Digging Into 3,3'-Dichlorobenzidine: History, Science, and the Ongoing Debate
Historical Roots and Discovery
Taking a hard look at the story behind 3,3'-Dichlorobenzidine, you see the push and pull between industrial progress and safety. Chemists first synthesized this compound in the early 20th century, at a time when the coloring industry was racing to deliver brighter, longer-lasting dyes. The molecule’s discovery marked a turning point—suddenly, manufacturers could produce vivid pigments for everything from textiles to plastics. The colorfast blues and greens found in many everyday items owe their existence to molecular tweaks like the simple addition of two chlorine atoms to benzidine. Chemists, often working in dimly lit labs, had no idea what risks accompanied these new compounds, because the typical mindset of that era rarely lingered on downstream effects. Society benefited from new products, but workers and communities near production sites faced unrecognized dangers.
Product Overview and Industrial Use
3,3'-Dichlorobenzidine goes beyond the world of chemistry textbooks. Its primary claim to fame involves the creation of azo dyes, especially those yielding green and blue hues that refuse to fade, even with repeated washing or outdoor exposure. Textile makers, plastics manufacturers, and ink producers have leaned heavily on this molecule. For years, you could spot objects colored with these dyes in homes, classrooms, and even medical labs. What’s troubling is that as demand grew, manufacturers prioritized high yields and economic production, but they overlooked the environmental and health footprints. Anyone familiar with dye factory stories from midcentury knows about spills, contaminated water, and inadequate ventilation that harmed workers and nearby residents alike.
Physical and Chemical Properties: The Science and the Risks
Handling 3,3'-Dichlorobenzidine means coming face-to-face with an off-white to pale yellow solid, often with a faint aromatic scent. It doesn’t dissolve in water, but it mixes well with organic solvents, making it easy to transport through production cycles. The molecule contains twin benzene rings linked by a nitrogen bridge, and chlorines attached at the 3 positions. This robust structure is why the pigment refuses to break down when exposed to light or humidity. Anyone who has spent time in a chemistry lab knows that stability is a double-edged sword—hardy molecules can stick around longer in the environment, drifting into soil and water for years. That persistence makes cleanup difficult and raises flags about long-term exposure.
Technical Specifications and How It’s Labeled
Labels on the drums or containers storing 3,3'-Dichlorobenzidine list its chemical formula—C12H10Cl2N2—and highlight hazard symbols warning of toxicity, carcinogenic potential, and ecological harm. Regulations in many countries, especially in North America and Europe, demand clear, upfront labeling. These descriptions aim to alert handlers and consumers. But paper labels often feel like a stopgap, especially when used in low-resource settings where training lags behind regulation.
Moments in Preparation and Synthesis
Turning benzidine into 3,3'-Dichlorobenzidine involves chlorination, typically with chlorine gas introduced in controlled bursts while keeping temperature and acidity within strict limits. Synthetic routes rarely look clean or simple—side products, volatile emissions, and left-behind reactants pose significant handling and disposal challenges. Few outside industry circles realize just how much waste a single batch can generate, or how poorly some plants managed their leftovers in the past. Only in the last few decades have production facilities faced pressure to close the loop and cut down on persistent pollution.
Chemical Reactions and Tweaks
In real-world labs, few chemicals serve a single purpose. Chemists learned how to modify 3,3'-Dichlorobenzidine, crafting dozens of derivatives by attaching or swapping out chemical groups. These modifications help dye-makers control hue, shade, and resistance to sunlight or abrasion. Despite all this ingenuity, every tweak carries risk—the basic structure resists breaking down in nature, making even new versions tougher to remove from waterways or soils. Each time a molecule gets altered, it introduces new questions about breakdown products and bioaccumulation that cannot be answered overnight.
Names Across Disciplines
In the chemical industry, a single compound can go by many names. 3,3'-Dichlorobenzidine might appear as DCBD, Fast Garnet Base, or simply Dichlorobenzidine in technical papers and safety data sheets. These aliases can cause misunderstandings in research and trade, leading to mix-ups in regulation or safety enforcement.
Keeping Things Safe: Practices and Standards
It’s no secret that working with 3,3'-Dichlorobenzidine carries health risks. Standards have changed a lot since the early days of open flasks and bare hands. Today, advanced ventilation systems, sealed processes, and strict personal protective equipment requirements dominate responsible factories. Employees need to understand protocols, not just follow checklists. Training and environmental controls must keep pace with research about the compound’s hazards. Governments from the US to the EU now restrict its use, and the list of banned applications keeps growing as evidence about its links to cancer and chronic respiratory illness piles up. Some companies still flout the rules, and regulators keep chasing violators, proving that regulation without enforcement turns safety into an empty promise.
Where Does It End Up?
Most 3,3'-Dichlorobenzidine turned up in dyes used in plastics, paper, textiles, and laboratory stain kits. While its popularity shrank due to health findings, remnants linger in old products and archived materials, especially where recycling never occurred. Because dyes don’t vanish easily, trace amounts show up in soil and rivers near former dye plants around the world. It’s a somber reminder that yesterday’s inventions cast long shadows—living near contaminated land, some communities have tracked spikes in rare cancers traced back to industrial pollution. Quitting cold turkey isn't always an option; industries that rely on high-performance pigments still wrestle with phasing out toxic compounds entirely.
Lab Research and Where Science Stands
Ongoing research aims to break down persistent molecules like 3,3'-Dichlorobenzidine safely, but no chemical fix has proven foolproof. Teams at universities and government labs analyze how these molecules interact with cells, looking for clues about how low doses might still harm genetic material or disrupt hormone systems. Studies often reveal that metabolites—what the body turns a compound into—can show up as even more dangerous than the original. This keeps questions open and funding flowing for both basic and applied research. Even the most advanced countries struggle to draw a line that weighs innovation against public health.
Learning More about Toxicity
The evidence on 3,3'-Dichlorobenzidine’s risks can't be ignored. Studies have linked it to bladder and liver cancers in both workers and lab animals. Reports surfaced over the years describing factory employees who developed rare illnesses, years or decades after spending time on dye production lines. These testimonials led to landmark restrictions and stricter monitoring at worksites. Animal studies and epidemiology have become critical tools driving these changes, but they also bring hard choices—sometimes regulations arrive late, after too many people have already suffered.
Looking Forward: What Comes Next?
The debate over 3,3'-Dichlorobenzidine reflects a far broader challenge: how to balance the needs of industry with the obligation to protect health and the environment. Cleaner synthesis pathways have started to emerge, and industries are now more willing to consider substitutes, even if that means higher costs or performance sacrifices. As awareness builds, pressure mounts to find safer alternatives for dyes and pigments, invest in real cleanup efforts at contaminated sites, and support long-term health studies for exposed communities. Successfully winding down use of risky chemicals takes global cooperation, not just piecemeal bans. Industries, regulators, and communities need to trust one another enough to see hard data and move faster when warning signs appear. Experience across chemical safety has taught that early action saves more lives and costs less than drawn-out debates where business interests try to outweigh science. Society can’t afford to wait until tomorrow to fix yesterday’s decisions.
What is 3,3'-Dichlorobenzidine used for?
What 3,3'-Dichlorobenzidine Means in Real World Terms
In most factories, 3,3'-Dichlorobenzidine shows up as a raw material that helps make up certain dyes and pigments. Workers have stirred it into the mix for decades, mostly in plants that build colorants for textiles, paper, and plastic. This compound offers solid color stability, which keeps items from fading in sunlight or washing out after repeated cleaning. That’s why manufacturers often stick to it for brilliant shades of yellow, orange, and green. The colors end up in everything from book covers and synthetic leathers to the bright wrappers you see on store shelves.
Nitty-Gritty Use Cases
Take a walk through a textile factory, and you’ll spot vats filled with these bright pigments, especially those based on what’s called “azo chemistry.” 3,3'-Dichlorobenzidine handles the load in such recipes, where it helps create vivid tones that don’t simply fade after a month of sunlight. Sometimes the paper industry uses it, adding the dye to make packaging and labels pop with color, giving companies more options to help their products stand out.
Then there’s plastic. Manufacturers blend the pigment into plastics that need bright hues—stuff like toys, automotive parts, and even kitchen products. All of this adds up to huge demand across several heavy industries, since consistency and visibility matter. Bright yellow road signs, school folders, vinyl upholstery—all may owe their hues to this single molecule.
What’s the Tradeoff?
Here’s the tough part. Evidence links 3,3'-Dichlorobenzidine with significant health risks. Multiple studies, including those summarized by the National Institute for Occupational Safety and Health (NIOSH), point toward a strong cancer risk after frequent or high levels of exposure. Workers who have handled the chemical in pigment production, especially in earlier decades before stricter safety measures got into place, have faced higher cancer rates in the bladder.
OSHA and other regulatory bodies flagged this risk years ago. Factories have had to protect employees with gloves, respirators, and better ventilation. Dangers don’t just end inside the building. Disposal of this kind of chemical sometimes pollutes soil and waterways if companies cut corners, creating lasting health risks for nearby communities.
Solutions from Experience and Research
Working on factory floors years ago, I saw first-hand how safety instructions changed once word got around about cancer links. The best-run shops replaced open mixing and pouring with sealed systems. Managers began to monitor exposure and cycled out anyone who worked with the chemical for too long. These steps dropped the risks, but didn’t eliminate them.
Some major dye users now turn to alternative, less toxic colorants. The replacement process runs into hurdles, since not every substitute offers the same color stability or price point. More research on plant-based and synthetic alternatives continues, but demand for cheap, high-performing colors keeps manufacturers coming back to what works—until laws or market forces push a big shift.
A Path Forward
Pressure mounts as more data rolls in about the risks. Regulators now call for better tracking of chemical flows, stricter waste handling, and stronger worker protections. Consumers can help by asking about the safety of dyes in products and pushing for clear labeling. Investment in green chemistry delivers real results, but takes time and funding to replace tough, reliable chemicals like 3,3'-Dichlorobenzidine. The challenge lines up clearly: Protect workers and neighbors now, search hard for safer ways to color our world, and make industry own up to long-term impacts.
Is 3,3'-Dichlorobenzidine hazardous to health?
Chemistry in Daily Life: The Straight Talk
Some chemicals hang around factories and labs, but folks keep hearing their names because they wind up in our world far beyond the test tube. 3,3'-Dichlorobenzidine fits this bill. Its name sounds like something out of a spelling contest, but behind the syllables hides a substance that shapes industries and, for many years, shaped lives in not-so-great ways. I first read about this compound working on a report about worker safety in colorant manufacturing. The pigment industry uses it in making dyes, especially in textiles, plastics, and printing inks. Companies chased color and profit, and, for a long stretch, nobody asked tough questions. Today, the echo from those years still rings out in health and safety studies.
What Happens to Folks Around It?
Experience from the shop floor carries more weight than any textbook. Workers handling this chemical describe the dust—a fine, yellow haze that gets in hair, clothes, and lungs. Over the years, medical studies and government reviews started paying attention. I remember reading testimony from the 1980s: employees noted skin rashes, sore throats, and headaches after a day’s shift. Basic curiosity drove medical researchers to see if there was more beneath the surface. Turns out, there was. Those “everyday” symptoms weren’t the whole story.
Digging into the Risks
Data gathered by the National Cancer Institute, OSHA, and the World Health Organization ties long-term exposure to increased cancer risk. Cases of bladder cancer, one of the most documented effects, started showing up among former pigment plant workers. Animal experiments in labs gave similar results—exposure led to tumors. Risk isn’t limited to the factory either. Trace residues, fume inhalation, and improper disposal lead the chemical to water and soil around plants. I’ve seen local reports where creek tests near dye manufacturing sites picked up traces years after plants shut down.
Why Should This Matter?
People sometimes forget the human story behind chemistry. “Out of sight, out of mind” doesn’t work with substances built to last. Families living near old industrial sites aren’t choosing, yet run the daily risk. Protective gear and safety guidelines, if ignored, mean trouble sooner or later. In my experience, clear labeling and strict storage measures cut daily incidents. Companies investing in worker education and cleanup don’t just avoid lawsuits—they protect people.
Facing the Problem Head-On
Solutions start with awareness. Regulations—like OSHA’s workplace standards and the Environmental Protection Agency’s hazardous waste rules—went a long way, but not every business follows best practice. Testing local water for chemical traces gives city officials information they need to warn communities. Clean-up of contaminated sites, though expensive, stops the spread. Substitutes for this compound exist, but switching takes willpower and upfront investment. Whenever my team took the time to re-train staff on chemical safety—even walking the line with them, showing emergency showers and explaining symptoms—people listened, and near-misses dropped off.
Keep Pushing for Health
Everyone deserves to work and live without worrying about chemical fallout. I’ve seen small changes—like proper gloves, exhaust fans, and color-coded bins—make workplaces safer overnight. Stories of injury and illness brought about real change, but no one should have to wait for headlines. Chemicals like 3,3'-Dichlorobenzidine forced whole industries to rethink the hidden cost of color and convenience. The lesson sticks for every sector: cutting corners ends up costing more. Knowing what’s at stake and staying honest about the risks means building workplaces, and neighborhoods, that last.
What are the safety precautions when handling 3,3'-Dichlorobenzidine?
Why Pay Attention?
Safety in any lab or industrial setting means more than just going through the motions. I’ve watched both new and seasoned workers underestimate chemicals that don’t look dangerous at a glance. 3,3'-Dichlorobenzidine isn’t something to shrug off. People use it mainly in dye production, but contact brings some real health risks—skin, eyes, and especially breathing it in or letting it linger on your skin. The science is pretty clear: exposure has ties to cancer and serious organ damage. Stepping into this reality means taking precautions that don’t cut corners.
Personal Protection: The Layer Between You and Hazard
Back in my first chemistry job, my supervisor drilled one message home—treat your safety gear as part of you. Gloves aren’t an afterthought, they’re a frontline defense. For this compound, nitrile gloves work well. If I know splashes are likely, I’d grab my goggles and a face shield. Long sleeves and a lab coat keep skin covered. Respiratory protection can feel uncomfortable, but working without it around powders or open processes isn’t worth the risk. A simple paper mask won’t do. Properly-fitted, chemical-rated respirators make the difference.
Handling and Storage: Habits That Matter
Good habits start at the storage cabinet. Every time I move or use this chemical, I check that it’s still sealed tight, labeled clearly, set away from incompatible materials. Storage space shouldn’t be near heat, moisture, or sunlight. In my experience, open containers or makeshift bottles create trouble—leaks, contamination, or even accidents if someone grabs the wrong jar. Secure containment isn’t fancy, just vital.
Housekeeping and Cleanup: The Devil’s in the Details
Spills happen. What matters is having a plan. I always keep spill kits close, and I train myself to use them without hesitation. For 3,3'-Dichlorobenzidine, dry spills get scooped with care—never swept, since dust kicks right up. Wet spills call for absorbent pads. All cleanup materials go straight into marked hazardous waste bins. Soap and water aren’t enough; chemical-specific cleaners reduce residue and hidden exposure. I never skip handwashing, no matter how small the task.
Medical Vigilance and Emergency Response
Getting exposed doesn’t always mean instant symptoms. That’s what makes regular checkups important, especially in workplaces handling substances like this. I remember a colleague ignoring a skin rash that turned out serious. One overlooked spot caused weeks of trouble. Workplaces should keep safety showers and eye wash stations clear and in working order. In my years around chemicals, rapid first aid and transparency about incidents have avoided a lot of regret and long-term health problems.
Culture of Safety: Beyond Rules
Real safety goes deeper than checklists or posters on a wall. In my best workplaces, people spoke up when they saw shortcuts and looked out for each other. Training isn’t just a box to tick. It should push for real understanding of why steps exist and what can happen if ignored. Encouraging everyone to report near-misses helps catch problems early. I’ve seen more progress through open conversation than rigid enforcement alone.
Looking Ahead
Risks tied to 3,3'-Dichlorobenzidine aren't going away soon. Some countries call for strict limits or bans. Until substitutes take over, careful handling and respect for the dangers remain the best shield. For anyone working with this chemical, know your procedures, trust your gear, and never think a shortcut saves time. It rarely does.
How should 3,3'-Dichlorobenzidine be stored?
Why Storage Matters for 3,3'-Dichlorobenzidine
3,3'-Dichlorobenzidine is not a household name, but plenty of labs and factories know this chemical for what it is—a precursor for dyes and pigments, and a substance flagged for its serious risks. Once you’ve handled it, you start to see storage is not just a procedural checklist. It's about protecting workers, the building, and the broader environment.
This isn’t paranoia. The Environmental Protection Agency classifies 3,3'-Dichlorobenzidine as a probable human carcinogen. Chronic exposure may cause bladder cancer and other health problems. The dust or vapors can irritate the skin, eyes, and respiratory tract. Handling practices reflect more than bureaucracy—they protect real lives.
Common-Sense Practices in Storage
I spent time working in a chemical processing lab, where 3,3'-Dichlorobenzidine was often kept in small labelled bottles. We relied on practices set by OSHA along with what years of experience taught our supervisors. A cool, dry, well-ventilated area makes the most sense for storage, away from sunlight and direct heat sources. Moisture, heat, and light can degrade chemicals or cause unwanted reactions, so you go for a sturdy, labeled container. Anyone who has seen a chemical shelf ruined by a leaking cap knows how quickly that oversight turns into a nightmare cleanup.
Glass or high-density polyethylene containers work best. Nobody wants a weak cap or an unlabeled jar—mistakes multiply fast in these situations. The storage room itself should feature fire-resistant walls in case things get out of hand. Some labs use separate cabinets for carcinogens, under lock and key, with the access code strictly controlled.
Proper Labelling and Records
Accurate, visible labeling doesn’t just follow laws or guidelines; it prevents panic during an emergency. Common sense says the label needs the chemical name, concentration, hazard warnings, and date received. Outdated or faded labels can cost precious seconds in a spill or fire, and that’s no small thing. Record keeping tracks shelf life and ensures nothing gets forgotten until it’s past prime. In some workplaces, a digital inventory system tracks chemicals, cuts down on human error, and simplifies audit trails. Regular checks by supervisors catch old or deteriorating stocks before they turn into big problems.
Ventilation and Employee Training
Most folks who’ve spent time in a lab know: ventilation keeps everyone safer. Fume hoods or exhaust systems pull vapors out, especially when you have volatile or toxic chemicals. Storing 3,3'-Dichlorobenzidine near the door or emergency exit never ends well. The smarter move is to keep it far from busy passageways, lowering the chance that an accident pulls in more people than necessary.
Training sticks with people long after safety posters come down. Anyone handling or storing this chemical should know how to use gloves, goggles, and lab coats, and what to do in a spill or fire. Refresher sessions and drills show workers that safety isn’t a one-and-done. Fact is, experience builds confidence and reduces mishap rates. OSHA and NIOSH offer clear guidelines, and any workplace ignoring recommendations risks big trouble.
Future Directions and Solutions
Most organizations reduce risk by scaling back unnecessary stock levels. Smaller amounts, rotated more frequently, lower the odds of costly accidents. Advancing technology brings better containers, new absorbent materials, and even remote monitoring that alerts staff to temperature or humidity changes in real time. Companies switching away from 3,3'-Dichlorobenzidine altogether, when possible, show a forward-thinking approach, but not everyone can swap it out so quickly.
Good storage isn’t technical jargon or paperwork. It’s a set of habits and decisions that put safety before convenience. The real trick isn’t just knowing the guidelines—it’s living them, every shift, every procedure, every day.
What is the chemical structure and formula of 3,3'-Dichlorobenzidine?
Digging into the Formula
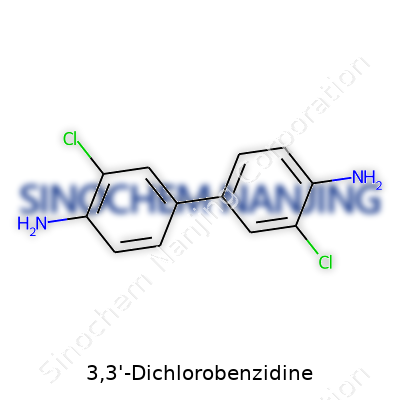
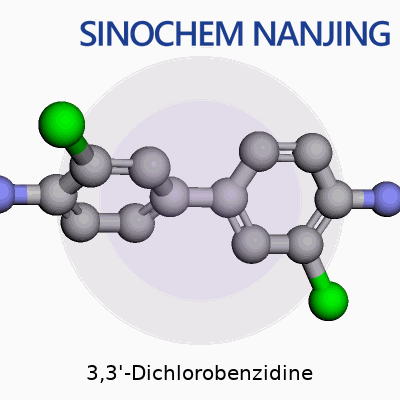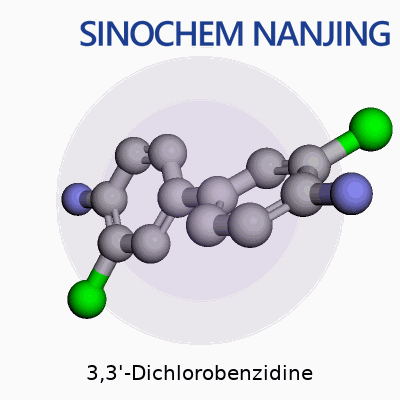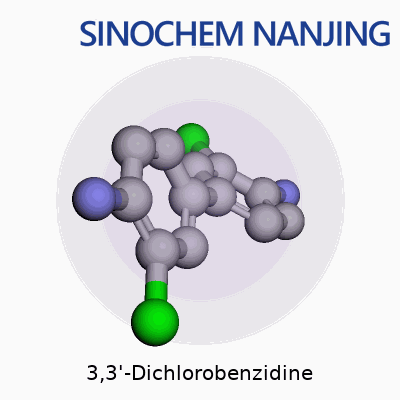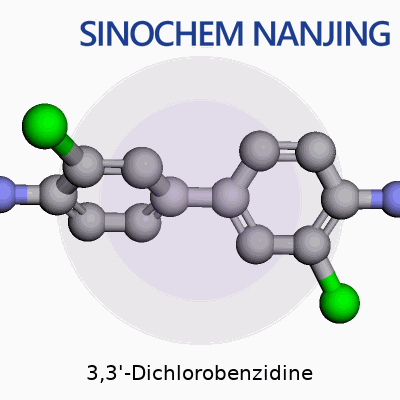
3,3'-Dichlorobenzidine stands as a foundational chemical for folks navigating colorant manufacturing, especially in dyes and pigments. The molecular formula is C12H10Cl2N2. It looks simple at first glance, but there’s more under the hood. The structure features two benzene rings tethered by a hydrazine bridge, with chlorine atoms sticking out at the third carbon on each ring. The full name—3,3'-dichloro-4,4'-diaminobiphenyl—gives a hint about those amine groups decorating either side.
More Than Just Numbers
It helps to step beyond theory. Think about each benzene ring as a six-sided playground. Each third carbon, counting from the point where the rings meet, hosts a chlorine atom. Stick an amine group (–NH2) at each fourth carbon, and you’ll start to picture its basic skeleton. The bridge between the two rings is the hydrazine connection (–NH–NH–), which doesn’t just flex the structure, it pushes this compound deep into the business of industrial organic chemistry.
Why Structure Sets the Tone
Back in my early days working in a college chemistry lab, drawing out structures like 3,3'-Dichlorobenzidine gave clarity to lab safety and chemical handling. Those chlorine atoms toughen up the molecule, making it stick around longer in the environment. While that trait works for long-lasting colorfast dyes, it turns into a headache for regulators and workers exposed in the workplace. Studies confirm its persistence and how it breaks down slowly, causing more concern over environmental and health risks.
Real-World Impact
Public health data led the International Agency for Research on Cancer to flag 3,3'-Dichlorobenzidine as a possible carcinogen. Factory workers, especially in lower-regulated sectors worldwide, still come into contact with it. Once I spent a summer job helping set up fume hoods at an old dye plant, and I saw firsthand how legacy chemicals like this one stick around. Unprotected skin exposure and inhalation risks stack up where safety corners get trimmed. These stories repeat anywhere cost savings trump health protections.
Learning and Solutions
The way forward depends on choices at every level of industry and regulation. Switching to less toxic dye-building blocks makes a difference but demands investment and retraining. Ongoing research now pushes for alternative compounds with reduced environmental impact. On the workplace side, strict engineering controls, protective gear, and real-time exposure tracking go a long way. In my experience, labs and plants that put in the effort see fewer health issues and better morale. Government agencies, through stronger enforcement and clearer public reporting, can push industry to phase out the most problematic uses.
Looking Beyond the Formula
The formula C12H10Cl2N2 captures the raw ingredients, but the full story comes from understanding both the structure and the real-life challenges tied to the chemical. Whether used in a manufacturing setting or entering the wider environment, knowledge and action together make a difference in reducing risk and sparking change. Each decision—right down to the safety gear worn in the plant—gets us closer to a safer chemical world.
| Names | |
| Preferred IUPAC name | 3,3'-dichloro-[1,1'-biphenyl]-4,4'-diamine |
| Other names |
3,3′-Dichlorobenzidine 4,4′-Dichloro-1,1′-biphenyl-2,2′-diamine o,o′-Dichlorobenzidine 4,4′-Dichlorobiphenyl-2,2′-diamine 3,3-Dichloro-4,4′-diaminobiphenyl |
| Pronunciation | /ˌθriːˌθriː daɪˈklɔːrəˌbɛnˈzɪdiːn/ |
| Identifiers | |
| CAS Number | 91-94-1 |
| Beilstein Reference | 120924 |
| ChEBI | CHEBI:81946 |
| ChEMBL | CHEMBL156210 |
| ChemSpider | 10235 |
| DrugBank | DB13875 |
| ECHA InfoCard | 100.005.853 |
| EC Number | 202-109-0 |
| Gmelin Reference | 77159 |
| KEGG | C14385 |
| MeSH | D03U7GVL5S |
| PubChem CID | 7341 |
| RTECS number | KH8575000 |
| UNII | IA46B1J9ZT |
| UN number | UN2579 |
| Properties | |
| Chemical formula | C12H10Cl2N2 |
| Molar mass | 253.09 g/mol |
| Appearance | Light gray to purple solid |
| Odor | odorless |
| Density | 1.5 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.8 |
| Vapor pressure | 0.00016 mmHg (25°C) |
| Acidity (pKa) | 13.70 |
| Basicity (pKb) | pKb = 11.86 |
| Magnetic susceptibility (χ) | -73.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.693 |
| Viscosity | 1.1 cP (25°C) |
| Dipole moment | 1.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 65.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3334 kJ/mol |
| Hazards | |
| Main hazards | Suspected of causing genetic defects. Suspected of causing cancer. Harmful if swallowed, in contact with skin or if inhaled. Causes skin and eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H350: May cause cancer. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P261, P264, P270, P273, P280, P301+P312, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2 Health:2 Fire:2 Reactivity:2 |
| Flash point | Flash point: 241°C (466°F) |
| Autoignition temperature | 500°C |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD50 oral (rat) 2830 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 2500 mg/kg |
| NIOSH | 197-09-5 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 25 mg/m3 |
| Related compounds | |
| Related compounds |
Benzidine 3,3′-Dimethylbenzidine 3,3′-Dimethoxybenzidine 3,3′-Dichlorobenzidine dihydrochloride o-Tolidine |

