3,4-Dichlorophenol: A Closer Look at Its Role, Risks, and the Road Ahead
Historical Development
Scientists started isolating and experimenting with chlorophenols like 3,4-Dichlorophenol in the early marches of the twentieth century. Back then, curiosity about how chlorine bonds to aromatic rings set off a wave of discoveries, challenging old views on chemical stability and reactivity. This compound joined its relatives as a byproduct of pesticide and antiseptic manufacturing. Over decades, industries found new reasons to synthesize these compounds, moving from accidental byproducts to intentionally produced substances for various applications. As regulatory scrutiny sharpened on pesticide residues and chemical byproducts in water and soil, 3,4-Dichlorophenol never strayed far from the spotlight in laboratory and policy discussions.
Product Overview
3,4-Dichlorophenol stands out because of its distinct dual chlorine structure attached to a phenol ring. This layout doesn’t just shape its chemical behavior—it’s why scientists, manufacturers, and regulatory groups maintain intense interest in it. The substance serves as a starting point for building more complex chemicals, especially herbicides, pharmaceuticals, and disinfectants. In my own experience seeing environmental testing results, I watched laboratories chase trace detections of compounds like this, knowing they told stories about upstream manufacturing or accidental leaks, stories that matter for community health and ecosystem resilience.
Physical & Chemical Properties
Anyone who gets a whiff of 3,4-Dichlorophenol knows the scent turns noses up—a sharp, medicinal odor drifts from its pale, off-white crystals. This chemical melts at about 68°C and boils around 209°C, numbers that anchor its usability in a range of processes. It dissolves better in organic solvents than in water, which changes how it behaves once loose in the environment. These physical quirks don’t just fill data sheets; they affect cleanup crews, wastewater engineers, and researchers who track this compound’s movement through air, soil, and water.
Technical Specifications & Labeling
Clear labeling and straightforward technical data matter, especially with a chemical that lands in both industrial and research settings. A container must bear more than a name. Purity, sourcing, batch, and handling guidelines—details that sometimes get overlooked—stand between safe handling and dangerous mistakes. I’ve witnessed confusion in plants where old stock or mislabeled drums triggered hazardous mixing incidents. Full chemical identification—CAS number, synonyms, and hazard symbols—prevents confusion in busy environments and supports responsible use. Clear documentation also helps companies meet national and international shipping rules, reducing cross-border legal headaches and ensuring safety through the supply chain.
Preparation Method
Synthesizing 3,4-Dichlorophenol usually starts with chlorination of phenol, often under controlled temperature and pressure. Controlling the reaction tweaks the position and number of chlorines on the aromatic ring. By adjusting catalysts, temperature, and chlorine flow, chemists steer away from undesired byproducts and minimize waste. Factories today integrate recovery and purification steps to squeeze every bit of usable product from raw materials. Where processes once pumped out complex waste streams and air emissions, technology has matured to close those loops, whether through activated carbon scrubbing, solvent recovery, or improved reactor design.
Chemical Reactions & Modifications
3,4-Dichlorophenol doesn’t stand still chemistry-wise. It forms bonds, reacts with bases or nucleophiles, and can get transformed into ethers, esters, or other chlorinated derivatives. These modifications carve paths to herbicides, antiseptics, or pharmaceutical intermediates. Chemical engineers and researchers try to harness reactivity, not just for cost-cutting, but for improved performance and reduced side effects. For example, tweaking the chemical opens paths to less persistent environmental residues or more targeted crop protection agents. Every tweak carries responsibility, so experimentation walks hand-in-hand with monitoring and risk assessment to keep surprises contained.
Synonyms & Product Names
Depending on the literature, a reader might see this compound listed as 3,4-DCP, 3,4-Dichlorophenol, or a more descriptive name like 1-Hydroxy-3,4-dichlorobenzene. Synonyms sometimes lead newcomers astray, especially when databases, government reports, or supplier catalogs pull from a range of naming conventions. Standardized chemical identifiers help trace these compounds through regulatory filings, scientific studies, and global trade, acting like a passport at every node of its journey. Standardization matters, and with it comes accountability and clarity for everyone up and down the supply chain.
Safety & Operational Standards
Workplace safety professionals spend plenty of time thinking about 3,4-Dichlorophenol for good reason. Skin contact or inhalation can set off irritation or worse, depending on exposure level and duration. Personal protective equipment isn’t a formality—it’s standard issue. Storage demands cool, dry, and well-ventilated spaces to reduce the chance of leaks or fires. Spill response training, emergency showers, and chemical fume hoods aren’t nice-to-haves; they make the difference between a safe workplace and a local news headline. National standards and international guidelines exist for handling, storing, and disposing of this chemical, and experienced operators know the margin for cutting corners fades quickly when safety is at stake.
Application Area
3,4-Dichlorophenol has carved out a central spot in synthetic chemistry, environmental monitoring, and the design of herbicides and fungicides. Pesticide manufacturers appreciate how its structure serves as a stepping-off point to more complex agrochemicals. Water treatment facilities track it as a residue and elimination target. Researchers studying breakdown pathways in soil, water, or sewage sludge often use 3,4-Dichlorophenol as a reference point or model contaminant. My time working alongside environmental scientists taught me that keeping tabs on this class of compounds reveals big-picture trends—how product stewardship, regulation, and process optimization change chemical footprints in our rivers and food supplies.
Research & Development
Ongoing research circles around detection, degradation, and risk reduction. Advanced analytical tools sharpen our ability to detect trace amounts in the toughest samples, from river sludge to air filters. On the engineering side, efforts gear toward green chemistry—ways to use fewer hazardous inputs, recycle waste, and coax nature’s own microbes to break down stubborn residues. Universities and policy coalitions sponsor projects that close the gap between discovery and regulation, linking new science with real-world risk reduction. Whether developing next-generation analytical techniques or closed-loop waste recovery, the main goal emerges: keep utility, slash risk.
Toxicity Research
The downsides of chlorophenols like 3,4-Dichlorophenol show up in toxicity studies with living organisms. This compound can disrupt aquatic ecosystems, prompting regulatory bodies to set tight discharge and residue limits. Animal studies point to potential impacts on liver, kidneys, and neurological systems, and occupational health records stress the need for proper exposure controls. While the sharpest risks stem from direct industrial use, even trace residues in water worry communities and consumers. Continuous research puts these concerns under the microscope, updating what we know and how we respond—because science never stands still, and neither do new findings on chronic or acute toxicity.
Future Prospects
Looking ahead, the fate of 3,4-Dichlorophenol will intertwine with advances in cleaner chemistry, streamlined manufacturing, and science-based regulation. Companies face tighter environmental reporting rules, and this pushes new investment into detection, capture, and remediation. Demand for safer chemical alternatives picks up as market expectations and regulatory scrutiny sharpen, nudging both researchers and industry leaders toward new approaches and less hazardous cousins. Wastewater treatment facilities continue to scale up removal methods, harnessing biotechnology and improved catalysis. By investing in science education, stronger oversight, and cross-sector partnerships, society edges closer to solutions where the benefits of chemicals like 3,4-Dichlorophenol carry less risk and more accountability—because progress on the ground comes from a mix of vigilance, know-how, and ongoing conversation between those who make, use, and live alongside these substances.
What are the main uses of 3,4-Dichlorophenol?
A Closer Look at Where 3,4-Dichlorophenol Plays a Role
3,4-Dichlorophenol may sound like a complicated compound, but you’ll find it in more places than you might expect. Over the years, the chemical industry has relied on it to keep things running smoothly, especially in the production of other chemicals that drive many industries. Its power lies in starting reactions that eventually shape agriculture, pest control, and even consumer products.
Agricultural Chemicals: Not Just Another Ingredient
Walking into any farming supply store, you’ll notice a wide range of crop protection products. Some of those, especially older herbicides and fungicides, owe their performance to molecules that start with 3,4-Dichlorophenol. The substance stands out as a building block for creating different types of pesticides. Without it, the process of synthesizing certain herbicides would become much more challenging. It acts as a kind of “starter kit” for more complex chemicals that help keep weeds and destructive fungi in check. This means it has contributed to big jumps in farming yields and food quality, even though the push toward sustainable farming sometimes raises hard questions about chemicals in agriculture.
Intermediate in the Chemical Industry
Every big chemical plant depends on reliable intermediates. 3,4-Dichlorophenol fills that job well. The compound serves as a halfway point in the chain reaction that produces dyes, plastics, and drugs. Take antiseptics, for instance. Many bacteria-fighting substances rely on this phenol during manufacture. It makes sense why chemists stick with it; handling controlled reactions with 3,4-Dichlorophenol lets them fine-tune outcomes more predictably. That same predictability shows up in colorants, especially dyes for fabrics. Industries invest in this molecule not because of nostalgia but because it delivers the consistent outcomes required for large-scale production. The reality is that few other molecules can do the job at its price and reliability.
Protecting Consumer Goods from Decay
Mold and mildew tend to find their way into all sorts of places, from shoe closets to lumber yards. Preservatives derived from 3,4-Dichlorophenol can keep them at bay, giving goods a much longer shelf life. Lumber, in particular, benefits from treatments involving related chemicals, ensuring that boards don’t rot before reaching construction sites. Some cleaning products—especially older formulas—borrow the structure of this compound to fight off bacteria. This helps cut down on spoilage and improves safety for everyday consumers.
Weighing Risks and Looking Toward Safer Paths
No discussion about these uses can skip over the environmental and health questions. Studies have shown that 3,4-Dichlorophenol doesn’t easily break down in water or soil. Trace levels have turned up in rivers and streams, pointing to the need for tighter handling procedures and better cleanup technology. Workers who regularly come into contact with it should use protective gear, as long-term exposure may affect liver and kidney health. Some research has suggested connections with hormone disruption in wildlife.
Regulators in many countries push for careful monitoring and treatment of waste containing this substance. Some companies now search for greener alternatives, relying on enzymes or new synthesis routes that create less persistent byproducts. Finding better approaches that keep both people and nature in mind takes hands-on experience and honest cooperation across the supply chain. Building on what works—and learning from what hasn’t—will help promote safer practices while still reaping the benefits that 3,4-Dichlorophenol brings to modern life.
Is 3,4-Dichlorophenol hazardous to health?
Hazard Lurking in Unfamiliar Places
I remember helping my dad repaint an old fence as a teenager. He reached for a bottle labeled with chemicals I couldn’t pronounce, including 3,4-Dichlorophenol. He just shrugged and told me to wear gloves and keep the can away from my face. Scenes like this never left me: we brush up against compounds every day, not realizing their invisible dangers.
3,4-Dichlorophenol rarely pops up in everyday conversations. It comes from the world of industrial chemistry, where it’s used to make pesticides, disinfectants, dyes, and even as an intermediate for some pharmaceuticals. Because of those uses, traces show up in soil, water, and even the food chain. Studies have found it in rivers near factories, stormwater runoff, and dust in buildings. Once it’s out in the world, there’s no magic broom to sweep it away.
How It Gets Into Our Lives
I’ve learned that these chemicals tend to stick around. 3,4-Dichlorophenol resists breakdown, floating up into the air or leaching into the water supply. If it gets inside us, it doesn’t just pass through harmlessly. Some research in the journals of environmental science points to effects on the liver and kidneys. Even low-level, ongoing exposure may irritate eyes or the lining of the nose. In animal tests, long-term contact links up with changes in organ tissue and disruptions to the way hormones work.
Children get a bigger hit for their size. They crawl across floors, dig in playground dirt, and touch their faces hundreds of times a day. Certain studies suggest that kids living near industrial sites face a higher risk for skin problems, allergies, or trouble with their immune systems if these chemicals float in the dust.
What the Science Tells Us
A lot keeps changing. For years, toxicologists said that only very high doses would create trouble. More recent data gives a different picture. Even chronic, low-level exposures may add up, especially for workers in pesticide or dye manufacturing. Reports from the National Institutes of Health show a pattern: extended skin contact or inhaling fumes can inflame tissue or aggravate asthma. The International Agency for Research on Cancer hasn’t labeled 3,4-Dichlorophenol as a confirmed human carcinogen, but uncertainty lingers. Science sometimes takes years to catch up to what people living near polluted waterways already suspect.
Everyday Precautions and Safer Paths
My own approach changed after reading case studies and news reports about small towns near waste sites. People got tired of headaches and rashes and organized for stricter rules. Precaution beats regret. Anyone using products with confusing labels should dip into safety data sheets and always slap on gloves and goggles. Good ventilation goes a long way in workshops or home sheds. Washing hands thoroughly after gardening or painting makes sense, especially before meals.
Communities should fight for water tests, demand companies tighten disposal practices, and ask local leaders to monitor air and soil. Programs that help factories capture spills cut risks for everyone. Every time neighbors press for transparency and hold polluters to account, kids in that town breathe a little easier.
Staying alert to these hidden hazards doesn’t mean living in fear—it’s about knowing what’s out there and pushing for commonsense protections.What is the chemical structure of 3,4-Dichlorophenol?
Breaking Down the Molecule
Imagine a benzene ring, a familiar hexagon made of six carbon atoms, often used in textbooks to introduce chemical structures. Now, take that ring and stick two chlorine atoms onto it. That’s what 3,4-dichlorophenol looks like. The two chlorine atoms each bond to the third and fourth positions on the ring, while a hydroxyl group (–OH) attaches to the first spot. This kind of chemical layout changes how the compound behaves, especially compared to a simple phenol or other isomers with different chlorine placements.
Why Structure Makes a Difference
Chemists spend a lot of time talking about where certain atoms sit on a molecule. The reason is simple—arrangement influences everything from reactivity and toxicity to how a substance moves through the environment and inside living bodies. In the case of 3,4-dichlorophenol, those two chlorine atoms make the compound more persistent than plain phenol. Chlorine atoms bump up the molecule’s resistance to breakdown, making it stick around in soil and water longer.
Some might remember a high school chemistry lab, mixing up solutions and watching colors change, maybe not realizing that shifting just one element on a ring can mean the difference between a useful disinfectant and a persistent pollutant. Back in university, I saw how small molecular tweaks turned a harmless-looking compound into one flagged for tight regulation. 3,4-Dichlorophenol helps illustrate that lesson. Its stiffness against natural decay raises flags in environmental monitoring.
The Real-World Impact of 3,4-Dichlorophenol
Environmental scientists know this compound as both a breakdown product and a raw material. It's created during the manufacture of certain herbicides and insecticides, so it sometimes drifts into water or soil near industrial sites. Tests have picked up 3,4-dichlorophenol in streams downstream from chemical plants. Over time, the chlorinated molecule lingers, sometimes even showing up in drinking water.
People living near these contamination points might not see any immediate changes, but public health workers have reason to keep a watchful eye. Regular exposure to chlorinated phenols has triggered concerns. Studies link them to irritation and sometimes worse, especially if exposure lasts a long time. The presence of the –OH group makes the compound reactive in certain chemical settings, leading to complex behavior involving both the body and the world outside.
Moving Toward Safer Spaces
Over the past decade, clean-up crews and researchers have teamed up to hunt down these stubborn chemicals and lower their risks. One of the first steps often means understanding the contaminant at the molecular level—figuring out how those chlorine atoms affect both toxicity and persistence. That knowledge helps engineers design filters and microbes capable of breaking down these tough molecules.
From experience on community engagement panels, I’ve seen that trust grows where information flows freely. Knowing what 3,4-dichlorophenol looks like, and how its structure sets it apart, gives people a sense of what they’re facing when news of a spill or contamination pops up. The chemical structure isn’t just a diagram on a page: it influences everything downstream, from health advisories to the approach used in groundwater treatment.
Seeking Solutions
Creating safer environments starts at the molecular drawing board, but it draws strength from open communication and practical local action. Along with stricter release controls on industries handling chlorinated phenols, community groups can support ongoing monitoring, and investment in remediation technology pays off in cleaner water and healthier neighborhoods. Understanding the map of a molecule like 3,4-dichlorophenol offers real-world value as science, daily life, and policy goals meet.
What are the safe storage and handling procedures for 3,4-Dichlorophenol?
Clear Labeling and Storage Setup
3,4-Dichlorophenol doesn’t belong on an unsupervised shelf or anywhere near food, drink, or common-use materials. It calls for a dedicated cabinet that resists both corrosion and leaks. People who work in labs or storage rooms know there’s no room for shortcuts here—this stuff has a strong odor and can be harmful. Seal the container well, keep the label bold and clear, and store it far from heat or sunlight. Sturdy shelving that withstands the weight and possible corrosion means less risk of container breakage. Never stack heavy items above chemicals like this, since drops and spills can turn a routine afternoon into a medical emergency.
Personal Protective Equipment: Real-World Barriers
Anyone who’s ever spilled even a mild irritant on their hands knows gloves are non-negotiable. With 3,4-Dichlorophenol, chemical-resistant gloves—nitrile or neoprene, not the thin latex kind hanging out in many office kitchens—are the way to go. Safety goggles should become second nature. Think back to a time your eyes watered from a strong cleaning product—this chemical acts much faster. Overstocked labs might tempt workers to move fast, but no schedule beats basic safety. Long sleeves make sense, even if they’re less comfortable during warmer days. A lab coat and closed-toe shoes create another barrier that makes a difference if a splash happens.
Ventilation and Spill Response
Odors from 3,4-Dichlorophenol linger, and breathing them in can feel rough on the lungs. Fume hoods are essential during handling sessions. Improvised fans only blow fumes around. If you’ve worked with volatile chemicals before, it’s easy to spot those minor headaches and scratchy throats that signal poor airflow. This risk drops when jobs happen inside proper ventilation systems.
Accidents still happen, and spill kits shouldn’t gather dust. Absorbent materials, scoops, heavy-duty bags—these need to stay close at hand. In the event of a spill, people nearby rely on fast cleanup to minimize both individual harm and larger contamination. Supervisors need to insist on immediate cleaning, not “wait until my hands are free.”
Training and Documentation: Building Trust through Skills
Regulators like OSHA stress the importance of hands-on training, not just reading binders. Everyone who might open a bottle of 3,4-Dichlorophenol should know the risks and firsthand steps for an emergency. Regular drills keep routines sharp. Watching a practice run for an eyewash station or spill kit sure beats hunting through instructions for the first time during a crisis. SDS sheets—Safety Data Sheets—should never be hidden deep in a filing cabinet. Easy access means mistrust and confusion fade away.
Disposal Practices and Community Concerns
Disposal needs special care. People have different opinions about environmental rules, but no one wants dangerous chemicals washed down the drain. Licensed hazardous waste handlers deserve respect for taking on these burdens, since improper dumping damages local waterways and wildlife. If there’s ever a question about what to do with leftover material, asking the local environmental agency sets the right example. Good disposal habits reach far beyond the lab—neighbors downstream can feel the effects of a shortcut that took just seconds.
Why It All Matters
After years handling chemicals, it’s clear that storage and handling rules come from real-world lessons and tough experiences, not red tape. Following safe storage and handling procedures for 3,4-Dichlorophenol keeps people healthy and preserves trust with the community. The routines might seem tedious, but simple respect for the risks pays off every single day.
How should 3,4-Dichlorophenol be disposed of safely?
Understanding 3,4-Dichlorophenol
3,4-Dichlorophenol finds its use in creating pesticides and other chemicals. It doesn't just disappear into thin air after use. People who work with this substance know how strong it smells, and even a small spill can turn into a headache—literally and figuratively. EPA says it sticks around in water and soil, and it harms aquatic life. I once worked in a facility where we handled chemicals like this, and letting any of them touch drainpipes wasn't just a mistake—it risked a serious fine and more importantly, health.
Why Proper Handling Matters
Pouring this chemical into a sink or tossing it in with the trash poisons land and water. In my plant days, I saw what happened when folks weren't careful: brown ponds, sick wildlife, odd odors months later. Neighbors will notice and so will the authorities. Studies show 3,4-Dichlorophenol connects with kidney and liver issues in animals, and nobody wants those risks near home or city water supplies.
What Works for Real Disposal
Incineration at a qualified facility remains the most effective approach. This isn't just burning in any old fire—specialized incinerators turn dangerous waste into harmless ash by keeping temperatures extremely high and controlling oxygen. On-site burning never offers enough control; backyard methods pump toxic smoke into the air. Chemical companies pay for transport to these licensed places—it's not just about rules, it's about being responsible.
Collection companies make this job easier by picking up waste in approved drums and managing all the paperwork. These folks use specific containers and seal them tightly, because spills during transit bring their own set of headaches. I always checked lids twice, because any spill on a hot summer day means an evacuation and a hazmat call.
Reducing Exposure and Risks
Wearing gloves and goggles sounds like common sense, but accidents still happen. At our site, even janitorial staff learned to identify chemical drums and never sweep around them carelessly. OSHA training covers this in detail, and most plants post guides around hazardous waste areas. I’ve learned that almost every mistake in storage and disposal results from taking shortcuts. Having a checklist, even if it feels like overkill, protects everyone in the building.
Tools and Resources to Rely On
The EPA and state agencies publish clear lists of certified waste handlers. Every chemical shipment should travel with a manifest—it tracks where it goes and who received it. Companies selling 3,4-Dichlorophenol often suggest handlers, but double-checking credentials online is worth the extra five minutes. Many regions host annual hazardous waste drop-offs for small businesses and even individuals, helping people avoid risky dumping.
So much of this comes down to respect: for the law, for the neighborhood, and for your own skin. Mistakes turn into big stories fast, and cleanup never comes cheap. Investing the time and money upfront keeps disasters from happening, and that’s worth more than a quick fix.
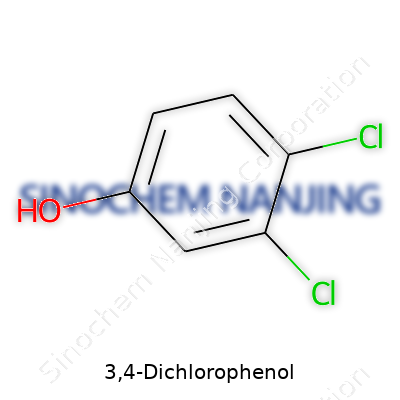
| Names | |
| Preferred IUPAC name | 3,4-dichlorophenol |
| Other names |
1,2-Dichloro-4-hydroxybenzene 3,4-DCP 4-Hydroxy-1,2-dichlorobenzene |
| Pronunciation | /ˌθriː ˌfɔː ˌdaɪˌklɔːrəˈfiːnɒl/ |
| Identifiers | |
| CAS Number | 95-77-2 |
| Beilstein Reference | 1209222 |
| ChEBI | CHEBI:34615 |
| ChEMBL | CHEMBL14678 |
| ChemSpider | 7136 |
| DrugBank | DB04588 |
| ECHA InfoCard | The ECHA InfoCard of product '3,4-Dichlorophenol' is: **03be5fa0-1e86-41ab-8461-8ab8a7c42bfb** |
| EC Number | 3.3.1.6 |
| Gmelin Reference | 8591 |
| KEGG | C01575 |
| MeSH | D002556 |
| PubChem CID | 3459 |
| RTECS number | SN1575000 |
| UNII | 9DPX91T41P |
| UN number | UN2022 |
| CompTox Dashboard (EPA) | DTXSID5020697 |
| Properties | |
| Chemical formula | C6H4Cl2O |
| Molar mass | 163.00 g/mol |
| Appearance | Colorless to yellow crystalline solid |
| Odor | Phenolic odor |
| Density | 1.5 g/cm³ |
| Solubility in water | moderately soluble |
| log P | 2.8 |
| Vapor pressure | 0.007 mmHg (25°C) |
| Acidity (pKa) | 7.9 |
| Basicity (pKb) | “8.52” |
| Magnetic susceptibility (χ) | -63.6e-6 cm³/mol |
| Refractive index (nD) | 1.578 |
| Viscosity | 1.34 mPa·s (at 20 °C) |
| Dipole moment | 2.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 152.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -171.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1052.0 kJ/mol |
| Pharmacology | |
| ATC code | D08AJ06 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS06, GHS07 |
| Pictograms | GHS06, GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H318, H400 |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P310, P321, P332+P313, P362+P364 |
| NFPA 704 (fire diamond) | 3*2*2* |
| Flash point | 77°C |
| Autoignition temperature | 330°C |
| Explosive limits | Explosive limits: 2.2–12% |
| Lethal dose or concentration | LD50 oral rat 283 mg/kg |
| LD50 (median dose) | LD50 (median dose): 330 mg/kg (oral, rat) |
| NIOSH | B087 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 3,4-Dichlorophenol: 5 ppm (skin) |
| REL (Recommended) | 0.5 mg/L |
| IDLH (Immediate danger) | The IDLH (Immediate Danger) of 3,4-Dichlorophenol is "50 ppm". |
| Related compounds | |
| Related compounds |
Phenol 2,4-Dichlorophenol 4-Chlorophenol 3-Chlorophenol 2,4,6-Trichlorophenol |

