3,3'-Dimethoxybenzidine: A Detailed Commentary
Historical Development
3,3'-Dimethoxybenzidine has roots stretching back over a century, tracing its path through the colorful history of synthetic organic chemistry. In the early 1900s, rapid industrialization pushed chemists to explore aromatic amines and their derivatives for new dyes and pigments. This compound emerged during those times as part of a wider search for robust intermediates to serve the growing textile and print industry. Demand for colorfastness in fabrics and efficiency in synthetic processes made discoveries like 3,3'-Dimethoxybenzidine valuable. Chemists weren't thinking of environmental impact or toxicity during its birth; the priorities focused on performance and production scale. The chemical structure offered just the right properties for diazo coupling reactions, which became vital in making vibrant blues, violets, and greys, propelling its industrial relevance. Over time, 3,3'-Dimethoxybenzidine found itself surrounded by regulatory debate and health concerns, but its historical roots still shape how industries approach development and safety today.
Product Overview
Producers have used 3,3'-Dimethoxybenzidine as a key intermediate in the manufacture of certain dyes and pigments, especially those meant for textiles and inks. Its ability to serve as a precursor in azo-coupling reactions, where vivid colors arise from linking aromatic rings, has made it a staple in the dye sector for decades. Besides coloring fabrics, it has shown up in formulations for inks, plastics, and paper. Chemists looking for a reliable starting point for specific chromophores often turned to this particular benzidine derivative because methoxy groups on the rings offer electronic tweaking, leading to sharp, consistent color outcomes. That's a big reason it stayed relevant across shifting demands in manufacturing.
Physical & Chemical Properties
Taking a look at 3,3'-Dimethoxybenzidine in its pure form reveals a pale brown or yellowish crystalline solid, though batches sometimes lean toward off-white depending on purity and preparation. It's not very soluble in water but dissolves reasonably well in organic solvents like ethanol or acetone. A molecular formula of C14H16N2O2 translates to predictable behavior in most reactions: methoxy groups slow down oxidation compared to the simpler benzidine, yet keep the backbone ready for substitution and further modification. The aromatic structure carries two amino groups in para positions relative to each other, both shielded by the methoxy rings—a factor that keeps reactivity in check and helps tailor downstream reactions. Its melting point can provide a quick quality check, with minor deviations flagging possible impurities.
Technical Specifications & Labeling
Anyone handling or sourcing this compound needs to keep an eye on assay levels—purity above 98% for technical applications is standard for most labs and factories. Consistent color and crystalline form act as quick markers to gauge the handling environment's quality; clumping, off-odors, or significant dusting might signal storage issues or cross-contamination. In my experience, proper labeling not only tracks concentration or batch but also ensures traceability, which becomes critical during contamination scares or quality audits. Each container must show clear hazard markings aligned with local and international regulations, especially in labs or shipping docks, since oversight in this area can bring sudden shutdowns or shipment rejections.
Preparation Method
Making 3,3'-Dimethoxybenzidine draws from classic organic chemistry. The process starts with the nitration of dimethoxybenzene to generate dinitro derivatives. These intermediates undergo catalytic reduction, often using iron filings and hydrochloric acid. Careful attention to temperature and pH prevents over-reduction or unwanted byproducts. Some plants have moved to catalytic hydrogenation to improve efficiency, but traditional reduction routes still dominate, especially in regions where infrastructure lags behind. The entire sequence demands robust ventilation and chemical waste management—metallic iron, strong acids, and leftover organics line up as pollution risks all along the chain. Unless operators follow careful handling and containment, residues make their way into local groundwater or emissions.
Chemical Reactions & Modifications
With two primary amino groups, 3,3'-Dimethoxybenzidine gives chemists a versatile handle for further synthesis. Diazotization tops the list—conversion to bis-diazonium salts opens routes to a wide range of azo dyes, offering reliable color stability and shade strength. Coupling these salts with phenols or naphthols produces colorants used in everything from synthetic fibers to leather. The methoxy substituents slow down oxidative degradation and offer possibilities for downstream halogenation or coupling with sulfonic acids, tuning both solubility and vibrancy. For researchers tracking toxicity, the way those methoxy rings interact with metabolic enzymes becomes a key question: the same electronic effects that make the compound useful in chemistry also affect how live tissue handles it.
Synonyms & Product Names
Across industry literature and product labels, this compound often shows up under different names. Some lab catalogs list it as ortho-Dianisidine, 3,3'-Dimethoxy-4,4'-biphenylenediamine, or Dianisidine. In pigment manufacturing, older trade names still persist in technical reference documents, which sometimes complicates training and compliance checks. I've seen teams miss critical safety bulletins simply because product names differed from those on official warning lists. Taking the time to cross-check synonyms can make a big difference in preventing accidents.
Safety & Operational Standards
Handling 3,3'-Dimethoxybenzidine brings serious health and safety considerations. Routine gloves and lab coats don't offer enough protection when larger-scale operations come into play. Studies over the years have raised concerns about its links to carcinogenicity and other long-term toxic effects. Proper ventilation, chemical-resistant gloves, and designated fume hoods are non-negotiable during weighing, mixing, or transferring. Safety data sheets call for prompt spill management and strict waste disposal to keep exposure low, both for workers and for communities downstream from production sites. Regulatory landscapes in North America and Europe enforce mandatory monitoring—airborne dust, trace residues in water, and direct skin contact all sit under the microscope. I've seen first-hand how small lapses mushroom into big problems, from health scares to regulatory penalties.
Application Area
Most uses for 3,3'-Dimethoxybenzidine revolve around colorant synthesis. Textile mills once churned out vast quantities of cotton and synthetic blends dyed with its derivatives, since these colorants blended well and offered sharp hues that resisted fading under sunlight or washing. Printer ink formulas relied on the compound's consistency, while early plastics colored with these dyes set new standards for durability in toys, furnishings, and packaging. Environmental awareness and the rising tide of health concerns have started to shift industry preferences, pushing some companies toward alternative intermediates or greener routes, especially in regions governing hazardous substance use more tightly. Still, pockets of demand persist, with specialized pigment manufacturing retaining some dependence on this classic aromatic amine.
Research & Development
Research on 3,3'-Dimethoxybenzidine splits into two main branches: making process chemistry more efficient, and tackling the health risks. Industry-driven R&D focuses on tweaking yields, cutting byproducts, and harnessing new catalysts to shrink energy usage or boost selectivity. Some academic labs investigate how subtle variations in methoxy positioning can nudge color profiles or affect downstream reactivity. On the safety front, modern toxicologists apply sensitive detection tools to track even minute exposures, using biomonitoring and real-time sensors rather than waiting for health complaints to surface. In my own work, advances in chromatography and spectroscopy finally allow for mapping out breakdown routes in biological systems, revealing weak points where safer substitutes might eventually cut in. Knowledge sharing and open publication of results has grown more important, since regulatory agencies and consumer watchdogs look for transparency in both methods and outcomes.
Toxicity Research
Toxicology never lets 3,3'-Dimethoxybenzidine off lightly. Early animal studies flagged concerns about its breakdown into reactive metabolites, which target the liver, bladder, or kidneys. Epidemiological research, especially among dye workers from factories active in the 1960s-1980s, traced links to increased cancer risk. The slow pace of regulatory action often means exposure levels get debated in the press, with unions and advocacy groups calling for stricter cutoffs or outright bans. As detection methods sharpen, researchers have found lower and lower traces cropping up in factory effluent and even in household dust. For me, the most critical challenge lies in balancing the scientific evidence against real-world exposure: how much risk do trace residues pose, especially once these colorants filter through market chains? Industry reforms push better containment, worker blood monitoring, and investment in alternative chemistries—yet decades of use ensure that conversations around legacy contamination and long-term risk stay alive.
Future Prospects
Looking ahead, the road for 3,3'-Dimethoxybenzidine divides into two directions: managed use with heightened controls, or an eventual phase-out as safer options become available. More governments take a hard line on aromatic amines tied to health problems, especially when consumer goods or water tables show up as endpoints. Companies committed to sustainable growth push R&D toward drop-in replacements or new dye processes that skip known hazards. Efforts to clean up existing environmental contamination could draw on better filtration, waste treatment, and biomonitoring—fields now drawing donations and startup interest. Experience tells me that industries with long memories often resist rapid change, but rising consumer awareness and biomonitoring tools cut down the space for high-risk chemicals to linger. Those who adapt early tend to shape regulation and win credibility with both customers and employees. As industry, regulators, and researchers learn from past mistakes, new collaborations between sectors might finally see safer, more sustainable pathways open up.
What is 3,3'-Dimethoxybenzidine used for?
Color Runs Deeper Than You Think
Most people pick up their favorite colored pens or slip on a pair of blue jeans and never stop to ponder where those deep, lasting colors come from. Years spent around manufacturing plants have shown me that the dyes behind our everyday fabrics and inks have a story, often starting in a chemistry lab, with compounds only a handful of folks understand. 3,3'-Dimethoxybenzidine belongs on that list. This chemical helped shape modern dyes and inks, but it didn’t come without concerns.
The Role in Dye Manufacturing
During my college summers, I worked at a textile mill in the Midwest, watching how batches of white fabric transformed into everything from ocean blue shirts to burgundy work jackets. 3,3'-Dimethoxybenzidine’s biggest claim to fame sits in the world of azo dyes. It acts as a key building block, giving a chemical structure that helps dyes stick, last through wash after wash, and resist sunlight. It isn’t the only ingredient that can make color stay, but its presence means reds and blues really pop.
Besides clothing, the same chemical sits at the base of dyes used for paper, plastics, and printing inks. Anyone who ever picked up a vibrant, razor-sharp marker back in school probably saw the results of such chemistry without even thinking about it. Manufacturers favor it for bright, consistent tones. Its legacy can even be traced in identification cards and fancy postal stamps.
Health and Environmental Concerns
Having seen the insides of dye plants, safety comes up plenty in breakroom conversations. Years ago, workers handled 3,3'-Dimethoxybenzidine like just another chemical, but later studies showed its dark side. Research published by the National Institute for Occupational Safety and Health linked it to bladder cancer. Even at low exposure, it doesn’t take much for risk to rise. It stands on official lists of hazardous substances in North America, Europe, and Asia.
Regulations started tightening, sometimes forcing companies to redesign entire product lines. Plants that once handled it by the drum now use closed systems, point-of-use ventilation, and advanced protective gear. Waste disposal has changed, too, after groundwater tests detected traces leaking from old facilities. Today, environmental engineers keep close watch over every drum and tank, knowing the cost of careless handling.
Looking at Safer Alternatives
In the past decade, green chemistry has become more than a trend. Chemists work to develop new dye precursors, seeking options neither toxic nor persistent. Outfits with big sustainability pledges now ditch 3,3'-Dimethoxybenzidine in favor of safer aromatic amines or plant-based compounds. Still, finding replacements that match original colorfastness and intensity isn’t easy. Some companies turn back to natural dyes, though those don’t work for every application.
Pressure now falls on regulatory bodies to keep standards clear and on industries to invest in lab research. Regular health checks for workers, real transparency in ingredient lists, and waste tracking protect both people and the planet. In a business where color means everything, both the chemical’s history and the lessons learned from its risks shape how companies move forward.
Is 3,3'-Dimethoxybenzidine hazardous or toxic?
Understanding the Risks
3,3'-Dimethoxybenzidine, a chemical used in dye production and lab testing, rarely comes up in daily conversation. Still, for folks working in chemical plants, textile factories, or analytical labs, this compound should get their attention. I’ve known several industrial chemists who treat handling certain substances with extra caution—this one sits near the top of that list. Peer-reviewed research flags it as both acutely and chronically toxic, which means harm can come both from a big spill or from lesser, drawn-out exposures over the years.
What Makes It Dangerous?
Years back, while visiting a friend’s plant, I watched as workers handled dye intermediates with gloves, goggles, and rigorous cleanup. The company’s safety chief told me that 3,3'-Dimethoxybenzidine’s molecular cousins have left a long history of cancer and organ damage in their wake. As it turns out, the U.S. Environmental Protection Agency (EPA) lists benzidine-based dyes among probable human carcinogens. Studies connect these chemicals with bladder cancer rates that rise sharply among exposed workers over time.
The story doesn’t end at cancer. Some reports highlight how repeated contact can cause liver toxicity, skin inflammation, and sometimes respiratory irritation. This isn’t just chemistry on paper—multiple industrial accidents show how dust and fumes threaten lungs and skin if protections slip. Years of evidence can’t be ignored, and reputable organizations like the International Agency for Research on Cancer (IARC) flag this compound as possibly carcinogenic to humans.
Exposure and Real-World Consequences
For most of us, this compound isn’t showing up on grocery shelves. The trouble mainly lurks at the source: manufacturing plants, research settings, and environments recycling or disposing of dye wastes. If you grew up near a textile factory, as my uncle did, you might recall odd odors and murky wastewater. A 2020 survey of Eastern European dye factories found leaky drums, unprotected workers, and alarming airborne concentrations surpassing recommended limits.
People outside the lab or factory could still get exposed through contaminated water or residues. Improper dumping or spills don’t just go away—they seep into soil and eventually find their way to rivers. Newspaper investigations in India and Southeast Asia detail pollution and rising illness rates among nearby communities. The science backs this up: certain benzidine dyes linger in the environment and break down slowly.
What Can Be Done?
Holding companies accountable stands out as a core priority. Stronger workplace regulations on monitoring, maximum exposure levels, and regular employee health screenings stand to save lives. The U.S. Occupational Safety and Health Administration (OSHA) mandates a strict limit for benzidine exposure, but some regions fall short in enforcement or lack clear rules.
Switching to less toxic alternatives cuts risk at the root. Synthetic chemists continue searching for safer molecules for dyes and reagents. This takes research grants and a push from both customers and regulators. Regular environmental audits keep companies from cutting corners, and community groups play a role in holding polluters to account.
Easy solutions won’t fix historical pollution overnight, but demanding better protections pays off. It always struck me that good safety gear, clear training, and access to health care go much further than emergency responses after someone lands in the hospital. No one should face cancer or organ failure for a paycheck, and the science gives us enough reason to say: this hazard deserves respect and action, not ignorance or delay.
What are the proper storage and handling procedures for 3,3'-Dimethoxybenzidine?
Why Careful Handling Matters
3,3'-Dimethoxybenzidine isn’t a chemical you want to downplay. It’s got a real track record of health risks, and, over the years working in a few different labs, I've seen just how touchy things get when handling even small quantities. This compound appears as a pale yellow powder but it doesn’t take much to do harm: it’s recognized as a potential carcinogen. I often think about the lasting impact that a single careless move could have, both for individuals and teams. If you work with this stuff, you want to walk away at the end of the day with peace of mind.
Physical Setup for Storage
A big part of staying safe comes down to storage. I’ve always locked down 3,3'-Dimethoxybenzidine inside tight, clearly labeled screw-cap containers. Glass or high-quality plastic beats out anything porous; this isn’t a chemical you leave out for easy access. I keep containers in segregated cabinets away from direct light and heat — a must, since this compound degrades if you leave it exposed. Labs that work by the book rarely see cross-contamination, and the oversight mostly comes when people cut corners on storage or labeling. From personal habit, checking for signs of container wear or residue buildup has become second nature.
A Good Location: Out of Sight, Out of Harm
Storage has to mean more than tossing a jar on a shelf. A dedicated, ventilated chemical cabinet, set apart from acids, bases, or any oxidizers, helps prevent dangerous reactions. Flammable materials nearby just add risk. A lot of injuries can get traced back to storing incompatible chemicals side by side, or placing toxic compounds in everyday workspaces. Locks on cabinets make sense: only qualified staff get in, which shrinks the risk of mishandling or accidental exposure.
Handling Without Cutting Corners
Handling 3,3'-Dimethoxybenzidine means working behind the protection of a well-maintained fume hood. I never assume ventilation is just ‘good enough’ — regular checks with smoke sticks or airflow meters can save a lot of regrets down the line. Gloves go on before even opening the container, and nitrile or double-layer latex gloves do the trick. I won’t pick up a spatula or weigh paper without safety goggles, a lab coat, and face mask; one mistake can lead to skin absorption or inhalation, both are bad news. Even after handling, gloves come off the right way and go straight into a labeled hazardous waste bin.
Waste Disposal: Closing the Loop
Disposal gives plenty of opportunities for things to go wrong. All 3,3'-Dimethoxybenzidine waste — from gloves and wipes to leftover compounds — heads into sealed, labeled containers, away from regular trash. At every stop, the label lists the chemical’s name and hazard. As someone who’s witnessed improper dumping come back to haunt a lab, I push for everyone to know the local hazardous disposal protocols cold. The EPA and OSHA hold sites accountable here for good reason.
Training and Culture
Anyone can read a safety data sheet, but a culture of looking out for each other pays off. Colleagues who remind each other to wash hands, double-check PPE, or use designated storage keep everyone safer. From my own work, practice drills never feel over the top when the chemicals around can do serious lifetime damage. Management support matters, but so does personal responsibility. It often takes just one lapse to cause an accident, and that’s never worth the shortcut.
Final Thoughts on Responsibility
Chemicals like 3,3'-Dimethoxybenzidine demand real respect. The right container, the fume hood, and careful disposal aren’t just red tape — they’re steps that make sure each person gets to walk out of the lab at the end of the day happy and healthy. Shortcuts may save time in the moment, but they rarely save anyone in the long run.
How should 3,3'-Dimethoxybenzidine be disposed of safely?
Why 3,3'-Dimethoxybenzidine Demands Attention
3,3'-Dimethoxybenzidine shows up in labs mostly around dye production, chemical synthesis, and research settings. This stuff looks pretty harmless at first glance—just a white to tan powder—but stepping carelessly around it leads to health problems. Authorities and scientists call it a probable human carcinogen. Chronic exposure can threaten organs like the liver and bladder, plus it’s tough to shake from the body’s system. From personal experience in the field, even brief exposure in an unventilated space left me with headaches and a sore throat.
Real Hazards, Not Just Red Tape
Rules around hazardous chemicals grow out of real harm, not just government box-ticking. The dust from this chemical lingers, riding on your clothes or floating in the air. Disposal often gets rushed, but every shortcut can bring environmental damage and personal risk. A single carelessly handled beaker or a trash bag mixed in with street garbage risks seepage into the ground or the water supply. These stories don't come from regulatory pamphlets—I've seen colleagues scramble after realizing a waste bin ended up in the regular dumpster pickup, and cleanup got ugly.
Best Practice: More Than Just “Throwing It Out”
Disposing of 3,3'-Dimethoxybenzidine demands planning. Dumping it down the drain or mixing it with household garbage just spreads the problem further. In the workplace, the procedure starts with containment. All spills need to hit absorbent pads and go straight to a sealed, labeled waste drum. I remember a case where someone used plain old trash liners, and the chemical dust punched through like tissue paper—better choices save effort in the end.
Professional disposal routes offer the only safe answer. Hazardous waste contractors know the legal ins and outs. A reputable service guarantees secure containers, transportation, tracking, and incineration or chemical neutralization in a closed-loop facility. When I coordinated a cleanup in a small teaching lab, the contractor showed me exactly how every batch reached destruction—no mystery about where the waste ended up.
People Power: Training and Vigilance
No amount of caution on disposal helps if people handling the waste don’t spot the danger. Staff working with 3,3'-Dimethoxybenzidine need gloves, face protection, and lab coats that never leave the site. Handwashing cuts the risk, but so does sticking to a strict “no food, no drink, no shortcuts” rule in rooms where the chemical gets stored or handled. I’ve led safety briefings where one risky habit—eating lunch over the bench sink—nearly turned into a long illness.
Labeling every container and documenting every transfer or disposal step protects workers, janitorial staff, and the public. Sometimes employees feel shy about reporting spills or broken containers but, more than once, candor about a small incident prevented bigger disasters down the road.
Stronger Systems for a Healthier Environment
There’s an urgent need for small labs and teaching spaces to get access to disposal resources. Costs mount quickly, but sharing pick-ups, pooling resources, and lobbying for local collection days make safe disposal less burdensome. Sticking with best practices helps everyone—my experience points to fewer accidents, fewer clinic visits, and a safe space for daily work. The pathway starts with recognizing the risk and treating every disposal as a serious health task. Ignoring the rules sets up a chain of avoidable consequences, but choosing the safer route benefits your team and the wider community.
What is the chemical structure and molecular formula of 3,3'-Dimethoxybenzidine?
Getting to Know the Compound
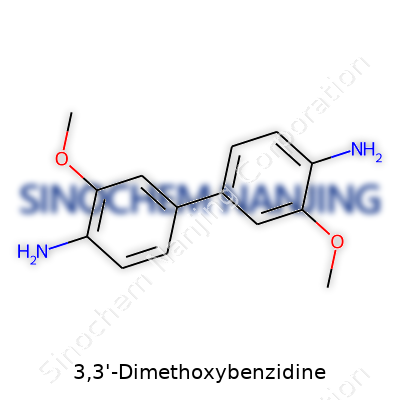
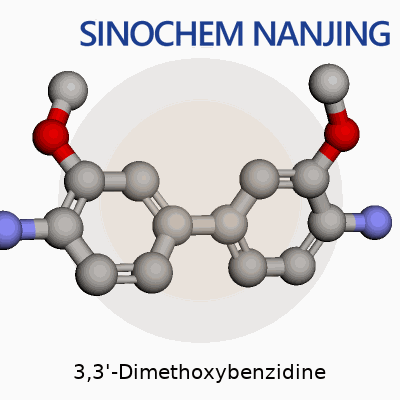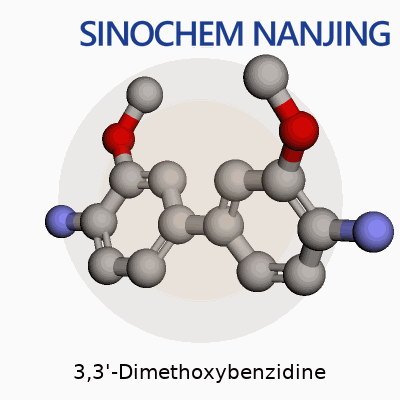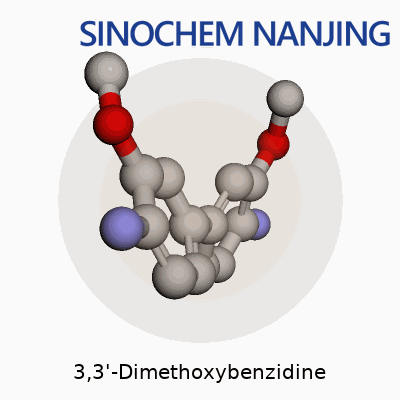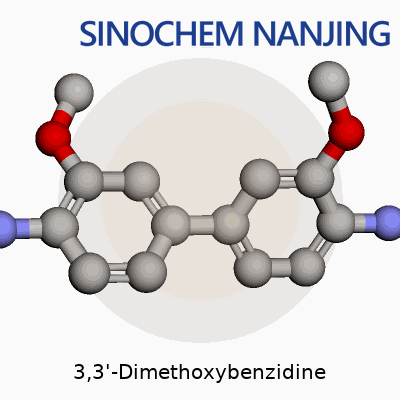
3,3'-Dimethoxybenzidine sits on the list of aromatic amines used in dye chemistry and lab research. Its molecular formula is C14H16N2O2. The structure builds on a benzidine core—meaning two benzene rings joined by a nitrogen-nitrogen bond. Both rings pick up a methoxy group (-OCH3) at the meta-positions relative to the amine link. Picture two flowering vines linked at the root, each carrying a bouquet of methoxy and amine groups; that's the essence of 3,3'-Dimethoxybenzidine.
Chemical Structure and Where It Leads
In this molecule, each benzene ring carries a methoxy group at the third position. The amine nitrogen bridges stand at positions one and one'. The setup: 1,1'-biphenyl-4,4'-diamine, decorated with methoxy groups at positions 3 and 3'. The result is a slightly sweet-smelling, crystalline solid—yellow to brown—which is common for its class.
The connectivity means each benzene ring shows this: a methoxy (-OCH3) at spot 3, an amine (-NH2) at spot 4, and a bridge connecting positions 1 on both rings via an –NH–NH– bond.
Why Structure Matters Beyond the Lab
Growing up in a family of chemistry teachers, I learned that small changes in a molecule can flip its properties or applications entirely. Add a methoxy, move a bond—these steps shift color, solubility, binding, and, sometimes, risk. That's true here. The methoxy groups boost solubility in certain solvents, giving dye chemists a softer touch when creating azo dyes. In textile and paper industries, this translates into longer-lasting colors that don't bleed out after a few washes.
3,3'-Dimethoxybenzidine entered lab life for its use in analytic chemistry to spot trace metals and as a core piece to synthesize more complex compounds. It never stayed in the limelight like benzidine or aniline, but those subtle changes in the molecular shape made it valuable in smaller, important ways.
Health Concerns and Safer Practice
Aromatic amines sit in a tricky spot when it comes to health. Many in this group, including 3,3'-Dimethoxybenzidine, have shown links to cancer in animal studies. I remember seeing old chemistry manuals with little to no warning about these risks. Today, we know better. Labs use fume hoods and gloves. Workplaces keep exposure low and track waste carefully.
OSHA classifies it as a potential occupational hazard. In one review, exposure led to concerns about bladder cancer after long-term use. Handling calls for careful tracking, disposal in chemical waste, and constant air monitoring if it shows up in large-scale manufacturing.
Room for Solutions
Chemists have pushed for safer alternatives in dye work, gradually replacing older aromatic amines with safer molecules. Digital records trace who handles stocks and make it easier to recall or substitute hazardous substances. Regular health checks for workers and high-efficiency air filtration slashed accidental exposure.
For students and researchers, learning about this compound highlights the need to balance chemical innovation with health and safety. Sometimes, reading an old structure in a textbook reminds me how every twist or turn in a molecule writes a new story in both applied science and workplace safety.
| Names | |
| Preferred IUPAC name | 3,3'-Dimethoxybiphenyl-4,4'-diamine |
| Other names |
Fast Blue B Base o-Dianisidine |
| Pronunciation | /ˌθriːˌθriː daɪˌmɛθ.ɒk.si bɛnˈzɪd.iːn/ |
| Identifiers | |
| CAS Number | 119-90-4 |
| 3D model (JSmol) | `3Dmol:'CC1=CC(=CC(=C1)N)OC.CC1=CC(=CC(=C1)N)OC'` |
| Beilstein Reference | 146881 |
| ChEBI | CHEBI:120390 |
| ChEMBL | CHEMBL1580057 |
| ChemSpider | 8901 |
| DrugBank | DB12983 |
| ECHA InfoCard | 03e0eaf8-cfe5-4c31-91d4-3f84ae23e979 |
| EC Number | 202-430-6 |
| Gmelin Reference | 85664 |
| KEGG | C06547 |
| MeSH | D02.455.426.559.389 |
| PubChem CID | 7090 |
| RTECS number | DU8050000 |
| UNII | 5A633G2P8W |
| UN number | UN1609 |
| Properties | |
| Chemical formula | C14H16N2O2 |
| Molar mass | 242.29 g/mol |
| Appearance | White to pale pink crystalline solid |
| Odor | Odorless |
| Density | 1.21 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.99 |
| Acidity (pKa) | 14.31 |
| Basicity (pKb) | 11.47 |
| Magnetic susceptibility (χ) | -73.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.669 |
| Viscosity | 1.06 cP (23°C) |
| Dipole moment | 3.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 274.6 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3219 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | May cause cancer. Causes skin and eye irritation. Suspected of causing genetic defects. May cause an allergic skin reaction. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02,GHS06,GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H317, H319, H351, H410 |
| Precautionary statements | P261, P280, P301+P312, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0- suspected carcinogen |
| Flash point | > 235°C |
| Autoignition temperature | 570°C |
| Lethal dose or concentration | LD50 (oral, rat): 3200 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 3000 mg/kg |
| NIOSH | DJ1225000 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Aniline p-Phenylenediamine Benzidine |

