3,3,5-Trimethylhexamethylenediamine: Industry Cornerstone and Lingering Questions
Historical Development
Ever since nylon’s birth in the 1930s, the world found itself eager for high-performance materials that could push back against the limits of older plastics and coatings. By the late 20th century, the chemical industry had shifted toward building longer, stronger polymers, and 3,3,5-Trimethylhexamethylenediamine began to show up on the radar. Chemists soon realized that its unique structure — a hexamethylenediamine backbone decked out with three methyl groups — carved out new territory for tailored polyamides. If you talk with folks who remember those years, they trace a direct line from increased demand for heavy-duty plastics in automobiles and electronics to the rising popularity of this diamine.
Product Overview
Many folks know 3,3,5-Trimethylhexamethylenediamine simply as TMHDA, and if you work in specialty chemistry, this is not some obscure lab reagent. It’s a colorless to pale yellow liquid at room temperature, but it's what it does in combination with other building blocks that matters most. TMHDA acts as a key raw material for certain engineering plastics, especially select grades of nylon and polyurethanes. Anyone building molecular chains for manufacturing automotive components, coatings, or adhesives knows that TMHDA’s presence can change the mechanical and thermal profile in the end product.
Physical & Chemical Properties
TMHDA brings a certain structure to the table: two primary amine groups anchored on a branched 9-carbon skeleton, three of which are methyl substituents. This structure keeps the melting point relatively high for a liquid diamine, and helps dial up chemical resistance in the polymers where it's used. Hydrogen bonding potential is robust, which comes in handy during polymerization. Those who’ve handled it in the lab report it to have a pungent odor; so working with TMHDA is never a “set it and forget it” exercise — you know what you’re working with. The molecular weight runs higher than simpler diamines, bumping up the viscosity, which brings its own challenges in industrial mixing but also grants more control in certain recipes.
Technical Specifications & Labeling
Labels matter, especially when you’re hauling reactive chemicals from one plant to another, or shipping barrels across the globe. TMHDA often arrives with regulatory labels warning of skin and respiratory hazards, and because it’s not as commonly used as ethylenediamine or hexamethylenediamine, extra attention goes into quality control. Purity levels sit above 98 percent for high-end applications. Storage often demands air-tight drums, temperature regulation, and rigorous documentation to meet occupational standards. Even back in the warehouse, you can’t help but notice the consistent attention to how every kilogram is tracked and handled to prevent accidental exposure or contamination.
Preparation Method
TMHDA doesn’t show up in nature, so it falls to organic synthesis. The usual route starts by alkylating hexamethylenediamine, slotting in three methyl groups onto the chain in a controlled way. Industrial chemists favor routes that limit unwanted byproducts, mostly because costs and downstream purifications can pile up fast. In big plants, reactors are filled under an inert atmosphere, reagents metered with precision, and the product worked up with solvent extractions or distillation. Old hands in the industry sometimes compare it to orchestrating a complicated recipe — get a single addition out of order, and your batch doesn’t pass muster for polymer-grade production.
Chemical Reactions & Modifications
Amines are workhorses in industrial chemistry, and TMHDA is no exception. Its primary amine groups welcome acylation, alkylation, and condensation reactions. Polyamide production pairs it with dicarboxylic acids in melt polymerization, producing chains that carry those methyl kinks down the backbone for greater flexibility and toughness. TMHDA can also be cured with epoxy resins — a move popular for high-performance coatings and adhesives. You see the payoff in applications where typical aliphatic amines might lose their edge under heat or chemical attack. And because it gives polymer chemists a handle for further functionalization, some R&D groups explore grafting or block copolymer strategies with TMHDA as the anchor.
Synonyms & Product Names
It’s never enough to keep track of just one name in the chemical business. Depending on the supplier, you might see 3,3,5-Trimethylhexamethylenediamine listed as TMHDA, 3,3,5-trimethyl-1,6-hexanediamine, or by various catalog numbers. Customs and regulatory documents often refer to CAS registry number 25620-58-0. For those scanning safety data sheets, these alternate names can keep shipments and inventories out of legal hot water, but on the production floor, most just say TMHDA.
Safety & Operational Standards
Nobody I’ve met gives short shrift to safety precautions around TMHDA. Direct skin or eye contact can cause burns or long-term irritation, and vapor inhalation causes real trouble for the lungs. OSHA and EU regulators impose tough occupational exposure limits, so everyone in storage or handling wears gloves, goggles, and sometimes respirators. Written procedures dictate spill responses, ventilation needs, and emergency showers. Many facilities run regular, unannounced drills not just to check compliance but to keep folks sharp, as even small mistakes can mean costly downtime or worse, serious injuries. Just as important, those at the end-use plants—working with the finished plastics—also look for residual monomer specifications to make sure worker and consumer exposure stays well below regulatory thresholds.
Application Area
The average car on today’s roads contains more plastic than those built fifty years ago, and TMHDA plays an unassuming part in that shift. Manufacturers turn to hexamethylenediamines like TMHDA as a chain extender for specialty nylons. This brings out a balance of flexibility and strength, which keeps parts like radiator end tanks, air intake manifolds, and component housings from cracking under stress. You’ll spot it in coatings, too, especially powder coatings where scratch and chemical resistance matter. In adhesives and sealants, its presence improves bond strength and resistance to weathering. More recently, it’s earned a place in composite development for wind turbine blades and electric vehicle battery housings, reflecting demands for materials that handle high-stress, high-heat environments while keeping weight down.
Research & Development
Stepping into an R&D lab where chemists are experimenting with TMHDA, you sense the impatience for breakthroughs. They test new polymer pairings, aiming to expand the diamine’s role into areas dominated by older, less efficient monomers. Projects focus on boosting high-temperature stability and limiting moisture uptake. Some explore TMHDA as a backbone for dendritic polymers or as a bridging unit in block copolyamides. Research from university-industry partnerships often digs deeper into reaction mechanisms, seeking pathways to make synthesis cheaper or recycling easier. The goal isn’t just to squeeze out better performance, but to address persistent challenges like lowering residual monomer content in finished plastics, or adapting new formulations to meet mounting demands for sustainable manufacturing.
Toxicity Research
Concern over health and environmental impact keeps TMHDA under scrutiny. Existing studies show that, like most alkylated diamines, it can cause moderate to severe irritation with acute exposure. Chronic effects remain an area of interest, especially as regulatory bodies push for more data on long-term low-level exposure, both in factory settings and in downstream consumer contexts. Environmental persistence and degradation products receive close watching, particularly because amines can be tough for wastewater treatment plants to neutralize. Some animal studies suggest high doses may affect organ systems; for now, industry watches new findings closely, as future regulations could tighten exposure limits or require reformulation of downstream plastics.
Future Prospects
Folks in specialty polymer development keep one eye on innovation and the other on sustainability trends. Demand for lighter, tougher, and more heat-resistant plastics appears set to climb, with electric vehicles, renewable energy, and miniaturized electronics leading the charge. TMHDA stands as one of several diamines with the backbone and reactivity needed for next-generation materials. Yet rising public and regulatory interest in the environmental cost of plastic production and disposal hangs over its future. Many believe the path forward means building in more closed-loop processes for TMHDA-based products, designing plastics that last longer without leaching hazardous monomers, and scaling greener synthesis routes. If recent years are any guide, technical progress will keep pace with policy, provided researchers, manufacturers, and regulators all row in the same direction. The conversation around TMHDA remains open, shaped at the laboratory bench, on the factory floor, and across the meeting tables of those weighing cost, performance, and safety for tens of thousands of products that hinge on its chemistry.
What is 3,3,5-Trimethylhexamethylenediamine used for?
Building High-Performance Plastics
Everyone uses plastics without a second thought. From cars to smartphones, these materials shape everyday life. What many people might not realize is that some plastics get their strength and longevity from specialty ingredients. 3,3,5-Trimethylhexamethylenediamine pops up here. Chemical companies rely on it to make polyamides—the backbone of nylon plastics—and certain coatings and adhesives. This diamine delivers toughness and heat resistance, features automotive engineers value. Decades ago, car bumpers often cracked or got brittle over time. Newer plastics, built using ingredients like this diamine, hold up in cold and heat. This results in lighter vehicles that cut fuel use while keeping drivers safer on the road.
Electronics and Devices Rely on It
Electronics manufacturers have their own reasons for wanting durable plastics. Circuit boards face heat, moisture, and physical stress. 3,3,5-Trimethylhexamethylenediamine helps make coatings that lock out water and keep circuits working. Without this kind of chemical innovation, cracked cases and failed connections would show up more often in phones, laptops, and even solar panels. It’s frustrating to toss out electronics because of avoidable failures. With better coatings and adhesives, devices last longer, offering some relief against waste and rising prices.
The Coating Factor in Heavy Industries
Protective coatings and sealants hold big industrial sites together. Oil refineries, chemical plants, and pipelines need coatings that don’t peel or corrode. Many formulas for these coatings depend on amines like 3,3,5-Trimethylhexamethylenediamine. My uncle, a maintenance engineer at a refinery, once showed me how these coatings saved hundreds of thousands in repairs after a harsh winter. A tiny shift in the way amines react with other ingredients makes the difference between a coating that flakes off and one that hangs on for decades. In practice, it’s not so much about chemistry as it is about fewer pipe leaks and lower risks for workers on site.
Environmental and Safety Checks
The very features that make 3,3,5-Trimethylhexamethylenediamine so useful—strength and chemical resistance—also mean extra care is needed. Handling these chemicals safely takes training and strict guidelines. Using gloves and proper ventilation isn’t optional in plants processing this compound. That’s not just lab talk; a neighbor once had a bad skin reaction from a warehouse spill involving a similar diamine. Workplace safety rules serve as a real buffer against long-term health issues.
The environmental side matters, too. Regulations like REACH in Europe keep a close eye on how much of these ingredients end up in soil and water. Companies have to report usage and invest in spill prevention. Their compliance goes beyond paperwork; it cuts the risk of long-term damage to local communities. People working with these chemicals, or living near factories, trust authorities and companies to handle both production and disposal smartly.
Balancing Progress and Responsibility
Advanced chemicals push industry forward, but they come with a responsibility to protect both people and the planet. Supplying better plastics and coatings means nothing if health and safety get skipped. Future improvements in green chemistry, safe substitutes, and improved waste treatment offer hope for solutions that don’t require trade-offs. Practical experience shows there’s no shortcut: investing in safety and environmental controls pays off, at home and out in the wider world.
What are the safety and handling precautions for 3,3,5-Trimethylhexamethylenediamine?
Why Chemical Safety Hits Home
Every time I hear someone talk about chemical safety in an offhand way, I remember the old days working in a small paint shop. We learned about risks the tough way. 3,3,5-Trimethylhexamethylenediamine, a mouthful of a name, deserves real respect. Once you put on those gloves and open a container, your habits matter. A day with sore skin, headaches, or trouble breathing, and nobody has to tell you twice. Keeping hands bare or skipping the goggles just doesn’t fly. This chemical can burn skin and set off asthma attacks. A splash in the eye becomes a trip to the ER. Stories travel fast on shop floors, and every one has a lesson baked in.
Practical Precautions—From Someone Who’s Been There
Nobody likes separating tasks from comfort, but small routines save big headaches. Nitrile gloves outlast latex when working with amines. The wrong glove or a crack in the surface means the chemical can creep through and cause irritation. In shops I’ve worked in, we checked gloves for holes every morning. Goggles should seal against the face. Open collars and loose sleeves tempt splashes, so buttoning up a lab coat keeps skin covered. Even tough folks often forget about shoes—canvas sneakers won’t stop a spill. Closed leather shoes serve better protection.
Some think the smell is just annoying, but that harsh odor means vapors. I’ve watched coworkers scoff at respirators only to regret it at lunch, coughing more than eating. A cartridge respirator, fitted for organic vapors, cuts risk. If the ventilation fan is running low, windows need to open, or the job stops until air gets cleared up. Hot days make fumes rise faster. I once watched a colleague open a drum indoors on a humid afternoon and pay for it with a pounding headache. Windows, exhaust fans, and breaks outside keep the body happy and the mind focused.
Storing Chemicals Right—Lessons From Ruined Floors
I’ve seen floors and metal shelves bubble and rust under careless storage. A leaky bottle tucked under a sink led to sticky shoes for a week. Coming in on Monday to that mess sticks with you. Only containers designed for corrosive chemicals deserve space in the storeroom. Tightly sealed lids, labeled bottles, and separate shelves help prevent cross-contamination. Mixing up labels costs money and health both. A misplaced bottle gets forgotten and someone discovers it the harsh way. Spill kits belong close by, not buried in a cabinet. It only takes a few seconds to knock a bottle over.
People Protect People
The best safety stories come from teams looking out for each other. Rough mornings, distractions, and a busy warehouse make it easy to miss a step. Sharing reminders on gloves, goggles, or that half-open vent duct helps everyone. Training sessions sometimes feel like a drag, but updates keep memories fresh. An old instructor once reminded us chemicals don’t care if you’re in a hurry. Personal monitoring—itching, redness, or funny smells—signals problems before warnings on a wall do. It comes down to routines, trust, and never getting complacent around amines.
Raising the Bar Across Industries
Regulations from OSHA set out clear ground rules. Places with strong safety cultures see fewer injuries and less lost product. I’ve seen labs track improvement just by posting reminders and rewarding safe habits. Real investment comes from management providing fresh gear and staying fair with breaks. When workers feel heard about their safety, they bring more care to the bench. Any business looking to stay in this field a long time makes safety a backbone, backed by daily action and honest conversations.
What is the chemical structure and formula of 3,3,5-Trimethylhexamethylenediamine?
Chemical Structure and Formula
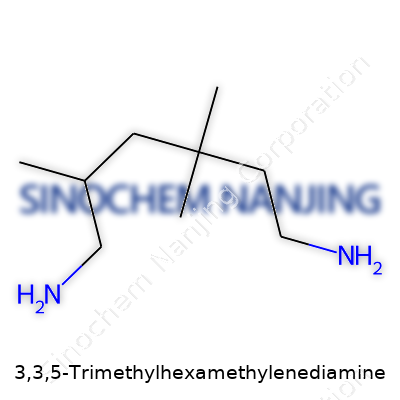
3,3,5-Trimethylhexamethylenediamine goes by a mouthful of a name, but its structure tells a clear story. This molecule belongs to the family of aliphatic diamines. Its formula reads: C9H22N2. Visualize a straight chain of six carbon atoms; now, substitute three of those positions—on carbon three and carbon five—with methyl groups (–CH3). Place amine groups (–NH2) at both ends. What you get is a compound that branches out in ways that change not just its look on paper but also its behavior in real-world applications.
What Makes its Chemistry Relevant
Organic chemistry rarely gets attention outside classes or labs, but practical use teaches the value of understanding these details. Taking the structure of 3,3,5-Trimethylhexamethylenediamine as an example, the methyl groups attached to the main carbon chain change more than the molecule’s shape. They impact physical features like melting point, boiling point, and even solubility. The presence of two amine groups turns this from just another hydrocarbon into something far more reactive and useful in synthesis.
I’ve seen firsthand how details like this play out for businesses making adhesives or coatings. The robust nature of this diamine, thanks to the extra bulk of those methyl groups, results in improved resistance to water and harsh chemicals. In polymer manufacturing, this means creating materials meant to last longer or handle more demanding conditions—job sites, machinery, even medical equipment.
Why Knowing the Structure Really Matters
It’s easy to gloss over chemical names, but no one cares about chemistry until a product breaks down before it should. The methyl branching in 3,3,5-Trimethylhexamethylenediamine doesn’t just change its reactivity; it makes things tougher and longer-lasting. Products exposed to weather, chemicals, or heavy wear benefit from this molecular architecture. Specialty plastics, high-performance composites, and advanced coatings all lean on compounds like this to perform when generic materials would fail.
Addresses Industry Needs and Safety Concerns
With any chemical, safety deserves attention. Exposure to diamines can bother skin, lungs, and eyes, so those who work with these chemicals need good ventilation, gloves, and a solid understanding of safe handling. Regulations in place demand up-to-date safety data sheets. Training matters. Nobody wants a chemical burn or sensitization after a shift.
Environmental rules around production and disposal grow stricter each year. Companies producing and using 3,3,5-Trimethylhexamethylenediamine invest in closed systems and waste treatment. Reducing emissions and controlling residue keeps both people and local ecosystems safer. From my time working around industrial coatings, I’ve noticed how even small process changes—better storage or cleaner disposal—can cut down on accidents and surprises.
Looking Toward Innovation
Demands for tougher, lighter materials from cars to electronics bring a steady drive for smarter chemistry. Formulators look for components that give polymers new strengths. Both research and market trends keep 3,3,5-Trimethylhexamethylenediamine on the radar. By studying the impact of each detail on a molecule, chemists find ways to improve not just a single product, but sometimes whole industries.
What are the storage requirements for 3,3,5-Trimethylhexamethylenediamine?
Understanding the Risks and Real-Life Challenges
I remember the first time I worked with 3,3,5-Trimethylhexamethylenediamine in the lab. The smell alone reminded me why attention to safe storage isn’t just for regulators, but also for the folks unpacking drums, using pumps, or cleaning up when no one else wants to. The chemical looks like a harmless liquid, sure, but underneath that clear appearance hides plenty of punch: this stuff can burn skin, mess with your lungs, and, if it gets hot enough, might even feed a decent fire. It’s not smart to keep it “just anywhere.”
Safe Temperatures and Container Choices
This amine doesn’t like temperature swings. Store it too warm and pressure can build inside containers, making leaks more likely. Anyone unlucky enough to open a swelling drum knows that pop can set the tone for a bad day. Cool, dry rooms—preferably below 30°C—fight this problem. Refrigeration isn’t necessary unless local climate really bakes, but direct sunlight? Don’t even think about it. Sun heats tanks up fast and this liquid, left alone, breaks down and releases vapors that attack the nose and lungs almost instantly.
Steel drums with tightly sealed lids are the go-to choice if you ask manufacturers. Plastics sometimes work, yet not all plastics stand up to this amine over months. Polyethylene works for shorter-term storage, but I’ve seen cheap plastic get brittle and crack after several months. Seals matter more than most expect—one loose ring and you’ll know by smell before anyone needs to check the label.
Moisture, Air, and Compatibility Hazards
Humidity creates its own headaches. If water finds a way inside, this amine can create pressure or react with impurities, leading to cloudiness or degradation. The stuff works much better if left in the dark and in airtight drums, ideally under a blanket of dry nitrogen if the operation is big enough to justify the extra step.
Don’t store it near acids, oxidizing agents, or even random cleaning chemicals. Spills mix easily on floors and odd reactions aren't as rare as folks pretend. One night I watched a small spill “fizz” on concrete after a bottle of bleach cracked—it was just a few centimeters, but it cleared out the room for hours.
Safety at Eye-Level: Labels, Gear, and Fire Rules
Accurate labels help new staff avoid mistakes. You’d be surprised how many people rely on hand-written duct tape tags in old shops. I’ve seen workers ignore standard black-and-white labels, yet no one misses a bright red “DANGER—CORROSIVE.”
Personal protective gear isn’t negotiable. Gloves, goggles, and long sleeves—these are the basics. I’ve met folks with skin burns because “the drum was sealed” and they thought it couldn’t splash. Accidents find the lazy and the rushed.
Don’t ever stack this chemical next to flammable solvents or under electrical cords. Fire regulations rule for a reason: this liquid, if it leaks, makes other accidents much worse. A foam fire extinguisher should wait close by, not hidden in a far corner.
Looking Ahead: Preventing Tomorrow’s Problems
Real safety grows from habits and a little paranoia. Rotating stock stops old material from degrading. Inspections every month pick up on rusty rings, weepy seals, or labels that have peeled away from humidity. Training shouldn’t stop at the safety video; hands-on drills and honest storytelling about close calls teach lessons in a way manuals never reach.
Is 3,3,5-Trimethylhexamethylenediamine hazardous to human health or the environment?
Toxicity Risks in Workplaces
Factories and labs using chemicals like 3,3,5-Trimethylhexamethylenediamine run into real challenges. This is a clear irritant. Even a quick splash can redden eyes, cause coughing, and burn skin. From what I’ve seen, workers feel the sting if protective gloves and goggles aren’t worn. Companies making epoxy resins or polyurethane systems often use this compound. Over years, some studies found repeated exposure led to asthma-like symptoms and trouble breathing. Occupational Safety and Health Administration doesn’t have a firm limit on this specific chemical, but the body’s signals are clear—dizziness, headaches, rashes. It pays to take these warnings seriously; safety data sheets point out the risks, but folks in the shop have to actually follow them.
Persistence in the Environment
It can be tempting to ignore what goes down the drain in an industrial setting, but that stuff resurfaces. 3,3,5-Trimethylhexamethylenediamine hangs around in water. Researchers in a few European rivers traced the chemical long after factory discharges had stopped. Plants and fish don’t break it down well. In some cases, small aquatic creatures struggled to reproduce or died off. This matters; if you like to fish or swim, no one wants persistent residue up the food chain. There’s no perfect clean-up. Prevention works better than any filtration or neutralization later on.
Regulation and Community Awareness
Most neighbors never hear the names of compounds being used a mile from their homes. Yet, in 2019, a German facility faced protests because residents didn’t get clear answers about fumes escaping the plant. Transparency matters. Regulators push for emergency plans and stronger notification systems. Europe’s REACH regulation lists similar amine compounds for restriction because once these chemicals get loose, they’re hard to track and manage. American laws lag behind, often letting companies decide what’s “safe enough.”
Keeping People and the Planet Safe
Practical safety means real action: Install closed systems so workers never have to touch the liquid. Good ventilation cuts airborne particles. I’ve seen workplaces put timed alarms for regular air checks, not just annual ones. Local communities pressured companies for public testing of groundwater and air; that kind of participation forced improvements. Simple substitution sometimes works; swapping out this diamine for less hazardous cousins in products, especially paints or adhesives, lowers the risks.
Better Decisions for Industry and People
Companies win trust when they involve workers in hazard reviews. Spreading information, using plain language, and giving workers tools to ask questions help everyone. Cleaner technology—closed pumping, sealed storage, smart sensors—pays off, both for health and profit. Local environmental groups do good work, offering independent testing when official answers come slow. Without pressure, safer alternatives tend to stay overlooked, because switching chemicals often brings upfront costs. Still, the price of ignoring health warnings or polluted water casts a longer shadow.
References and Trustworthy Sources:- National Center for Biotechnology Information – Hazardous Substances Data Bank
- European Chemicals Agency – Substance Information
- OSHA Safety Guidelines for Chemical Handling
- Peer-reviewed studies on aquatic toxicity and human exposure
| Names | |
| Preferred IUPAC name | 2,2,4-Trimethyl-1,6-hexanediamine |
| Other names |
3,3,5-Trimethyl-1,6-hexanediamine 2,2,4-Trimethyl-1,6-hexanediamine Isophorone diamine IPDA |
| Pronunciation | /ˌθriː,ˌθriː,ˌfaɪv traɪˌmɛθ.ɪlˌhɛk.səˌmiːθ.ɪlˈdiː.əˌmiːn/ |
| Identifiers | |
| CAS Number | 25620-58-0 |
| Beilstein Reference | 581158 |
| ChEBI | CHEBI:130209 |
| ChEMBL | CHEMBL3180536 |
| ChemSpider | 37269 |
| DrugBank | DB16360 |
| ECHA InfoCard | 30-211-545-1 |
| EC Number | 254-075-1 |
| Gmelin Reference | 102143 |
| KEGG | C18806 |
| MeSH | D017965 |
| PubChem CID | 91789 |
| RTECS number | XZ3150000 |
| UNII | EB4RS58M46 |
| UN number | UN2289 |
| CompTox Dashboard (EPA) | DTXSID8020823 |
| Properties | |
| Chemical formula | C9H22N2 |
| Molar mass | 170.31 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Ammonia-like |
| Density | 0.84 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | 2.13 |
| Vapor pressure | 0.02 hPa (20 °C) |
| Acidity (pKa) | 10.72 |
| Basicity (pKb) | pKb = 3.31 |
| Magnetic susceptibility (χ) | -0.0004 |
| Refractive index (nD) | 1.453 |
| Viscosity | 13.6 mPa·s (25 °C) |
| Dipole moment | 2.14 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 292.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -146.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6266.7 kJ/mol |
| Hazards | |
| GHS labelling | **"GHS02, GHS05, GHS07, GHS08"** |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H317, H412 |
| Precautionary statements | Precautionary statements: P261, P264, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P310, P312, P321, P330, P333+P313, P362+P364, P363, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 71°C |
| Autoignition temperature | 255 °C (491 °F; 528 K) |
| Explosive limits | 1.0–6.9% |
| Lethal dose or concentration | LD50 Oral Rat 910 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 910 mg/kg |
| NIOSH | UU8225000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.07 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Hexamethylenediamine Isophorone diamine 2-Methylpentamethylenediamine 2,2,4-Trimethylhexamethylenediamine |

