The Evolving Story of 3,4-Dihydroxybenzaldehyde: More Than Just a Compound
Historical Development
The earliest studies of 3,4-Dihydroxybenzaldehyde trace back to a period when organic chemists grew fascinated by the chemical makeup of naturally occurring phenolic compounds. Rooted in research from the late nineteenth and early twentieth centuries, scientists realized that certain compounds found in plants shared similar structures, including the basic aromatic ring and aldehyde group seen in 3,4-Dihydroxybenzaldehyde. The compound became a touchpoint in the investigation of lignin degradation products, which eventually led to the broader understanding of plant metabolism and microbial pathways in environmental science. Through the decades, methods for extraction, purification, and characterization of phenolic aldehydes advanced, and 3,4-Dihydroxybenzaldehyde moved from a footnote in plant biochemistry to a prominent reagent with its own research following. If you ask a chemist who spent years tracing breakdown pathways in plant tissue, the road from obscure structure to tool in the laboratory has left a mark on environmental chemistry and natural products research.
Product Overview
3,4-Dihydroxybenzaldehyde, with its two hydroxyl groups and an aldehyde function, originally found relevance in understanding how living systems process phenolic compounds. Today, it’s found in bottles on research shelves and sometimes as an intermediate in industrial settings. The compound serves multiple functions, from helping synthesize larger bioactive molecules to acting as a standard in chromatography experiments. Manufacturers now offer it at varying purity levels, targeting laboratories and companies engaged in the intricate synthesis of pharmaceuticals, dyes, and specialty chemicals.
Physical & Chemical Properties
This reddish-brown crystalline solid melts just shy of 115°C and dissolves in common organic solvents like ethanol and acetone. The scent of benzaldehydes isn’t as pronounced here, but it still reminds me of the almond-like aroma you get from related compounds. The molecular structure—an aromatic ring with an aldehyde at the 1-position and hydroxyls at the 3- and 4-positions—drives its chemical behavior. Reactivity springs mostly from the aldehyde group, while the hydroxyls not only influence solubility but also enable hydrogen bonding, affecting how the compound behaves in solutions and mixtures. The dual functionalities enable diverse chemical reactivity, which partly explains its popularity in organic synthesis and experimental design.
Technical Specifications & Labeling
Selling or storing 3,4-Dihydroxybenzaldehyde demands clear identification of its properties on labels. Researchers rely on these details to ensure correct usage and avoid mix-ups. Labels report identity, purity (usually upwards of 98 percent for research-grade stocks), and often include the CAS number. I’ve seen labs require these labels integrate hazard information and handling instructions, not only for compliance but also to prevent accidents. Transparency about potential chemical impurities matters, since trace contaminants influence how a reaction proceeds or how analytical equipment responds.
Preparation Method
Synthetic routes to 3,4-Dihydroxybenzaldehyde have evolved with the times. Traditional methods involve the oxidation of vanillin or related starting materials, often using oxidants like chromic acid. Those working toward greener chemistry have begun to revisit methods that reduce waste and avoid harsh reagents. For instance, researchers now lean more on catalytic systems or enzymatic transformations, some drawing from biotechnology tools. The shift to milder oxidation can generate the aldehyde without over-oxidation to the acid, preserving the sensitive hydroxyl groups. In personal lab work, finding a balance between reaction yield, cost, and environmental consequences always becomes the tricky part; faced with regulations and waste-disposal headaches, researchers look for ways to improve not just the product but the process too.
Chemical Reactions & Modifications
The two hydroxyl groups give 3,4-Dihydroxybenzaldehyde its strong reactivity. In the lab, this means opportunities for further functionalization: etherification, esterification, or even complex cyclizations. The aldehyde can react to form Schiff bases, hydrazones, or serve as a stepping stone for other condensation reactions. Some chemists, myself included, appreciate this compound’s role in synthesizing natural product analogues or probing new medicinal targets. Nucleophilic attacks on the aldehyde and radical substitutions started as textbook exercises but have evolved with modern synthetic goals. Powering both basic academic projects and ambitious pharmaceutical syntheses, these reactions keep 3,4-Dihydroxybenzaldehyde relevant far outside simple test-tube experiments.
Synonyms & Product Names
Those new to the compound often stumble across a stack of synonyms. “Protocatechualdehyde” stands out in older texts, especially in natural product chemistry. Other synonyms recurring on safety data sheets and chemical catalogs include 3,4-dihydroxybenzenecarboxaldehyde. Some vendors use trivial names or abbreviations, like DHBA. The diversity of names sometimes leads to mistakes or duplicate orders, so checking CAS numbers has become second nature in procurement and lab management.
Safety & Operational Standards
Routine lab work with 3,4-Dihydroxybenzaldehyde demands care. Contact with skin or mucous membranes brings irritation risks, and inhaling powders or vapors leads to respiratory discomfort. Chemical hygiene rules always apply—protective gloves, goggles, and fume hoods, every time. Waste streams containing 3,4-Dihydroxybenzaldehyde require special disposal, especially since local environmental agencies look closely at how phenolic waste gets managed. Safety training isn’t just about box-ticking; workers and students need to know where the risks lie, and emergency procedures demand regular drills. Oversight bodies have toughened enforcement, and the costs of slip-ups in compliance run high. Consistent lab audits and up-to-date chemical inventories reduce exposure and potential incidents.
Application Area
You find 3,4-Dihydroxybenzaldehyde cropping up in quite a few application fields. Pharmaceutical researchers value it for building larger bioactive molecules, especially agents showing antioxidant or anti-inflammatory effects. In analytical chemistry, it works as a comparator or marker in chromatography, and those developing environmental sensors sometimes use it as a reference standard to calibrate instruments. Specialty chemical producers explore its reactivity for niche dye synthesis and plastic additives. Antioxidant formulations for food and cosmetics research look to phenolic compounds, provoking rigorous debate about safety, extraction methods, and purity. The spectrum of use keeps growing, driven both by curiosity and real-world need.
Research & Development
A few decades ago, R&D around 3,4-Dihydroxybenzaldehyde centered mostly on natural product analysis and method development. Now, the focus has drifted toward customizing the compound’s chemical backbone to pursue better pharmaceuticals and biological probes. Investigation into enzyme-mimetic activity has grown, and modified derivatives show promise as imaging reagents in diagnostics. Efforts to make synthesis safer, cheaper, and more sustainable never stop. The future may belong to those who coax the compound into new therapeutic routes or invent low-impact production systems; those breakthroughs rest on the shoulders of earlier generations who mapped out its baseline chemistry.
Toxicity Research
The conversation about toxicity remains ongoing—and necessary. Early bioassays on cell lines and small animal models told an incomplete story, particularly considering the differences between phenolic compounds with slight structural tweaks. Chronic exposure concerns continue to drive studies, aiming for a clearer picture of genotoxicity, mutagenicity, and systemic toxicity. Regulatory agencies in the US, Europe, and Asia keep reviewing and updating their stance as new data arrives. Any researcher using the compound in formulations or biological tests stays responsible for tracking and understanding these updates, and for designing experiments around safety constraints. Handling 3,4-Dihydroxybenzaldehyde with respect comes from knowing its risks, not only to workers but also to downstream users and the environment.
Future Prospects
The next steps for 3,4-Dihydroxybenzaldehyde look set to build on decades of groundwork. Green chemistry principles push researchers to develop methods that avoid heavy metals and hazardous waste, especially for upscaled production. Biomedical research remains intrigued by new derivatives as potential treatments for inflammation, infection, and even cancer. Sensing technologies pick up speed too, with next-generation assays relying on the precise behavior of phenolic aldehydes as markers or signal generators. There’s a sense that collaboration between chemists, toxicologists, regulatory specialists, and engineers will define the next chapter for this compound. From my years seeing research priorities shift, progress rarely comes from one direction alone. Meaningful innovation emerges from shared expertise, incremental advances, and an eye kept firmly on both safety and purpose.
What is 3,4-Dihydroxybenzaldehyde used for?
What’s Behind This Chemical?
I remember the first time I saw a bottle labeled "3,4-Dihydroxybenzaldehyde" on a shelf in the university chemistry lab. The name looked intimidating, but most people know it as protocatechualdehyde. This compound shows up in more corners of industry and daily life than most folks expect.
Value in Medicine and Health
Researchers and drug makers often look for inspiration in nature, and this is where 3,4-Dihydroxybenzaldehyde shines. Plants like onions and green tea naturally produce this compound. Studies show it doesn’t just pop up by accident—plants seem to use it as a natural defense. For humans, it brings antioxidant and anti-inflammatory effects. Some scientists explore its effects in controlling diabetes and fighting cardiovascular diseases. While testing moves slow (the science world loves precision), what’s clear is that this chemical’s structure gives it a shot at blocking harmful reactions inside the body.
There’s also real excitement about fighting bacteria. Hospitals wage war against resistant bugs. Early findings suggest that 3,4-Dihydroxybenzaldehyde can halt the growth of some tough bacteria. It isn’t a cure-all but works as a tool in this ongoing battle, maybe giving doctors another round in the fight against infections driven by resistance.
Talking About Food and Flavor
Food scientists keep a close eye on natural preservatives. Freshness matters, and so does shelf life. This is where protocatechualdehyde steps in. It shows real promise in keeping food from spoiling, especially by preventing the effects of oxidation (which ruins oils and fats). Cooking often breaks down nutrients, but incorporating plant-based compounds helps slow that process. It keeps flavor intact just a little bit longer. Nobody wants to eat oxidized oil chips.
Some flavor chemists also experiment with this compound to develop new tastes. It’s not one you’ll spot on your spice rack, but people who care about taste keep exploring its properties, particularly when searching for plant-derived ingredients.
Industrial Applications: Dyes and Polymers
Dye and polymer manufacturers also use 3,4-Dihydroxybenzaldehyde. This chemical makes a good building block for synthesizing more complex molecules. For folks in textile production, it helps create stable, vibrant dyes. People expect their colored fabrics to stick around after a few washes, and molecules like this one help make that possible. This is the kind of work that goes mostly unnoticed, but without these innovations, we’d be stuck with fast-fading shirts and dull fibers.
Future Potential and Sustainable Chemistry
Science keeps digging deeper into plant-based chemicals. Demand for green chemistry grows every year. We’ve seen major health scares tied to synthetic additives; society’s appetite for natural solutions won’t fade soon. 3,4-Dihydroxybenzaldehyde could take a bigger role in making products that both work well and carry less baggage for the environment. Finding new extraction methods, or better yet, engineering plants to yield more, could lower costs and open doors. It’s about balancing innovation with responsibility.
What Matters Most
My own run-ins with research and medicine show that small molecules, those that rarely get headlines, can drive real change. Protocatechualdehyde may not grab attention on its own, but it sits at the crossroads between nature, science, and human health. Its journey has already begun, and people across several fields are watching to see where it leads next.
What is the chemical structure of 3,4-Dihydroxybenzaldehyde?
A Close Look at the Chemical Backbone
3,4-Dihydroxybenzaldehyde carries a straightforward name, but beneath it lies an elegant structure known in labs and textbooks as protocatechuic aldehyde. It forms a planar aromatic ring studded with two hydroxy groups set at the third and fourth positions, and an aldehyde group tucked right at the first carbon. In plain talk, visualize a hexagonal benzene ring with two –OH groups and one –CHO group. Scientists call this arrangement “1,2-dihydroxy-4-formylbenzene,” but the simplicity of its five-ring carbon structure plus the three functional substituents is what makes this molecule so distinctive.
The “Why” Behind Its Significance
Compounds like 3,4-Dihydroxybenzaldehyde don’t just float around in glassware—they’re building blocks in both nature and industry. Many plant-based antioxidants rely on these kinds of hydroxybenzaldehyde cores for their ability to neutralize free radicals. My own kitchen shelf holds green tea and some herbal tinctures loaded with polyphenols, compounds with similar chemistry. That’s more than health food hype. In my experience, researchers in food chemistry and pharmacology pore over molecules like this hoping to mimic or harness their effects for practical use.
Traditional herbal medicine uses ingredients sourced from roots and bark that contain this compound or close relatives, explaining why some folk remedies have real bite. The "hydroxy" bits (the –OH groups) and the "aldehyde" lend these molecules the power to form hydrogen bonds and participate in electron-donating action. That’s the scientific bedrock for their radical-scavenging, metal-chelating, and inflammation-dampening skills.
Applications and Potential
Laboratories keep tweaking derivatives of 3,4-Dihydroxybenzaldehyde. The goal relates to finding substances that work better as antioxidants or anti-inflammatory agents, or that help block enzymes that cause chronic diseases. Data from the Journal of Pharmaceutical and Biomedical Analysis reveal studies tying this core structure to cellular protection and even cardiovascular support. Pharmaceutical chemistry sees possibilities in clever modifications around the aromatic ring, aiming for new drugs with greater activity and fewer side effects.
Industrial applications reach beyond medicines. In textile processing and as dye intermediates, this compound’s reactivity simplifies the addition of other functional groups. Years back, I tagged along with a small lab that tested aromatic aldehydes in natural dye formulations—turns out replacing a single atom on the ring changes dye brightness and staying power, in ways synthetic chemists keep trying to predict with computers and experiment with in practice.
Tackling Production Challenges
One stubborn problem involves production. Extraction from plant materials doesn’t scale well and leaves impurities. Chemical synthesis using safe, cost-conscious solvents could help, provided researchers pay close attention to green chemistry best practices. A few teams have started using biocatalysts—enzymes that churn out aromatic aldehydes more precisely and with less waste. Early studies from chemical engineering departments are promising, but funding and upscaling still hold back full commercial success.
Sharing this knowledge, chemistry teachers can help students appreciate the balance between nature and technology. Instead of tossing obscure formulas onto the board, tie that structure to the antioxidant-packed foods students eat or the colorful shirts they wear. Chemical structure isn’t off in some isolated lab; it shapes visible, tangible things around us, guided by simple patterns set by a handful of atoms on a six-membered ring.
Is 3,4-Dihydroxybenzaldehyde hazardous?
What Is 3,4-Dihydroxybenzaldehyde?
Most people haven’t heard of 3,4-Dihydroxybenzaldehyde. This chemical shows up in science labs and sometimes pops up in research connected with medicines and antioxidants. The name might sound scary, but every chemical tells its story through how people use it and what the research shows.
Everyday Uses and Exposure
If you work in a lab, you might come across this compound in experiments about oxidative stress or metabolism. It can even be a building block when making certain drugs. The average person at home is unlikely to bump into it unless tinkering with serious chemistry. Still, factories and labs handling this compound have to think hard about exposure. Even common procedures like mixing, transferring, or even accidentally spilling can leave workers exposed.
What the Science Says about Safety
Before people started talking about chemical safety, I spent quite some time around industrial solvents and acids. Chemical labels, glove boxes, and even good ventilation didn’t always stop minor accidents. Looking at 3,4-Dihydroxybenzaldehyde specifically, research shows it can cause irritation to the eyes and skin. That’s true for plenty of organic chemicals, but this one deserves respect in both small-scale labs and larger industrial setups because it doesn’t take much for irritation to happen.
Breathing in dust or fumes can bother the nose and throat. For those spending years around chemicals—think of chemistry grad students or plant workers—repeated exposure may bring on more than just a quick rash or red eyes. Chronic effects remain under-studied, which always makes me wary. There’s a general scientific rule of thumb: if not enough is known, don’t take unneeded risks.
Data and Authority Guidance
Authoritative databases like PubChem and safety documents track chemicals for a reason. Government agencies in charge of workplace safety—OSHA and NIOSH—always err on the side of caution. If a material, including 3,4-Dihydroxybenzaldehyde, doesn’t have mountains of evidence saying “don’t worry,” policies treat it as a potential hazard.
I learned early on to respect Material Safety Data Sheets (MSDS). According to data from safety registries, this compound carries warning signs as an irritant. Regulatory bodies have not flagged it as a carcinogen, but there still isn’t a long list of chronic toxicity studies. If you handle it, gloves, safety glasses, and fume hoods aren’t overkill—they’re common sense.
Reducing the Hazards
Labs and chemical plants can’t afford to get careless. Proper storage in sealed containers, clear labeling, and sturdy personal protective gear make a big difference. If a spill happens, cleaning it up right away, and knowing where the nearest eyewash or shower sits, can save a lot of pain. Training isn’t a box to check—it's how accidents get caught early.
Better information helps too. Researchers and safety officers need backing from up-to-date toxicology studies. Companies and policymakers who encourage more study into mid-tier chemicals like this one—ones that aren’t household names—might catch risks before they grow expensive or dangerous. The best solutions start by staying honest about what we know and recognizing gaps where uncertainty still lingers.
What are the storage conditions for 3,4-Dihydroxybenzaldehyde?
Paying Attention to Storage—Why It Matters
3,4-Dihydroxybenzaldehyde helps researchers dig deeper into organic chemistry and biochemistry. Like many reactive chemicals, it can lose stability if ignored on the shelf. Chemical storage isn’t just an academic drill—one careless day can hurt results or even people. I’ve seen what happens when teams stash sensitive compounds without thinking about temperature swings, humidity, or sunlight. Expensive sample goes to waste, and projects move backward.
Factors Impacting 3,4-Dihydroxybenzaldehyde Storage
Temperature swings destroy sensitive structures. This compound breaks down with repeated heating and cooling cycles. Keep it at a steady, cool range—2 to 8°C works for both the short and longer haul. Most research fridges at universities and pharma labs hit this sweet spot without any extra setup.
Moisture brings trouble. A leaky cap or humid air speeds up unwanted reactions. Dry solids pull in water from the air. Store the compound in airtight amber bottles lined with dry liners or PTFE seals. Open only as much as you need, then seal up tight again.
Sunlight makes things worse. UV can trigger photodegradation, which means fragments in the molecule break apart and disappear. Nobody wants strange byproducts showing up in NMR or HPLC data. Choose amber glass over clear bottles and stow bottles in a cabinet away from windows.
Practical Steps for Better Lab Safety
I’ve noticed that clear labels save headaches down the line. Write the chemical name, date received, and re-seal date. If you spot crystals caked on the inside or dots of color changing, think about making a fresh stock.
Don’t mix open chemicals in one spot. Cross-contamination spits out impurities you won’t spot till it’s too late. Use a designated secondary container, such as a plastic tray or sealed metal box.
Fume hoods act as a good buffer zone when you measure or aliquot powders—even short contact with the open air cuts down on the risk of breathing in fine dust or exposing the bottle to ambient moisture.
Reliable Checks and Personal Routine
I run through basic inventory every few weeks. If I see a bottle with old labeling or something kept at room temperature, I double-check for stability issues. This habit caught minor spoilage more than once, letting me avoid bad experiments and wasted time.
Every research team faces cuts to time or supplies, but the cost of losing a batch or botching data can sink tight budgets faster than any careful handling protocol. Investing a few minutes right after delivery—transferring compounds to the right vials, updating the logbook, double-sealing bags—often makes the difference.
Troubleshooting and Solutions
If a batch changes color or forms unexpected crystals, I consider sending a small sample for re-analysis instead of guessing. Some companies and universities maintain shared cold rooms and humidity-controlled storage; using these shared spaces prevents individual slip-ups.
Ordering smaller sizes or splitting bulk material into small vials right away limits how much fresh compound gets exposed every time. Close communication within the team makes sure no one leaves a cap loose or forgets a sample out on the bench.
Small steps, carried out routinely, build up a safer and more reliable lab. Good storage practices for 3,4-Dihydroxybenzaldehyde don’t require fancy equipment—just a little organization and respect for unpredictable chemistry.
Where can I buy 3,4-Dihydroxybenzaldehyde?
Understanding What You’re Buying
It’s not every day someone goes hunting for 3,4-Dihydroxybenzaldehyde. Most people outside a chemistry lab have never heard that string of syllables, but researchers, medical labs, and specialty manufacturers keep a steady demand going for this aromatic compound. In plain language: this isn’t something you can pick up at a drugstore or supermarket. The compound carries its importance in organic synthesis, antioxidant studies, and pharmaceutical development.
Going Straight to the Source: Chemical Suppliers
Reliable chemical suppliers form the backbone of sourcing specialty chemicals like this one. Sigma-Aldrich, Thermo Fisher Scientific, Tokyo Chemical Industry, and Santa Cruz Biotechnology keep it in stock. These companies have earned their reputation for rigorous quality and accountable sourcing. Sourcing through them means you buy exactly what’s described, along with full documentation — safety data sheets, purity specifications, and batch testing results. That’s critical for any research that may influence products placed into the world, particularly where safety can’t be compromised.
Check for Verification and Compliance
No one should expect easy ordering. Companies put up layers of verification for a reason. Confirm your business license or research credentials before you try to place an order. These measures keep dangerous chemicals out of the wrong hands, but they also ensure your order isn’t delayed or denied. I had a colleague forget to upload his institution’s tax exemption letter — order got flagged, shipment stalled for weeks. A minor oversight, but it set the whole research timeline back. Keep all paperwork handy before making that first click.
Everything Comes Down to Purity
Every batch varies, and purity levels can heavily influence results. Researchers checked two different suppliers for a recent synthesis; one batch clocked in at 98%, the other at 95%. That tiny three percent gap changed the color, yield, and solubility in solution. Asking for certificates of analysis isn’t nitpicking. It’s the best way to head off repeat mistakes and unpredictable outcomes.
Don’t Overlook Regional Distributors
Major suppliers operate on a global scale, but it pays to check for authorized regional distributors. These local suppliers often handle smaller orders and can speed up shipping with inventory based near your lab or business. Several times I’ve dealt with US-based reps for a European supplier, which trimmed weeks off the wait. You’re more likely to get someone on the phone who can troubleshoot an order or answer technical questions quickly, especially if customs paperwork starts to look complicated.
Sustainable and Responsible Sourcing
Every industry faces pressure to minimize environmental impact and adopt safer handling. Chemical manufacturing doesn’t escape that. Responsible companies field questions about ethically sourced materials and proper end-of-life disposal every day. Some vendors post sustainability practices right on their product pages; others will send a full report if you ask. With many grants and institutions demanding proof of responsible sourcing, that’s no longer just a thing for activists or PR departments. It’s one more box to tick before signing off on a purchase order.
Weighing Price Against Service
Searches for better prices pop up all the time. No one wants to overpay, but you do get what you pay for. Off-label or gray-market sources seem tempting until you realize the risk isn’t worth it — contaminated or misrepresented chemicals undercut months of work, and those mistakes rarely get caught until it’s too late. I know groups who learned the hard way, spending extra to repeat studies with authenticated materials.
Final Thoughts
Finding 3,4-Dihydroxybenzaldehyde takes patience, credentials, and clear communication with trusted suppliers. Factoring in documentation, purity, local distribution, and responsible manufacturing makes the difference between a reliable supply and a research headache down the road. Trust the companies with a track record, ask for clear records, and press for transparency — long after the shipment shows up, those choices shape the integrity of your results.
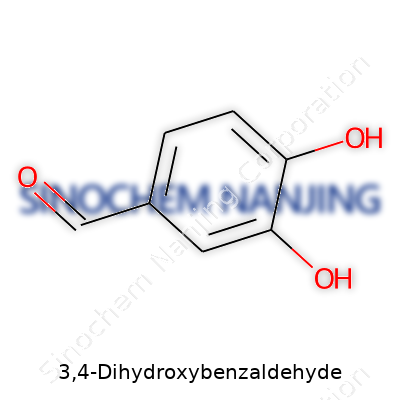
| Names | |
| Preferred IUPAC name | 3,4-dihydroxybenzaldehyde |
| Other names |
Protocatechualdehyde 3,4-Dihydroxybenzal PCA |
| Pronunciation | /ˌθriˌfaɪv.daɪˌhaɪˈdrɒksiˈbɛn.zælˌdə.haɪd/ |
| Identifiers | |
| CAS Number | 139-85-5 |
| 3D model (JSmol) | `3Dmol '3,4-dihydroxybenzaldehyde': CNc1ccc(O)c(C=O)c1` |
| Beilstein Reference | 1701694 |
| ChEBI | CHEBI:17997 |
| ChEMBL | CHEMBL502411 |
| ChemSpider | 7409 |
| DrugBank | DB04260 |
| ECHA InfoCard | 03b07bd3-39d1-4068-829e-cdb8e10e35d5 |
| EC Number | 205-012-7 |
| Gmelin Reference | 52970 |
| KEGG | C00533 |
| MeSH | D000072658 |
| PubChem CID | 86948 |
| RTECS number | DO6825000 |
| UNII | OPB015GH5K |
| UN number | 2811 |
| Properties | |
| Chemical formula | C7H6O3 |
| Molar mass | 138.12 g/mol |
| Appearance | Off-white to beige crystalline powder |
| Odor | Odorless |
| Density | 1.37 g/cm3 |
| Solubility in water | Soluble in water |
| log P | 0.52 |
| Vapor pressure | 0.000083 hPa (25 °C) |
| Acidity (pKa) | 8.42 |
| Basicity (pKb) | 13.13 |
| Magnetic susceptibility (χ) | -66.0x10^-6 cm^3/mol |
| Refractive index (nD) | 1.663 |
| Viscosity | 18 cP (25°C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 196.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -320.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1173.0 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | `GHS07` |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| Flash point | 163°C |
| Autoignition temperature | 245 °C |
| Lethal dose or concentration | LD50 (oral, rat): 2200 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2200 mg/kg (rat, oral) |
| NIOSH | WI6040000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m³ |
| Related compounds | |
| Related compounds |
Catechol Vanillin Protocatechuic acid Gallic acid Syringaldehyde |

