3,4-Dimethylaniline: From Chemical Bench to Industry Benchmarks
Traces from the Laboratory: Historical Development
The story of 3,4-dimethylaniline weaves through more than a century of chemical research. Its connection to the wider class of aniline derivatives dates back to the days when dye chemists scrambled to craft new hues in the burgeoning textile trade. Early synthesis pinpoints to foundational organic chemistry work in the late 19th and early 20th centuries, as chemists peeled apart the benzene ring to squeeze extra carbon atoms onto its skeleton. Progress in coal tar refinement and aromatic chemistry placed 3,4-dimethylaniline on radars, not just as another molecule but as one loaded with industrial possibilities. What used to be an academic curiosity grew into a building block for specialty dyes, agricultural products, and pharmaceuticals, each era adding new layers to its portfolio.
What Is 3,4-Dimethylaniline?
Known sometimes by the names 3,4-xylidine or 3,4-dimethylbenzenamine, this organic compound plays a double role: part laboratory staple, part industrial cog. It rolls in as a pale yellow liquid, sometimes crystallizing at cooler room temperatures. A faint, characteristic odor pegs it squarely in the aromatic amine category. This compound isn’t rare or expensive, but its value comes from adaptability. It’s used in the synthesis of dyes, pharmaceuticals, and agrochemicals, with applications built on the ring's ability to soak up new functional groups.
Physical and Chemical Properties: Small Changes, Big Impact
3,4-dimethylaniline stands out for its simplicity. It has a molecular formula of C8H11N and a molecular weight that simply nudges past 121 grams per mole. Boiling at about 230 degrees Celsius and melting just above room temperature, this compound navigates most chemical processes without much fuss. Its solubility in water stays predictably low, while organic solvents, especially ethanol, ether, and chloroform, dissolve it readily. Its density and vapor pressure fall within range for aromatic amines, so storage and transport rarely present extraordinary hurdles. The -NH2 group on the benzene ring, juxtaposed with methyl groups, boosts both electronic and steric factors; these modifications let researchers tinker with downstream reactions and tailor synthetic pathways.
Technical Specifications and Labeling Practices
Lab bottles may carry “3,4-xylidine” or “C8H11N” on the labels. Purity typically lands north of 98 percent, a nod to how critical contamination can be in fine chemical work. Labels include hazard statements for skin, eye, and respiratory irritation, mainly because aromatic amines have a track record for causing trouble on contact or with inhalation. Though no amount is considered negligible, trace impurities can derail dyes or interfere with pharmaceutical intermediates, where downstream effects snowball. So, it makes sense to respect these standards and routinely test incoming batches for quality.
Preparation: Making 3,4-Dimethylaniline Happen
Few routes reach this compound more directly than selective methylation or from reduction of 3,4-dinitrotoluene. Chemical textbooks often cite nitration of xylene, followed by catalytic hydrogenation, as a classic method. In practice, success rides on balancing temperatures and reaction time to ward off over-reduction or the creation of unwanted isomers. Advances in catalytic technology and solvent selection have squeezed more yield from these reactions, shrinking the cost gap with competing intermediates. For academic researchers, these syntheses hone skills with filtration, distillation, and solvent washes—a set of hands-on techniques that seem simple but hide subtle pitfalls.
Chemical Reactions and Tweaks in Functionality
The secret sauce with 3,4-dimethylaniline lies in what’s possible after it’s made. It’s a workhorse for condensation reactions, coupling with acid chlorides, sulfonyl chlorides, and even aldehydes. The methyl groups steady the ring, which makes for selective substitution and oxidation without the aniline wandering off in side reactions. Derivatives flow smoothly from classic Sandmeyer reactions, diazotization, or Friedel–Crafts acylation—each adjustment unlocking a new application. Over the years, chemists have built a catalog of related molecules, chasing everything from longer dye chains to the earliest hints of pharmaceutical activity.
Synonyms and Product Names: Keeping Tabs on Identity
Anyone flipping through catalogs will find “3,4-xylidine,” “3,4-dimethylaminobenzene,” and several international nomenclature twists. Translating across regulatory borders, you’ll still run into IUPAC names and trade brands. These variants reflect both local labeling laws and historical quirks in chemistry. For research, it helps to stick with the CAS registry number, which bounces between papers and supply chains, providing one clear pointer in a world of shifting names.
Getting Serious about Safety and Standards
No one with lab experience glosses over the risks tied to aromatic amines. 3,4-dimethylaniline can irritate skin and mucous membranes and pose hazards through inhalation. Safety guidelines call for gloves, fume hoods, and tight storage containers, not because regulations say so but because experience shows what can go wrong. Chronic exposure brings questions about carcinogenicity and organ toxicity, and old hazmat reports from industry accidents underscore the need for careful practice. Disposal routines focus on neutralization and incineration, and spill cleanups demand more than a mop—they need a clear head and a solid plan.
Where Does It Go? Application Areas
The biggest markets for 3,4-dimethylaniline run through dyes, pigments, and color developers. In textiles, it’s woven into the supply chain as a dye precursor, mostly for shades that call for stability and resistance. Pesticide chemists rely on it for core scaffolds that boost bioactivity or even change how an insecticide targets pests. Pharmaceutical labs use it as both a reagent and an intermediate, setting the backbone for active ingredients or diagnostic tools. Beyond large-scale industry, it finds a spot in polymer research, photography products, and, occasionally, as a stabilizer or additive wherever thermal endurance matters.
Diving into Research and Development
No one interested in sustainable chemistry overlooks the fact that aromatic amines have come under regulatory and environmental spotlight. Research groups revisit old synthesis routes with fresh ideas for waste reduction and green solvent use. The race for better catalysts echoes a broader industry push for energy savings and cleaner emissions, and 3,4-dimethylaniline gets swept along in this trend. Advances in computational chemistry help map reactivity and predict new uses, whether that’s a path to novel pharmaceuticals or better pigments. Development teams chase higher selectivity and improved yield, hunting for innovations that don’t trade safety or cost for efficiency.
Toxicity Research: Looking Past the Lab Bench
Public health and environmental scientists keep a close watch on aromatic amines, and 3,4-dimethylaniline earns its place in review articles and conference talks. Studies flag irritation, delayed toxicity to organs, and—at very high doses—risks of mutagenicity. Animal studies inform workplace limits, and occupational health guidelines reflect hard-earned lessons from decades of industrial handling. Analytical chemists have improved detection limits in water and soil, but complete answers about long-term fate in ecosystems stay elusive. For communities near large production facilities, it matters whether these chemicals leach from the plant or stick around after disposal. Weighing these risks, policy makers are slow to list them as high-priority hazards, but research keeps plugging away, often in step with public concern.
Scanning the Horizon: Where 3,4-Dimethylaniline Could Go
Markets move under the surface, shaped by changing regulations and demand for sustainable products. 3,4-dimethylaniline’s future hinges on better synthesis, safer handling, and perhaps a shift toward renewably sourced feedstocks. Researchers talk about new extraction techniques, bio-based aromatic amines, and tighter integration into closed-loop manufacturing. In the lab, future work looks set to focus on minimizing waste streams and taming any lingering environmental risks. The pressure to balance industrial need with stewardship won’t loosen, and chemists face a dual challenge: keep the benefits coming while closing the door on ongoing toxicity and environmental concerns. For now, this molecule keeps a steady place—not front-page, but firmly rooted in the practical backbone of chemistry.
What are the main uses of 3,4-Dimethylaniline?
Chemistry Beyond the Textbook
Plenty of synthetic chemicals live far from the public eye, but they quietly keep many industries running. 3,4-Dimethylaniline is a good example of that. It looks like a simple chemical at first: a colorless to pale yellow oily liquid, sharp smell, nothing fancy. Yet its uses ripple through agriculture, pharmaceuticals, dyes, and beyond. I once chatted with a chemist who explained that compounds like this don’t headline scientific breakthroughs, but they keep the gears turning in places we usually forget.
Pesticides: From Lab Synthesis to Fields
Most people connect farm chemicals with record harvests or food prices, but not many see the chain of chemicals behind the scenes. When I researched supply chains for crop protection products, 3,4-Dimethylaniline kept popping up as a key ingredient. Manufacturers use it to make several herbicides, especially those targeting broadleaf weeds. Factories in Asia and Europe pump out tons annually, feeding the global agricultural system. Its role in these products is more than a supporting actor—it often forms the backbone of molecules that disrupt unwanted plant life. Without access to such basic building blocks, modern farming would struggle to fight off competition from tough weeds.
Dyes and Pigments: Colors Crafted From Chemistry
Textiles owe their bright, lasting colors to the workhorse chemicals that dye makers depend on. I worked briefly at a textile plant, and I remember the shelves lined with drums that smelled acrid but produced some of the world’s favorite hues. 3,4-Dimethylaniline helps synthesize azo dyes, which account for a huge share of the reds, oranges, and yellows in fabrics and plastics. These dyes don’t just make clothes pop—they show up in inks, plastics, and coloring agents that mark everything from toys to packaging.
Pharmaceutical Precursors: Health Care’s Often-Unseen Foundation
The pharmaceutical story runs deeper. Before a medicine reaches a pharmacy shelf, it has to go through a jungle of chemical reactions. Some antihistamines, anesthetics, and other drugs start with molecules like 3,4-Dimethylaniline. Manufacturers rely on this chemical to construct bigger, more complicated structures found in tablets or syrups. It’s an interesting twist—people swallowing pills to recover from illness owe a quiet debt to a base chemical produced halfway across the world.
Concerns and Solutions: Handling With Respect
Working around chemicals brings up responsibility. Studies show that 3,4-Dimethylaniline carries toxicity, both for workers and in the environment. Regulators in the US, EU, and Asia set clear exposure limits based on real health data, aiming to protect factory staff, surrounding communities, and end-users. My old safety trainer hammered home the need for good ventilation and protective gear in plants dealing with aromatic amines, and the advice sticks years later. Companies now invest in closed systems and regular monitoring to prevent leaks and protect people.
Looking Forward
Staying informed about chemicals like 3,4-Dimethylaniline helps everyone from factory workers to consumers make smarter decisions. New research explores safer alternatives and better ways to capture or treat waste—because innovation isn’t just about making more, but making better. Real progress means safer workplaces, cleaner water, and a world where even unsung chemicals do their job without making headlines for the wrong reasons.
What is the chemical structure and formula of 3,4-Dimethylaniline?
The Foundation: What Makes Up 3,4-Dimethylaniline
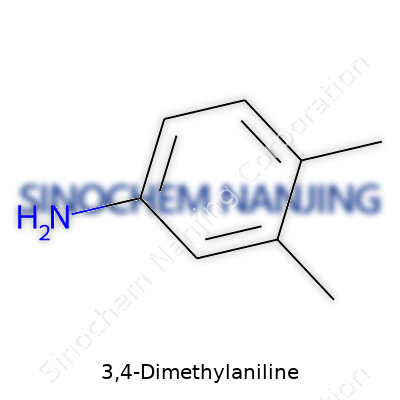
3,4-Dimethylaniline grabs some attention among chemists for its clear, direct chemical identity. Its structure includes a benzene ring at the core—a common backbone for countless organic compounds in the laboratory. That ring holds three attachments: two methyl groups and an amino group. The methyls hug the ring at the 3 and 4 positions, while the amino group hangs on at the 1 position. This tight arrangement shapes how the molecule behaves and reacts with other substances.
The chemical formula carries no surprises: C8H11N. The way the elements line up matters a lot, especially if you have seen the consequences of isomers in drug reactions, dyes, and agricultural chemicals. Even shifting a methyl group by just one carbon slot on the ring can change toxicity, solubility, and compatibility with other compounds. I’ve seen this kind of impact play out in quality control labs, where even minute changes disrupt the process.
Structure at the Atomic Level
Look up the structure in any reputable database—PubChem, ChemSpider, or the Merck Index—and you get a consistent answer. The amino group (–NH2) lies at the 1 position. From there, methyl (–CH3) branches sit at the 3 and 4 positions around the benzene ring. I remember sketching out such diagrams in organic chemistry class to hammer in the fact that position means everything in aromatic chemistry. Get the orientation wrong, your synthesis goes off track, and the properties can shift away from what industry or research demands.
The chemical formula C8H11N spells out eight carbons, eleven hydrogens, and one nitrogen. That single nitrogen lets 3,4-Dimethylaniline interact in certain reactions that pure hydrocarbons can’t touch. For example, it can take part in forming azo dyes—a staple for textile coloring. The presence of that amino group points to basicity, where it can grab onto protons, making it handy for downstream syntheses and chemical derivatization.
Real-World Implications
Labs use this compound to make colorants, medicines, and rubber processing chemicals. I’ve worked in chemical manufacturing sites where the safe handling rules for methylated anilines got highlighted at every safety briefing. Minor changes in aromatic amine structures can bump up health risks—higher potential for skin absorption or allergenic properties. The concern isn’t theoretical; see enough production incidents and it hits home quickly how a functional group or substituent changes the real-world hazard profile.
For anyone dealing with chemicals in research or industrial settings, knowing precise structures avoids costly errors. Label mistakes have led to shelf mix-ups, and confusion between similar-looking names has caused failed syntheses and safety scares. Resources like the GHS database confirm that compounds with methyl and amino groups arranged differently often call for different controls or even trigger different regulations.
Sticking to Reliable Information
High-quality research hinges on reliable chemical references. Authoritative sources double-check each detail; reputable journals and chemical catalogs rarely skip structural diagrams. I learned quickly to lean on those resources after seeing school lab accidents linked to misidentified bottles. Every step taken to verify not only chemical formulas but also their underlying arrangements—especially with substances like 3,4-Dimethylaniline—pays off in both experimental reliability and personal safety.
Is 3,4-Dimethylaniline hazardous or toxic?
Looking at 3,4-Dimethylaniline: What We Know
Everyday life brings us face to face with chemicals. Some sound familiar, others don’t, and names like 3,4-dimethylaniline can catch even folks with a science background off guard. This chemical pops up in certain manufacturing processes and research settings. It holds a spot in industrial life for making dyes and pigments, but things get personal once questions rise about its safety.
Hazards: Fact Over Hype
I’ve spent years reading chemical safety data for work, and many substances with “aniline” in the name make me stop and double-check. 3,4-Dimethylaniline is no exception. Reliable data shows it can cause some skin and eye irritation. Breathing in vapors or dust brings health risks. There’s solid research around aromatic amines—and this group often trips alarms for toxicity. Some cause methemoglobinemia—a word that means the blood has trouble carrying oxygen. Workers report headaches, dizziness, and blue-tinged skin from exposure to similar chemicals.
Lab tests point to genotoxic and carcinogenic effects. Rodent studies note a higher risk of certain cancers if the animals get regular doses over time. No chemical gets labeled “carcinogen” lightly. Research keeps pointing toward risk, especially with repeated long-term exposure.
Not Just via the Skin: Multiple Exposure Paths
On industrial floors, accidents can send chemical vapors into the air. Skin contact brings another risk. Chemical safety guides—trusted sources like the National Institute for Occupational Safety and Health—urge personal protection. This means gloves, eye wear, good ventilation, and never letting the stuff touch bare skin. At home, nobody finds this in cleaners or food. But for anybody around industrial settings, the risks sit close by.
Why It Matters: Real-Life Consequences
All this science talk turns real when stories surface: dizziness after a spill, rashes, or the stress of long-term risk. As a young lab tech, I watched a co-worker get sent to the nurse after a bungled chemical transfer. Blood tests came back fine, but nobody enjoyed waiting for the results. Even seasoned workers admit: toxicology results change how they treat everyday tasks—I learned to scrub up and gear up before handling anything with a warning label.
Room for Solutions
No silver bullet erases chemical risk, but action makes a difference. Industries swap hazardous materials for safer ones whenever possible. Where substitutions stall, tight process controls and worker training step up. Regular air testing and medical checkups turn into real protections—not just lines in a rulebook. Increased transparency around what’s getting used keeps people honest about risk.
On a practical note, education drives safer outcomes. Anyone in a lab or plant benefits from knowing which chemicals lurk in bottles and pipes. Workers need clear labels, honest hazard communication, and the gear that actually does the job. As research grows, better controls enter the scene, helping workplaces limit health impacts not just for today’s employees, but for everyone down the line.
What safety precautions should be taken when handling 3,4-Dimethylaniline?
True Hazards and Real-World Experience
Coming face to face with 3,4-dimethylaniline in a lab puts safety into sharp focus. This chemical plays a role in dyes, pesticides, and medicines, but its use never feels routine. Years back, I watched a colleague develop severe dermatitis from a simple spill that seemed minor—a reminder that every chemical demands respect. According to the International Agency for Research on Cancer, aniline compounds may carry carcinogenic risk, so regular carelessness stacks up into real harm.
Personal Protective Equipment is Non-Negotiable
Direct skin contact equals trouble. I always pull on thick nitrile gloves, not flimsy latex, because chemicals like this find their way through thin materials too fast. Lab coats with tight cuffs and chemical splash goggles are part of my routine, not an afterthought. Splashing even a drop near the eyes or mouth leads to burns or possible absorption. On one rough day, not wearing a face shield meant a single splash during transfer cost someone their eyesight.
Ventilation: Lessons Learned the Hard Way
I remember headaches and coughing spells from working in cramped, poorly ventilated corners of research labs. Volatile amines like 3,4-dimethylaniline give off fumes that make air quality plummet. Strong local exhaust—ducted fume hoods with regular filter checks—has saved my lungs more times than I care to count. Never trust a make-do setup; even homemade vent pipes backed up with every opening of a fume hood can drag residues back toward you. A broken sash or clogged filter is a silent threat, not just a minor inconvenience.
Storage and Spill Response—Day-to-Day Discipline
Leaving bottles of 3,4-dimethylaniline out on benches creates chaos fast. Locked cabinets designed for flammable and toxic substances keep both the chemical and staff safe. After a spill, preparedness separates safe labs from dangerous ones. In my experience, not all absorbents grab liquids with the same tenacity—dedicated chemical spill kits with proper neutralizing agents are a smart investment. Keeping clean-up plans written, visible, and practiced makes panic less likely.
Health Monitoring and Emergency Planning
Working with any aromatic amines brings chronic risks, not just acute exposure. Early in my career, I shrugged off mild headaches—years later, I saw charts linking repeated exposure to liver and blood disorders. Regular health check-ups, blood work, and straightforward reporting keep risks in the spotlight. Emergency showers and eyewash stations sitting fully functional near every work station can mean the difference between recovery and lifelong injury.
Training and Culture
No chemical safety plan works if only some people follow it. Consistent, hands-on training builds habits that protect everyone. Peer reminders, open discussion of near-misses, and leadership taking health and safety seriously make the difference. Encouraging openness means small problems get solved before they become disasters.
Fixing What’s Broken
Investing in high-quality ventilation, sturdy equipment, and regular training costs far less than the price of medical bills or lost productivity. Suppliers should deliver up-to-date safety data sheets, and users need to keep them close. Smart labs check their equipment themselves and don’t rely on memory. Experience shows that the most routine work can bring the biggest danger without daily vigilance.
How should 3,4-Dimethylaniline be stored and disposed of?
Storing with Respect for Safety
3,4-Dimethylaniline does not belong in the average household shelf or a poorly ventilated closet at work. From my days working alongside lab techs, storing materials like this calls for a healthy dose of respect. For starters, this chemical sits among aromatic amines, which have histories filled with toxic exposures and potent health risks. Keeping it safe starts with a tight-fitting lid on a chemical-grade container. Never decant it into random jars or any plastic that can degrade. Metal, amber glass, or certified chemical storage containers last longer and cut out unnecessary risks.
Store it in a well-ventilated chemical cabinet dedicated to organics, away from sunlight, sparks, and heat sources. These cabinets act as a barrier, contain spills, and help keep flammable vapors away from fire hazards. Strong ventilation matters because fumes from aromatic amines carry more than an unpleasant odor—they can cause headaches, respiratory irritation, and, over time, much worse. Never let this kind of substance sit next to acids or oxidizers. Even a small accident between incompatible chemicals can end with fire or toxic fumes.
Why Security and Monitoring Matter
Theft and misplacement of chemicals do not just spell regulatory headaches—they pose a real threat to human health. People have used basic storage mistakes as stepping stones to harmful incidents. Facilities with regular audits, secure access, and up-to-date inventories put a hard stop to these problems. Security measures protect both workers and the local environment. Simple checks—like keeping an eye on the integrity of a seal, checking for label smudging, and securing cabinet locks—go a long way.
Disposal Decisions Reflect Values
Pouring 3,4-Dimethylaniline down a drain or dropping it in the garbage is never just a shortcut. It is a potential disaster for water systems, wildlife, and neighborhoods. Such chemicals break down slowly and sometimes make their way into drinking supplies. Ethical labs and responsible folks consult hazardous waste guidelines and call in certified chemical waste contractors. I’ve seen organizations cut costs by cheap disposal, only to face massive cleanup bills when contamination comes to light years later.
Safe disposal starts at the source. Collect all spill residues and used gloves in sealed containers labeled clearly with the chemical’s full name. Local hazardous waste programs usually have scheduled pickups. Municipalities spell out exactly where and how to hand over these types of chemicals. Some regions require documentation about volume, container type, and origin. Following these steps respects both regulations and the neighbors who trust nearby facilities to do the right thing.
People and Policies Make a Difference
Chemicals like 3,4-Dimethylaniline do not get safer with wishful thinking. People who work around it rely on robust training programs so everyone understands why rules matter. Labels showing hazard symbols, spill kits placed close by, eyewash stations, and fire extinguishers all represent layers of protection. It takes more than a rulebook to build a safety culture—communication and continual education cut through complacency. Regular drills and real discussions about past mistakes help keep everyone alert.
Industry regulations, like those set by OSHA and the EPA, did not appear by accident. They reflect painful lessons learned from earlier decades, where chemical spills and careless handling poisoned rivers and sickened communities. Strong internal policies, coupled with respect for local rules, ensure that short-term choices do not turn into long-term disasters. For anyone handling 3,4-Dimethylaniline, safety is the outcome of daily habits, talk, and transparent action.
| Names | |
| Preferred IUPAC name | 3,4-dimethylaniline |
| Other names |
3,4-Xylidine 3,4-Dimethylbenzenamine |
| Pronunciation | /ˈθriːˌfɔːr daɪˌmɛθɪl əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 108-69-0 |
| 3D model (JSmol) | `3d:cc1ccc(cc1N)C` |
| Beilstein Reference | 1635063 |
| ChEBI | CHEBI:28079 |
| ChEMBL | CHEMBL15924 |
| ChemSpider | 66253 |
| DrugBank | DB14111 |
| ECHA InfoCard | 03eae4b5-848e-400a-9a66-9852829581c3 |
| EC Number | 202-494-5 |
| Gmelin Reference | 84138 |
| KEGG | C01579 |
| MeSH | D002167 |
| PubChem CID | 8651 |
| RTECS number | BX8575000 |
| UNII | 5172N42B63 |
| UN number | 2810 |
| CompTox Dashboard (EPA) | 3,4-Dimethylaniline |
| Properties | |
| Chemical formula | C8H11N |
| Molar mass | 121.18 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | aromatic |
| Density | 0.98 g/mL |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.19 mmHg (25°C) |
| Acidity (pKa) | 4.66 |
| Basicity (pKb) | 11.02 |
| Magnetic susceptibility (χ) | -68.7 · 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.571 |
| Viscosity | 3.10 mPa·s (25 °C) |
| Dipole moment | 2.04 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 181.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 86.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4082 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D03BA04 |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic in contact with skin, causes skin and eye irritation, may cause respiratory irritation, suspected of causing cancer. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H301, H311, H331, H315, H319, H317, H351, H373, H410 |
| Precautionary statements | P261, P273, P280, P302+P352, P304+P340, P308+P311, P312 |
| NFPA 704 (fire diamond) | 3,4-Dimethylaniline: "2-2-0 |
| Flash point | 135 °C |
| Autoignition temperature | 630°C |
| Explosive limits | Explosive limits: 1.2–7% |
| Lethal dose or concentration | LD50 oral rat 940 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2340 mg/kg (oral, rat) |
| NIOSH | NT1225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 3,4-Dimethylaniline: "2 ppm (10 mg/m3) (skin) |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Aniline o-Toluidine m-Toluidine p-Toluidine 3,5-Dimethylaniline 2,4-Dimethylaniline 2,6-Dimethylaniline N,N-Dimethylaniline |

