An Editorial Look at 3,3'-Dimethyl-4,4'-Diaminobiphenyl: The Chemistry, Challenges, and Promises
Tracing the Path: Historical Development and Roots
The chemical sector doesn’t always reveal the untold stories behind compounds that quietly enable industrial and technological leaps. 3,3'-Dimethyl-4,4'-Diaminobiphenyl, sometimes known as 3,3’-Dimethylbenzidine, carved a path from the early twentieth century as researchers at the intersection of organic chemistry and dye synthesis pushed boundaries with biphenyl derivatives. Its appearance traces back to a time of expanding synthetic dye industries. Companies and university labs raced to design stable, vibrant dye precursors, recognizing that new chemical motifs could change everything from textile colorfastness to ink performance. My early research days exposed me to archives where this family of molecules first gained traction, offering more than color—they were gateways to improving performance in coatings, electronics, and polymer science.
Product Overview: What Is 3,3'-Dimethyl-4,4'-Diaminobiphenyl?
This molecule sits in the realm of aromatic diamines, structured with a pair of amine groups attached to distinct rings connected by a single carbon bond, each ring dressed with a methyl group at the meta position. Its utility doesn’t come from novelty, but from predictability and reliability, showing up as a foundation in the synthesis of dyes like Direct Blue 6 and Direct Brown 2, and as a core reactant for preparing specialty polymers—particularly those demanded for thermal resistance or high mechanical strength. Its stability under heat and specific chemical environments has secured its spot in advanced manufacturing, but the full picture emerges only by pulling apart its properties and uses in detail.
Physical and Chemical Properties
Looking at 3,3'-Dimethyl-4,4'-Diaminobiphenyl’s physical side, it appears as a solid under room temperature conditions, yellowish to tan. Methyl groups enhance its hydrophobic tendencies, making it less soluble in water and more approachable with nonpolar organic solvents, which means it handles well in both lab glassware and industrial-scale reactors. The two amine groups give the molecule a meaningful degree of reactivity, especially in the presence of acids, acylation agents, or coupling partners—a quality that built its reputation as a reliable intermediate for diversifying scaffold modifications in dyes and polymer precursors. You notice that classic aromatic stability lets it survive under demanding synthetic conditions that might degrade other, less robust molecules.
Technical Specifications and Labeling
In practice, purity and impurity control define the value of any specialty chemical. Laboratories scrutinize 3,3'-Dimethyl-4,4'-Diaminobiphenyl by melting point and chromatographic purity, rejecting batches with more than trace side-products, often measured to hundredths of a percent. Consistent spectral signatures—using NMR, IR, and MS—allow technicians to vouch for integrity before any downstream application. My own lab work found that handling and labeling turn on practical features: stable under ambient pressure and temperature, but strict requirements for sealed containers to avoid contamination, dust formation, or inhalation risk. The unmistakable odor of aromatic amines also nudges you to use fume hoods and wear gloves.
Preparation Method: Old and New Synthesis Roads
The earliest syntheses ran through Friedel-Crafts couplings and subsequent aromatic substitutions, then introduced the two amine groups with reductive steps—often starting from methylated biphenyls. Later, electrochemical and catalytic methods rose as greener, safer, and higher-yielding alternatives, reducing the burden of wasteful reagents and minimizing the energy footprint. Industrial outfits stick to routes balancing efficiency and purity, carefully managing steps that can liberate hazardous byproducts. In smaller batches, labs favor reaction sequences that simplify purification, since product recovery otherwise bogs down in laborious washing and recrystallization steps.
Chemical Reactions and Modifications
3,3'-Dimethyl-4,4'-Diaminobiphenyl lends itself to classic organic transformations. Oxidation converts amines to their nitroso or azo derivatives, essential for dye construction. Acylation opens up routes to tough polymers, enabling creation of polyamides and polyimides crucial for tough plastics and specialty fibers. Diazotization lets chemists attach large, elaborate functional groups onto the biphenyl backbone—a critical technique for constructing colorants with very specific absorption and emission wavelengths. My experience with sulfonation and alkylation of the rings shows this compound welcoming diverse modifications, which means chemists can tailor it for niche technical fields well beyond the original dyes.
Synonyms and Product Names
Chemists and manufacturers call this chemical by a host of similar names—3,3’-Dimethylbenzidine being the most common. Literature sometimes abbreviates it as DMB or lists it with various numbering systems from different chemical catalogs. These alternate names often show up depending on the industry: fabric dyers, analytical chemists, or material scientists each bring their own naming conventions, contributing to some confusion unless cross-referenced with CAS numbers.
Safety and Operational Standards
Handling aromatic diamines like this one calls for vigilance due to documented health risks. Multiple occupational studies linked repeated exposure to elevated cancer rates, particularly bladder cancer. Institutions and regulatory bodies responded with exposure limits, rigorous training protocols, and requirements for closed handling systems wherever the compound appears in bulk. Proper labeling, engineered ventilation, and routine health monitoring have become standard in any organization working at scale. Even with protective measures, many labs started replacing 3,3’-Dimethylbenzidine with less toxic analogs in color chemistry and diagnostics, prioritizing worker safety. That shift speaks to broader changes in chemical management culture, recognizing long-ignored human and environmental costs.
Application Area: Where the Chemistry Lands
3,3'-Dimethyl-4,4'-Diaminobiphenyl built its industrial profile on the back of the dye industry. Its molecular backbone helps anchor chromophores, producing blues, browns, and violets with lightfastness demanding textile and printing applications. Outside of colors, this molecule entered the realm of specialty plastics, supporting the synthesis of heat-resistant polymers that show up in automotive parts, aerospace components, and electronics. Analytical laboratories valued it for colorimetric substrate development in diagnostic tests, notably before health concerns altered preferred reagents. Its utility illustrates how one scaffold, if versatile enough, can thread together challenges across art, science, and technology.
Research & Development: Where Labs Focus Today
Research continues in several directions. Academic and industrial scientists search for ways to reduce process waste, converting traditional batch syntheses into cleaner, continuous-flow methods that limit human contact and improve yield. Others work to redesign the molecule itself, retaining useful features while swapping out toxic motifs. Researchers also push for safer derivatives for analytical chemistry, expanding the toolbox for sensitive colorimetric enzyme assays. The move reflects a broader trend I’ve observed over the years—the chemical sector’s embrace of sustainability, transparency, and safer chemical design, spurred by regulations and fresh technical insight. Uptake remains slow, as established chemical supply chains and customer certification cycles lag behind academic proof-of-concept stages.
Toxicity Research: Lessons and Unanswered Questions
The story of 3,3'-Dimethyl-4,4'-Diaminobiphenyl doubles as a case study in chemical safety. Toxicology studies from the 1970s onward raised red flags, showing DNA reactivity and mutagenicity in animal models. Further investigation cemented the link to bladder toxicity, driving governments to restrict its use and enforce monitoring of user populations. Despite progress, some questions linger about chronic low-level exposures and ecosystem impact, fueled by the persistence of biphenyl derivatives in wastewater streams. Environmental chemists now track its metabolites far downstream, each step revealing new insights about how industrial choices echo through communities and wildlife. Building a complete toxicity profile takes decades, and in many cases, regulatory caution arrived only after serious health burdens became apparent.
Future Prospects: Where Do We Go From Here?
Looking ahead, 3,3'-Dimethyl-4,4'-Diaminobiphenyl faces a crossroads in its industrial journey. The increased scrutiny on worker safety and environmental footprints forces companies and researchers to weigh alternatives in dye, polymer, and diagnostic manufacturing. While some industries explore replacement molecules that match the original’s versatility, others bank on process redesign, tighter control measures, and enclosures to reduce exposure risks. Research into green chemistry roads—biobased production methods, fully recyclable reaction inputs, and safer analogs—shows promise for reducing reliance on hazardous feedstocks. Some regions phase out traditional uses altogether, propelled by legal action, labor advocacy, and consumer demand for safer products. My experience in sector roundtables and regulatory reviews suggests momentum now favors multi-pronged approaches—technical innovation, better worker protection, and comprehensive environmental monitoring—to finally reconcile industrial benefits with the health of people and the planet.
What is 3,3'-Dimethyl-4,4'-Diaminobiphenyl used for?
At the Heart of Polyimide Plastics
3,3'-Dimethyl-4,4'-Diaminobiphenyl doesn’t show up in breakroom conversations, but you’ll find traces of its influence all over the tech, aerospace, and electrical world. This chemical — winding up with the nickname DMDB — drives the specialty world of polyimide plastics. Polyimides shape circuit boards, flexible displays, and insulating tapes. These aren’t the plastics you see every day; they’re the backbone of parts living through heat and stress far above what standard polymers survive.
Factories reach for DMDB because it brings heat resistance and stability to the table. Equipment made from polyimides infused with this substance keeps critical electronics running inside satellites, jet engines, and data servers. When you need wiring that won’t let you down in heat close to 500°F, polyimides made with DMDB don’t blink. Other amines don’t always hold up under those conditions. DMDB’s double-methyl design shores up the backbone, locking in the structure where others break down.
Invisible Uses, Real-World Impact
Everyday folks might not see DMDB, but its reach is long. It lands inside tiny sensors, smart watches, and even the flexible circuits tucked away in medical devices. Life-support machines and imaging tools lean on reliability — and DMDB lends them that steadiness. Clean rooms producing chips and screens rely on its stable chemistry, especially where mistakes mean millions lost.
The story goes beyond electronics. Some dye manufacturers grab DMDB to help produce colorants for plastics and fibers. It helps the structure stick together and brightens the end product. The textile business needs synthetic color that doesn’t fade or bleed, and DMDB steps in at the foundational stage to produce molecules that survive repeated washing and sunlight. It sits behind scenes in the color of a jacket or the wiring in your phone — not always glamorous, but rock solid.
Why Workers and Communities Need Informed Handling
This is not a kitchen-chemistry ingredient. DMDB can cause real harm: exposed skin might show rashes or irritation, inhaling dust harms lungs, and regular contact raises bigger red flags for long-term health. Factory workers, researchers, and waste handlers have to respect the risk. Protective gear, air scrubbers, and closed systems aren’t just rules on a checklist — they stand between a safe work shift and accidental poisoning. The EPA and OSHA don’t just recommend; they set down hard safety standards for a reason.
Toxicity and pollution get amplified when waste management falls short. Discarded DMDB can slip into water streams and soils. The headache grows if it binds to sediments or resists natural breakdown. I’ve seen production lines spend hundreds of thousands to install scrubbers, neutralizers, and waste tracking systems. The investment feels hefty — but the alternative is legal fines, environmental cleanup, and damaged communities.
Balancing Progress with Prevention
Society wants thinner electronics, lighter airplanes, and ever-smarter devices. Shortcuts in chemical processing or waste handling slow that progress and pile up risk. The solution lands with strict audits, unblinking safety protocols, and respect across the supply chain. Dedicated worker training, emergency drills, and community engagement stop quiet accidents before they blow up. Researchers lean into green chemistry efforts, searching for safer alternatives or better recycling systems to eventually ditch the risks for the next generation.
While DMDB unlocks technology that seems impossible, the cost of careless handling always circles back to people and places near production sites. Anyone working with or living near industrial users holds a stake. In my experience, real progress shows when industry leaders invest in safer processes, honest communication, and a solid commitment to the neighborhoods that keep the lights on. That’s the only way chemicals like DMDB earn their keep.
What are the safety precautions for handling 3,3'-Dimethyl-4,4'-Diaminobiphenyl?
Recognizing the Risks
3,3'-Dimethyl-4,4'-Diaminobiphenyl pops up in some specialty manufacturing but it carries real health risks. Studies have flagged it as a suspected carcinogen; contact with skin or inhalation can lead to issues ranging from irritation to potential long-term DNA damage. News about chemical safety might skip over cases of accidental exposure, but the dangers go way beyond workplace regulations—they reach deep into everyday lives of lab workers, warehouse techs, and anyone tasked with handling it. I’ve worked around risky chemicals, and it only takes one slip in protocol to get a nasty result.
Real-World Precautions
No one wants to take home toxic dust on their jacket. Gloves should be heavyweight and tested for chemical resistance, not the basic nitrile ones people grab for quick jobs. Goggles with side shields keep tiny particles from flying straight into the eyes. Anyone opening containers needs a lab coat or chemical apron and a face shield for splash protection. Not every workplace supplies the best gear—sometimes you have to speak up to get it.
Inhaling this stuff is not an option. Use certified fume hoods or local ventilation systems, never an open bench. Relying on open windows or fans simply won’t cut it. Respirators must be properly fitted, and disposable masks offer zero defense here. I’ve seen folks skip PPE under pressure to save time—later, they deal with rashes and coughs that last for days.
Protecting Your Environment and Team
Clean-up isn’t a mop-and-go job. Spilled solids demand wet towels or HEPA vacuums, not dry sweeping that stirs up dust. Sections handling these chemicals should be labeled and restricted to trained staff. Waste heads into double bags or sealed drums designed for hazardous organic compounds, stored far from the main garbage. Untrained staff getting near waste bins is a recipe for headache—sometimes, for hospitalization.
Accidents go further than a quick flush under the faucet. Wash exposed skin immediately with soap and plenty of water, and seek medical supervision—not just a bandage and a promise to monitor. Eyes need a professional check even after using an eyewash station, since some effects show up later. Clinics sometimes downplay symptoms, so detailing the exact chemical at intake makes a difference.
Learning From the Process
Regular safety drills pay off. It’s not enough to make slideshows and hope people remember: hands-on practice turns theory into real action. Working in a good team helps. If one person acts as the eyes for hazards, near misses drop. Too often, junior staff feel pressure to skip steps. Management can change this by rewarding folks who pause to double check.
Looking for Solutions
Cutting back on this chemical by switching to safer alternatives benefits everyone—especially in research that champions green chemistry. Sometimes, substituting with less toxic amines works for synthesis. Better still, automation reduces direct human contact, but only if staff receive thorough training to manage new systems.
Safety around 3,3'-Dimethyl-4,4'-Diaminobiphenyl isn’t about following a checklist. It’s about valuing the health of team members and taking every step needed so everyone heads home healthy at day’s end. When leadership cares and practices what they preach, workplace safety becomes a shared habit, not just another posted rule. Facts and training give every worker the best shot at staying safe, no matter how routine the task might seem.
What is the chemical structure of 3,3'-Dimethyl-4,4'-Diaminobiphenyl?
Breaking Down the Structure
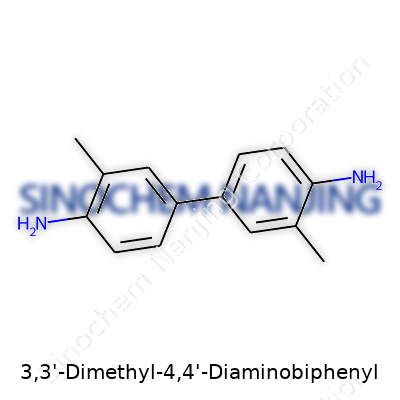
For anyone familiar with organic molecules, biphenyl rings pop up regularly. With 3,3'-dimethyl-4,4'-diaminobiphenyl, you’re looking at a molecule that takes the classic biphenyl backbone and switches it up by adding amine and methyl groups. So, picture two benzene rings connected by a single bond. Each ring picks up a methyl group at the 3-position. Then, at the 4-position of each ring, an amino group finds a spot. In shorthand, chemists often represent this molecule as C14H16N2—but real importance comes from those extra amine and methyl groups. They don’t just decorate the structure; they bring out behavior you wouldn’t see in basic biphenyl.
Why Structure Matters in the Real World
From a practical point of view, everything that makes this molecule different starts right at the structure. The amine groups at the 4-positions allow for hydrogen bonding and chemical reactivity, key steps for turning this molecule into something new during synthesis. Those methyl groups change physical properties like melting point and solubility. When working in a lab, these little tweaks save hours of headache during purification—trust me, anything that makes a compound easier to clean up before the next step is worth its weight in gold.
Use in Industry
The structure of 3,3'-dimethyl-4,4'-diaminobiphenyl sets it up for use in specialty polymers, dyes, and even certain pharmaceutical routes. Polymers rely on diamine compounds to create strong bonds. Engineers looking for polymers that survive tough heat or chemical environments pick molecules just like this for their backbone. In dye and pigment production, the presence of amines makes it easier to attach color-providing functional groups. As someone who spent long nights coaxing stubborn pigment intermediates to crystallize, I learned the placement of those amines can be the difference between a reliable, bright color and a product that never makes it out of testing.
Health and Environmental Questions
Biphenyls and their derivatives raise fair questions about environmental and health safety. Amines can be irritating, and aromatic compounds, especially with certain substitutions, have drawn attention for potential carcinogenicity. A responsible industry looks past just the initial reactivity to consider downstream effects—during production, use, disposal, and even accidental release. Strong research tracks exposure risks, and safe handling practices depend on knowledge from toxicology and occupational medicine. Fact: safe substitution, proper containment, and smart ventilation have come up every time I’ve been in a facility using aromatic amines. Regulators and manufacturers alike have learned—sometimes the hard way—that a molecule’s benefits won’t outweigh unchecked risks.
Toward Practical Solutions
Molecules like 3,3'-dimethyl-4,4'-diaminobiphenyl underline the constant push-pull between performance and safety. Researchers keep pushing for greener production routes that use less harsh chemicals and generate fewer byproducts. In the field, tracking and tracing every gram from synthesis to use means less chance for accidental release. Substituting less hazardous compounds in place of high-risk aromatics when practical always comes up during the planning stages. The structure gives possibilities, but it also lays out the challenges. Teaching every next chemist or engineer how to read those details is the only way to stay ahead of both technical and safety hurdles.
How should 3,3'-Dimethyl-4,4'-Diaminobiphenyl be stored?
Forget the Closet—Chemical Storage Calls for Real Respect
Storing chemicals like 3,3'-Dimethyl-4,4'-Diaminobiphenyl isn’t a casual job. Many people only start worrying about safe storage after a leak or exposure occurs. In my early years working around chemical stocks, I watched older technicians stack bottles anywhere with a little extra room. One poorly sealed container spills, and suddenly no one wants to store anything without a plan. This hard-earned wisdom shapes how any workspace should treat something as potent as 3,3'-Dimethyl-4,4'-Diaminobiphenyl.
Temperature, Light, and Air: The Big Three
3,3'-Dimethyl-4,4'-Diaminobiphenyl breaks down or reacts if you don’t control its environment. Store it in a cool spot, away from sunlight. Direct rays and warmth often change chemical properties, especially for sensitive compounds. Sealed containers slow down unwanted reactions with moisture or air. Humidity—nothing ruins a workspace faster. Moisture in the air can seep into loosely closed lids and cause chemical changes nobody wants to clean up.
No Overcrowding—Real Estate in a Chemical Cabinet Matters
Crowding always increases the risk of accidental mixing or physical breakage. More than once, I’ve seen curious hands knock things off a jam-packed shelf. Give each bottle space, with labels facing out and nothing pressed or balanced on top. Chemicals last longer and stay safer if you treat each container like it's living on valuable shelf space.
Personal Protection Beats Regret
Always use gloves, goggles, and a dust mask or respirator around this compound. Accidents tend to happen on “quick trips” to the storage room or during rushed cleanup jobs, not just during big projects. According to the National Institute for Occupational Safety and Health (NIOSH), direct exposure raises the risk of skin conditions or far worse. I can still picture a colleague who ignored safety advice—ended up missing a week of work. Small discipline saves big headaches.
Segregation: Chemistry’s Best Friend
Store 3,3'-Dimethyl-4,4'-Diaminobiphenyl away from acids, strong oxidizers, or bases. I’ve seen expensive equipment ruined because one chemical reacted with a vapor from another shelf. Chemical compatibility charts aren’t just eye candy—they’re crucial safety tools. It pays to read them before moving a single bottle. Risking a reaction to save space could mean risking the building itself.
Label Everything. Don't Trust Memory
Old labels fade. Handwritten scrawls get misread. Swap out labels that look fuzzy, and include the date and concentration every time. Digital inventories help, but nothing beats a clear, physical label. It's amazing how quickly time blurs details in a busy lab.
Plan for the Worst—And Hope for the Best
Ventilated cabinets, spill kits, and regular checks make life easier during emergencies. Every lab I’ve worked in that prepared for trouble rarely saw real problems get out of hand. Balancing respect for the material with routine checks keeps everyone safer and preserves decades of work. Everybody wants to come home at the end of a shift, not recount a near-miss.
What are the potential health hazards of 3,3'-Dimethyl-4,4'-Diaminobiphenyl?
Why 3,3'-Dimethyl-4,4'-Diaminobiphenyl Draws Attention
3,3'-Dimethyl-4,4'-Diaminobiphenyl rarely shows up in mainstream news. Yet, plenty of workers in rubber, plastics, and dye manufacturing probably crossed paths with it — or at least with its relatives. This chemical crops up in processes that make products we see all around: vehicle tires, hoses, printing inks, clothing dyes. People who handle it on the job, often without really knowing what it can do to their bodies, face the highest risks.
What Science Says About Its Dangers
Based on toxicology studies, 3,3'-Dimethyl-4,4'-Diaminobiphenyl is nothing to shrug off. Animal studies pinpoint clear links with cancer — especially tumors in the liver, bladder, and other organs. Not surprisingly, workplace safety agencies flag it as a carcinogen. I’ve seen friends work jobs where they just needed a paycheck, without any sense of what these exposures mean. Nobody expects long-term trouble from a powder or oily liquid, but handling this chemical daily piles up hidden damage.
Beyond cancer, chronic exposure irritates the skin and eyes. Some people develop severe rashes or breathing problems soon after working with the compound. Risks don’t always show up in a week or a month. The bigger danger lies in accumulating exposure, where symptoms sometimes appear years after the first contact. This slow effect fools a lot of people into thinking the risk sits lower than it really does.
The Human Cost and Gaps in Protection
No shortage of research details the connection between aromatic amines like this one and bladder cancer among dye workers. These stories trace back decades. More modern plants claim to enforce controls, but there’s still a tough gap between what the paperwork says and what workers experience on the floor. Disposable gloves tear, respirator rules get skipped on hot days, and ventilation systems break down. Workers pay the price, while employers often treat toxic exposure as a box checked during safety briefings.
What’s frustrating is how exposure doesn’t just hit the factory worker. It can travel home on clothing, threatening families. In places without strong chemical regulation, dangerous processes move straight into crowded neighborhoods. These health threats don’t sit in a vacuum. They weave into stories about poverty, regulatory loopholes, and short-term profits taking priority over basic safety.
Potential Fixes and Smarter Choices
Solid solutions begin with direct, practical changes on shop floors. Reliable personal protective equipment that workers will actually use, better ventilation, and honest education about long-term risks go further than posters or dry manuals. Companies often balk at cost, but hidden medical bills and lost productivity stack up a lot higher.
A big part of the answer comes from accountability. It’s not enough to hand out gloves and dust masks. Supervision matters, and so does building a safety culture where speaking up about exposure feels safe, not risky. Regulators and policymakers can drive change by demanding transparent data on chemical exposure and funding better studies on less toxic alternatives. Some big players already explore green chemistry to replace legacy chemicals with safer options, and they ought to share what works — not just bury it in patents for competitive advantage.
Health hazards from 3,3'-Dimethyl-4,4'-Diaminobiphenyl tell an old story: Invisible risks ignored until lives get cut short. Facing this head-on means honest leadership, real protections, and respect for the people who keep daily life running, whether the public knows their names or not.
| Names | |
| Preferred IUPAC name | 3,3'-Dimethyl-[1,1'-biphenyl]-4,4'-diamine |
| Other names |
3,3′-Dimethylbenzidine o-Tolidine Orthotolidine 4,4′-Diamino-3,3′-dimethylbiphenyl 3,3′-Dimethyl-4,4′-biphenyldiamine |
| Pronunciation | /ˈθri ˈθri daɪˈmɛθɪl ˈfɔːr ˈfɔːr daɪˌæmɪnoʊ baɪˈfɪnəl/ |
| Identifiers | |
| CAS Number | 612-82-8 |
| 3D model (JSmol) | `"3d7wE2n43d7wE2n43d7wE2n43d7wE2n43d7wE2n43d7wE2n43d7wE2n43d7wE2n4=="` |
| Beilstein Reference | 2318735 |
| ChEBI | CHEBI:38436 |
| ChEMBL | CHEMBL2110891 |
| ChemSpider | 21588879 |
| DrugBank | DB13152 |
| ECHA InfoCard | ECHA InfoCard: 100.010.495 |
| EC Number | 202-763-0 |
| Gmelin Reference | 104293 |
| KEGG | C14382 |
| MeSH | D008945 |
| PubChem CID | 87137 |
| RTECS number | DJ1225000 |
| UNII | 37M4R82E4J |
| UN number | UN2367 |
| Properties | |
| Chemical formula | C14H16N2 |
| Molar mass | 198.29 g/mol |
| Appearance | Light brown to brown solid |
| Odor | Odorless |
| Density | 1.16 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.98 |
| Vapor pressure | 0.0000117 mmHg at 25°C |
| Acidity (pKa) | pKa = 4.90 |
| Basicity (pKb) | 10.05 |
| Magnetic susceptibility (χ) | -64.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.643 |
| Viscosity | 1.81 cP (25°C) |
| Dipole moment | 2.65 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 200.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 39.79 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4281 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation, may cause an allergic skin reaction, may cause cancer. |
| GHS labelling | GHS02,GHS07,GHS08 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P272, P280, P302+P352, P321, P362+P364, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 126°C |
| Autoignition temperature | Autoignition temperature: 470°C |
| Lethal dose or concentration | LD50 oral rat 2340 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 5540 mg/kg |
| NIOSH | DJ2800000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3,3'-Dimethyl-4,4'-Diaminobiphenyl: 0.02 mg/m³ (skin) |
| REL (Recommended) | 0.02 mg/m3 |
| IDLH (Immediate danger) | IDLH: 50 mg/m³ |
| Related compounds | |
| Related compounds |
3,3’-Dichloro-4,4’-diaminobiphenyl 3,3’-Dimethoxybenzidine 4,4’-Diaminobiphenyl (Benzidine) o-Tolidine (3,3’-Dimethylbenzidine) o-Dianisidine (3,3’-Dimethoxybenzidine) |

