3,4-Dimethylhexane: Shaping Chemistry’s Present and Future
Historical Development
Curiosity in chemistry often circles back to hydrocarbons, especially the isomers of common alkanes. 3,4-Dimethylhexane represents a good example of branches in organic chemistry that sprouted as scientists began unraveling the influence of structure on performance, stability, and availability. During the 20th century, as chemists revisited the composition of petroleum fractions, the cataloging of branched alkanes like 3,4-dimethylhexane grew out of practical needs in fields ranging from fuels to synthetic chemistry. These projects reminded everyone that even minor changes—two methyl groups tacked onto a linear chain, for instance—could swing attributes and outcomes, whether refining gasoline components or mapping reaction pathways for bigger molecules later on.
Product Overview
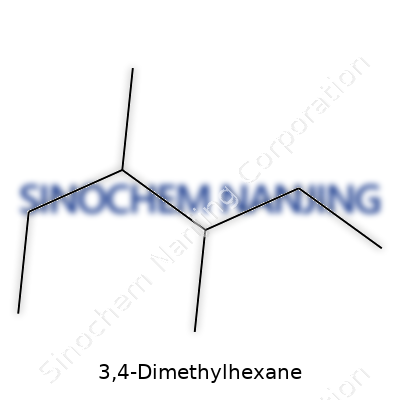
Diving into 3,4-dimethylhexane, it's a clear, colorless liquid holding eight carbons in its skeleton, set apart by methyl groups attached to its third and fourth carbon atoms. This arrangement takes it out of the shadows of more familiar straight-chain siblings, giving it unique behavior in chemical blends. For chemists who spend time hands-on in the lab, the smell, volatility, and viscosity all signal its difference right away. These tangible clues tell us how branching changes more than the textbook melting point—it means something real in a beaker.
Physical & Chemical Properties
3,4-Dimethylhexane doesn’t stray too far from the familiar traits of alkanes. It flows easily and acts as a nonpolar solvent, thanks to weak intermolecular forces. Branching tends to lower boiling points and increase octane, key for applications where combustion control matters. In practice, the boiling point hits an intermediate notch among its isomers, standing lower than n-octane but higher than more heavily branched cousins. For those troubleshooting mixtures, its density and refractive index can help confirm purity, while its flash point and vapor pressure come into play when storing or using it around ignition sources.
Technical Specifications & Labeling
On any bottle or lab drum, the label “3,4-dimethylhexane” tells people this isn’t a generic alkane. Standardized nomenclature drawn from IUPAC keeps everyone on the same page, whether ordering, inventorying, or running analyses. Trade and regulatory codes sometimes come attached for transport and safety paperwork. Observant users pay attention to these not just for compliance, but for safety and to avoid cross-contamination with other similar-sounding isomers.
Preparation Method
Synthesizing 3,4-dimethylhexane draws on solid textbook chemistry knowledge—branching out heptane, juggling methyl groups onto an appropriate precursor, or distilling and separating from broader hydrocarbon fractions. In the lab, one might start from a suitable hexane or heptane, bringing in methylation reactions under controlled conditions. Industrial streams, on the other hand, can yield this compound as a minor product when cracking larger hydrocarbons or refining crude oil. Separation relies on distillation and chromatography, both of which draw on careful temperature, pressure, and solvent control. The yield and purity often depend as much on process finesse as on the starting materials.
Chemical Reactions & Modifications
Alkanes generally avoid flashy chemical behavior, and branched ones like 3,4-dimethylhexane are no exception. Still, it takes on significance in reactions involving radical halogenation or in combustion studies, thanks to the altered structure and distribution of hydrogen atoms. Catalytic cracking, isomerization, or controlled oxidation experiments teach a lot about the branching effects on reaction mechanisms—useful knowledge when developing new synthetic routes for fuels or specialty chemicals. Experimenters keep an eye on factors like regioselectivity and yields, since branching often redirects where reactions kick off or how fast they run.
Synonyms & Product Names
Aside from the standard name, most chemists end up using descriptors like “C8H18” or shorthand such as “dimethylhexane” with the numbers dropped in casual conversation. This compound resists brand names, partly because it’s not a mass-market solvent or additive, but more of a reference and research-grade hydrocarbon. The strictest nomenclature makes sense in academic writing and regulatory paperwork, helping to keep potential confusion with other octanes at bay.
Safety & Operational Standards
Working with 3,4-dimethylhexane means respecting its flammability and volatility. Even an experienced chemist maintains a healthy respect for the way light alkanes catch fire—the flash point hovers uncomfortably low for complacency. Lab coats and gloves keep liquid contact to a minimum, good fume hoods whisk away vapors, and dispensing it from grounded, static-dissipating containers heads off accidents. Regulatory rules covering storage, handling, and disposal help set a baseline, though experienced users lean on habits built from training and mistakes. For transport, UN numbers and guidelines reflect lessons learned from decades of fire prevention and emergency response.
Application Area
Its main home stands as an internal standard or calibration substance in research laboratories, especially for chromatography. Branching patterns in its structure make it useful for understanding reaction dynamics or for teaching students about isomer effects in chemical properties. Oil and gas companies see it from time to time as a minor hydrocarbon in fuels, where it can inch up the overall octane rating. That octane boost never happens in isolation, but from a blend of many branched compounds, so 3,4-dimethylhexane gets attention when analyzing fuel compositions or developing additives for cleaner, higher-performing combustibles.
Research & Development
Chemists keep returning to molecules like 3,4-dimethylhexane in part to puzzle out how subtle tweaks in branching can steer combustion, emissions, and refinery processes. It acts as a reference point for computer models predicting fuel performance or for experimental explorations that test new catalysts and synthetic routes. In academic circles, researchers run studies looking at how these small hydrocarbons function under catalytic pressure or break down into useful fragments. These efforts feed new understanding, backing up regulatory frameworks, and improving commercial processes ranging from fuel production to specialty chemical synthesis.
Toxicity Research
Toxicologists and regulatory scientists haven’t raised major red flags about 3,4-dimethylhexane, based on current research and its similarity to other low molecular weight alkanes. That said, no one ignores the risks from inhalation or prolonged skin contact—occupational health standards require good ventilation and protective gear, if only to avoid discomfort from vapors or defatting of the skin. Chronic exposure studies often roll these compounds together, but as studies evolve, slight differences in structure sometimes show up in subtle metabolic responses in animals or people. Keeping new studies rolling in remains a priority for making sure handling standards stay up to date.
Future Prospects
Looking ahead, compounds like 3,4-dimethylhexane may not headline green energy projects, but they continue to fill essential gaps in research, regulatory work, and the development of smarter fuels and materials. The growth of computational chemistry leans on accurate reference data for a roster of molecules like this, while future fuel formulations may draw on what branching tells about knock-resistance, volatility, or emission profiles. More sustainable production methods—either by bioprocess or selective catalysis—could nudge this compound into new territory, aligning old-school hydrocarbon chemistry with emerging environmental priorities.
What is 3,4-Dimethylhexane used for?
Understanding 3,4-Dimethylhexane
Most people rarely hear about 3,4-Dimethylhexane, but it plays a role behind the scenes in several key industries. As a hydrocarbon in the family of alkanes, it shows up in products and processes that make modern living possible. Looking back on years spent in industrial labs, I saw the value in chemicals like this, especially in refining and fuel production.
Fuel and Energy Applications
Gasoline doesn’t just come from the ground ready to use. It relies on a balance of ingredients to give us the smooth-running engines people expect today. 3,4-Dimethylhexane acts as a component in specialized fuel blends. Its chemical structure helps raise the octane level of gasoline, which keeps engines running clean and efficient. Low-octane fuel causes knocking or pinging inside engines, a problem anyone with an older car might recognize. By adding compounds such as 3,4-Dimethylhexane, refiners produce fuels that resist knocking, extending engine life and improving performance.
Oil refineries break down crude oil into many parts, hunting for groups of molecules that work well together in gasoline. In practice, we combine chemicals like 3,4-Dimethylhexane to dial in the right performance for everything from family sedans to racing cars. During the oil crises of the 1970s, I remember seeing engineers scrambling to get every ounce of performance from each drop. That sense of ingenuity still exists—people in the industry rely on these refined hydrocarbons to stretch resources and reduce waste.
Importance in Chemical Research and Testing
Beyond fuel, research and testing labs value pure chemicals for controlled experiments. 3,4-Dimethylhexane serves as a standard reference hydrocarbon. Analytical chemists use it to calibrate instruments or to compare the properties of similar compounds. High-precision work in labs always demands reliable benchmarks. On busy testing days, nothing feels more satisfying than a clean chromatogram showing a peak right where it should be, thanks to well-characterized standards like this one.
Solvent and Industrial Use
This chemical performs as a solvent for certain types of chemical reactions. Because of its stability, it dissolves a range of substances without reacting. I remember working with both stubborn resins and greases—materials that fought every attempt at clean-up—until we tried hydrocarbons like 3,4-Dimethylhexane in our process. Cleanup grew faster, waste shrank, results improved. The right solvent makes jobs safer and more efficient, especially in manufacturing environments where time equals money.
Environmental and Health Concerns
While 3,4-Dimethylhexane supports big industries, workers should handle it carefully. Exposure to large amounts of hydrocarbons can harm health, often causing respiratory trouble or skin irritation. Industry guidelines recommend strong ventilation and protective gear. Companies must keep an eye on emissions, catching leaks before they reach dangerous levels. During several inspections, I saw firsthand how a simple checklist stops accidents before they start.
Better Solutions for Tomorrow
Lately, there’s a push for cleaner, more sustainable practices. Energy producers and chemical companies alike experiment with bio-based alternatives and strict environmental controls. As technology advances, replacements for petroleum-derived ingredients, including 3,4-Dimethylhexane, become more realistic. Smarter engineering and responsible work habits go a long way in protecting people and the planet.
Many folks never learn about the molecular building blocks behind daily life. Still, careful use and steady innovation around chemicals such as 3,4-Dimethylhexane make modern comfort possible while opening doors to safer, greener solutions.
What is the chemical formula of 3,4-Dimethylhexane?
Examining What Makes Up 3,4-Dimethylhexane
3,4-Dimethylhexane goes by a complicated name, but its makeup tells a pretty straightforward story. The core here: it's a hydrocarbon. Digging into its structure, you start with a straight chain of hexane—six carbon atoms linked together like beads on a string. With 3,4-dimethylhexane, things get a bit more interesting thanks to two extra methyl groups. These sit on the third and fourth carbons of the main chain. Stack those extra carbons onto the standard hexane skeleton, and you've got a special branch-shaped molecule. The chemical formula tallies up as C8H18.
Why Structure and Formula Matter
Looking at organic chemistry in high school, my attention often wandered until molecules started connecting to daily life. Hydrocarbons like 3,4-dimethylhexane form part of the gasoline in most vehicles. Getting formulas right is crucial—not out of routine, but because nature is picky. A single swapped carbon or hydrogen can mean a harmless substance like table sugar or a poison like methanol. C8H18 describes more than one molecule; it covers many isomers, all sharing the same count of atoms but shaped differently. Each one, including 3,4-dimethylhexane, brings its own properties to the table. Some burn smoothly, some don’t, and that matters to engine designers chasing the perfect fuel blend.
The Ripple Effect of Small Changes
It sounds simple, just two methyl groups added to a straight chain. That small tweak shifts the boiling point, changes how the molecule evaporates, and even tweaks the octane rating—a number that drives the performance of fuels on the road. I've seen people pour money into car modifications, obsessing over octane numbers, hoping to squeeze a little more power out of their engines. That all comes down to molecules like this one. You don’t need a laboratory coat to feel the effect.
Industry and Environment
In chemical manufacturing, details matter. If someone asked for “hexane,” and you delivered 3,4-dimethylhexane, you’d be giving them a molecule that won’t always perform the same role. Gasoline suppliers care not just about quantity, but the precise mix. Some isomers knock less in engines. These molecules help cut pollution by burning cleaner. Environmental rules push for fewer emissions, which ties right back to what’s inside the tank. Many times, a chemist’s day turns on these sorts of distinctions—getting the right substance at the right time.
Moving Toward Solutions
In my experience working alongside chemical engineers, small details often lead to big lessons. Clear labeling, investment in staff training, and deeper education for students help avoid costly mistakes with chemicals. Getting young scientists to see real-world stakes in learning formulas can spark careful habits for life. Tighter industry standards and transparent supply chains help, too. Missteps carry risks, whether it’s an engine breakdown or a safety issue at the plant. Keeping tabs on the structure and formula of compounds like 3,4-dimethylhexane is more than an academic exercise—it’s part of running a safer, greener, and smarter operation.
Fact Check: Chemical Formula
To nail down the chemical formula for 3,4-dimethylhexane: count the carbons in the main chain (six for hexane), add the two methyl groups (each with one carbon) placed on the third and fourth carbon atoms. All these carbons bind with enough hydrogens to reach four bonds each. That chemistry sums up to C8H18.
Is 3,4-Dimethylhexane hazardous or toxic?
The Real Risks Behind a Chemical Name
People hear “3,4-Dimethylhexane” and may assume the worst. The name rolls off the tongue like something out of a high school chemistry class, or maybe a warning on the back of a cleaning bottle. Yet behind every long chemical equation, there’s a story about how the substance gets used, what risks actually look like, and if anyone should really worry.
What Is 3,4-Dimethylhexane?
This compound belongs to the family called alkanes. Most folks know alkanes as ingredients in gasoline or lighter fluid. 3,4-Dimethylhexane is one of those tiny building blocks that help refine fuels to power engines. Almost nobody outside a lab or oil refinery keeps a jar of it on the shelf. This stuff usually shows up mixed with other hydrocarbons, mostly as part of gas blends or specialty solvents.
Toxicity and How Exposure Happens
Many straight-chain or branched alkanes share similar properties: they’re flammable, they evaporate easily, and too much can irritate the eyes, skin, or lungs. Swallowing the liquid—clearly not a smart move—can cause nausea and dizziness. Inhaling thick vapors in an unventilated workshop brings headaches or worse. The real damage happens through “chemical pneumonitis” if any alkane gets into lungs. This is a medical emergency, and it doesn’t matter much if you’re talking 3,4-Dimethylhexane or another hexane cousin.
Official bodies like the U.S. National Library of Medicine and the CDC group 3,4-Dimethylhexane with its fellow hydrocarbons. Compared to notorious toxins such as benzene or toluene, 3,4-Dimethylhexane does not attack the nervous system or bone marrow in small quantities. But breathing it for months or years in cramped, poorly ventilated spaces comes with worries. Factory workers might see the most risk if rules go ignored. Skin contact strips oil from the hands and can lead to dermatitis if left unwashed.
Long-Term Health and Environmental Concerns
So far, there’s no concrete evidence tying 3,4-Dimethylhexane to cancer or genetic mutations based on existing animal and cell studies. That said, nobody would recommend bathing in it or filling a closed garage with its vapor. Chronic exposure to any lower-boiling alkane may stress the kidneys, liver, or lungs over time. Nature doesn’t handle big chemical spills well: these hydrocarbons float on water, choke aquatic life, and take a while to break down if left uncared for.
Solutions Rooted in Good Practice
Hazards rise when people treat chemicals carelessly. Ventilation makes a major difference. Safety data sheets, which any reputable supplier must provide, spell out safe handling rules for substances like 3,4-Dimethylhexane. In oil refineries and labs, gloves, goggles, and closed containers serve as basics. Workers who follow these rules rarely see serious health effects from casual exposure.
The wider world cares less about the pure chemical than about products that contain it. Lawmakers focus more on broader fuel emissions. Still, spill response drills and chemical accident plans in industrial settings help reduce harm if something goes wrong.
What This Means for Ordinary People
Unless you’re working with fuel refineries or specialty labs, you’re not likely to bump into 3,4-Dimethylhexane by name. For those who do, it pays to respect those long lists of safety advice and to treat any chemical with the respect it deserves. Lessons from the past show that safe habits, air flow, and protective gear keep problems small and people safer, even with an unfamiliar name on the label.
What are the physical properties of 3,4-Dimethylhexane?
Everyday Chemistry: More Familiar Than You Think
3,4-Dimethylhexane sounds like something straight from a chemistry textbook. Truth is, this molecule belongs to the alkane family. Anyone who’s filled up a car with gas has interacted, even indirectly, with these sorts of hydrocarbons. The gasoline in your tank isn't just "gas": it's a wild mix of molecules, and 3,4-Dimethylhexane happens to be one of the many.
It’s a colorless liquid under normal conditions, which just means room temperature, same as milk or olive oil. Don’t expect it to smell pleasant; alkanes carry that sharp, gasoline-like odor. The molecular formula is C8H18, telling you it’s part of the same group as octane, which often pops up during debates about fuel quality.
Why Boiling and Melting Points Matter
This compound isn’t as volatile as the smallest alkanes, but it still has a boiling point in the ballpark of 113–115°C. Why care about boiling points? Refineries depend on this information. Lighter alkanes boil away quickly, leaving behind others that can serve different roles—this is what separates road-ready gas from jet fuel. It melts well below freezing, too, hovering somewhere around –90°C. So, unless you live at the South Pole or work in a cryogenics lab, it’ll stay liquid with ease.
How Density and Solubility Shape Everyday Uses
Stick 3,4-Dimethylhexane on a scale and fill up a container, you’ll get a density near 0.69 grams per cubic centimeter. It floats on water, just like oil, because its molecules don’t want to hang out with H2O. That makes it both a headache and a solution: a headache if it spills near groundwater (because it will spread and resist cleanup), a solution for blending and dissolving other hydrocarbons. Many of these liquid alkanes become solvents or get added to specific fuel mixtures, since they won’t mix with water but will blend with other organics smoothly.
Flammability and Safety in the Real World
Substances like 3,4-Dimethylhexane burn easily. Pour a little near an open flame and you'll see how fast it catches. Its flash point sits below room temp, making it risky to handle if folks get complacent. This property keeps safety officers and industry regulators busy: facilities design storage based on these numbers, and employees go through endless safety routines to avoid fires and inhalation risks. Anyone who’s worked in a lab knows the smell can fill a room quickly, and you learn to respect the fumes.
Making Practical Choices: Beyond the Lab Bench
This molecule doesn’t pop up in home cleaning products or flavors. Its physical characteristics—low viscosity, low density, flammability—push it into roles behind the scenes: laboratory research, industry control blends, fuel optimization. In my experience working with solvent mixtures, understanding a physical property is about knowing what could go wrong and how a substance might surprise you. That’s where molecular details stop feeling abstract and start mattering in day-to-day safety, product formulation, and environmental impact.
Room for Solutions
Plenty of industries run into trouble from volatile organics escaping into the air or leaking into soil. Better container technology, stricter protocols in shipping, and more responsive cleanup tools can cut down on accidental releases. For anyone storing, transporting, or blending fuels containing compounds like 3,4-Dimethylhexane, regular training and clear safety plans go further than fancy labels and data sheets.
How should 3,4-Dimethylhexane be stored and handled?
Looking Past Routine: The Real Stakes in Storage
Most people won’t ever see a drum of 3,4-Dimethylhexane up close, but that doesn’t mean the way it’s stored and handled lacks significance. Working in a chemical lab taught me early on that the small stuff carries lasting consequences. This liquid may look clear and harmless on the outside, but a little oversight goes a long way in turning a regular day sideways.
Fire, Fumes, and Feeling Safe
Let’s get direct: 3,4-Dimethylhexane isn’t just another chemical. It’s a hydrocarbon with high flammability. That alone calls for sharp attention. Toss it next to an open flame or even a spark, and you risk more than a ruined experiment. Storing this compound in a cool, well-ventilated spot makes sense. Concrete walls and steel storage cabinets are not just decoration in labs—they’re shields that cut down fire risk.
Years of accident reports and government data don’t lie. Vapors from these solvents build up fast, especially in cramped, unventilated spaces. I’ve worked somewhere where people assumed a fan in a window did the trick—until a vapor cloud formed and set off alarms. If proper vent hoods and explosion-proof equipment are available, there’s less to worry about.
No Substitutes for Good Containers
Some folks have a habit of “making do” with whatever jugs or bottles they find. That shortcut might look clever on paper, but there’s a real trade-off. Hydrocarbons love to eat through plastics over time, and poorly labeled or cheap containers open the door to leaks and confusion. Sturdy, tightly sealed metal or chemical-grade plastic bottles win out every time. Label everything, not for the inspectors, but for the next person who might grab the bottle without checking twice.
What Personal Protection Actually Feels Like
Spilling a little on your skin feels colder than you’d expect, then there’s the stinging. It’s easy to tell yourself gloves and goggles “slow you down,” but after you’ve watched a coworker rinse their eyes at the emergency wash, those few seconds of care seem minor. Nitrile gloves, goggles, and a lab coat become part of the work rhythm. Good habits here keep you out of awkward conversations with the medic.
Everyday Solutions Beat Fancy Tech
Not every lab or storage room has new ventilation or smart sensors, and accidents rarely give out warnings. Simple routines—logging containers in and out, keeping spills cleaned right away with absorbent pads, and storing incompatible chemicals apart—pay off in the long run. These routines didn’t just show up in safety manuals; they came from years of stories that didn’t make the news.
Accountability From Top to Bottom
Safety isn’t the responsibility of just specialists or managers. Anybody handling 3,4-Dimethylhexane can speak up, notice a missing label, or tighten a loose cap. I’ve seen more problems solved by people open enough to question a shortcut or ask for better gear than by lectures alone. Training refreshers help, but the culture around storage and handling matters more.
Building Trust on the Shop Floor
Authentic safety grows from trust—both in the rules and in the people who follow them. 3,4-Dimethylhexane may never trend as a headline chemical, but careless storage or handling always finds a way to make itself known. Fact-based routines, shared experiences, and a dose of humility keep businesses running, workers safe, and surrounding communities at peace.
| Names | |
| Preferred IUPAC name | 3,4-Dimethylhexane |
| Other names |
Diisopropylmethane 3,4-Dimethylhexan |
| Pronunciation | /ˌθriˌfoʊr.daɪˈmɛθ.əlˈhɛk.seɪn/ |
| Identifiers | |
| CAS Number | 2213-31-0 |
| Beilstein Reference | 1718733 |
| ChEBI | CHEBI:157283 |
| ChEMBL | CHEMBL1604767 |
| ChemSpider | 157172 |
| DrugBank | DB14057 |
| ECHA InfoCard | 100.152.362 |
| EC Number | 208-175-2 |
| Gmelin Reference | 87138 |
| KEGG | C21138 |
| MeSH | D016697 |
| PubChem CID | 11767 |
| RTECS number | MO2626000 |
| UNII | 4M6YQ84Z3T |
| UN number | UN1208 |
| Properties | |
| Chemical formula | C8H18 |
| Molar mass | 114.23 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.718 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 3.82 |
| Vapor pressure | 2.49 mmHg (25°C) |
| Acidity (pKa) | The acidity (pKa) of 3,4-Dimethylhexane is approximately 50. |
| Magnetic susceptibility (χ) | -72.03·10⁻⁶ cm³/mol |
| Refractive index (nD) | 0.767 |
| Viscosity | 0.674 mPa·s |
| Dipole moment | 0.00 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 347.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -198.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4321.3 kJ/mol |
| Hazards | |
| GHS labelling | GHS labelling: Not a hazardous substance or mixture according to the Globally Harmonized System (GHS). |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | No Hazard Statements |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P301+P310, P303+P361+P353, P304+P340, P312, P331, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 54 °C (closed cup) |
| Autoignition temperature | 221 °C (430 °F; 494 K) |
| Explosive limits | Explosive limits of 3,4-Dimethylhexane: 1.1–6.5% |
| LD50 (median dose) | LD50 (median dose) of 3,4-Dimethylhexane is 5000 mg/kg (rat, oral) |
| NIOSH | NM1570000 |
| PEL (Permissible) | PEL (Permissible) for 3,4-Dimethylhexane: No specific OSHA PEL established |
| REL (Recommended) | 200 mg |
| Related compounds | |
| Related compounds |
2,3-Dimethylhexane 2,5-Dimethylhexane 3-Ethylhexane 3-Methylheptane 2,4-Dimethylhexane |

