3,3'-Dichlorobenzidine Hydrochloride: Unpacking the History, Science, and Future Directions
Historical Development
Digging into the story of 3,3'-Dichlorobenzidine Hydrochloride, you reach back into the late 19th and early 20th century, when breakthroughs in coal tar chemistry fueled the rise of synthetic dyes. German chemists, looking to expand the palette for textile industries, picked up 3,3'-Dichlorobenzidine as a key raw material in azo dye production. As demand grew for stronger, brighter colors that all sorts of businesses could afford, this compound transformed how coloring agents were manufactured on a mass scale. It carried forward into the 1960s and 1970s, tying its name to the huge spread of plastics and coatings, both of which leaned heavily on its chemical backbone.
Product Overview
Chemists working with 3,3'-Dichlorobenzidine Hydrochloride get a vital intermediate. Walk into any dye or pigment lab, and you’ll find it used widely for producing pigments in printing inks, fabrics, and plastics. Companies that need consistent color quality grab this chemical for its reliable conversion into yellow and red pigments, critical in everything from industrial coatings to packaging inks. It’s also been tapped for specialty applications, including analytical reagents and complex sensor work, though these uses haven’t caught on as much as pigment manufacturing.
Physical & Chemical Properties
You recognize 3,3'-Dichlorobenzidine Hydrochloride by its white to light tan crystalline appearance, often giving off a mild, characteristic chemical odor that stays on lab gloves even after a wash. Its melting point lands between 225–229°C, showing its thermal stability when compared to similar aromatic diamines. The compound dissolves best in water, ethanol, and dilute acids, staying stable under normal handling but breaking down in strong oxidizing environments. With a molecular formula of C12H10Cl2N2·2HCl, it brings in a molecular weight north of 294 g/mol. The hydrochloride salt, compared with the free base, offers more manageable dusting characteristics, cutting down unnecessary exposure risks in manufacturing environments.
Technical Specifications & Labeling
Manufacturers relying on strict labeling practices, typically demand purity levels above 98%, with related impurities like 3,3'-Dichlorobenzidine and other chlorinated anilines capped at tenths of a percent. Standardized specifications also require moisture content tests, because too much water can derail both synthesis runs and finished pigment performance. The labeling normally displays batch number, production date, and clear hazard pictograms, to ensure transport and storage protocols stay airtight. Regulatory mandates in Europe, the US, and Asia force suppliers to provide detailed safety data sheets, identifying hazards linked to carcinogenicity, acute toxicity, and proper disposal measures.
Preparation Method
Production follows a classical chemistry route with roots in exhaustive scholarship: beginning with nitro derivatives of dichlorobenzene in a controlled reduction process, typically using iron filings or catalytic hydrogenation in acid media. Strict temperature surveillance and real-time monitoring stamp out byproducts and keep reaction yields high. Operators separate, purify, and crystallize out the hydrochloride salt by slow addition of hydrogen chloride gas, watching for pH benchmarks that ensure conversion. Industrial facilities lean into energy recycling steps and solvent recovery to meet growing sustainability standards from watchdogs and customers alike.
Chemical Reactions & Modifications
You see its chemical flexibility through reactivity at the amine groups. Diazotization converts it to a reactive diazonium state, forming the core bond in azo pigments—these reactions drive the colorants you find in road markings, plastics, and heavy-duty coatings. Its aromatic rings allow electrophilic substitutions, enabling functionalization for sensor probes or specialty dyes used in microscopy. Even beyond its main pigment uses, chemists sometimes modify its structure with sulfonation or halogenation, tailoring solubility or shifting optical properties for experimental applications.
Synonyms & Product Names
There’s an ocean of synonyms in industry paperwork—3,3'-Dichlorobenzidine Hydrochloride might turn up as DCBD·2HCl, 4,4'-Dichloro-o-tolidine Hydrochloride, or just “BASF Yellow Intermediate” in legacy catalogs. Older research references lean into names like “Dichlorobenzidine salt” or “C.I. 37050 Intermediate.” Cross-referencing product codes and trade names becomes a routine task for purchasing teams, given regulatory differences and international trade language quirks. This tangle of aliases only highlights the material’s roots across several sectors and decades of commercial life.
Safety & Operational Standards
The safety discussion never stays academic—handling 3,3'-Dichlorobenzidine Hydrochloride raises tough questions about workplace cancer risk, spill management, and environmental leakage. It’s classified as a Group 2B carcinogen by the IARC, meaning there’s evidence supporting its cancer-causing potential in humans. OSHA has set exposure limits, and engineering controls like fume hoods, rigorous personal protective gear, and closed-system handling now serve as non-negotiable standards. Training rounds focus on decontamination, acute spill scenarios, and emergency evacuation. Changes in legislation—such as stricter REACH controls or US EPA updates—keep compliance officers and plant managers on their toes. Waste management involves strict incineration protocols set forth by international agencies, avoiding waterway contamination.
Application Area
Printing ink and textile dye markets drive the largest demand curves for 3,3'-Dichlorobenzidine Hydrochloride. Classic yellow and red pigments owe their brightness and resistance to light or washing cycles to the chemical robustness of its derivatives. Beyond colorants, some research labs use its reactivity for analytical chemistry—both as a precursor and in sensor development for hazardous gas detection or metal ion sensing. Plastics manufacturing plants, especially in regions with less strict regulation, draw on its vivid coloration properties for everything from toys to furniture coatings. This broad reach brings with it industry involvement from safety-linked disciplines such as industrial hygiene, environmental engineering, and toxicology.
Research & Development
A lot of research focuses on improved synthetic routes that reduce hazardous waste, cut down worker exposure, and swap out environmentally persistent byproducts. Green chemistry initiatives at academic and industrial labs chase new reduction agents and process intensification, leveraging continuous flow systems and online monitoring to predict and control impurity formation. In the field of analytical chemistry, surface-modified versions allow new applications—such as material science blends or molecular imprinting for better chemical detection tools. Despite regulatory headwinds, pigment research teams keep working on substitutes or hybrid materials that keep color quality high but cut health risks.
Toxicity Research
There’s a daunting body of work connecting 3,3'-Dichlorobenzidine Hydrochloride exposure to increased risks of bladder and liver cancer, with animal studies confirming its metabolic activation to potent DNA-binding agents. Chronic inhalation, ingestion, or even skin absorption can pose sizable risks. Worker monitoring, frequent biological assays, and long-term epidemiological studies dig into the full extent of its impact on factory staff and downstream users. Toxicologists keep raising red flags about accidental environmental release, with aquatic organisms particularly sensitive to trace contamination. Calls continue for better real-time detection methods and exposure countermeasures—whether environmental remediation or systematic medical surveillance of at-risk populations.
Future Prospects
The industrial world stands at a crossroads: demand for high-performance colorants is not slowing down, but regulatory frameworks keep closing in. European markets, in particular, are leaning harder on alternatives and full phaseouts, though replacements still struggle to match cost and color intensity. Green chemistry breakthroughs could finally tip the balance, either through complete synthetic redesign or successful encapsulation of carcinogenic intermediates. Academic labs see new promise in modular dyes based on less hazardous building blocks. Stronger international cooperation, with synchronized risk assessment and data sharing, may cut duplication and speed up innovation cycles. For now, worker and environmental safety demand an all-hands-on-deck approach—smart engineering, continued toxicological tracking, and transparent supply chains all count in keeping this chemical from turning up in headlines for the wrong reasons.
What is 3,3'-Dichlorobenzidine Hydrochloride used for?
A Chemical With a Colorful Purpose
Mention 3,3'-Dichlorobenzidine Hydrochloride in a science class, and you’ll probably get a blank stare. Yet this compound plays a quiet but vital role behind the scenes in manufacturing—especially in the world of pigments and dyes. Spend enough years reading safety data sheets or touring a chemical plant, and it turns up on the radar often enough. Its main claim to fame is as a building block for certain colorants, above all, those deep and durable yellow pigments used in printer inks, plastics, and textiles.
Where It Fits in the Color Industry
The color in your favorite plastic food container or printer cartridge likely owes a debt to substances made from 3,3'-Dichlorobenzidine Hydrochloride. Its molecular structure lets chemists build high-performance organic pigments—think vivid yellow or green shades that stay bright after years of sunlight. Azo dyes, which dominate the market for synthetic pigments, use this chemical as a precursor. By tweaking the molecule, manufacturers can adapt pigment properties—maximizing brightness or toughness depending on the job.
If you’ve handled brightly colored textiles or ever worked near an offset printing press, there’s a good chance you’ve seen the color results from these manufacturing choices, even if you didn’t clock the chemistry behind them. The pigments created from this compound stand up well to heat and light, and that durability matters a lot in everyday products.
Health and Environmental Risks Raise Big Questions
Chemistry often brings tradeoffs. 3,3'-Dichlorobenzidine Hydrochloride stands as a stark example. On the one hand, the chemical enables bright, lasting colors in everyday products. On the other, its history of toxicity and potential to cause cancer in humans and animals cannot be ignored. Agencies like the US Environmental Protection Agency and the International Agency for Research on Cancer flag it as a possible or likely human carcinogen. This gives anyone handling the raw material reason to pause—engineers, plant workers, and waste management teams face serious risks if exposure limits are not respected. Skin contact, inhalation, and even contaminated water runoff all present hazards.
I’ve seen first-hand the level of attention paid to raw material handling and hazardous waste at facilities that use these intermediates. Workers suit up in heavy protective gear, safety officers monitor every ounce of dust and vapor, and waste management routines follow precise protocols. Even the best-run plants can deal with spills or accidental exposure, pushing everyone to stay vigilant. It’s not just workers at risk. Improper disposal could let harmful compounds leach into groundwater or soil. Trouble here doesn't stay isolated.
Drifting Toward Safer Alternatives
Manufacturers and regulatory agencies have not stood still. Faced with known risks, many companies phase out dyes built from 3,3'-Dichlorobenzidine Hydrochloride, preferring safer intermediates where practical. Research teams continue to hunt for new pigment technologies that deliver the same durability and brightness without the health hazards.
Rules keep tightening as evidence grows. In many regions, strict exposure limits force companies to redesign equipment, recycle solvents, and treat waste exhaustively before release. It makes production costlier, but few would argue against the moral and legal importance of safer workplaces and communities. Using every tool available from greener chemistry to better regulation, the push continues to color our world without endangering it in the process.
What are the safety precautions when handling 3,3'-Dichlorobenzidine Hydrochloride?
Why This Chemical Draws Special Attention
3,3'-Dichlorobenzidine hydrochloride pops up mostly in dye and pigment labs. Just seeing a drum of this stuff on the shelf rings alarm bells for anyone who’s ever read a material safety data sheet. There are plenty of chemicals with toothy warnings, but few sit under the microscope like this one. Researchers, techs, and anyone handling it must deal directly with cancer warnings on the label. The International Agency for Research on Cancer flags it as a potential occupational risk. That sobers up anyone, fast.
The Problem: Long-Term Harm Isn’t Obvious
You’d never notice anything wrong after a single afternoon in the lab with this powder. Problems bubble up over time, and mostly below the surface. Absorption through skin means you don’t always see or smell danger. Chronic exposure links directly to bladder cancer. Every health and safety officer shares the same experience: it’s easy to ignore rules around invisible threats. But nothing upends a routine like a diagnosis down the road.
Real-World Safety Advice
Work with 3,3'-Dichlorobenzidine hydrochloride feels different once you understand its risks. Anyone who reads credible case studies or listens to survivors knows skipping one glove change can stack up with consequences. Forgetting to zip up a lab coat, or trusting old goggles, can undo months of staying careful. Here’s what should be in daily habits:
- Always wear that full set of PPE: This means nitrile gloves, chemical apron, closed goggles, and a face shield for mixing powders or spills. Leather shoes with no open tops, tucked under pant legs, give another line of defense. Street clothes pick up more than you’d guess.
- Handle powders in a real fume hood: A spare cardboard box won’t stop invisible dust. It takes an actual hood, tested for airflow, to protect lungs.
- Check and double-check labeling: Nothing complicates a day like an unmarked jar. Every bottle needs solid labeling and dated logs for audits and safety rounds.
- Never work alone during mixing or spills: Two people in the room means a better chance of fast response if there’s a problem. Solo shifts with this chemical increase risk for everyone.
- Always practice proper waste disposal: This chemical can’t get washed down the drain. Close up waste containers, label them clearly, and hand over material to a licensed hazardous waste handler. Anyone who’s spilled a drop learns quickly that improper disposal wrecks both health and the environment.
Building a Culture of Safety
There’s a difference between ticking a box on a training sheet and actually creating a lab where people look out for one another. I remember starting out and seeing veterans who never let newbies open a bottle without walking through the process first. That culture saves lives. Old timers keep emergency showers working and remind folks how to handle splashes before anything ever happens. Posting emergency phone numbers next to the hood, running regular spill drills, and checking PPE for tears matter much more than the official posters on the wall.
Better Solutions Are Possible
Regulations help, but they don’t replace a crew who actually talk and keep tabs on gear and routines. Regular health screenings for those who handle these chemicals, clear reporting of close calls, and supervisor support keep everyone sharper. And as safer alternatives become available, switching over can cut harm to zero. I’ve seen labs move away from high-risk dyes because managers listened to the people on the front line. In the end, handling 3,3'-Dichlorobenzidine hydrochloride safely comes down to habit, honesty, and a willingness to speak up—because long-term health starts with what happens each day at the bench.
Is 3,3'-Dichlorobenzidine Hydrochloride hazardous or carcinogenic?
Understanding 3,3'-Dichlorobenzidine Hydrochloride in Everyday Context
A lot of chemical names sound like they belong in some far-off laboratory. But 3,3'-Dichlorobenzidine Hydrochloride shows up much closer to home than most people realize. This compound helps manufacturers create dyes for textiles, plastics, and even paper. Workers with paint-stained hands and those running massive dyeing vats might not recognize the name, but they've probably handled products colored with this chemical.
Risk Factors
Talking about cancer and chemicals always puts people on edge. The International Agency for Research on Cancer calls 3,3'-Dichlorobenzidine and its salts, including the hydrochloride form, “reasonably anticipated to be a human carcinogen.” That’s not alarmist. Studies show workers exposed over long stretches have a greater risk for bladder cancer. Evidence points to aromatic amines—the group of molecules 3,3'-Dichlorobenzidine Hydrochloride belongs to—as repeat offenders. The body absorbs them, often through skin contact, then tries to break them down. In doing so, dangerous by-products can damage DNA inside bladder cells. That’s how people can end up with a higher chance of cancer just by showing up to work each day.
Why Does This Matter?
Most everyday exposure comes indirectly, since finished consumer goods typically don’t have much remaining residue. Still, the bigger problem lands in the lap of factory workers and anyone cleaning up the sites where these chemicals were used—think old dye plants and Superfund sites. My family has roots in a mill town where folks used to talk about water that ran bright blue or yellow, depending on the dye batch that week. The stories about neighbors getting bladder or liver cancer were whispered, not broadcast. That lived experience stays with me. Even if technology improves, people always fill the gap and take the risk somewhere along the line.
What Steps Can Lessen the Risk?
Wearing gloves and protective gear keeps direct contact to a minimum. Good ventilation, closed systems, and regular cleanups prevent dust and vapor from building up, so inhalation exposures drop. Smart companies invest in monitoring and regular health checks for their employees. They also train workers on what these chemicals do to the body. No one wants health problems sneaking up years down the road.
Policymakers have the job of making sure chemicals flagged as carcinogens get extra attention. Regulations push industries to use safer alternatives if they exist. Take Europe’s REACH rules—authorities require companies to prove they’re handling substances safely, with documentation and substitution plans for dangerous chemicals. That kind of oversight gets results, but it needs enforcement muscle. Without regular inspections and real consequences, even the best-written rulebook gathers dust.
Looking Ahead
Replacing old chemical favorites with safer options usually costs money and plenty of effort. It’s worth the hassle if it means fewer cancer stories in families like mine. People building careers in industries using dyes deserve to know exactly what touches their skin and fills the air around them. Giving workers information, tools, and choices gives everyone a fair shot at a healthy future. That’s the least anyone can expect, no matter what color their shirt is at the end of the day.
What is the chemical formula and structure of 3,3'-Dichlorobenzidine Hydrochloride?
The Chemical Formula and Structure
3,3'-Dichlorobenzidine hydrochloride carries the formula C12H10Cl2N2·2HCl. Looking at its structure, two chlorinated benzene rings connect by a pair of nitrogen atoms sitting between them. This setup places chlorine atoms at the 3 positions on each ring, so both face away from the center. Two additional hydrochloric acid molecules form the salt, increasing its stability. The result is a compound that dissolves much more easily in water. That solubility makes a real difference in the lab, whether someone is running tests or making dyes.
Why Structure Shapes Use
This isn’t just dry chemistry. The layout decides what this compound can do. Putting chlorines at the 3-positions toughens the molecule against many forms of breakdown. It’s more resistant to oxidation and light, which matters for its workhorse role as an intermediate in dye production. Factories use it to create pigments for plastics, textiles, and inks. The actual salt form means handling it becomes less risky — dust stays down, and spills can be cleaned up with water. These details get noticed the most in manufacturing and research, where safety and efficiency count for more than a tidy formula on a page.
The Shadow Over Benzidine Derivatives
Back in the twentieth century, companies churned out derivatives like 3,3'-Dichlorobenzidine for colorfast dyes. Later on, researchers started to connect exposure to benzidine-based compounds with increased bladder cancer risk. Studies in the British Journal of Cancer and research from the National Toxicology Program show these links. Workers from dye plants reported more cases of urinary tract tumors than anyone expected. For someone spending years breathing particles or getting chemicals on the skin, regulations alone couldn’t erase that risk.
Regulations and Safer Alternatives
Regulators have stepped in to limit exposure. In the United States, OSHA sets strict workplace exposure levels. The European Union has flagged 3,3'-Dichlorobenzidine hydrochloride as a substance of very high concern. I’ve seen plants move over to automated, closed systems to keep workers out of direct contact. Safer alternatives for coloring—like azo compounds that don’t break down into carcinogens—are now chosen more often, especially for products that touch food or skin. Factories move to water-based pigment processing rather than the old types that put powder in the air.
Moving Toward Safer Chemistry
From my experience in lab supply, education and training make the biggest dent among workers exposed to legacy chemicals. Lab managers encourage gloves, splash goggles, and fume hoods for even small runs. Still, cleaning up the supply chain goes further — where raw materials get triple-checked for contaminants and substitutes become the new normal. Research keeps looking for pigments that can deliver the same bold color without the carcinogenic baggage. A big part of safe handling comes down to treating substances like 3,3'-Dichlorobenzidine hydrochloride with respect. It belongs in the hands of professionals with backup safety measures and nowhere near consumer goods. Change takes a while, but every step away from hazardous compounds like this adds up in healthier workplaces and safer products.
How should 3,3'-Dichlorobenzidine Hydrochloride be stored and disposed of?
What’s at Stake
3,3'-Dichlorobenzidine Hydrochloride shows up in some dyes and pigments, but its potential risks aren’t just a matter for a lab handbook. The Centers for Disease Control and Prevention (CDC) lists it as a probable human carcinogen, so regular exposure piles up the danger for workers and the environment. I remember hearing about a research facility hazmat team in Philadelphia that locked down their entire chemical storeroom because one misplaced bottle leaked—a mess no facility manager wants to handle. Accidents like that underscore why this chemical cannot be treated as just another bottle on a shelf.
Storage that Puts Safety First
People working with 3,3'-Dichlorobenzidine Hydrochloride keep it in tightly sealed, chemical-resistant containers. The right approach means using glass or high-density polyethylene, not just whatever’s around. Every container gets a clear label, because faded or missing tags invite confusion, which leads to mistakes nobody can afford. Store it far from food, strong oxidizers, acids, and open flames—this stuff doesn’t mix well with much.
Shelving belongs in a well-ventilated, cool, dry spot with limited access. Locked cabinets or separate rooms prevent unauthorized entry, which matters in busy facilities where people can wander in. Spills become less likely, and containment is easier, when chemicals are kept at or below eye level. Vent hoods also play a big part. I’ve seen techs check storage areas with regular walk-throughs, making sure no dust or residue builds up on counters and floors.
The Right Kiss-Off: Disposal that Respects Health and Regulations
You can’t toss leftovers in the regular trash. EPA rules treat 3,3'-Dichlorobenzidine Hydrochloride as hazardous waste. Facilities collecting the substance work with certified hazardous waste contractors, because municipal dumps or drain disposal pose unacceptable risk. Chemical spills end up contaminating groundwater and harming communities that barely have a say in what gets buried in their backyard.
Professional disposal usually means incineration at high temperatures in facilities equipped with scrubbers to neutralize harmful breakdown products. Landfilling as "hazardous solid waste" follows strict procedures—double-bagging, leak-proof drums, and truck transport with all the right paperwork. I’ve watched waste tracking in real time, barcodes and logs making sure nothing disappears into thin air. Being able to trace each container from storage to final disposal is not just box-ticking, it means accountability.
Better Practices Keep People and Places Safe
Training remains a frontline defense. Workers who understand what is in those bottles act quickly when anything seems off—smells, leaks, or mysterious residues trigger a response, not a shrug. This depends on regular safety drills, posted reminders, and supervisors who know how to lead by example. No shortcut or clever hack replaces honest communication and a willingness to call for help.
Finding safer alternatives to 3,3'-Dichlorobenzidine Hydrochloride underpins long-term safety. Businesses can press suppliers for less toxic substitutes, which is already happening at some textile and manufacturing plants. Until industry-wide shifts become the norm, real attention to safe storage and disposal helps prevent the sort of disasters no community—or company—should have to clean up.
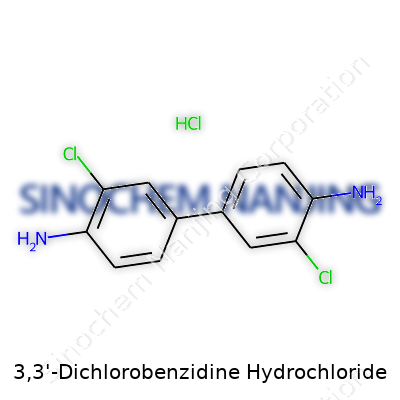
| Names | |
| Preferred IUPAC name | 3,3'-dichloro-[1,1'-biphenyl]-4,4'-diamine hydrochloride |
| Other names |
3,3’-Dichlorobenzidine dihydrochloride 3,3’-Dichlorodiphenyl-diamine dihydrochloride 4,4’-Dichloro-1,1’-biphenyl-2,2’-diamine dihydrochloride |
| Pronunciation | /ˌθriːˌθriː daɪˌklɔːrəˈbɛn.zɪˌdiːn ˌhaɪ.drəˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 612-83-9 |
| 3D model (JSmol) | `3d:JSmol|Clc1ccc(cc1N)c2ccc(cc2N)Cl.Cl.Cl` |
| Beilstein Reference | 72374 |
| ChEBI | CHEBI:86476 |
| ChEMBL | CHEMBL3254123 |
| ChemSpider | 82953 |
| DrugBank | DB13783 |
| ECHA InfoCard | ECHA InfoCard: 100.013.761 |
| EC Number | 209-345-4 |
| Gmelin Reference | 80653 |
| KEGG | C18397 |
| MeSH | D003639 |
| PubChem CID | 16132522 |
| RTECS number | KL3325000 |
| UNII | Y4Y7R61HVL |
| UN number | UN2579 |
| Properties | |
| Chemical formula | C12H12Cl2N2·2HCl |
| Molar mass | 296.55 g/mol |
| Appearance | Light yellow to light buff crystalline powder |
| Odor | Odorless |
| Density | 1.5 g/cm³ |
| Solubility in water | Soluble in water |
| log P | 2.7 |
| Vapor pressure | 1 mmHg (190°C) |
| Acidity (pKa) | -4.0 |
| Basicity (pKb) | 13.3 |
| Magnetic susceptibility (χ) | -83.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.656 |
| Dipole moment | 4.10 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.8 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | May cause cancer; toxic if swallowed, inhaled, or in contact with skin; causes skin and eye irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H350: May cause cancer. |
| Precautionary statements | P202, P220, P280, P308+P313, P405, P501 |
| Flash point | > 210 °C (410 °F) |
| Lethal dose or concentration | LD50 oral (rat) 1530 mg/kg |
| LD50 (median dose) | 2 gm/kg (oral-rat) |
| NIOSH | DT1750000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 3,3'-Dichlorobenzidine Hydrochloride: 0.1 mg/m³ |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
Benzidine 3,3′-Dichlorobenzidine 3,3′-Dimethylbenzidine 3,3′-Dimethoxybenzidine o-Tolidine |

