3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride: From Bench to Breakthroughs
Historical Development
Chemists in the early 20th century dug deep into aromatic amine chemistry. The search for new organic intermediates led researchers to biphenyl derivatives. Out of this push, 3,3'-Dimethoxy-4,4'-Diaminobiphenyl hydrochloride emerged as a significant player. Once laboratories realized two amino groups and two methoxy groups offered diverse reactivity, the compound started appearing in dye manufacturing patents and research papers focused on high-performance materials. Later decades saw it mentioned in pharmaceutical synthesis efforts as well as explorations of specialty chemicals. Each new publication, patent approval, or industrial adoption deepened understanding, creating a network of knowledge rooted in real-world experience and hands-on trial and error. Many positive discoveries happened through unexpected outcomes—practitioners know firsthand how messy chemistry can enrich knowledge charts.
Product Overview
Looking at 3,3'-Dimethoxy-4,4'-Diaminobiphenyl hydrochloride, one finds a fine solid crystal. The hydrochloride salt form makes it more stable and easier to handle compared to its free base. Folks in research and manufacturing appreciate its solubility pattern: it dissolves in water, partly due to the hydrochloride counterion, and exhibits moderate solubility in ethanol and other polar organics. The two amino groups give this compound versatile chemical hooks, which means synthetic routes can pivot in multiple directions. The product has attracted attention in textile dyeing, as well as specialty pigment work, thanks to its ability to act as a high-quality linker. Its harsh reactivity profile raised questions about stability but pushed ahead applications in resin and polymer production where reliability and strong bonds count.
Physical & Chemical Properties
Holding a white-to-off-white powder, lab workers notice a faint chemical odor—nothing overpowering, but unmistakable. Melting point measurements usually fall around 320°C with slight decomposition, so this isn’t a compound for low-temp operations. Molecular weight tips the scale at 325 g/mol for the hydrochloride salt, which matters both for stoichiometric calculations and for evaluating batch processing. Water solubility sets this material apart from many biphenyl derivatives, making cleanup and post-reaction workup more straightforward. Under standard storage—moisture-protected, room temperature—it offers decent shelf life without any obvious breakdown. The combination of two electron-donating methoxy groups and two readily reactive amino substituents creates a playground for chemical modifications.
Technical Specifications & Labeling
Labs and suppliers list a purity range near 98% or higher for research-grade lots. Structural formula and standardized nomenclature must appear on shipping paperwork. UN number labeling and hazard pictograms, especially those indicating potentially harmful amines, are common. Specifications sheets usually include details about melting range, identity confirmation (by NMR or IR), and batch lot records for traceability. Chemical abstract service (CAS) numbers, EC/EINECS identifiers, and storage guidance fill out the necessary documentation. Certificates of analysis often record moisture content, ash residue, and trace metal contamination levels for each delivery. Prerequisites like COA and MSDS are standard at professional supply outlets, ensuring everyone downstream knows what to expect in each drum, bag, or vial.
Preparation Method
Synthesizing 3,3'-Dimethoxy-4,4'-Diaminobiphenyl hydrochloride isn’t quick, and the route often starts from commercially available 3,3'-dimethoxy-4,4'-dinitrobiphenyl. Chemists reduce the dinitro precursor, usually via catalytic hydrogenation using palladium-on-carbon, though old-school methods with iron/acid or tin chloride appear in some older journals. The reduction step yields the diamine, which reacts directly with hydrochloric acid to form the more stable hydrochloride salt. Good filtration and recrystallization finish things off. Each step demands tight process control, especially during reduction, to prevent partial reactions or unwanted byproducts. Modern setups focus on maximizing yield and purity while minimizing residual heavy metals, volatile organics, or explosive intermediates. In many respects, these practical experience-driven improvements mark the distance between academic synthesis and real industrial production.
Chemical Reactions & Modifications
With two methoxy and two amino groups, creative chemists can run wild. N-acylation, diazotization, and oxidative coupling reactions shape this molecule into everything from advanced dye intermediates to bistable molecular switches. Amino groups participate in straightforward condensation reactions to make ureas and sulfonamides, while methoxy substituents help control reaction rates and increase resistance to unwanted oxidation or hydrolysis. For instance, sulfonation at the available rings adds water solubility, while crosslinking with aldehydes or isocyanates leads to higher-performance polymers. In the lab, modifications tend to target either greater lightfastness for pigments or improved compatibility for polymer systems. Small shifts in process variables—temperature ramp rates, reagent additions—reshape both yields and selectivity, an outcome that anybody who’s scaled up from flask to kilo-lab has learned firsthand.
Synonyms & Product Names
Anyone tracking chemical supply chains runs into alternate names: some call it bis(3-methoxy-4-aminophenyl) hydrochloride, while others label it 4,4'-Diamino-3,3'-dimethoxybiphenyl hydrochloride. International catalogs sometimes use translated versions, blending systematic and trivial naming approaches. Each alias traces back to the same core structure, so cross-referencing CAS numbers helps avoid misorders or dangerous mix-ups—no one wants an unexpected byproduct because of a catalog typo.
Safety & Operational Standards
Chemical handling standards leave no room for shortcuts. Based on its structure, 3,3'-Dimethoxy-4,4'-Diaminobiphenyl hydrochloride carries risk of skin and eye irritation. Industry and academic practice both call for gloves, goggles, and well-ventilated spaces. Some jurisdictions require local exhaust ventilation and work surfaces resistant to amine spills. Larger operations, like pilot-scale synthesis, mandate full-face shields and splash gear. Waste streams with aromatic amines must go through special treatment—neutralization and incineration, not down the drain. Proper labeling and secure storage protect against accidental exposure or cross-contamination, and safety datasheets drive home the need for spill kits and eye wash stations right in the lab. Support staff and senior scientists alike have stories of “near-misses” that make safety standards not just checkboxes, but actual lived practice.
Application Area
Most often, 3,3'-Dimethoxy-4,4'-Diaminobiphenyl hydrochloride ends up in the dye and pigment trade. Textile manufacturers value its direct reactivity when creating azo dyes with stable coloring and strong fastness. Advanced pigments for polymers and coatings incorporate this building block to harness both coloration and chemical resilience. Resin chemists take advantage of the diamine’s functionality to crosslink epoxies or urea-formaldehyde systems, boosting adhesive strength or heat resistance. Academic researchers pick it up as a model molecule for biphenyl-based ligand studies in catalysis. Postdocs working in green chemistry also experiment with its derivatives, exploring novel materials for use in solar panels and organic electronic devices. The sheer flexibility provided by its molecular features keeps it relevant across multiple industries, including niche areas like analytical chemistry and electroactive binder research.
Research & Development
Future-looking labs investigate new derivatives and tailor reaction conditions for sustainability. Teams seek to swap out heavy metals from the classic hydrogenation steps, searching for more environmentally benign reduction catalysts. Researchers develop methods to recycle reaction wastes and streamline purification. On the application front, collaboration between material scientists and synthetic chemists leads to improved polymers, dyes, or functional materials with lower toxicity or sharper performance. Cross-industry forums and academic conferences feature in-depth presentations on biphenyl diamine chemistry, with speakers sharing hard-won lessons—from avoiding process bottlenecks to unexpected regulatory challenges that can stall a good idea. A hallmark of responsible R&D involves documenting observations, both positive and negative, and channeling those experiences into improved process designs and safer alternatives.
Toxicity Research
Aromatic amines have long drawn regulatory scrutiny due to known toxicological risks, and 3,3'-Dimethoxy-4,4'-Diaminobiphenyl hydrochloride sits squarely in this debate. Early screening pointed to a potential for mutagenicity common among many biphenyl-derived amines. Animal studies indicate organ-specific effects at high exposures, with liver and kidney function affected most. In vitro models show DNA reactivity, prompting calls for more detailed epidemiological investigations. Some research groups run long-term exposure studies on wastewater treatment workers and plant staff who handle aromatic diamines across shifts. Monitoring protocols, including regular blood panels and urine tests for metabolites, feature in current best practices. Regulatory agencies demand limits on workplace air concentrations and push for improved ventilation and engineering controls. Ongoing research focuses on developing less hazardous analogues while advancing rapid analytical tests for environmental and worker safety monitoring.
Future Prospects
Trends in specialty chemicals always favor compounds with robust performance and lower risk. For 3,3'-Dimethoxy-4,4'-Diaminobiphenyl hydrochloride, new process improvements promise reduced waste and higher yields, trimming both financial and environmental costs. Material scientists look at ways to tweak its substituents, aiming for photoactive or charge-carrying variants to support next-generation batteries or organic semiconductors. As regulatory standards tighten, plenty of effort goes into lifecycle assessments and end-user safety, pressing researchers to design greener syntheses and safer disposal routes. Chemical manufacturers who invest in these upgrades open up fresh markets in high-tech coatings, greener dyes, or even molecular electronic components. Each innovation pushes the compound’s story forward, powered by practical experience, honest trial, and collective ambition to balance utility, safety, and sustainability.
What is 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride used for?
Where It Shows Up
3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride does not make the evening news or pop up in casual conversations, but it shapes medical progress in ways most people overlook. Lab workers know it best as a building block for synthesizing dyes, specialty chemical intermediates, and, in a big way, pharmaceuticals. Chemists focus on this molecule because its structure supports both industrial processes and medical research. I’ve talked to a few in the field who value it for its reliability when pushing new concepts in drug discovery.
Drug Discovery and Scientific Impact
Research teams see this compound as a gateway to developing anti-cancer treatments and therapies targeting chronic diseases. The scientific literature—especially studies published in organic chemistry and pharmacology journals—shows that the structure of 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride mimics structural features useful for binding to proteins and enzymes. This is crucial. Nearly all new drugs depend on fine-tuned molecular interactions, and having access to a molecule like this gives researchers a leg up. My own experience in academic research taught me that access to rare or specialty building blocks like this one determines how fast a project can move from idea to proof of concept.
Industrial Applications and Risks
The chemical industry also leans on this compound for creating pigments and dyes with specific shades and chemical properties. Certain tapestries and textiles, familiar in hospitals and laboratories, gain their distinct color from derivatives developed using this compound. Its double amine and methoxy groups give manufacturers more leeway in achieving stability and vibrance. Those working on the production floor keep close track of handling guidelines, since these aromatic amines can pose health risks if not managed correctly. Some related compounds have drawn scrutiny for their carcinogenic potential, and the safety data on 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride still raises caution.
The Challenge of Safe Handling
Workplace safety officers push for tighter controls. Gloves and respirators are not negotiable. Regulations differ by country, and compliance teams find themselves cross-checking local rules with updates from larger agencies like OSHA and the European Chemicals Agency. Looking back at my time supporting a regulatory team, the big surprise was the gap between what policies called for and what actually happened on factory floors. Until oversight tightens and safer alternatives prove practical, this compound demands respect and vigilance from anyone handling it.
Seeking Better Alternatives
One potential solution lies in designing and testing new molecules that do the same job with lower health risks. Green chemistry principles encourage companies to reevaluate older compounds and update production methods. Pharmaceutical labs push boundaries by searching for new building blocks. If suppliers listen to scientists calling for more sustainable options, it will improve long-term outcomes for everyone—researchers, factory workers, and end users who benefit from safer medicines.
What are the storage conditions for 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride?
Recognizing Hazards and Protecting Quality
Many people working in research, pharmaceuticals, or specialty chemical labs cross paths with complex compounds like 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride. Handling substances like these doesn’t just involve scientific curiosity—it carries responsibility. The specific structure of this hydrochloride salt means it responds to moisture, light, and heat in ways that can affect both safety and performance. If you leave it at room temperature on a cluttered bench with the cap loose, you don’t just risk wasting expensive material—you also face health risks and unreliable results. From my own time in academic labs, a single careless error with sensitive compounds always meant lost hours and dangerous surprises. Keeping this chemical in top condition starts with knowing what really matters in everyday storage practice.
Temperature and Environment
High temperatures shorten shelf life, speed up decomposition, and promote reactions you’d rather avoid. Your standard chemical refrigerator keeps material cool enough (2°C to 8°C) to reduce these risks. At normal room temperature, reactions with airborne moisture or contaminants speed up. If the label insists on dry storage at lower temperatures, take that seriously. Humidity creeps in faster than you think, and once water interacts with hydrochloride salts, changes begin at the molecular level—colors shift, debris forms, and, sometimes, dangerous gases escape. Sealed containers prevent this problem, but shoving them next to something like dry ice or an open source of heat always backfires. A dry, stable, moderately cool environment serves almost every batch of this compound well.
Light and Air Exposure
Light, especially direct sunlight or strong indoor lamps, degrades organic chemicals rapidly. Repeated exposure means the bonds that keep your molecule intact start to break down. Protecting sensitive substances often means more than just a closed cabinet. Store this chemical in an amber or opaque bottle—a tip I learned from a meticulous lab manager who never let photo-reactive compounds anywhere near a window. This practice grows even more important when the chemical is expected to sit for months between uses. Tight lids hold off air and environmental moisture, stopping the slow shift to degraded, toxic, or useless products. For a material that can release corrosive or irritating gases, hands-on experience taught me the wisdom of good sealing and frequent checks.
Labeling, Inventory, and Safe Handling
Many dangerous incidents start with poor labeling or casual attitudes. Every container benefits from a clear label with the chemical’s name, concentration, date opened, and specific hazards. In busy workspaces, good records keep colleagues safe and ensure nobody accidentally grabs a bottle thinking it’s something else. Never underestimate the power of a monthly inventory check. Storing this ingredient far from incompatible substances—like strong bases, oxidants, or anything reactive—makes all the difference. Simple shelving strategies prevent leaks or spills from touching other chemicals, eliminating chain reactions before they have a chance to begin.
Reducing Risk and Keeping Work Safe
Sparks, static, or open flames don’t belong near storage areas. Many compounds in this category—amines and hydrochloride salts—can emit toxic fumes or catch fire when handled carelessly. Spill kits and proper ventilation, including fume hoods, offer real protection. Wearing gloves, eye protection, and lab coats is a habit worth forming, not just a rule to check off. In my own lab, every time someone skipped these basics, near-misses happened. You protect people and science by storing and using chemicals with intention and vigilance.
Simple Practices for Consistent Results
Store 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride away from sunlight, at controlled cool temperatures, and in tightly sealed, moisture-proof bottles. Keep it labeled, log every opening, and keep inventory streamlined. Regular checks, attention to compatibility, and safe workspace layout prevent both material loss and dangerous mistakes. Good habits pay back tenfold in reliable research and workplace health.
What is the chemical structure of 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride?
Digging Into the Molecule
Every so often, a chemical name looks like it belongs in a spelling bee, not a lab notebook. This one, 3,3'-Dimethoxy-4,4'-Diaminobiphenyl hydrochloride, sounds complicated, but its structure speaks volumes about the value it brings to chemistry and industry. The base compound starts with biphenyl, two benzene rings joined at their centers. Chemists often use biphenyl as a backbone to anchor different functional groups. Attach two methoxy groups at the '3' positions of each ring and two amino groups at the '4' positions and you get a versatile molecule. Once hydrochloride joins, its applications stretch even further—from dye manufacturing to specialty polymers and maybe something that ends up in an electronics lab.
The Core Structure: Breaking Down Its Skeleton
Biphenyl draws a simple line in chemistry—a pair of six-membered carbon rings tethered together. Now picture each of those rings with a methoxy group (-OCH3) on carbon number three and an amino group (-NH2) on carbon number four. Positions matter; swapping groups changes the entire behavior of the compound. This exact arrangement means each benzene ring offers both electron-donating groups, which tunes the chemical’s reactivity.
Hydrochloride as a salt form usually comes from reacting the parent amine with hydrochloric acid, delivering better stability and making handling friendlier for the folks in the lab. The hydrochloride salt often dissolves better in water, which speeds things along during synthesis or purification steps.
Why Structure Dictates Usefulness
Small changes in functional groups send shockwaves through what a chemical can do. Methoxy groups encourage certain reactions, pushing electrons into the aromatic system, and helping stabilize intermediates. The amino groups crank up reactivity even more. As someone who’s spent days wrestling with similar biphenyls, I remember how tiny changes transformed dyes' brightness or shifted melting points.
This molecule isn’t just a static skeleton. Industrial chemists value such compounds because they offer options—buttons to push that affect everything downstream. A more water-soluble salt means less dust, fewer losses to the wind, and better control over dosing in daily production. Whether synthesizing advanced pigments or breaking ground in organic semiconductors, this versatility changes the game.
Potential Challenges and Places for Improvement
Working with biphenyls always carries environmental baggage. Their persistence in the ecosystem, historical links to toxic PCBs, and the need for cleaner production keep them surrounded by a cloud of scrutiny. Green chemistry offers a roadmap: look for renewable feedstocks, reduce waste during halogenations or aminations, and test new solvents that spare workers’ lungs. Realistically, nobody turns a petrochemical giant on a dime, but every process that squeezes out a little more efficiency or lowers emissions pays off somewhere along the line.
Safer alternatives don’t always leap to the front. But by studying precisely how methoxy and amine groups work on the biphenyl core, researchers might design molecules that perform the same job without lingering in the environment or breaking down into something hazardous later.
Direct Impact on Innovation
Lab work with this compound brings home how a single molecule can open doors. From stronger colors in textile dyes to tweaks in electronic materials, the underlying structure matters more than marketing buzzwords. Solutions will come from clear-eyed analysis of these chemical details and the will to keep pushing for safer, smarter methods in manufacturing and research.
Is 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride hazardous or toxic?
Why Chemical Safety Matters for Workers and Communities
Few people think about chemicals like 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride outside of labs or factories, but these kinds of compounds shape real-world health in ways that don’t show up in the headlines. With a tongue-twisting name and a complex molecular structure, this substance usually pops up in specialty chemistry, dye production, or polymer research. Those who handle it need to stay alert to the risks, even though they might never see their names in any news story. Safety here isn’t a checklist — it’s a real shield for the people breathing and working nearby.
Understanding the Health Risks
You won’t find this chemical on a kitchen shelf, and that’s for good reason. Dimethoxy-diaminobiphenyl compounds share similarities with other biphenyl derivatives known for hazardous properties. Animal studies on close relatives, especially certain aromatic amines, point to potential cancer risks. Long-term exposure — especially by inhaling dust or through skin contact — raises concern over carcinogenic outcomes and chronic toxicity. The Occupational Safety and Health Administration updates guidance regularly, reminding companies to stay current on what is known about chemical hazards. Some overseas guidelines flag this substance as potentially mutagenic. That can mean damage down at the DNA level after repeated unprotected mishandling.
During my early days in industrial research, I watched seasoned technicians treat aromatic amines with a respect reserved for rattlesnakes. Double gloves, respirators, closed-loop extraction systems — not just for show but because even mild slip-ups can cost someone their health years later. Nobody shrugs off the risks after enough headlines about dyes leading to unexpected cancers in retired workers.
The Environmental Angle
Runoff and improper disposal have a way of finding their path into water tables and open fields. Once these chemicals enter soil or water, they can stick around, affecting small aquatic life and trickling up the food chain. Common wastewater treatments often don’t capture complex organic compounds very well, and the ones that do cost money, requiring real investment from companies. Ignoring the environmental load rarely stays a quiet issue — communities often react when health patterns shift near chemical plants.
What Better Safety Looks Like
People working around these sorts of chemicals deserve real-time monitoring, health checks, and protective gear. It takes more than a couple of warning labels. Direct ventilation, sealed handling systems, and thorough training prove their worth shift after shift. Regular air monitoring catches leaks or spills before someone gets sick. No one wants to look back on their career and discover their biggest risk came from the thing they handled every day.
Industry can choose greener alternatives when they become available, slashing the need for hazardous amines in new processes. Some research labs switched out these chemicals entirely, finding sustainable routes for colorants or polymers. None of that progress happens by accident. Pushing for safer options means funding, fresh chemistry, and honest communication about what’s really at stake. Every improvement, big or small, starts with admitting the risks don’t just belong to “someone else.”
What is the purity and available packaging size of 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride?
The Story Behind Chemical Purity
In my days working at a small specialty lab, conversations about the quality of chemicals always turned heads because purity can shape the results of every experiment. 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride, a mouthful in name but critical in use, tells its own story. For anyone walking into a lab and reaching for a reagent, those tiny details on the label carry big meaning.
Research-grade chemicals are expected to meet or beat standards, but the journey from supplier data to a researcher’s bench reveals plenty. The purity for 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride typically registers upwards of 98%. Ask any synthetic chemist and they’ll confirm: that two-percent margin can swing results. Leftover impurities won’t just muddy data; they can create safety hazards or kill a project’s momentum altogether. I saw this play out firsthand, watching colleagues retrace months of work to uncover a contaminant hiding in a supposedly “pure” sample. Purity, even when it sneaks into the decimals, changes the game.
Sizing Up the Supply
Not every scientist needs a drum of materials, and most don’t get budgets that stretch that far anyway. This compound, in my experience, lands in packaging that suits real-world demand. Small bottles, typically 1 gram, 5 grams, and 25 grams, cover everything from screening a reaction to beginning preclinical work. The market doesn't usually flood buyers with kilo options unless there’s a custom run, which costs more and takes time.
Why these particular sizes matter comes down to working style. In academic labs, nobody wants to lose sleep over an open bottle oxidizing on the shelf, so a 1-gram vial feels right. Industrial groups, running bigger batches, might reach for larger quantities but usually want to negotiate directly with suppliers to lock in freshness and quality. I’ve sat in on these calls. Scientists haggle less for price, more for guarantees about batch traceability, storage practices, and the data that comes with the product. The wrong labeling or a missing certificate of analysis means the material might go completely unused.
Evidence for Responsible Choice
Sourcing chemicals like 3,3'-Dimethoxy-4,4'-Diaminobiphenyl Hydrochloride draws on more than catalog shopping. PubChem and Sigma-Aldrich list purity specs, and research papers set benchmarks—hard data that lab managers use to choose suppliers. Regulatory bodies like the FDA put pressure on suppliers to audit quality more than ever, especially for molecules destined for drug discovery. The demand has never been higher for consistent and pure samples with every new shipment. During a short stint in a quality lab, I learned to scan for not just the chemical formula but full traceability—batch number, date of production, even specific impurity profiles when available.
Moving Forward—Practical Steps
Anyone deciding on a chemical supplier starts by requesting up-to-date certificates with every lot. Open conversation between buyer and supplier helps clarify packaging needs and shelf life. Keeping a running log of how different brands perform can save trouble down the road. Buying from certified companies with transparent quality-control practices makes a difference, not just for data but for safety. In my practice, I always pushed for pilot samples before committing to bulk orders, no matter how urgent the timeline felt. In science, speed without care slows everything down eventually.
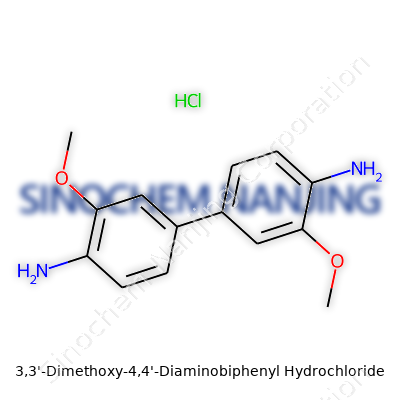
| Names | |
| Preferred IUPAC name | 4,4'-Diamino-3,3'-dimethoxy-1,1'-biphenyl dihydrochloride |
| Other names |
Fast Blue B Salt Fast Blue BB salt o-Dianisidine diamine hydrochloride Dianisidine diamine hydrochloride |
| Pronunciation | /ˈθriː ˈθriː daɪˈmɛθ.əks.i ˈfɔːr ˈfɔːr daɪˈæm.ɪ.noʊ.baɪˈfiː.nɪl haɪˌdrɒ.kləˈraɪd/ |
| Identifiers | |
| CAS Number | 73814-82-3 |
| Beilstein Reference | 365246 |
| ChEBI | CHEBI:94479 |
| ChEMBL | CHEMBL3324661 |
| ChemSpider | 26632478 |
| DrugBank | DB08798 |
| ECHA InfoCard | 100.027.594 |
| Gmelin Reference | 83392 |
| KEGG | C18708 |
| MeSH | D04.210.500.247.249.374.875.210 |
| PubChem CID | 19510 |
| RTECS number | SG4375000 |
| UNII | K8R6Y8SQJ3 |
| UN number | This product does not have a UN number. |
| Properties | |
| Chemical formula | C14H18ClN2O2 |
| Molar mass | 308.79 g/mol |
| Appearance | Light yellow to brown powder |
| Odor | Odorless |
| Density | 1.295 g/cm³ |
| Solubility in water | Soluble in water |
| log P | 0.51 |
| Acidity (pKa) | 2.8 |
| Basicity (pKb) | 4.67 |
| Magnetic susceptibility (χ) | -65.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.641 |
| Viscosity | 450 cP (25°C) |
| Dipole moment | 7.1 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 357.56 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261-P280-P305+P351+P338-P337+P313 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: |
| Flash point | > 234.9 °C |
| Lethal dose or concentration | LD50 oral rat 679 mg/kg |
| LD50 (median dose) | LD50 (median dose): 285 mg/kg (oral, rat) |
| NIOSH | Not listed |
| PEL (Permissible) | 'PEL (Permissible Exposure Limit) Not established.' |
| REL (Recommended) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Aniline Benzidine 3,3’-Dimethoxybenzidine 4,4’-Diaminobiphenyl 3,3’-Dichlorobenzidine |

