Thinking Through 3,4-Dichloronitrobenzene: More than Just a Chemical Compound
Historical Development: The Roots and the Realities
Some chemicals grab more headlines than others, but 3,4-Dichloronitrobenzene isn’t one of them. It emerged during a time when the chemical industry started pushing boundaries in synthesis, especially in the mid-20th century. Early research into nitroaromatics had mostly targeted explosives or dyes, and slowly, substituted benzenes like this one found a place thanks to their versatility. My own experience in academic labs showed that very few reactions were truly new under the sun; tweaks and substitutions like adding those chlorine atoms at the 3 and 4 positions carried big effects in downstream chemistry, changing both the process safety and the environmental impact. Rather than chasing brand-new molecules, a lot of progress came from modifying old basics. That mindset brought 3,4-Dichloronitrobenzene into wider use, driven by the need for sophisticated intermediates with manageable hazards.
Product Overview: Why Chemists Still Care
In most synthetic benches, 3,4-Dichloronitrobenzene counts as neither exotic nor obsolete. Its main draw? Providing reactive sites in handy spots—two chlorines and a nitro group, all nicely spaced, serve as convenient handles for later transformations. You see it show up in the recipe book for pharmaceuticals, plant protection agents, and specialty dyes. Every company has its own scale and approach, but the reason for sticking with 3,4-Dichloronitrobenzene consistently circles back to reliability and reactivity. Realistically, chemists keep using what works—especially when safety and cost matter, and this one often scores well on both counts compared to more volatile analogues.
Physical & Chemical Properties: Not Glamorous, Just Functional
From my experience, physical characteristics can make or break a compound’s shelf life in the average lab. 3,4-Dichloronitrobenzene crystallizes in yellow solid form, with melting points generally reported in the ballpark of 70 to 75°C. It has just enough solubility in organic solvents like ethanol and ether to stay useful, but not so much that it causes headaches with storage or accidental spills. It has low vapor pressure, so it doesn’t stink up the workspace as quickly as some older nitro compounds. One thing that professionals regularly watch: under the right (or wrong) conditions, that nitro group holds potential for both reduction and substitution, so detailed knowledge of its reactivity profile helps prevent accidents, especially in scale-up situations.
Technical Specifications & Labeling: Practical Concerns
Working with 3,4-Dichloronitrobenzene, technical purity always comes into play. In production, most batches arrive at claimed purities above 98%, since trace impurities in substituted benzenes can carry forward into final products, complicating both regulatory filings and process consistency. Most people in the chemical industry prefer suppliers who can document batch traceability and include proper labeling with hazard pictograms and risk codes as required by global regulations. Small mistakes in labeling cause big headaches down the line. Experience tells me that taking time upfront to double-check this paperwork prevents a lot of regulatory drama or failed quality audits later.
Preparation Method: Chemistry Without Shortcuts
On the synthetic side, professionals generally start from chlorinated benzenes then introduce the nitro group using controlled nitration techniques. Direct nitration sometimes leads to an ugly mix of isomers, so many have developed safer and more selective routes by tweaking reaction temperature, acid concentration, and even the order of reagent addition. It’s become clear that while selectivity doesn’t come for free in aromatic substitution, dozens of papers exist on optimizing yields and purity for this specific compound. In operations, nobody wants runaway reactions or too many halogenated byproducts, so old-school carefulness—like strict control of addition rates and temperatures—remains essential. Chemists who cut corners here end up with either an impure product or an unsafe process, neither of which wins any favors in production or EHS reviews.
Chemical Reactions & Modifications: More Than a Starting Block
Looking at reactivity, 3,4-Dichloronitrobenzene holds up its end as a flexible intermediate. The chlorine atoms allow for nucleophilic aromatic substitution, opening the route to a wide range of amine, ether, or even thiol derivatives. Reducing that nitro group unlocks pathways to amine-containing products—core features in pharmaceuticals and agrochemicals. From my time in the lab, some modifications remain straightforward on small scale but turn trickier under scale-up, especially if heat management and thorough washing aren't on point. That’s the reality for many chemical intermediates: they behave in the flask, but they teach lessons the hard way in kilo labs or at ton scale. Rigorous standard operating procedures (SOPs) matter far more here than any theoretical reactivity chart could suggest.
Synonyms & Product Names: Navigating Supplier Lingo
From reviewing dozens of MSDS sheets and vendor catalogs, 3,4-Dichloronitrobenzene hides behind plenty of synonyms—an occupational hazard for chemists. It turns up as 1,2-Dichloro-4-nitrobenzene in some regions, or as “DCNB” when brevity beats clarity. Anyone ordering or cross-referencing must read the fine print. A slip-up can bring the wrong isomer, wrecking weeks of work. Recognizing these naming quirks comes with time, but the short lesson is: double-check the CAS number, not just the name.
Safety & Operational Standards: Keeping the Process Honest
Organic nitration brings real hazards, and 3,4-Dichloronitrobenzene sits among the less threatening but still notable cases. Direct skin and eye contact needs to be avoided; it’s possible to get by with gloves and goggles, but industrial operators rely on fume hoods and good ventilation, especially with heating. Combustibility doesn’t often cause trouble if dust isn’t allowed to accumulate, but thermal decomposition can kick up toxic gases. Regulatory agencies like OSHA and ECHA recommend standard precautions that many experienced chemists take for granted: personal protective equipment, chemical fume extraction, and rigorous training for handling and disposal. My years in industry showed that complacency sets in most often during repeated batch work—routine creates blind spots, so frequent refreshers and incident debriefings keep standards high.
Application Area: Real World Uses and Limitations
In the real world, this compound fits best as a stepping stone rather than a finished product. Pharmaceutical and agrochemical sectors tap into its dual chlorines and nitro group to construct molecular complexity from a common building block. Colorant and dye industries blend it into synthesis schemes for specialty pigments. Direct consumer exposure sits low, since the compound rarely exits the chemical plant as-is, but its footprint in global supply chains can’t be ignored. Everyday products, from medicine to crop protection, often owe a debt to such intermediates, even if few outside the sector recognize the name. Regulations in North America, Europe, and increasingly in Asia have forced manufacturers to keep tighter tabs on purity, waste, and worker safety, which on balance, has pushed the industry toward cleaner and more transparent production.
Research & Development: Beyond the Basics
Much current research trends toward greener synthesis, pushing for fewer solvents, lower emissions, and safer reaction conditions. Labs hunt for catalytic or mild conditions that reduce both cost and environmental load. With regulatory pressure mounting around halogenated aromatics, universities and companies share a growing interest in modifying classic steps or swapping in biocatalytic options. It's not just about solving technical puzzles—it's about keeping a license to operate in a world with increasing scrutiny. Younger chemists, in my experience, drive some of the best innovation here, questioning old dogmas and testing secondary reactions that industry veterans might dismiss. A fair bit of work continues in discovering downstream uses or safer analogues with similar performance in pharmaceuticals or dyes, reflecting the mix of curiosity and necessity that powers the best labs.
Toxicity Research: Learning from Hard Lessons
Toxicology of 3,4-Dichloronitrobenzene doesn't leave much room for complacency. Animal studies show dangers when exposure exceeds a safe threshold, particularly to the liver and kidneys. Chronic low-level exposure carries more risk for chemical plant workers than for the public. Acute overexposure can cause methemoglobinemia—a risk chemists watch for with most nitroaromatics. Papers from regulatory bodies and independent toxicologists give clear guidelines on limit values and acceptable exposure times, which translates to real operating habits in the workplace: break rooms away from process areas, close attention to PPE, and good hygiene before leaving the plant or lab. Experience tells me nothing drives home the need for vigilance like seeing a coworker struggle through a chemical accident, so toxicity studies aren’t academic abstractions—they guide practice daily.
Future Prospects: Where Do We Go From Here?
Sustainable chemistry has become the new watchword. The future for compounds like 3,4-Dichloronitrobenzene depends on shrinking their environmental footprint at every stage, from synthesis to waste handling. Next-generation approaches seek out less hazardous starting materials, safer solvent profiles, and robust recycling practices for both solvents and byproducts. Digitalization and process automation inch the industry toward fewer human error risks, and constant improvements in sensor technology support better leak detection and exposure limits. Creative minds will keep finding applications in pharma, agriculture, and materials science, provided the sector meets society’s growing demand for cleaner, safer production. A rigorous focus on research, transparency, and ongoing education is what keeps people, both inside and outside the industry, protected and informed about the chemicals shaping tomorrow’s world.
What is 3,4-Dichloronitrobenzene used for?
Chemical Building Block with Far-Reaching Impact
Step inside any specialty chemicals plant and you’ll catch a whiff—sometimes literally—of how foundational molecules like 3,4-dichloronitrobenzene shape much bigger processes. From my time following the chemical sector, I've seen how such ingredients serve as silent engines, pushing development across textiles, agriculture, and pharmaceuticals. This compound starts with a chlorinated, nitro-laced benzene ring, which acts like a toolkit for deeper chemical transformations. Scientists and technicians leverage its reactive spots to craft everything from crop protectants to dye intermediates that color much of the fabrics we take for granted.
Pesticides and Beyond: A Core Role in Crop Security
This molecule forms an important part in creating certain herbicides and insecticides. In big farming regions, where yield means everything, safeguarding crops against bugs and weeds does more than fill global supermarkets—it keeps prices stable and rural economies afloat. The chemistry here isn’t just about spraying toxins; it’s a long journey starting with molecules like 3,4-dichloronitrobenzene, modified downstream to target pests or halt fungal damage. The agricultural chemicals made possible by this compound support food systems worldwide.
Dye Industry: Fueling Colorfast Textiles
Vivid shades on clothes might begin their tale inside a beaker with this chemical. Dye manufacturers build on 3,4-dichloronitrobenzene to synthesize colorants that last through wash after wash. Color stability matters for fashion brands and customers, so consistency here ripples outward, affecting both quality and trust. A significant share of synthetic dyes owes some properties to these foundational substances, showing how materials science shapes what eventually shows up in our wardrobes.
Pharmaceuticals and Fine Chemicals
Drug discovery often borrows intermediates from industrial chemistry. Researchers use 3,4-dichloronitrobenzene to build more complex molecules with precise medicinal activity. The pharmaceutical industry thrives on reliable, cost-effective building blocks, and this compound fits that bill—delivering raw structure that steps up to treat diseases or manufacture new therapies. Its value stretches beyond just being a feedstock; the molecule’s versatility allows scientists to work faster and target evolving health challenges.
Environmental Concerns and Safer Practices
My conversations with environmental advocates always circle back to chemical safety. Handling and disposal of compounds like 3,4-dichloronitrobenzene call for real vigilance. Long-term pollution and workplace exposure pose health risks. Industrial regions facing contaminated soil know the stakes. Industry and regulators invest in better containment, responsible waste treatment, and greener chemical processes. More companies place sensors and detection systems to limit accidental leaks during storage or synthesis. The shift toward green chemistry also encourages finding safer alternatives and recycling used materials to cut down hazardous waste.
Moving Forward
With sharper regulatory oversight and consistent attention to safe production, the future use of 3,4-dichloronitrobenzene should focus on reducing environmental burden without halting innovation. Industry leaders can work directly with research institutions to build safer pathways or develop biodegradable pesticides and colorants, pivoting from traditional methods that leaned on this chemical. Transparent communication about its risks and uses gives communities a seat at the table and pushes everyone toward more sustainable manufacturing. The challenge lies in balancing the benefits this chemical brings with the responsibility to manage its hazards at every stage.
What are the safety precautions when handling 3,4-Dichloronitrobenzene?
Understanding the Risks
Factories and research labs keep their shelves stocked with all sorts of chemicals. One of them—3,4-Dichloronitrobenzene—carries more risk than most folks realize. This stuff hides in many chemical synthesis setups, and it doesn’t take much exposure for irritation to start. Eyes sting. Skin burns. Nose and throat feel raw. I’ve watched coworkers get headaches or queasy from breathing just a small whiff of spilled powder. Staying cautious matters not only for personal health but also for everyone around you.
Personal Protection Is a Must
Everyone who handles this chemical should suit up. No shortcuts, ever. Chemical-resistant gloves—nitrile or neoprene—work far better than latex. Long-sleeved lab coats or aprons keep skin covered, and safety goggles actually prevent serious accidents. More than once, I’ve seen colleagues save their eyesight because their glasses acted as a shield. Not feeling like a hero, just doing the basics, saved their day. For respirable dust or if fumes start spreading, a properly fitted respirator makes a big difference. Few people remember that standard cloth masks don’t filter out these particulates, but a cartridge-type respirator usually handles the job.
Ventilation Beats High-Tech Gadgets
Some labs chase after expensive filtration systems. Sure, those help, but just working under a fume hood knocks exposure way down. Hood fans and exhaust vents direct vapors away—this isn’t fancy technology, just smart worksite design. I’ve noticed fewer accidents and complaints about smells in places where folks keep hoods running, even for small batches or transfers. Opening windows feels old-school, but cross-ventilation still helps outdoors or in field labs without proper infrastructure.
No Eating or Drinking Near Chemicals
Lunch breaks tempt everyone, especially during exhausting shifts. Forgetting a water bottle on the bench might seem harmless. In truth, eating or drinking in the work area opens up another route for toxic chemicals to sneak into your body. Once, a slice of pizza left unattended on a nearby bench got dusted during a messy transfer. No one wants to admit they almost swallowed a chemical, but it happens. Designating clean spots for food keeps mistakes down.
Chemical Storage and Cleanup Basics
Storage mistakes create headaches, and sometimes real disasters. Always keep 3,4-Dichloronitrobenzene in airtight containers, clearly labeled, away from heat and sunlight. Separate acids and strong bases—accidental mixing can get violent. From experience, keeping incompatible substances apart reduces mid-day scramble and minor emergencies. Cleanup plans shouldn’t just gather dust in a binder. Absorbent pads, appropriate waste bins, and a working eye wash sink make spills less frightening. Cleaning with water alone won’t cut it; residue sticks around. Trained staff recognizing not to use a soaked rag for everything lowers cross-contamination risks.
Training and Routine Checks
The best safety gear on earth won’t mean much if nobody remembers why or how to use it. Hands-on drills deliver more than online quizzes. Colleagues watch out for one another during transfers and disposal, and less-experienced folks ask for help instead of guessing. Regular checks for leaks and proper waste container labeling have prevented accidents in every lab I’ve worked. Sharing real stories of minor incidents gets people’s attention more than reading rules on a sheet.
The Real Payoff
Safety around strong chemicals like 3,4-Dichloronitrobenzene comes down to discipline and team habits. Everyone deserves to clock out in the same shape they arrived. It’s not about looking tough—it’s about going home healthy.
What is the chemical formula and molecular weight of 3,4-Dichloronitrobenzene?
The Building Blocks: Chemical Formula and Molecular Weight
Genuine understanding in chemistry comes from digging into the nuts and bolts. For 3,4-Dichloronitrobenzene, the chemical formula stands as C6H3Cl2NO2, with a molecular weight measured at 192.00 g/mol. These numbers are not just trivia; they hold real meaning for anyone working with or studying this compound. The correct molecular formula tells you exactly what atoms are present, and how many of each make up one molecule. Accuracy here means safety for labs and reliability for industries.
Knowing What's Inside Matters
Misidentifying something as basic as a formula or molecular weight has tripped up many projects. Years ago, in my undergraduate lab, a miscalculation of molar mass led to a failed synthesis that threw off an entire research week for my team. These numbers echo beyond the page: they're packed with consequences for dosing, reactivity, and environmental impact. For instance, in pharmaceutical research, the balance between efficacy and toxicity pivots on the smallest differences in molecular structure and weight.
Why Exact Data Drives Safer and Smarter Chemistry
Handling 3,4-Dichloronitrobenzene without knowing its formula means gambling with physical and health risks. This compound, like many nitroaromatics, can present health hazards. Reliable formula and weight data ensure the right calculations for storage, disposal, and reaction setup. Oversight here often leads to bad news in safety reports. Having precise information becomes a shield against common laboratory slips and industrial accidents.
The Reach Beyond the Laboratory Bench
The impact of accurate molecular data spreads into environmental science. Incorrect formulas distort predictions of how a pollutant moves through soil or water. I’ve watched researchers at environmental agencies battle with cleanup logistics after a misreported compound formula led to flawed models. Lessons like these stick: data accuracy up front saves resources and sharpens responses in real-world scenarios.
Fighting Misinformation with Expertise
Today’s world drowns in chemical information. Not every data sheet on the internet delivers the goods. A minor mistake on a popular database can echo through academic papers, industry protocols, and regulatory documents. Skill and cross-referencing with primary sources help create a safer, more reliable information environment. In my experience fact-checking chemical labels and reference materials, even trusted vendors have mixed up similar-sounding compounds. Vigilance matters, especially for chemicals like 3,4-Dichloronitrobenzene that see use across research and production lines.
Solutions: Trust but Verify
Cross-checking chemical data in multiple peer-reviewed sources stands as a safeguard against mistakes. Laboratories can encourage this by keeping updated copies of reference texts and using digital databases with rigorous editorial oversight. Bring in experience, too. Consulting with a chemist or toxicologist adds another layer of review that software cannot replace. For people new to the field, mentorship and double-checking calculations before ordering or using unfamiliar chemicals stay key. Good habits early keep batch records clean, inventories clear, and workplaces safer.
Takeaways for All Involved
Solid chemical knowledge, built on accurate numbers, keeps projects on track and people protected. The formula C6H3Cl2NO2 and the molecular weight of 192.00 g/mol for 3,4-Dichloronitrobenzene mean more than figures on a page. They are the foundation for science done right and the frontline of safety in every lab and production setting.
How should 3,4-Dichloronitrobenzene be stored?
Understanding the Risks
3,4-Dichloronitrobenzene, a yellowish solid with a sharp smell, plays a role in making dyes, chemicals, and drugs. Despite its usefulness, it can harm both the body and the environment. Skin or eye contact causes irritation, and breathing in its dust or vapors may leave you coughing or feeling dizzy. Accidental spills could pollute water or soil. Treating this chemical with care is no different from how skilled workers treat other flaky or powdery industrial chemicals: with respect, routine, and the right setup.
Picking the Right Container
Sturdy packaging creates the first barrier. Steel drums with airtight lids or high-density polyethylene containers offer tough resistance to leaks or cracks. Weak or thin plastics barely stand up to the corrosive nature of chlorinated solids. Leaving a lid ajar or using a dented drum raises the odds of moisture sneaking in; cakes or clumps become a pain, making dosing and mixing unreliable or even unsafe.
Keeping Cool and Dry
Heat or humidity messes with most organic chemicals. 3,4-Dichloronitrobenzene breaks down or reacts more easily as the temperature climbs or as dampness seeps in. Stocking bags or drums in direct sun can push up the risk of decomposition or pressure buildup. Experienced handlers rely on shaded warehouses or temperature-controlled rooms. Below 30°C, dry, and away from drains or direct sunlight—that setup keeps conditions stable.
Ventilation Prevents Buildup
Vapors and dust gather quickly in closed rooms. Too many people treat “storage room” as a catch-all, but poor airflow lets fumes linger. A fan or mechanical vent does more than just cool the air. It removes stray dust or fumes, cutting the danger for workers. If someone spends time in the storage space, seeing moving air and smelling nothing strange offer reassurance that the setup works.
Labeling and Segregation
3,4-Dichloronitrobenzene shares some traits with other aromatic nitro compounds. Mixing storage with strong acids, alkalis, or oxidizers could lead to unplanned reactions. Bright, chemical-resistant labels warn any user of what sits inside. Old habits, like writing with a marker straight on the drum, invite errors if labels smudge or fall off. Modern storage always leans on printed, laminated tags.
Training Staff and Keeping Records
No storage system works without people paying attention. Unguarded curiosity can lead to spills, and guesswork about safe limits—such as stacking too high or moving containers carelessly—creates unnecessary drama. Regular training and easy-to-access instruction sheets never waste time. OSHA standards, as well as country-specific rules in the EU and Asia, demand such records and signage for a reason. Knowing emergency plans for leaks or exposure, and running drills once in a while, reinforces safer habits every time a new shipment arrives.
Avoiding Shortcuts
Proper storage avoids expensive cleanups or occupational injuries. Investing in reliable shelving, spill kits, and regular floor checks pays off. Many have learned the hard way that rushing saves nothing and only creates headaches later. Talking with peers about storage problems often uncovers better methods and can even reduce insurance premiums.
The Bottom Line
Safe storage doesn’t grow out of rules alone. It relies on habits, organization, and respect for the hazards involved. Every smart move—right container, climate control, clear labels, fresh air—lays another brick in the foundation of a safer workplace. Taking a few extra minutes to double-check details means more security for people, property, and the environment.
What are the potential health hazards of 3,4-Dichloronitrobenzene exposure?
An Unseen Risk in the Workplace
Factories and labs handling chemicals often carry an invisible cost for workers. 3,4-Dichloronitrobenzene isn’t a name people usually see outside safety sheets, but for anyone spending time around paints, dyes, or pesticides, it’s a silent player. Direct exposure can happen fast. A spill on skin, a faint cloud missed by a faulty mask—these simple slip-ups open the door to a pile of health issues.
How Contact Hits Health
I once toured a dye plant where the supervisor described splitting headaches and dizziness, not realizing fumes were the cause. Common symptoms show up as eye and throat irritation, or coughing that clings for days. Long-term exposure digs deeper. It can hurt the liver and kidneys—organs responsible for cleaning the blood. Workers sometimes chalk up tiredness and stomach problems to life’s stress, but these signs may point to something nastier working below the surface.
Inhaled vapors reach the bloodstream fast. Even a small amount triggers sensitivity in some people, flaring up rashes or making breathing miserable for asthmatics. Frequent contact breaks down skin, dragging out healing time for minor cuts and letting more chemicals sneak through.
Cancer Risks and Scientific Evidence
Scientists sound the alarm after testing chemicals like these for years. 3,4-Dichloronitrobenzene belongs to a wider group suspected for cancer links. Animal studies show tumors after repeated exposure, pushing safety agencies to keep a close eye. It doesn’t sit on shelves alone; it often turns up with other industrial toxins, creating a risky mix. Research digs into DNA changes in exposed cells, which build a possible case for cancer over years.
One large study pulled together health records from chemical plant workers and found liver enzyme spikes and, in rare cases, some types of blood cancers. This isn’t just about what’s possible. It’s a real pattern traced through years of records. In countries with lax regulations, these cases climb higher. Living near a dumping site or working in unregulated factories drives up daily intake until the signs become hard to ignore.
What Can Make Things Safer?
It starts with honesty about the real risks. Training sessions save lives, not just to tick a box but to make sure workers know the difference between fatigue from a bad night’s sleep and a warning sign from exposure. Good ventilation stood out in every safe plant I visited. Simple fans paired with chemical filters cut levels inside buildings fast. Face masks, gloves, and eye protection aren’t just accessories—they give real, proven results when used right.
Regular health checks help too. Blood tests can show trouble before symptoms hit hard. Plants with strong medical monitoring catch liver problems early, steering workers away from deep damage. There’s also a strong push for manufacturers to swap out hazardous chemicals for safer alternatives. Some have switched ingredients in dyes or pesticides to lower-risk materials, proving that progress doesn’t mean picking between safety and a working business.
Community pressure carries more weight than people think. Sharing findings, showing health records, and telling stories in plain language lights a fire under both companies and local officials. If the public knows about unsafe handling and demands better, companies notice.
Every worker deserves to finish a shift without wondering what might show up in a blood test years down the line. Honest talk, sound science, and some good old-fashioned vigilance go a long way toward turning silent risks into manageable problems.
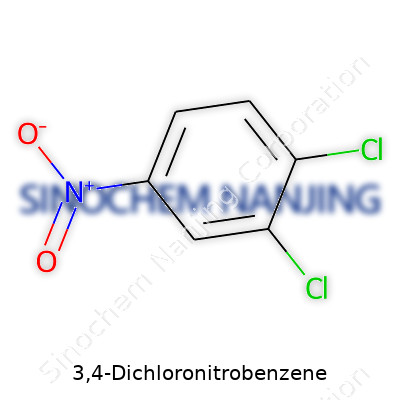
| Names | |
| Preferred IUPAC name | 1,2-Dichloro-4-nitrobenzene |
| Other names |
1,2-Dichloro-4-nitrobenzene 3,4-Dichloronitrobenzol 3,4-DCNB |
| Pronunciation | /ˌθriː,ˌfɔːɹ.daɪˌklɔːroʊˌnaɪtroʊ.bɛnˈziːn/ |
| Identifiers | |
| CAS Number | 99-54-7 |
| Beilstein Reference | 1221187 |
| ChEBI | CHEBI:34496 |
| ChEMBL | CHEMBL20260 |
| ChemSpider | 2777 |
| DrugBank | DB13267 |
| ECHA InfoCard | ECHA InfoCard: 100.006.365 |
| EC Number | 202-746-6 |
| Gmelin Reference | Gmelin 174247 |
| KEGG | C13960 |
| MeSH | D004006 |
| PubChem CID | 8506 |
| RTECS number | DJ8575000 |
| UNII | EW9FSD4T4O |
| UN number | UN1578 |
| Properties | |
| Chemical formula | C6H3Cl2NO2 |
| Molar mass | 202.00 g/mol |
| Appearance | Pale yellow crystalline solid |
| Odor | Odorless |
| Density | 1.6 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.98 |
| Vapor pressure | 0.0027 mmHg (25 °C) |
| Acidity (pKa) | 13.53 |
| Basicity (pKb) | 9.90 |
| Magnetic susceptibility (χ) | -70.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.596 |
| Viscosity | 1.509 mPa·s (at 25 °C) |
| Dipole moment | 3.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 185.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -28.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1682.5 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, in contact with skin or if inhaled. Causes skin irritation. Causes serious eye irritation. May cause damage to organs through prolonged or repeated exposure. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335, H411 |
| Precautionary statements | P261, P264, P270, P271, P273, P301+P312, P304+P340, P305+P351+P338, P312, P319, P330, P337+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3,4-Dichloronitrobenzene: 2-2-2-☠ |
| Flash point | 138 °C |
| Autoignition temperature | 570°C |
| Explosive limits | Lower: 0.9%, Upper: 5.7% |
| Lethal dose or concentration | LD50 oral rat 575 mg/kg |
| LD50 (median dose) | LD50 (median dose): 283 mg/kg (oral, rat) |
| NIOSH | KN0175000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 50 mg/m³ |
| Related compounds | |
| Related compounds |
Nitrobenzene 1,2-Dichlorobenzene 2,4-Dichloronitrobenzene 3,5-Dichloronitrobenzene 3,4-Dichloroaniline 3,4-Dichlorophenol |

