3,4-Dichloroaniline — Progress, Pitfalls, and Prospects
Historical Development
Walking through the corridors of chemical history, 3,4-Dichloroaniline has a story that blends both innovation and controversy. This compound entered synthesis labs near the start of the twentieth century, not long after the rise of the broader family of aniline derivatives. Chemists unlocked a path to chlorinated aromatics during a surge in dye production and pesticide innovation, with 3,4-Dichloroaniline standing out thanks to its versatility in further synthesis. It found work early on as a building block for agricultural chemicals. There was excitement around its technical promise long before regulators thought to ask hard questions about its safety and long-term effect on soils, water, and workers.
Product Overview
3,4-Dichloroaniline looks like a pale solid, often creamy white to light brown, with a distinct, somewhat unpleasant smell. For many years, big chemical plants churned out this material for use in synthesis of dyes, herbicides, and pharmaceuticals. Its popularity stems from its molecular simplicity and the way its chlorine substitutions influence later chemical reactions. This isn’t some rare, exotic reagent, but an industrial intermediate linking the upstream world of base chemicals to the downstream needs of crop protection, colorants, and, increasingly, active pharmaceutical ingredients.
Physical and Chemical Properties
The solid form of 3,4-Dichloroaniline has a melting point just above room temperature, making storage fairly straightforward in most climates but causing concern in summer shipping or for facilities without climate control. The compound dissolves well in organic solvents like ethanol or acetone, and not particularly well in water. Its chlorinated aromatic structure resists easy breakdown by light or mild oxidizers, lending both a stability prized by formulators and a persistence that worries environmental watchdogs. As with most anilines, the presence of the amine group makes the molecule reactive enough to support a range of follow-up modifications.
Technical Specifications and Labeling
Technical purity for commercial batches of 3,4-Dichloroaniline typically runs high, but careful labs check for traces of isomers and related anilines as routine contaminants. Labels warn about its toxic effects, highlighting danger to aquatic life and possible skin sensitization for exposed workers. While regulations on its labeling stiffened over the years, inconsistencies between countries pop up often. This creates headaches for exporters and sometimes lets batches cross borders with labeling that downplays crucial risks. Mistakes don’t just cause regulatory fines— they have real potential to harm people who work with or around this chemical daily.
Preparation Method
Earlier generations of chemists leaned on direct chlorination of aniline to make this compound, juggling temperature and solvent choice to get the right substitution pattern. Nowadays, the route often turns to reduction of 3,4-dichloronitrobenzene, relying on catalytic hydrogenation or iron in acidic media. This switch brought better yields and less byproduct, but side wastes still present environmental burdens. Synthesis remains affordable on a mass scale, but end-of-pipe treatment of wastewater loaded with chlorinated intermediates creates ongoing disputes between manufacturers and surrounding communities. No silver bullet has emerged for making this process “green” by modern expectations.
Chemical Reactions and Modifications
With two chlorine atoms already built in, 3,4-Dichloroaniline acts as a launchpad for many other molecular transformations. N-acylation produces amide derivatives found in older photography and modern pharmaceutical chemistry. Diazotization opens up a world of azo dyes, still staples across textiles worldwide even after a century of debate over dye effluent. Further substitution or coupling turns this core structure into fungicides, anti-malarials, and countless research compounds. Its electron-withdrawing chlorines tune the reactivity, making modifications more selective but also slowing down some reactions. This tunability— coupling stability with directed reactivity— built its legacy as a chemical backbone.
Synonyms and Product Names
3,4-Dichloroaniline goes by a handful of names, none more scientific than its number-based IUPAC moniker: 3,4-dichlorobenzenamine. Trade and regulatory circles sometimes shorten this just to DCA. Those in the agrochemical sector know it as a key intermediate in the synthesis of various active ingredients, though product names generally reference the downstream branded formulations rather than this underlying building block. The proliferation of synonyms across documentation, labels, and research creates confusion for newcomers and leaves room for costly mistakes.
Safety and Operational Standards
Handling 3,4-Dichloroaniline safely means respecting its power as much as its utility. Its toxicity for aquatic organisms and proven health risks for workers have brought tighter controls in recent decades. Protective gear and robust local ventilation belong in any workspace handling open product— there’s no shortcut here, and “just a trace” is enough to raise alarms for chronic exposure. Compliance audits visit producers and downstream users alike, yet enforcement lags enforcement, especially in areas where regulatory agencies are stretched thin. Global trade only amps up the need for consistent standards and real accountability. No one wants another high-profile contamination case where plant workers or nearby rivers pay the price for shortcuts.
Application Area
The real demand for 3,4-Dichloroaniline stems from its role making herbicides, notably diuron and linuron, themselves major tools in controlling weeds in fields and along public rights-of-way. Some modern medicines also trace a chemical path back to this intermediate. Dyestuff creators, especially in legacy textile regions, built whole color offerings around azo compounds produced with 3,4-Dichloroaniline as the starting point. Environmental regulators never let these uses off the hook, since residues in water and crops remain an ongoing headache. Here’s the tension: agriculture and chemical industries rely on this chemistry to keep yield and profits up, but the cost to ecosystems and public health pushes researchers to find replacements or better containment solutions.
Research and Development
Academic and industrial labs keep tinkering— with greener syntheses, less hazardous byproducts, and clearer understanding of its downstream chemistry. Advances in catalysis offer glimmers of hope for cleaner preparation. Analytical chemists focus on tracking trace contamination, helping regulatory agencies flag hotspots before they fester into full-blown crises. Computational modeling also lends a hand, predicting both synthetic pathways and environmental fate. Broader adoption of high-throughput experimentation promises faster answers, but the social impact side— local trust, long-term health studies— still demands direct, human-driven research. Investments from governments and big firms aim for dual wins: the economic stability of the chemical industry, and a safer footprint for communities and workers.
Toxicity Research
After so many decades on the scene, 3,4-Dichloroaniline has amassed a thick file of toxicology studies. Animal trials and environmental screening showed clear warning signs: bioaccumulation in aquatic life, skin and eye irritation in exposed workers, and likely links to liver and kidney damage with long-term exposure. Water monitoring near chemical plants and downstream from major agricultural operations find this molecule and its breakdown products at worrisome levels. Public health authorities in some countries now scan for it as part of routine drinking water checks, though detection limits and reporting lag behind actual risk in poorer settings. Chronic exposure issues still haunt remediation efforts nearby old dump sites. There’s no turning back the clock, so focus shifts to better sampling, transparency, and support for those hurt before modern standards landed.
Future Prospects
Eyes remain on the evolution of both regulations and real alternatives to 3,4-Dichloroaniline-dependent chemistry. Some agrochemical players pivot to enzyme-based weed killers or focus on rotating crops to lessen herbicide use outright. Dye manufacturers, pressured by major retailers and new laws, experiment with plant-based colors and future-proof molecules that break down quickly in water. Technologies that capture process waste at the factory or break down residues in situ before they reach waterways inch toward affordability and scale. At the same time, chemical engineers rethink classic synthetic routes, searching for cleaner, less chlorine-intensive pathways. Though demand stays steady in traditional markets, the strongest scientific voices call for a future where 3,4-Dichloroaniline moves from staple to specialty— used only where no safer, cleaner substitute gets the job done. Real change won’t just come from breakthroughs in a lab, but from society’s willingness to prioritize health and ecosystems over chemical output for its own sake. The next chapter depends less on new patents and more on how regulators, firms, and communities work together to balance real economic gains against long-term stewardship of land, water, and public trust.
What is 3,4-Dichloroaniline used for?
The Quiet Workhorse in Everyday Chemistry
3,4-Dichloroaniline sometimes drifts through the background in talks about chemical building blocks. People chalk it up as just another complex name on a label, but its uses reach further than expected. My first brush with it happened during a summer spent helping a family friend at a local water treatment testing lab. For me, that string of chemicals on bottles all looked the same, but this one kept popping up for pesticide research. I didn’t realize until much later how it fuels dozens of products many of us touch or eat every day.
More Than a Lab Ingredient
At its core, 3,4-Dichloroaniline acts as a stepping stone in chemistry. The largest demand comes from making herbicides, especially the well-known linuron and propanil. These chemicals keep weeds under control in fields of carrots and rice. Companies use this compound because small changes to the molecule make all the difference in how the final product behaves in soil. Farmers end up with tools for better yields. But it doesn’t stop there. This same compound finds its way into fabric dyes, pigments for plastics and ink, and some rubber chemicals as well. Each time I toss out old sports shoes with faded soles, I can picture someone in a factory using this chemical in the process.
Touching on Safety and Health
Problems show up because 3,4-Dichloroaniline doesn’t just disappear. There are real concerns about its presence in waterways and the soil. When I talk to my chemist friends, they mention the headaches regulators face tracking residues in food crops and runoff. Its persistence pushes lab teams to run detailed tests. Studies have shown chronic exposure in rodents led to some worrying outcomes, like liver and kidney changes. Agencies like the Environmental Protection Agency flagged it as a possible concern for long-term health, especially for workers handling it every day. Gloves and protective gear aren’t optional in plants where it gets processed. In a strange twist, traces of this chemical sometimes stick around from the very breakdown of certain pesticides, spreading the issue beyond its initial use.
Environmental Questions
Communities living downstream from big farms or factories sometimes hear about 3,4-Dichloroaniline in relation to polluted wells. It can last a long time in water and sediment. That means local wildlife feel the effects far into the future. One group of researchers I shadowed for a story found low levels turning up in fish tissue. Sensitive aquatic species end up as warning signs long before people notice anything is off. Some watchdog groups now press both companies and agencies for deeper monitoring and new research into better breakdown methods.
Rethinking the Approach
I often hear from experts that switching out key industrial chemicals takes patience and new ideas. Rule changes alone won’t get us far without stronger partnerships between government, independent scientists, and the chemical sector. Some green chemists are testing ways to redesign molecules for faster breakdown. Tech firms look for water treatment upgrades that handle these stubborn pollutants. Communities tracking their drinking water offer valuable insights about where these problems start. Even the simple act of reading labels, voting for stronger standards, or supporting farms and brands working to phase out risky chemicals matters. Real progress means connecting the dots between farms, industry, regulators, and the people and places relying on clean water, safe food, and trust in what flows downstream.
Is 3,4-Dichloroaniline hazardous to health?
Why People Should Care About 3,4-Dichloroaniline
Talk to anyone in chemical manufacturing, they’ll mention 3,4-Dichloroaniline popping up as a key building block for dyes, pesticides, and pharmaceuticals. Someone working near these chemicals—or even those living next to a plant—deserves to know what exposure really means. Breathing or touching chemicals has life-long impact, even when you cannot see or feel it right away.
Stories from the Field and Science
I’ve spoken with workers who spent years mixing pigments, and some stories stay with you. Imagine after a long shift, your skin itches or your eyes burn. The body reacts to exposure quickly, and chemicals like 3,4-Dichloroaniline do not disappear when you wash your hands. Short-term contact causes skin and eye irritation, and if you accidentally breathe dust or fumes, you might cough or feel lightheaded.
Look at animal studies—rats and mice inhaling or eating even small amounts show damage to liver, kidneys, and blood. Researchers point out that 3,4-Dichloroaniline can break red blood cells, dropping the oxygen your body moves around. The International Agency for Research on Cancer lists chemicals like this as having a suspicion for cancer risk, though more research is still ongoing.
Real-World Exposure
Walk through a plant floor during production and you see powdered dyes and pesticides loaded on trucks, and the dust clings to everything. There’s risk for workers, but also local families if chemicals escape through water or air. Farmers using pesticides with 3,4-Dichloroaniline handle concentrate that gives off fumes, and sometimes it runs off into groundwater or streams.
Tests from environmental agencies find this compound in river sediment and soil where factories discharge waste. Over time, wildlife absorbs it, and this can lead to smaller populations or mutations. Community drinking supplies picking up trace amounts might pose health hazards, especially for children or those with weaker immune systems.
Protecting Health and Finding Solutions
There’s plenty people can do. Training workers about gloves, goggles, and proper ventilation cuts risks. Proper chemical storage, spill plans, and clean-up routines make a huge difference. Many factories replace older equipment that leaks or breaks easily, ensuring less contact for staff.
Strict environmental rules help protect towns near factories. Plants must treat waste, monitor air and water, and regularly report leaks. Community groups push for cleaner technology, including alternatives for dye and pesticide synthesis. The chemical industry responds when people stand together and demand change. For farmers, special guidance on safer handling stands out. Switching to less toxic pest and weed treatments also helps both workers and rural health.
Why the Conversation Should Continue
Every household deserves clean water and safe air. Over time, minor exposures build up. Health authorities urge erring on the side of safety, pushing for regular health checks, workplace monitoring, and transparency in chemical use. Researchers continue to track long-term effects so future generations have answers we might not.
3,4-Dichloroaniline isn’t a household name, but it affects more lives than people realize. Whether you live by a factory, work with dyes, or grow crops in treated fields, keeping people informed keeps everyone healthier and safer.
What are the storage requirements for 3,4-Dichloroaniline?
Understanding the Hazards
Anyone who’s worked around labs or chemical storage sheds recognizes the need for solid practices when dealing with compounds like 3,4-Dichloroaniline. Used in manufacturing dyes, pharmaceuticals, and pesticides, this compound doesn’t seem like much on paper. Under the wrong conditions, though, it brings up safety risks that nobody wants to deal with: respiratory irritation, skin contact problems, and chronic toxicity over time. Growing up near a busy agricultural area, I’ve seen firsthand what happens when hazardous products aren’t stored right—strong odors, ruined protective gear, even folks needing medical attention for exposure.
Key Storage Principles
Every bottle and drum demands respect. The first rule: keep it cool, dry, and ventilated. Moisture promotes slow breakdown and unexpected hazards. Warm air does nobody any favors—higher temperatures make volatile compounds more active, which can cause both slow leaks and rapid, dangerous jumps in vapor levels. For 3,4-Dichloroaniline, stick to a temperature below 30°C, ideally closer to ambient, and keep it away from sunlight or any bright artificial lighting. The warehouse in my last lab had a row of shaded racks ten feet from any heater, and that simple move cut down on product loss and smell immediately.
Separation and Segregation
This chemical doesn’t get along well with oxidizing agents, acids, and strong bases. Sitting it next to bleach, nitric acid, or even certain cleaning products can lead to nasty surprises. Chemical safety data backs this up: even minor contact with oxidizers can ignite fires or cause toxic fumes. Every storage map or cabinet should clearly label incompatible substances. Segregation is more than a simple rule—it matters for everyone from the delivery staff to the senior researchers.
Container Integrity
Choose glass or HDPE (high-density polyethylene) containers. Metal corroded right on the shelf in one older facility I visited, allowing dark liquid to seep and stain an entire cabinet. Screw caps and seal rings become the frontline of defense. Regular checks for cracks and loose lids prevent larger accidents down the line. Over time, vapors can eat at rubber or cheap plastic stoppers, which means containers need routine inspection and prompt replacement.
Labeling and Documentation
Too often, faded or missing labels cause confusion or worse: chemical mishaps. On one job, a mislabelled bottle led to hours wasted as supervisors hunted for the right neutralizer after a small spill. Use waterproof labels, clear warning symbols, and keep detailed records of storage dates and amounts. Everyone on shift should know where the chemical sits and how much holds in stock, which makes emergency response smoother.
PPE and Access Control
Access isn't for everyone. Only trained folks should handle storage areas, wearing gloves, goggles, and long sleeves. Extra care makes a huge difference, especially since skin contact brings its own list of problems. I’ve seen the result of shortcuts—itchy rashes, wasted hours with first aid kits, and, once, a trip to the ER. Limiting entry, keeping staff up to date on procedures, and running regular drills all help avoid preventable trouble.
Emergency Preparedness
Spill kits, eye wash stations, and ventilated storage rooms serve as backup plans. I once worked in a place that kept neutralizing materials next to the chemicals. We learned quickly: move them, add signage, and create a routine for monthly drills. Based on safety statistics, fast, confident action limits both harm and paperwork after an incident.
How should 3,4-Dichloroaniline be handled safely?
Hands-On Safety: More Than Just Gloves
Anyone who has spent time handling chemicals knows that certain names carry more weight in the lab or on the production floor. 3,4-Dichloroaniline falls into that camp. It serves a purpose in dyes, pesticides, and other industrial staples, but you won’t see anyone jumping at the chance to work with it unguarded. The reason is clear: this compound poses real health risks, including skin and eye irritation, respiratory issues, and possible links to carcinogenic activity.
From years of working around hazardous substances, I’ve seen firsthand what happens when people take shortcuts. A colleague had a bad day with a related aniline compound — a split glove meant blistered fingers. Not a scare story; just a lesson learned. Direct skin contact with 3,4-Dichloroaniline invites burns, rashes, and longer-term health worries. So, heavyweight nitrile or neoprene gloves, chemical splash goggles, and lab coats become second nature. Not because the rulebook says so, but because experience proves light gear or shortcuts can mean painful recovery or worse.
Air Quality in the Real World
Breathing in the dust or fumes from 3,4-Dichloroaniline gives more trouble than just a cough. The chemical can linger in lungs and airways, and over time, that means real risks — not just a day of discomfort, but potentially issues that follow folks home. Ventilation isn’t just a box to tick. A simple fan won’t cut it; purpose-built fume hoods or extraction systems genuinely make breathing easier and safer.
Workers often get used to the presence of chemical smells, but no nose works as a substitute for good detectors and airflow systems. The Occupational Safety and Health Administration (OSHA) presses for regular monitoring, and I’ve seen places get cited for letting dust settle instead of clearing the air. It’s not paperwork — it’s real protection.
Unseen Risks: Storage and Waste That Matter
3,4-Dichloroaniline doesn’t play well with sunlight or open air. A leaky lid or a poorly labeled drum can turn a safe workplace into a danger zone overnight. Metal containers with strong seals, stored in shaded, cool rooms, help keep the substance inert and contained. Up-to-date labels and inventory logs mean everyone knows what they’re picking up and when it entered storage.
Disposal looks like someone else’s job — right up until it isn’t. Dumping leftovers or contaminated gear in standard trash brings legal trouble and, more importantly, community health hazards. Trained hazmat teams handle disposal, often arranging for incineration or approved chemical treatments. Environmental Protection Agency (EPA) guidelines come into play, and anyone tempted to cut corners only has to look at the fines and cleanup bills on public record.
Building Better Habits: Training Everyone
Training for workers must go beyond a once-a-year slide deck. Real safety comes from hands-on drills, regular “what-if” walkthroughs, and open lines for reporting near misses. I’ve seen teams that talk through close calls — not to punish, but to prevent. That sort of transparency actually reduces injuries and boosts morale.
3,4-Dichloroaniline sticks around in the environment, and it doesn’t forget the mistakes of the past. Responsible teams review safety data, stay current on local and federal rules, and hold each other accountable. Safe handling isn’t a checklist — it’s a habit learned by people who respect experience and want everyone to get home with their health intact.
What is the chemical formula and structure of 3,4-Dichloroaniline?
Breaking Down the Chemistry
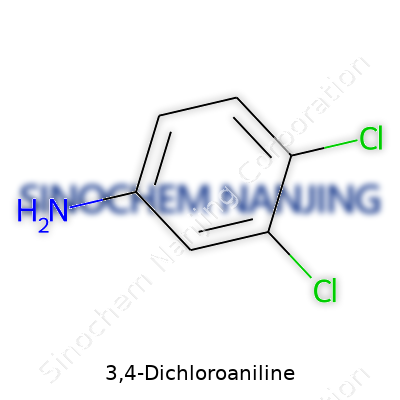
3,4-Dichloroaniline grabs attention not just because of its formula, but also because it pops up in real, everyday situations for those who work with chemicals or environmental analysis. Its straightforward chemical formula, C6H5Cl2N, paints a clear picture: this compound branches off from the aniline family, but takes a twist with two chlorine atoms gripping the benzene ring. You’ll find them clinging to positions three and four, which gives this chemical its name.
I’ve handled a range of aromatic amines in my own time in the lab, and it’s always surprising just how much a simple switch — like adding chlorine atoms — changes what a molecule does and how it acts. For 3,4-Dichloroaniline, the extra chlorine impacts not just its physical character, but its role in bigger industrial and environmental stories.
Visualizing the Structure
At the molecular level, 3,4-Dichloroaniline keeps things recognizable. You’ll see a benzene ring as its skeletal core. At spot number one, the amino group (-NH2) signals the “aniline” part. Then, the third and fourth carbons on the ring each grip a chlorine atom. Line it up on paper, and the structure tells you right away why this chemical shows up in so many synthetic pathways — the pattern makes it ideal for manufacturing pigments, pharmaceuticals, and especially pesticides.
The bulky chlorine atoms on the ring and the presence of the amino group bring out chemical properties quite distinct from plain aniline. Naming conventions aren’t just for show; they reflect real differences. The placement of those chlorines influences electron distribution in the molecule, and that means changed reactivity. That’s something you can see first-hand in organic synthesis: you might hope to attach another group, but those chlorines put up roadblocks or steer reactions in new directions.
Why This Compound Matters
This isn’t just a textbook molecule. 3,4-Dichloroaniline gets used heavily in producing agricultural chemicals, dyes, and drug intermediates. Plenty of herbicides and fungicides owe some part of their activity or resistance to variations of this compound. Farmers, chemists, water safety teams — they all cross paths with 3,4-Dichloroaniline, whether they realize it or not.
Its use doesn’t come without downside. The molecule’s strong structure means it doesn’t break down easily. Years back, I worked on trace pesticide analysis in river samples near farm fields, and more than once, 3,4-Dichloroaniline popped up in results. If you’re dealing with water quality or soil safety, this substance hits home as a marker of contamination. That sticks in your mind when you know how persistent organics travel far from where they started. Long-lasting residues build up in ecosystems, hitting aquatic life and even working their way up the food chain.
Looking for Smart Solutions
Tackling the issues around 3,4-Dichloroaniline involves responsibility at several levels. You can’t just ban all useful chemistry, but you can push industry and research towards safer alternatives. Stronger guidelines for responsible disposal and careful monitoring around crop fields go a long way. In the lab, better synthesis plans and greener chemistry keep hazardous byproducts from reaching waterways. Driving awareness starts with understanding why this formula and structure matter, not just memorizing them. That kind of practical, hands-on knowledge lays the groundwork for chemical safety and smarter use down the line.
| Names | |
| Preferred IUPAC name | 3,4-dichlorobenzenamine |
| Other names |
3,4-Dichlorophenylamine 3,4-Dichloro-benzenamine 3,4-Dichloroanilin 3,4-Dichloraniline |
| Pronunciation | /ˌθriː,ˌfɔːr.daɪˈklɔːroʊ.əˈnɪliːn/ |
| Identifiers | |
| CAS Number | 95-76-1 |
| 3D model (JSmol) | `3D model (JSmol)` string for **3,4-Dichloroaniline**: ``` NC1=CC(Cl)=C(Cl)C=C1 ``` This **SMILES** string can be used to render the 3D structure in JSmol. |
| Beilstein Reference | 922833 |
| ChEBI | CHEBI:34498 |
| ChEMBL | CHEMBL14217 |
| ChemSpider | 7676 |
| DrugBank | DB14089 |
| ECHA InfoCard | 03aab33c-6a2b-48c5-83ec-f75fb47dc6d4 |
| EC Number | 202-456-5 |
| Gmelin Reference | 7172 |
| KEGG | C06591 |
| MeSH | D002601 |
| PubChem CID | 7921 |
| RTECS number | BX8575000 |
| UNII | 80T2D6T48A |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C6H5Cl2N |
| Molar mass | 162.04 g/mol |
| Appearance | White to slightly beige crystalline solid |
| Odor | aromatic |
| Density | 1.43 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.67 |
| Vapor pressure | 0.0025 mmHg (25°C) |
| Acidity (pKa) | 3.51 |
| Basicity (pKb) | 7.70 |
| Magnetic susceptibility (χ) | -63.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.604 |
| Viscosity | 1.33 mPa·s (25 °C) |
| Dipole moment | 2.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 151.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -56.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3606 kJ/mol |
| Pharmacology | |
| ATC code | N01AX10 |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic in contact with skin, toxic if inhaled, causes damage to organs through prolonged or repeated exposure, very toxic to aquatic life |
| GHS labelling | GHS05, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301, H311, H331, H373, H411 |
| Precautionary statements | Precautionary statements: P261, P264, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P330, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-2-2 Health:2 Flammability:2 Instability:2 |
| Flash point | 138°C |
| Autoignition temperature | 630°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 650 mg/kg |
| LD50 (median dose) | LD50 (median dose): 650 mg/kg (oral, rat) |
| NIOSH | RN0162 |
| PEL (Permissible) | PEL: 0.5 mg/m3 |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | IDLH: 100 mg/m³ |
| Related compounds | |
| Related compounds |
2,4-Dichloroaniline 3,5-Dichloroaniline 2,3-Dichloroaniline 2,6-Dichloroaniline 4-Chloroaniline 3-Chloroaniline Aniline |

