Heat up a flask of 3,4-Lutidine, and you’ll be dealing with not just sharp fumes, but a liquid that can catch fire easily. Fires can spread fast if containers tip in warm labs, sending plumes into the air and raising the stakes for everyone nearby. Fire department guides list it as a moderate fire risk, with enough volatility to make anyone rethink storage on crowded shelves.
Dumping or accidental releases run into environmental hurdles. 3,4-Lutidine sticks around in soil and groundwater, resisting natural breakdown. Fish exposed to it in lab tanks show signs of stress, even at levels far below obvious pollution. Chemical safety references stress the need for careful handling and disposal, as the compound can leach into local water supplies and affect aquatic life.
Safety Precautions and Solutions
Chemists trust gloves, goggles, and fume hoods for good reason. Direct contact and inhalation are real risks with 3,4-Lutidine, so protective equipment isn’t optional. Emergency showers and eyewash stations should stay close at hand in any space where it’s used. Adding proper labeling and secure storage helps keep track of the risks, making sure nobody grabs a bottle without knowing what’s inside.
Manufacturers and lab managers face a real challenge. They train staff not just on what to do in a spill, but how to prevent mistakes in the first place. Good ventilation, written protocols, and regular reviews make a big difference. Some newer labs aim for greener chemistry by picking safer solvents or swapping to less toxic alternatives. It brings home a lesson from experience — the best way to stay safe is to treat every unknown as risky, and to keep records of what works and what doesn’t.
Why It Matters
Hazards at work might look small at first, but one slip can shift the story fast. Reports from research parks and teaching labs share tales of minor accidents that barely missed bigger disasters. Open lines of communication between users, safety officers, and local medical staff keep everyone ready when something does go wrong. Curiosity about the chemicals people use pays off, especially for those who might not see the risks at first glance. Anyone who’s reused a bottle or skipped a glove learns quickly that comfort gets replaced by regret in the world of hazardous chemicals.
References:- National Institute for Occupational Safety and Health (NIOSH): 3,4-Lutidine Info
- PubChem Open Chemistry Database, Compound Summary for 3,4-Lutidine
- European Chemicals Agency (ECHA): 3,4-Lutidine Hazard Assessment
How should 3,4-Lutidine be stored and handled?
Understanding 3,4-Lutidine Risks
3,4-Lutidine is a pyridine derivative used in research and chemical manufacturing. It’s flammable, gives off a strong odor, and can irritate the skin or eyes. I’ve worked with similar organic compounds in laboratory environments, and it’s clear that rushing this process causes trouble. Even a slight spill or mistake prompts uncomfortable reactions or lost productivity. Anyone tasked with handling this chemical should prioritize safety and take these facts seriously.
Setting Up Storage Areas
A good storage habit makes all the difference. Always opt for a well-ventilated, cool, and dry environment. Direct sunlight or heat sources can trigger vaporization or fire. I’ve seen a colleague dismiss these basics, only to discover warped, leaky bottles and the accompanying smell a week later. Metal cabinets with flame-resistant designs work best. Open shelving or plastic containers stay away from because the risk of spill and breakage jumps up.
Store 3,4-Lutidine away from acids, oxidizers, or anything that could create a hazardous reaction. These substances react sharply, and an unexpected chemical fire brings more damage than most realize. Keep the original container tightly sealed. Repackaging leads to confusion, misplaced hazard labels, and sometimes, dangerous chemical interactions with the new material. Safety data sheet instructions from trusted suppliers lay out clear recommendations about storage temperature and incompatible materials.
Smart Handling Practices
Gloves and goggles are not just for show. I once noticed a coworker skimp on protective gear during cleanup. Their skin felt the sting for hours, and the incident made a lasting impression. So, go with nitrile gloves and a face shield when pouring or moving this liquid. Lab coats or long sleeves reduce accidental skin contact. Fume hoods prevent inhalation of volatile vapors, and even a small whiff causes headaches or worse symptoms in some people.
In case of spill, never use water since it spreads the chemical farther. Instead, reach for absorbent materials like sand or vermiculite, and ventilate the area right away. Place any used cleanup materials in designated hazardous waste bins to avoid cross-contamination.
Disposal and Environmental Care
Disposing of 3,4-Lutidine according to environmental rules isn’t just a formality; it’s about protecting both your work site and the broader community. Many waste companies won’t touch improperly labeled or mishandled waste. Partner with licensed hazardous waste services for disposal. Never pour the substance down the drain or put it in regular trash—local water supplies face serious harm from trace chemical residues.
Check the expiry date before use. Old or degraded 3,4-Lutidine produces byproducts that increase safety risks. Use secondary containment trays under all storage shelves; I learned this the hard way after a ruptured bottle led to a time-consuming cleanup. These trays catch small leaks before they escalate into full-blown emergencies.
Building Safer Work Habits
Training matters. Companies that run regular safety sessions see fewer accidents and less downtime. Encouraging workers to report leaks or unsafe practices empowers everyone. Careful documentation of chemical movements and inventory checks keeps surprises to a minimum.
3,4-Lutidine, like most specialized lab chemicals, rewards careful planning and respect for standard operating procedures. With the right setup and attentive habits, storage and handling won’t become major hurdles in the lab or industrial site.
What is the molecular formula and structure of 3,4-Lutidine?
What Chemists Know About 3,4-Lutidine
Science never stops finding new ways to use old compounds, and 3,4-Lutidine stands out in a busy lineup of pyridine derivatives. If you’ve ever worked in a laboratory, its distinct smell is hard to forget; there’s a reason chemists keep bottles tucked away in ventilated hoods. Dig around the molecular details a little, and you’ll find that 3,4-Lutidine is nothing more than a pyridine ring hugged by methyl groups at the 3 and 4 positions. Its molecular formula is C7H9N.
The Structure: Where Atoms Fall Into Place
Think of pyridine as the backbone, a six-membered ring with one nitrogen atom in place of a carbon. For 3,4-Lutidine, a methyl group (CH3) attaches to the third and fourth carbon in the ring. This substitution pushes the molecule toward a sweet spot—enough reactivity for organic synthesis, but with a touch of laziness compared to its punchier cousin, pyridine itself. In textbook diagrams, you’ll see the nitrogen at the top, then count down to carbons three and four where the methyls stick out.
Real-World Importance Beyond the Lab Bench
You can spot 3,4-Lutidine just by opening an old chemistry cabinet, as its pungent, fishy odor tends to escape even tightly capped bottles. That sharp scent actually hints at its volatility, meaning it doesn't linger long if spilled. While the compound doesn’t carry the hazards of some amines, that doesn't mean it’s entirely safe—skin contact irritates, and inhalation never leads to anything good. My own time as a lab assistant taught me to treat it with respect, always donning gloves and ensuring hoods ran before uncapping any bottle.
The importance of 3,4-Lutidine doesn’t stop at academic curiosity. It finds a home in research labs during synthesis of pharmaceuticals, dyes, and as a building block for ligands in metal-catalyzed reactions. Pharmaceutical companies often tweak molecules using methylated pyridines like this—it can shift biological activity or influence how a drug molecule slips through lipid membranes. If you look at the medicinal chemistry literature, there’s a long trail of research papers citing its use as a base or reactant in creating new chemical entities.
Potential Issues and Responsible Use
While handling 3,4-Lutidine, safety keeps events from heading south. Poor ventilation or a careless spill means headaches—literally and metaphorically. Laboratories must stay vigilant, and carrying out a risk assessment is no mere box-ticking exercise. I remember more than one rookie underestimating the need for protective eyewear until a minor splash taught a hard lesson.
Proper disposal is another concern. Strong regulations exist for disposing of pyridine derivatives, since they don’t just vanish in the environment. Waste must run through solvent recovery or incineration, never down the drain. Institutions and industry partners need clear protocols so neither worker health nor the environment pays the price for sloppy habits.
Future solutions likely involve green chemistry. Substitutes with less environmental risk—or improved ways to recover and recycle—could make a difference. Attention to solvent alternatives might also shrink the chemical’s footprint. Until then, vigilance and education remain the main defenses against accidents or mishandling.
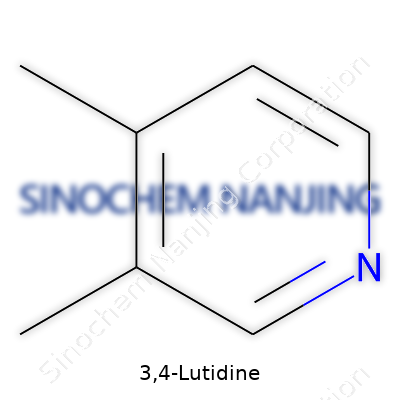
| Names | |
| Preferred IUPAC name | 3,4-dimethylpyridine |
| Other names |
3,4-Dimethylpyridine γ,δ-Lutidine |
| Pronunciation | /ˈθriːˌfɔːr.ljuːˈtɪdiːn/ |
| Identifiers | |
| CAS Number | 504-58-1 |
| 3D model (JSmol) | `3D;JSmol;C1=CC(=CN=C1)C` |
| Beilstein Reference | 505928 |
| ChEBI | CHEBI:28837 |
| ChEMBL | CHEMBL1431 |
| ChemSpider | 5048 |
| DrugBank | DB01874 |
| ECHA InfoCard | 100.007.853 |
| EC Number | 203-594-7 |
| Gmelin Reference | 79376 |
| KEGG | C01573 |
| MeSH | D016697 |
| PubChem CID | 10547 |
| RTECS number | OV3675000 |
| UNII | DL60J1XR3D |
| UN number | UN2656 |
| CompTox Dashboard (EPA) | urn:epa.compound:000944 |
| Properties | |
| Chemical formula | C7H9N |
| Molar mass | 107.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | pyridine-like |
| Density | 0.925 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | 0.83 |
| Vapor pressure | 0.82 kPa (at 20 °C) |
| Acidity (pKa) | 6.70 |
| Basicity (pKb) | 6.77 |
| Magnetic susceptibility (χ) | -52.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Viscosity | 0.86 mPa·s (20°C) |
| Dipole moment | 1.34 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 179.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -14.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –4560.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H301, H311, H331, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P303+P361+P353, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 41 °C |
| Autoignition temperature | 515 °C |
| Explosive limits | 1.1–7.4% |
| Lethal dose or concentration | LD50 oral rat 400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 400 mg/kg |
| NIOSH | SN8750000 |
| PEL (Permissible) | PEL: 2 ppm (skin) |
| REL (Recommended) | REL (Recommended Exposure Limit): 2 ppm (10 mg/m3) |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
2,4-Lutidine 2,6-Lutidine 3,5-Lutidine Pyridine 2-Picoline 3-Picoline 4-Picoline |

