3,3'-Diaminobenzidine Hydrochloride: More Than Just a Lab Reagent
Historical Development
3,3'-Diaminobenzidine Hydrochloride, often simply called DAB, has a track record in life sciences and diagnostic research dating back to the early 1970s. At a time when researchers searched for sensitive, specific chromogenic substrates for peroxidase enzymes, DAB quickly turned heads due to its reliability and ability to provide a visible, brown, insoluble precipitate. Across immunohistochemistry, its adoption meant clear visualization of target proteins in tissue sections, a major leap over previous dyes that washed away or gave a fuzzy outline. Early pioneers saw that DAB's stable reaction products gave pathologists and scientists a window into cellular details that had been blurry or out of reach. Laboratories worldwide still turn to DAB solutions even as newer, flashier markers compete for shelf space.
Product Overview
This compound shows up in labs as a light tan to brown powder, most often supplied in tightly sealed, opaque bottles due to its sensitivity to light and oxygen. Unlike ready-to-use kits that cut corners, pure DAB hydrochloride gives researchers the control they need over concentration and purity, both crucial for reproducible results. My time in academic and clinical labs taught me that consistency in reagents like DAB builds trust in experiment outcomes, especially in high-stakes environments like cancer diagnostics or forensic laboratories.
Physical & Chemical Properties
DAB hydrochloride appears as a crystalline powder, easily distinguished by its hue. Its solubility in water allows for simple solution preparation, yet its stability remains threatened by prolonged exposure to air and light, which can prompt autosoxidation or degradation. The hydrochloride form improves shelf stability compared to the free base, a lesson learned over years of field use. Chemically, it contains two amine groups that serve as the focal points in peroxidase-driven reactions, with a benzidine backbone providing rigidity and enabling it to form robust precipitates.
Technical Specifications & Labeling
Manufacturers specify DAB hydrochloride with purity levels above 98%, as lower grades introduce risk of background staining or unreliable chromogenic response. Product datasheets lay out critical identifiers: molecular weight (230.1), empirical formula (C12H14N2·2HCl), melting point (above 250°C, with decomposition), and clear hazard labeling in compliance with global standards (including GHS, OSHA, REACH). Correct storage conditions (cool, dry, dark) and batch tracking numbers provide a safety net for users needing to trace results or ensure consistency. Labels always warn about the compound’s suspected carcinogenic nature—a point I learned to respect every time I donned gloves and mask.
Preparation Method
Labs typically dissolve DAB hydrochloride in distilled water or buffer just before use. A fresh, filtered working solution maintains sensitivity in peroxidase detection experiments. Stock solutions stored for more than a few days often lose potency as the chemical gradually oxidizes. Some research groups optimize solubility with mild acidification, but strong acids accelerate breakdown. It's not unusual to see protocols suggest gentle heating under nitrogen or sealing tubes tightly to stave off unwanted side reactions. Years in the lab taught me you can’t cheat time or oxygen with DAB—freshly made always wins.
Chemical Reactions & Modifications
The heartbeat of DAB’s value lies in its peroxidase-mediated oxidation, generating an intense brown precipitate wherever the enzyme is present. The chemistry doesn’t stop there, though. Many labs enhance the signal by treating tissues with metallic salts—nickel and cobalt are common choices—post-DAB development. These modifications deepen the color or shift its hue, helping distinguish co-localized targets in complex samples. Some creative researchers attach biotin or fluorophores to DAB analogs, expanding its use into multiplexed imaging. Every new twist in chemical modification must balance greater visibility against risks like over-background or cytotoxicity, a challenge chemists and pathologists grapple with every new protocol iteration.
Synonyms & Product Names
Product catalogs carry 3,3'-Diaminobenzidine Hydrochloride under names like DAB HCl, Benzidine, 3,3'-Diamino-, Dihydrochloride, and CAS number 7411-49-6. This jungle of synonyms sometimes trips up procurement staff, especially with lookalike compounds on the shelf. Clear education and robust supply chain management prevent mix-ups. I’ve seen research teams grind to a halt over a mislabeled bottle, costing days of work and throwing results into question.
Safety & Operational Standards
Decades of experience in research settings hammered home the importance of safety with DAB. Classified as a potential carcinogen, contact with skin or inhalation carries distinct risk. OSHA and NIOSH recommend PPE that includes gloves, eye protection, and fume hoods. Waste must go in labeled, closed containers, destined for hazardous material disposal. My lab always included DAB in chemical safety training, spelling out accidental exposure protocols and emphasizing good lab habits. Institutions that cut corners find themselves at regulatory risk and, more importantly, put staff on a collision course with preventable exposure or contamination incidents.
Application Area
DAB proved its worth in immunohistochemistry, providing sharp, stable stains used in clinical diagnostics of cancer biomarkers like HER2 or p53. Pathologists rely on crisp, brown nuclear or cytoplasmic dots that persist through tissue mounting, storage, and even archiving. Beyond diagnostics, DAB shows up in enzyme-linked immunosorbent assays (ELISAs), western blots, and electron microscopy. Even plant biologists leverage it for mapping peroxidase activity in crops. Law enforcement labs sometimes use it in forensic detection of blood traces. My forays into neuroscience put DAB at center stage for tracing neural circuits: nobody forgets the moment a pattern of labeled cells emerges out of background, telling a story at the cellular level.
Research & Development
Research doesn’t stand still, and DAB remains both a staple and a springboard. New detection systems push for safer, cleaner alternatives with lower toxicity, yet few match DAB’s visual clarity and permanence. Scientists create hybrid protocols pairing DAB with automated staining robots or integrating it into multiplexed digital pathology workflows. Advances in reagent purification and packaging have lengthened shelf-lives and made daily handling less hazardous. I keep tabs on green chemistry teams reengineering DAB analogs to block formation of harmful byproducts—these innovations don’t just promise lower regulatory hurdles, they also improve lab air quality for everyone on the frontlines of experimental work.
Toxicity Research
Evidence mounts asserting DAB’s carcinogenic potential, mostly underlined by its connection to benzidine-derivative toxicity. Recent toxicology studies, some looking decades into animal exposure, reinforce calls for alternative stains when possible. Regulatory agencies cite both acute and chronic risks, including skin irritation, mutagenicity, and increased long-term cancer risk. Despite clear hazard data, reliable substitutes fail to dislodge DAB entirely from its pedestal, especially in settings where diagnostic fidelity transcends risk calculations. Risk management boils down to enforcing airtight operational controls, keeping exposure to the bare minimum—those who skimp just add names to injury logs.
Future Prospects
Few lab reagents combine proven results with ongoing risk quite like DAB. Its legacy endures because it solves real-world diagnostic challenges, connects today’s pathologists with yesterday’s breakthroughs, and anchors so many standard protocols that the life sciences would look unrecognizable without it. The future may see DAB giving ground to engineered chromogens with built-in safety features, yet no new contender has lasted decades under the microscope’s gaze quite like this chemical. Continued investment in green chemistry, user education, and robust regulatory frameworks can shepherd a new generation of stains while respecting the lessons learned from DAB’s storied career. For the lab worker, staying sharp, following best practices, and keeping an eye on new developments will help balance DAB’s utility with the safety imperative that shapes every good laboratory.
What is 3,3'-Diaminobenzidine Hydrochloride commonly used for?
The Heartbeat of Diagnostic Science
Hospitals and research labs run on more than just smart people and steady nerves. Reliable chemicals give doctors and scientists the tools they need to untangle mysteries in the body—sometimes on a microscopic level. Among these chemicals, 3,3'-Diaminobenzidine Hydrochloride (often called DAB hydrochloride) stands out. Most people won’t see this name outside a technical manual, but its impact stretches across medicine, biology, and even crime labs.
DAB in Immunohistochemistry: More Than Just a Stain
Pathologists look for clues in tissue samples, trying to figure out what’s happening at a cellular level. DAB hydrochloride helps turn invisible signals into clear, brown stains on microscope slides. In a common process called immunohistochemistry, antibodies—often likened to biological detectives—attach to specific targets such as cancer proteins. DAB reacts with these antibodies, producing a crisp brown color wherever those target molecules show up. The process allows doctors to actually see the spread or location of disease, not just guess based on symptoms.
Real experience at a histology lab taught me that without reliable stains, everything slows down. Diagnoses get delayed. It’s not just about coloring tissue. The reaction between DAB and peroxidase (an enzyme used in these tests) becomes a chain that links medical science to real patient answers. If doctors misread a slide, lives hang in the balance. DAB offers strong contrast and stays stable under the microscope. That clarity gives confidence in tumor diagnosis, autoimmune disorders, and infections.
The Role of DAB Hydrochloride in Research and Forensic Labs
Colleagues in neuroscience and plant biology depend on DAB hydrochloride as well. In neuroscience, tracking tiny fibers in the brain takes a good stain that doesn’t fade. Researchers tracing protein buildup in Alzheimer's or Parkinson's patients bank on the same brown marker. In forensic science, DAB’s use goes even further. Blood stains at a crime scene, even faint ones, become visible with DAB-based tests. Cops and crime scene investigators use these reactions to confirm the presence of blood, helping turn cold leads into solid evidence.
Safety and Ethical Considerations
DAB hydrochloride isn’t just helpful; it carries risks. The chemical can be toxic or carcinogenic with repeated exposure. Labs that use DAB, including ones where I trained, enforce glove-and-mask protocols. Waste disposal gets special attention since pouring leftovers down the drain could harm the environment. Some agencies now look for substitutes, but none give the same strong, lasting color—at least not yet. Awareness and good habits matter as much as the latest microscope.
Exploring Alternatives and Solutions
Chemists work on safer replacements. Digital imaging offers another path—using software to enhance faint signals instead of pushing harsher chemicals onto slides. Dedicated scientists worldwide keep testing new options, but few deliver DAB’s consistency. As new dyes and digital tricks improve, labs can reduce chemical usage and exposure. Collaboration between industry and academia will help bring safer choices onto the market.
Every discovery in the lab ripples outward. Decisions about chemicals like DAB hydrochloride aren’t just technical—they tie into health, safety, and the speed at which doctors and detectives uncover answers. Talking about what works, sharing better practices, and demanding safer solutions shapes the kind of progress everyone wants in medicine and science.
Is 3,3'-Diaminobenzidine Hydrochloride hazardous or toxic?
What 3,3'-Diaminobenzidine Hydrochloride Does
Lab workers and histologists often reach for 3,3'-Diaminobenzidine Hydrochloride, better known as DAB, to make the invisible visible. It’s the golden-brown color you see revealing cells in tissue samples under a microscope. DAB comes up in thousands of research papers—especially in cancer, neuroscience, and autoimmune disease research. Most folks outside a medical or research lab won’t run into it, but for people who do, paying attention to its hazards matters for health and safety.
Hazards: It’s Not Just a Stain
DAB isn’t harmless. Decades of toxicology studies—including animal tests—show it can damage DNA. The U.S. National Toxicology Program lists DAB as "reasonably anticipated to be a human carcinogen." That’s strong language. Skin or eye contact, inhalation, or accidental swallowing puts people at risk. Even with short exposure, some people report rashes or respiratory irritation. Once DAB enters the body, enzymes can break it down into compounds that damage cells, raising the chance of mutations. Anyone who works directly with the powder or solution is closer to that danger.
Facing Risks Directly
Some labs still treat DAB as just another chemical. I’ve seen lab benches with open containers, wrinkled gloves, and untrained helpers moving fast. That’s risky. Major safety incidents don’t always make the headlines, but accidental splashes and low-level exposure can add up. Local rules—like labeling, fume hood use, and hazardous waste disposal—reduce that risk, but only if folks take them seriously every day, not just when auditors visit. Young researchers with limited safety training might not hear frequent reminders about DAB’s dark side.
Staying Safe Day-to-Day
The clearest path to safety starts with practical steps and honesty about the dangers. Gloves, goggles, and a proper lab coat block most direct contact but don’t cover every risk. Vapor and fine particles call for a well-maintained fume hood. Open bottles should never stay outside containment. Waste—swabs, pipette tips, gloves, and old samples—can’t go in the regular trash. Landfills don’t break down these chemicals safely. That usually means yellow hazardous waste bins and pickup by licensed vendors. Even washing glassware needs rules, so no traces go down the sink.
Rethinking Stains and Processes
Research brings advances, but sometimes stubborn habits in science slow down innovation where safety is concerned. Some newer chromogenic or fluorescent stains can replace DAB without the same cancer link. Transition doesn’t happen overnight; budgets, protocols, and training each play a part. Institutions and suppliers have to evaluate alternatives with real science, not just marketing, and support labs in making the change. Nobody wants new hazards hiding in a new bottle, so solid toxicology data matters before swapping out reagents.
Building a Culture of Safety
Skills and habits in the lab often pass from older researchers to younger ones. Safety conversations can lag behind advances in research. I know from experience: reading old protocols covers experimental steps but rarely focuses on exposure risk. Real progress comes from leaders making time for drills and sharing updated information. People work best when they know why a rule exists, not just what to do. DAB has made vital discoveries possible, but keeping people healthy means weighing new options and doubling down on education and vigilance.
How should 3,3'-Diaminobenzidine Hydrochloride be stored?
Why Pay Attention to Chemical Storage?
Working with chemicals in a lab never feels routine. Each substance comes with its quirks, and 3,3'-Diaminobenzidine Hydrochloride (DAB hydrochloride) stands out for several reasons. Anyone who's worked with this powder knows it reacts easily to moisture and light. If left in the wrong conditions, DAB loses its punch, ruins experiments, and, more importantly, becomes a safety risk. Ignoring proper storage wastes supplies and threatens safety in shared labs. Responsible handling not only protects results but also those working nearby.
Common Problems from Poor Storage
DAB hydrochloride acts as a sensitive compound. Exposure to air, moisture, or sunlight turns it brown or makes it clump. This isn’t just annoying. It shows the material has changed, which compromises research and diagnostics. A friend once overlooked a slightly open lid, only to find a useless, oddly colored mass inside that cost hours in sample prep. Imagine that frustration multiplied across dozens of labs worldwide. Science doesn’t move forward that way.
Optimal Conditions to Maintain Quality
To stay effective, DAB hydrochloride belongs in a cool, dark place. I keep mine in tightly sealed amber bottles, tucked in a desiccator or a refrigerator at about 2–8°C. Direct sunlight causes the compound to degrade, so dark storage always beats clear glass containers. Some labs skip the refrigerator and store it in a temperature-controlled room, but any place hotter than room temperature risks early breakdown. Moisture in the air invites chemical reactions, so a dry container with a silica gel packet does the trick.
Why Labeling and Security Matter
Mislabeling a chemical container looks harmless until someone grabs the wrong bottle by accident. Safety routines in my lab involve clear, legible labels with contents, date received, and a hazard icon. A fine powder like DAB deserves this respect, given its links to health risks if inhaled or touched. I keep DAB behind a locked cabinet, reserved for trained staff, since it isn’t something trainees should handle unsupervised. In my early lab days, I saw someone forget gloves while refilling a solution—rashes and anxiety soon followed. No experiment is worth that risk, especially when better habits could have stopped it.
Reducing Waste and Protecting Staff
Every time a batch of DAB hydrochloride degrades from poor storage, a lab loses money and time. Too many labs treat “use by” dates as loose guidelines, but fresh stock works better and keeps experiments on track. Regular inventory checks help catch old reagents before they ruin test results. I put periodic reviews into my schedule, setting reminders to dispose of anything past its prime. This approach stops problems before they start and sets an example for others on the team.
Final Tips for Safe Lab Practice
Good habits around DAB hydrochloride benefit everyone from students to experienced researchers. Keep it cold, dry, and dark. Handle it only with gloves, and always recap bottles right away. Label everything clearly, and lock up the stock. Confidence in results grows from this sort of discipline, and colleagues who share your space appreciate the care. Reliable science and safer workplaces start with these steps.
What is the recommended method for disposing of 3,3'-Diaminobenzidine Hydrochloride?
What’s At Stake with DAB Hydrochloride
3,3'-Diaminobenzidine Hydrochloride (DAB Hydrochloride) shows up in labs as a trusty chromogen for immunohistochemistry. It helps researchers reveal what’s going on at the cellular level. But the same properties that make it useful mean you can’t just toss leftovers in the regular trash. A little bit of information goes a long way: DAB is a potential carcinogen. The dry powder itself and the residue from staining both present risks to both health and the environment.
I still remember the keen warnings from my days in academic research: always put DAB waste in dedicated containers, never let it go down the drain. It wasn’t about following rules for the sake of it. We wanted to protect ourselves and the city water supply. Some old habits stick because they matter.
Why the Usual Trash Bin Fails
Chemical waste isn’t regular garbage. When labs toss DAB Hydrochloride into ordinary bins or down the drain, those residues travel. They can slip into groundwater, start unwanted chemical reactions at waste facilities, and harm the folks handling that waste. The Material Safety Data Sheet for DAB tells a clear story: this chemical doesn’t just break down harmlessly.
Some institutions in the past ignored those warnings, leading to environmental headaches. In the US, the EPA categorizes DAB as hazardous. Disposal through improper channels led some cities to install extra filters and fight chemical contamination downstream. These lessons cost millions in cleanup, not to mention the cost in safety lapses.
Better Ways to Dispose of DAB Hydrochloride
Responsible disposal starts even before you open the bottle. Order only what you need for the next few months. Reducing surplus means lower risk and less waste. If a lab winds up with expired or unused DAB, the next step involves partnering with certified hazardous waste handlers.
Some universities and hospitals have their own disposal teams. They’ll sweep in with special collection bins: leak-proof, marked with clear hazard symbols, and dedicated exclusively for DAB and related chromogens. These bins keep the waste contained until it moves out of the building. A chemical-resistant liner inside prevents spills, and a tight-fitting lid keeps volatile dust where it can’t do any harm.
Major waste handlers transport DAB waste offsite in accordance with hazmat guidelines. At that point, the chemical meets its end in a high-temperature incinerator, monitored for toxic byproducts. Incineration at over 1200°C leaves little to chance, breaking DAB into stable ash and safe exhaust. Some waste companies report every step electronically, allowing labs to trace the journey from bench to final disposal. This kind of accountability builds trust in the system.
Personal Take: Safety Culture Trumps Shortcuts
Everyone wants to get on with their research or clinical work without mountains of red tape. Yet, a few extra minutes pouring waste into a secure container saved me a world of trouble—and kept the lab off the auditor’s radar. Building a habit of checking labels and following institutional guidance is worth more than hope or luck.
For anyone working with DAB Hydrochloride today: connect with your environmental health office. Ask for clear instructions and follow them, even if it means an extra trip down the hall. It’s about responsibility, not just compliance. Besides, the person who comes next will thank you for leaving a safe and clean workspace behind.
What are the handling precautions for 3,3'-Diaminobenzidine Hydrochloride?
Why Safety Around DAB Matters
Working in a lab brings plenty of rewards, but it also puts professionals in contact with substances that can do harm when handled carelessly. 3,3'-Diaminobenzidine Hydrochloride, usually called DAB, plays a key role in immunohistochemistry for staining—it lets researchers see what’s going on at a molecular level. But DAB deserves respect. It’s classified as hazardous. Research from the National Institutes of Health and European Chemicals Agency counts it as a possible carcinogen and a material that causes skin and respiratory irritation.
Personal Experience in the Lab
Days spent in pathology and molecular biology labs taught me that cutting corners with chemicals like DAB only invites problems down the road. Skin contact, swallowing dust, or just breathing in the powder can have lasting effects. Colleagues once neglected proper scarfing procedures, and they got rashes and persistent coughs, a hard-won lesson that even small mishaps can mean weeks of medical attention.
Staying Protected: Simple Steps Matter Most
Don’t let experience lull you into complacency. Always use gloves and eye protection. Lab coats should fit snugly and cover as much skin as possible. Face masks and well-fitted goggles serve as valuable barriers against accidental splashes and inhalation. Wash hands right after working with DAB, even if gloves were used every step.
Never use regular work surfaces for weighing or mixing DAB. Work in a chemical fume hood. Fans, air filters, and proper ventilation matter, especially when dealing with powders and strong-smelling substances. Build habits—check that airflow is steady and don’t open reagent bottles outside that safe zone.
Storage and Spill Control
Keep all DAB containers tightly sealed, stored in a cool, dry, and well-marked spot away from incompatible chemicals. Labels should be clear and easy to read. Lab workers shouldn’t reach blindly into shelves—organization saves time and, more importantly, prevents surprise exposures. Once, I saw a container mislabeled after a hurried restock, which created confusion that could have gone badly wrong.
Spills happen anywhere people work with powders or liquids. A small DAB spill turns into an emergency without quick, methodical response. Use absorbent pads or chem-spill kits, not paper towels, which may push the substance around. Wear a mask and gloves as you work. Dispose of everything in a hazardous waste container. Even minor residues left behind create risks for those who take over your workspace later in the day.
Hazard Communication: Don’t Work Alone
One of the best safeguards in chemical handling is an informed team. Safety data sheets must sit close by, and staff meetings should review the risks, especially for newcomers. Visual reminders—a sign over the workbench, for instance—keep hazards front and center. If anyone feels a tingle or cough after exposure, report and record it right away, no exceptions.
Better Habits, Safer Labs
Strict rules around DAB handling don’t get invented just to create paperwork. Keeping labs clean, well-labeled, and well-ventilated protects the health of every researcher, technician, and maintenance worker who passes through that space. It supports the long run—people stay healthier and research keeps moving forward. My own routine these days is simple: take extra time on setup, work with intention, and don’t leave cleanup to someone else.
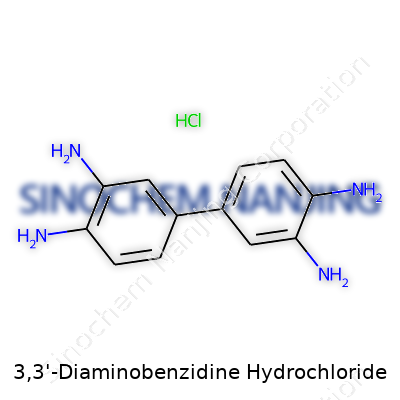
| Names | |
| Preferred IUPAC name | 3,3'-Diaminobenzidine dihydrochloride |
| Other names |
DAB Diaminobenzidine tetrahydrochloride 3,3’-Diaminobenzidine tetrahydrochloride 1,1’-Biphenyl-3,3’-diamine, hydrochloride DAB HCl 3,3’-Diaminobiphenyl dihydrochloride |
| Pronunciation | /ˈθriː ˈθriː daɪˌæmɪnoʊˈbɛnziːn haɪˌdrɒklaɪd/ |
| Identifiers | |
| CAS Number | 7411-49-6 |
| Beilstein Reference | 136478 |
| ChEBI | CHEBI:9505 |
| ChEMBL | CHEMBL1636 |
| ChemSpider | 10174769 |
| DrugBank | DB11447 |
| ECHA InfoCard | ECHA InfoCard: 100.028.860 |
| EC Number | 220-420-2 |
| Gmelin Reference | 126134 |
| KEGG | C06814 |
| MeSH | D03.633.100.221.173.221 |
| PubChem CID | 17449 |
| RTECS number | CZ0175000 |
| UNII | ZN58Z1566X |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C12H16Cl2N4 |
| Molar mass | 229.12 g/mol |
| Appearance | Light brown to brown crystalline powder |
| Odor | Odorless |
| Density | 1.28 g/cm³ |
| Solubility in water | Soluble in water |
| log P | “-2.2” |
| Acidity (pKa) | 9.19 |
| Basicity (pKb) | 11.18 |
| Magnetic susceptibility (χ) | -23.0×10⁻⁶ cm³/mol |
| Dipole moment | 4.78 D |
| Pharmacology | |
| ATC code | V04CX40 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause allergic skin reaction, suspected of causing genetic defects, may cause cancer. |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS05,GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P280, P305+P351+P338, P404, P501 |
| NFPA 704 (fire diamond) | 1-2-0 Health: 1, Flammability: 2, Instability: 0, Special: |
| Lethal dose or concentration | LD50 Oral Rat 472 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 464 mg/kg |
| NIOSH | UW40000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
Benzidine 4-Chloro-o-phenylenediamine o-Phenylenediamine 2,3-Diaminotoluene |

