3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride: Insights and Commentary
Historical Development
Chemists started looking at biphenyls and their derivatives in the early 20th century, mostly chasing stronger, more versatile materials for dyes and polymers. Early patents focused on simple diaminobiphenyls, then tweaks added methyl groups. By the 1960s, 3,3'-dimethyl-4,4'-diaminobiphenyl hydrochloride marked an important step. Industrial syntheses aimed to improve yields and keep out impurities common with multi-step aromatic chemistry. Demand in pigment and specialty rubber production pushed research, simply because the resulting materials held color better and ages didn’t yellow as quickly. Without those early advances, the specialty coatings and fibers folks rely on today might not have developed the same way.
Product Overview
This compound sounds a little imposing, but at heart it’s a light yellow crystalline solid, stable under most reasonable lab conditions. Chemists in dyes, electronic parts, even biomedical engineering value it for the way its amine groups interact with other aromatic rings. People often choose it as a reaction intermediate, thanks to its reactivity and ability to anchor other groups. Experience in process labs shows that careful control of its hydrochloride salt keeps the dust down and makes its dispersal less risky, which matters since uncontrolled particulates in air can create both health and fire hazards.
Physical & Chemical Properties
Look at its structure—a rigid biphenyl backbone, two methyls jutting off the rings, and a couple of amines at the para positions. In practice, this means it melts between 280 and 290°C and dissolves pretty well in polar solvents, but stands up to both heat and weak oxidizers better than the parent biphenyl diamine. Hydrochloride salt formation improves handling: powders clump less, solutions stay more stable, and contamination risk drops. Many labs check every incoming batch for melting point consistency and off-odors, since an off-profile usually means decomposition or poor storage.
Technical Specifications & Labeling
Check the bottle in any research lab and one thing jumps out: labeling details matter. Besides stating purity (most products range from 97 to 99%), reputable manufacturers provide lot number, moisture content—sometimes as “loss on drying”—and maximum allowable levels of heavy metals. Granule or powder form, storage at room temperature, and strong advice to keep it sealed away from direct sunlight repeat across safety data sheets. Smart buyers read Certificates of Analysis against in-house standards, since regulatory compliance rests on paper as much as on actual product.
Preparation Method
Production usually starts with methylation of biphenyl, followed by nitration and careful reduction. Methyl groups don’t just show up; their introduction sometimes calls for harsh conditions, and leftover reactants can cause real headaches downstream. Post-nitration reduction relies on catalytic hydrogenation under controlled temperature, where any slip can set off unwanted side reactions or reduce yield. Once the diamine forms, bubbling hydrochloric acid gas produces the stable hydrochloride salt, with filtration and drying steps to pull out the cleanest product. Any industrial chemist knows the pains of optimizing for both purity and scale—especially since customers in the electronics or dye business absolutely hate variable results.
Chemical Reactions & Modifications
Those two amine groups make the compound a workhorse for further functionalization. Crudely put, they let you stick on acyl groups, form azo bonds in dye work, or build bigger aromatic systems for electronic uses. Oxidation, diazotization, and even cross-coupling reactions—each one works differently according to pH, temperature, and solvent. Process chemists juggle these reactions constantly, tuning conditions to get target molecules without side-products, because purification costs rise fast if the chemistry drifts out of control. Recent literature still chases greener reaction conditions without giving up yield, since scaling up sometimes exposes safety or waste issues missed at bench scale.
Synonyms & Product Names
Ask five chemists, get five different names. Some call it 3,3'-dimethylbenzidine dihydrochloride, others refer to it as ortho-tolidine hydrochloride, and certain catalogs list obscure code numbers. Trade names change by supplier, and researchers switching vendors often double-check structures to avoid misordering. Labels matter. One lab disaster years ago involved a misidentified drum, and that mistake forced a full cleanout and months of testing to prove the facility safe. Internal naming discipline has improved, but mixing up closely related biphenyl diamines remains a risk if teams don’t check documentation carefully.
Safety & Operational Standards
Anyone handling aromatic amines knows to take precautions. Both short-term exposure and chronic low-level contact with compounds like 3,3'-dimethyl-4,4'-diaminobiphenyl hydrochloride raise health risks. Protective gloves, dust masks, even full cartridge respirators make sense in synthesis labs and production facilities. Eye protection goes without saying. Spills must be handled promptly—absorbent pads, wet-wiping to minimize dust, and thorough ventilation. Safety data sheets (SDS) call out skin sensitivity, possible carcinogenicity, and environmental hazards, especially because breakdown products can persist in wastewater. Local regulators check disposal protocols; violators face stiff penalties, so responsible labs set clear procedures and refresh staff training at least yearly.
Application Area
Industrial users reach for this compound mostly in pigment and dye production, where its amine groups help form stable, lightfast colorants. The electronics industry uses modified biphenyls for certain circuit board materials, taking advantage of stability at high temperatures. Research into specialty polymers, particularly those needing rigid aromatic backbones, keeps finding new uses for 3,3'-dimethyl-4,4'-diaminobiphenyl derivatives. Environmental monitoring sometimes uses it as a test compound—it shows up in some protocols measuring trace nitrite, for example. My time working in an analytical chemistry lab hammered home how a single new application can drive production scale unexpectedly, stretching supply chains and forcing quality teams to meet new, tighter purity demands.
Research & Development
A lot of current R&D focuses on making its derivatives safer or more sustainable. Chemists work to build more biodegradable polymers using biphenyl diamine cores, hoping these new materials can match the performance of the old hydrocarbons without long-term pollution risks. Some groups pursue analytical uses, tweaking the aromatic amines for better sensitivity in sensors. Academic teams push for synthesis routes that eliminate harsh reagents or reduce solvent use, driven both by green chemistry targets and tightening regulations. In industry, stability tests keep uncorking surprises—UV light and ozone sometimes degrade products faster than expected, spurring runs of accelerated testing and forced storage tests.
Toxicity Research
The track record for aromatic amines isn’t great. Animal studies and limited occupational surveys suggest elevated cancer risks from repeated exposure, though exact dose-response data remains patchy for rare derivatives like this one. Many countries classify it as a suspected carcinogen, spurring extra labeling and strict worker monitoring. Water solubility adds challenges: any leak or improper disposal risks marine contamination, and researchers have noted disruption of aquatic organisms’ reproduction cycles at even low concentrations. While acute toxicity symptoms—like mucous membrane irritation—show up fast, the deepest worry lies in what traces of these compounds do over years or decades. There’s ongoing pressure from environmental agencies and advocacy groups to either ban or tightly regulate all related biphenyl diamines. Labs find themselves balancing productivity with responsibility, looking for both technical fixes and safer operational habits.
Future Prospects
Demand isn’t going away soon. Functional dyes, high-performance pigments, certain plastics, and even analytical reagents all draw on this chemistry. Still, the push for safer, greener alternatives grows louder each year. Some start-ups investigate renewable origin aromatic amines or crack catalytic cycles that bring compostable polymers into the mainstream. Smart R&D money looks for formulations needing smaller super-active doses or entirely new end uses outside of familiar pigments and plastics. Legal and regulatory changes could quickly reshape the market; proactive firms already invest in alternatives or partner with universities to stay ahead of safety rules. The real test comes as new risks or side-effects emerge—no chemist wants another asbestos-style legacy, so listening to early-warning studies and locking in solid data handling practices matter as much as any flashy new breakthrough.
What is the primary use of 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride?
Peeling Back the Layers in Practical Chemistry
You don’t often hear anyone drop the name 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride over dinner. Even in chemistry circles, only certain researchers or industrial chemists ever handle it. Spend enough time in a lab or chemical manufacturing plant, and you learn which raw materials get used nonstop and which stick to more specialized jobs. This one lands squarely in the second camp, but its impact runs deeper than most realize.
Why the Dye Industry Relies on This Compound
Step inside a textile factory, and you see the story: rich reds, deep blues, intricate patterns. Many synthetic dyes in those fabrics draw life from base chemicals like 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride. This material feeds into the process for building azo dyes, one of the biggest families of colorants used to color textiles, plastics, and inks. That’s the main ticket—the compound supplies intermediate building blocks for dye synthesis. If you follow the chemical steps, it acts as a diamine for coupling with other materials, forming vivid, stable colors.
Factories value this chemistry for practical reasons: control, consistency, and bright, lasting colors that stand up to repeated washing. One reason it won’t ever disappear from industry shelves is simple—there’s still no cheaper, cleaner substitute that gives the same results across so many color formulas. Over the decades, dye engineers have tweaked the recipes, but this ingredient holds its ground.
Beyond Dyes: The Small Shadows in Polymer Chemistry
Dive a bit deeper, and you find this compound also has a part in specialty polymers. Some plastic manufacturers use it for fine-tuning polymers that need specific thermal or electrical traits. It lets chemists pinpoint just the right flexibility or heat resistance, especially for electronics or high-performance coatings. Plastics and coatings made this way often end up in cars, appliances, or complicated gadgets.
Weighing Up Health and Environmental Risks
I worked once with a team tasked with reviewing old dye recipes for toxicology and environmental risk. One hard lesson: compounds like these rarely show up on the front page, but they matter for worker safety and downstream pollution. Many aromatic amines (the broad chemical family here) show links to toxicity or carcinogenic behavior if handled poorly.
A few landmark studies and occupational reports show why real attention pays off. Handling this biphenyl means proper gloves, fume hoods, and careful disposal. Discharging waste into water sources ends up accumulating in soil and rivers, affecting everything from fish to future crops. Growing restrictions in Europe and the US mean tighter limits, more regular testing, and research into alternatives. Despite the drive for safer chemicals, the reality is these old recipes stick around where cost and performance matter more than easy substitution.
Tracking Solutions: Safer Handling and Sustainable Chemistry
Getting beyond the risks starts with education and accountability. Regular safety training, honest labeling, and checks at every handoff cut down on accidents. Some startups and academic labs have started publishing new routes to replace troublesome amines in dyes or polymer work, and industry watchdogs push for green chemistry initiatives. Still, every chemical swap means reworking what factories know and trust—which takes years and real investment.
Even as stricter rules roll out, demand for bright, bold colors in everyday goods sticks around. Part of the solution comes from creating better systems for waste handling and investing in more sustainable chemical research, so that stories about toxic byproducts and worker harm grow rarer each year.
What are the storage conditions for 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride?
Straight Talk on Chemical Storage
Few things in the lab set off more alarms than improper chemical storage. Each compound comes with its own quirks, and 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride (let's call it DMABP-HCl) is no different. Anyone who has handled specialty amines knows moisture, light, and heat remain the big threats. Sometimes it feels like the simplest slip-up — a loose cap or an unlabeled jar — snowballs into bigger risks for everyone nearby. People trust that chemicals are being locked away with care, and it’s worth diving into what that really takes with something like DMABP-HCl.
Why This Stuff Matters
DMABP-HCl finds its way to research spaces for a reason. Chemistry professionals count on its reactivity and purity. Those who have worked with aromatic amines remember the issues: many break down when stacked next to oxidizers or left in humidity. A single exposure to open air can set off decomposition, turning a useful tool into a safety hazard. Health studies over the past two decades point at chronic exposure risks, ranging from skin irritation to more severe health effects over time. Nobody wants to deal with a slow-release hazard hiding on a forgotten shelf.
Solid Habits for Storage
Glass containers with tight-fitting lids do the heavy lifting against leaks and spills. Some labs use colorless bottles, but I’ve found amber glass gives extra insurance against stray light. Fluctuating temperatures breed condensation — so keeping the climate cool and steady matters. I remember once sweating over a box left near a vent; that moisture warped labels in less than a week, making a simple inventory check a scavenger hunt.
Dedicated shelving away from acids, bases, and oxidizers stops incompatible mixes from causing fires or toxic fumes. It’s tempting to stack jars anywhere open, but good storage draws clear boundaries. Labels don’t just help with ID; they also include hazard warnings and the date of receipt, keeping accountability front and center. I’ve seen too many labs with faded or handwritten notes that can’t be read in an emergency. Digital logs back up hard labels for added traceability.
Avoiding Moisture and Air Problems
Moisture control makes all the difference with hydrochloride salts. Desiccators — those ever-present glass chambers with silica gel — keep things bone dry. Anyone handling chemicals in a coastal or humid city soon learns that even quick transfers outside the desiccator can spoil sensitive compounds. Some labs use those small, reusable packet desiccants inside jars. That redundancy pays off, especially if the main chamber gets busy or overcrowded.
Fire and Disposal Safety
Even if DMABP-HCl doesn’t catch flames as easily as solvents, treating it with respect helps everyone breathe easier. Stash it away from ignition sources. Maintain up-to-date safety data sheets within arm’s reach. Used containers and waste need to hit proper hazardous disposal streams, not regular trash bins. This point gets hammered home during audits for a reason — mixing industrial waste with common garbage can bring fines or worse, cause hospital trips from cross-contamination.
Looking to the Future
New guidelines from regulatory bodies keep shifting as knowledge grows. Staying current means hitting up recent chemical safety updates, and never relying on memory from training ten years back. In my own work, I run refresher sessions and label checks monthly. It keeps me sharp and catches small problems before they endanger a whole team. No shortcut replaces regular vigilance. Good storage culture forms the backbone of any lab that values safety and progress in equal measure.
What is the chemical structure and molecular weight of 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride?
Chemical Footprint: Structure and Weight
Sitting in a research lab, pipette in hand, I have watched curiosity turn into facts under a microscope. Not every molecule tells its secrets easily, but 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride draws attention right from its structure. This compound stacks together two benzene rings by a direct carbon-carbon bond. It's not just a simple biphenyl. Methyl groups decorate the 3 positions on each ring, like small signposts, with each 4-position bearing an amine group. Pouring in hydrochloric acid adds the hydrochloride salt, stabilizing the whole package for handling and synthesis. The chemist in me appreciates these deft tweaks—the dimethyl and diamino configuration creates unique reactivity and function.
The molecular formula looks like this: C14H18N2·2HCl. Calculating the molecular weight brings a result of about 285.2 g/mol for the base, but when combined with hydrochloride (two chloride ions and two extra hydrogens), the total value climbs to about 358.24 g/mol. Accurately weighing out this compound in the lab involves trusting those numbers and knowing exactly what lands on the scale. Skip the math, and you risk botching an entire synthesis or analysis.
Where Structure Meets Use
Benzidine derivatives like this don't just live on a shelf. People draw on them for serious work in synthetic chemistry, dye manufacture, and even some pharmaceutical pathways. The tweaks in structure—each methyl, each amino—change how aggressively it can react, what color it brings, and how it survives in a mixture. For decades, biphenyl diamines paved the way for deeper knowledge about electronic interactions, resonance, and how small changes in structure can mean large swings in function. I’ve watched reactions shift color before my eyes, the result of swapping one group for another or changing ring placement.
Real concern shadows these uses. Aromatic amines show up on lists of chemicals under scrutiny for toxicity and mutagenicity. The story doesn’t stop at pretty molecular drawings. In industries, safety goggles and gloves mark the day-to-day dance with these molecules. Regulatory documents tie each step. During a graduate project, I spent more time reviewing MSDS pages than actual benchwork some weeks, not because I love paperwork, but because the risks are real. Limiting exposure and managing waste demands diligence and respect for the molecule’s structure as much as its theoretical value.
Facing the Future: Solutions Rooted in Evidence
We don’t just ponder chemical structure for idle curiosity. Folks in industry and academia track molecular weights and ring configurations because new rules keep arriving. Better understanding lowers the odds of accidents and environmental fallout. Substitution and green chemistry lie on the horizon; finding less hazardous alternatives drives curiosity in young chemists. Process chemists regularly tweak molecules to keep useful properties while sidestepping the properties that put safety under pressure. Improving detection technology, pushing for tighter air and water monitoring, and investing in personal safety gear become part of daily life, not just afterthoughts. The effort to minimize risks attaches straight to knowledge about structure and weight, making these facts far more than textbook trivia.
Is 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride hazardous or toxic?
Understanding the Substance
3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride does not pop up on most people’s radar unless you work in a lab or an industrial setting where specialty chemicals take center stage. This compound, usually manipulated behind strict safety lines, can show up in some polymer or dye processes. Whenever you see a compound with multiple rings, methyl groups, and amine functions, a red flag on health always waves somewhere in the background. My own experience with similar organoamine derivatives often boiled down to one rule: keep exposure as close to zero as possible.
Routes of Exposure and Health Risks
Dust in the air, careless glove use, or that splash when weighing powder—every one of these means risk. Evidence from toxicology studies suggests many aromatic amines, especially ones with extra methyls, pose real hazards. They do not just wind up as throat or nose irritants; many interact with DNA and cell machinery, raising cancer risks. The hydrochloride part means it comes as a salt, and does not magically make it safe. In practice, that only changes how it dissolves or moves around in your workspace.
Handling this chemical with unprotected skin can lead to burns or delayed allergic reactions. If you breathe the dust, there is a chance for lung and nose irritation. Some lab workers have reported skin rashes and even more severe reactions after weeks of exposure, with cumulative effects showing up in behavior and memory. Nobody wants to play around with something that can knock health sideways, even if the process is lucrative.
Worker Safety and Cancer Concerns
Cancer agency databases, such as those run by the International Agency for Research on Cancer (IARC), have logged various substituted biphenyls as possible carcinogens. While not every variant carries the same level of threat, chemicals in this family share similar red flags. Multiple published studies trace the links between amine-containing biphenyls and bladder or liver cancers. That drives home the point: limit handling, reduce airborne dust, ensure airtight containers, and lean on engineered controls.
Personal anecdote: in one chemical plant job, we replaced open scoops with enclosed transfer systems, and respiratory problems nearly vanished overnight. This demonstrates the effectiveness of simple controls over theoretical risk assessment. Relying on personal protective equipment alone does not stack up; processes and design matter even more for real safety.
Safe Handling and Mitigating Risks
Every chemical deserves respect, and this one more than most. Storage in sealed containers, collection of dust with vacuum systems, and regular medical checks for exposed staff pay dividends. I remember the best plants always ran on repeated training sessions and open channels for reporting symptoms. That built a real safety culture, not just a checklist of good intentions.
Substituting such hazardous substances with less toxic alternatives, especially in dyes or polymers, sets a practical roadmap for companies aiming to protect workers. If replacement is not possible, layering controls—think fume hoods, automated weighing, and protective suits—becomes non-negotiable. Safety data sheets should never gather dust, and workers should know exactly what procedures exist in case of exposure or spills.
Weighing the Value and the Hazard
Discussing value versus hazard gets personal quickly. Living with lingering doubts about that headache or rash makes no paycheck feel secure. Prioritizing health and safe practices while considering the science behind each compound forms the most honest, trustworthy workplace. Science keeps advancing, and one day, we’ll cut out such hazards entirely. Until then, the best defense is vigilance and making the safer choice every time a tough decision about chemicals lands on the table.
What is the recommended method for handling and disposing of 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride?
The Risks Overshadow Convenience
Every time I’ve worked with aromatic amines in a laboratory, extra steps always come with the territory. 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride isn’t any different. Even though it seems stable on the shelf, the potential hazards sit right beneath the surface. Studies show links between some aromatic amines and severe health conditions, including cancer. A compound like this doesn’t just pose respiratory and skin hazards — it lingers on surfaces and can stay stable long enough to cause trouble days after a spill.
Without strict personal protective equipment — gloves made from nitrile, safety glasses, fitted respiratory protection, and a dedicated lab coat — every movement in the lab turns risky. Washing hands isn’t enough. Anyone who has cleaned up after a careless spill can confirm: these chemicals love to cling to anything porous. That’s why confined, ventilated workspaces such as fume hoods become non-negotiable. It’s not paranoid; it’s what experience teaches after enough close calls.
Storage: More Than Just a Label
People sometimes try to save space by shoving specialty chemicals onto crowded shelves. From my time running college stockrooms, I learned a quick shortcut here can backfire fast. 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride needs a sealed, labeled container — ideally glass — kept in cool, dry conditions away from acids, oxidizers, and food. Grouping with incompatible substances increases accidental risks, a lesson hammered home by more than one minor incident over the years.
Disposal Done Right
No amount of optimism replaces careful disposal. This isn’t a “just toss it” situation. Standard drains or general trash bins can’t handle chemicals with toxic and persistent properties. Waste companies spell it out: treat aromatic amine hydrochlorides as hazardous waste, keep them isolated, and document everything. Properly designed waste streams keep these chemicals out of waterways, soil, and the food chain. Ignoring those steps brings real-world harm.
In my early years, I saw what happens when staff skip paperwork or mix incompatible lab waste. I’ve watched waste containers swell from pressure and vents struggle to keep up with untracked chemicals. Most cities and institutions require hazardous waste pick-up. Signing chemicals in and out of a central log holds everyone accountable. Waste must sit in leak-proof, labeled containers, away from anything else, until a licensed team takes over. Anything less turns an inconvenient job into a full-blown safety incident.
Why Care? It Matters to All
People sometimes roll their eyes at strict procedures. Still, good handling habits don’t just shield the researcher; they protect janitors, waste haulers, and finally the wider environment. The way we manage chemicals like 3,3'-Dimethyl-4,4'-Diaminobiphenyl Hydrochloride draws a pretty clear line between safe science and carelessness. Even small decisions — putting on gloves, documenting use, storing chemicals up high — add up over the long haul.
Some universities and companies hold regular waste awareness sessions. They push for more training, smarter lab design, and easier access to material data sheets. Real progress starts at the bench and, in my experience, with each of us refusing to take shortcuts. No one ever regrets taking an extra minute for safety, but plenty have regretted the opposite. By making chemical handling a priority, people and the planet stay safer in ways that regulations alone can’t guarantee.
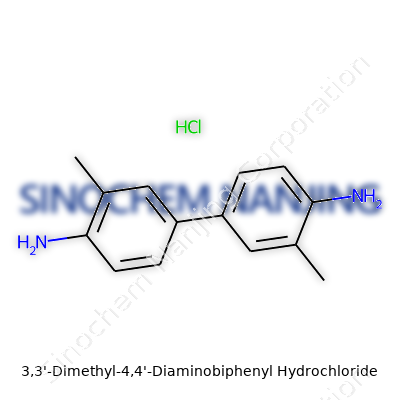
| Names | |
| Preferred IUPAC name | 4,4'-Diamino-3,3'-dimethyl-1,1'-biphenyl dihydrochloride |
| Other names |
3,3’-Dimethylbenzidine dihydrochloride o-Dianisidine dihydrochloride 3,3’-Dimethylbenzidine hydrochloride 4,4’-Diamino-3,3’-dimethylbiphenyl dihydrochloride o-Benzidine, 3,3’-dimethyl-, dihydrochloride |
| Pronunciation | /ˈθriˌθri daɪˈmɛθɪl ˌfɔːrˌfɔːr daɪˈæmɪnoʊˈbaɪˌfɛnɪl haɪˌdrɒklaɪd/ |
| Identifiers | |
| CAS Number | [612-68-6] |
| Beilstein Reference | 1204297 |
| ChEBI | CHEBI:51464 |
| ChEMBL | CHEMBL3709862 |
| ChemSpider | 71716 |
| DrugBank | DB16675 |
| ECHA InfoCard | 07b478a9-39d5-4d5c-98b2-929a898d5c89 |
| EC Number | 211-413-2 |
| Gmelin Reference | 82340 |
| KEGG | C06710 |
| MeSH | D014222 |
| PubChem CID | 20804622 |
| RTECS number | DU8050000 |
| UNII | DF807UM784 |
| UN number | Not regulated |
| Properties | |
| Chemical formula | C14H18ClN2 |
| Molar mass | 286.82 g/mol |
| Appearance | Light yellow to brown powder |
| Odor | Odorless |
| Density | 1.15 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.7 |
| Acidity (pKa) | 3.70 |
| Basicity (pKb) | 3.20 |
| Magnetic susceptibility (χ) | -4.2×10^-6 cm³/mol |
| Refractive index (nD) | 1.658 |
| Dipole moment | 4.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 222 J K⁻¹ mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS05,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P333+P313, P362+P364, P363, P501 |
| Flash point | > 222.3 °C |
| Lethal dose or concentration | LD50 oral rat 3000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 350 mg/kg (oral, rat) |
| NIOSH | Not established |
| PEL (Permissible) | PEL: Not established |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Benzidine 3,3′-Dimethoxybenzidine 3,3′-Dichlorobenzidine 4,4′-Methylenedianiline 4,4′-Oxydianiline |

