Digging Deeper into 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride: Past, Present, and Future
Tracing the Roots: A Walk Through Development
Chemistry has a way of creating a bridge between history and innovation, and the story of 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride proves this every step of the way. Dating back decades, diazonium salts shook up dyes and organic synthesis, and as researchers got their hands on more complex versions, like the one we’re talking about here, new doors opened in both science and industry. I remember sitting in a graduate seminar, marveling at the evolution from simple diazonium chemistries into functionalized molecules with tailored groups. The addition of a 2-hydroxyethoxy moiety and a pyrrolidinyl twist shifted this compound into a category with new solubility, reactivity, and safety profiles—not just a textbook curiosity, but a serious player in labs and manufacturing floors alike. This sort of progression reflects how chemical science relies just as much on incremental advances as on big eureka moments.
What the Product Stands For
3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride falls into the diazonium salt family but sets itself apart through two features: its functionalized benzenediazonium core and its pairing with zinc chloride. The hydroxyethoxy group gives it a certain friendliness toward polar solvents and a leg up in stability compared to more old-school diazonium salts. Tossing pyrrolidinyl into the ring means altered electron-donating properties, which can change the way this molecule interacts with nucleophiles or forms azo compounds. Zinc chloride does more than act as a counterion—it can tweak reaction rates and yields, which anyone who’s struggled with finicky diazotization reactions can appreciate.
Physical and Chemical Character
Think about handling something granular but not dusty, a product that doesn’t aggravate allergies or float in the air like chalk. This compound generally appears as a pale powder with a faint odor, dissolving readily in water thanks to the hydroxyethoxy group, making life easier for anyone who has spent hours coaxing reagents to mix without clumping. Its stability at room temperature extends shelf-life, addressing one of the key frustrations with less forgiving diazonium compounds that break down quickly, sometimes explosively. I’ve watched researchers debate storage best practices, and here, a desiccator or airtight container takes care of business without drama.
Going Beyond the Label: Technical Specs and Marking
Labeling practices now face stricter oversight than ever, not just for regulation but also for transparency and workplace safety. Labels for compounds like 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride highlight concentration, batch specifics, and protective handling icons. Anyone who’s ever had to backtrack through a poorly labeled shelf realizes that clear, comprehensive labels prevent headaches and lost research time. Chemical companies and research institutions rising to that challenge promote a safer, more reliable experience whether in academic or industrial settings. I’d argue that improved labeling mitigates unnecessary exposure risks and avoids wasted materials, which resonates especially in the wake of increasingly tight budgets and stricter waste management policies.
On the Bench: Preparation and Synthesis
Getting this compound on the bench involves a careful diazotization of a substituted aniline, which calls for an even hand and good temperature control. A careful addition of sodium nitrite under acidic conditions converts the starting aniline, after which the zinc chloride stabilizes the resulting benzenediazonium, decreasing the risk of premature decomposition. This route offers an improved safety margin over some older copper or silver analogs, especially in teaching labs or smaller pilot plant contexts. The shift toward water as a reaction medium, enabled by the hydroxyethoxy group, reduces harmful by-products, which I remember being a strong point in a safety audit years ago. Consistent yields depend on buffered pH and slow reagent combinations—anything less invites a messy reaction flask.
Reactions and Tweaks: Chemical Playgrounds
Chemists often look for new ground to cover in functional group modifications, and this molecule fits right into that spirit. Its diazonium group acts as a launching pad for nucleophilic aromatic substitution, enabling the formation of azo dyes, bioconjugates, and coordination complexes with surprising ease. The hydroxyethoxy and pyrrolidinyl modifications aren't just window dressing—they tune reactivity, sometimes helping to direct substitution patterns on the aromatic ring. I once saw a team turn what looked like a routine diazotization step into a starting point for anti-corrosive coatings by matching the electronic properties of the substituents with the intended metal substrate. The zinc chloride component keeps these transformations tidy; without it, by-products would balloon, making cleanup a serious chore.
Going by Many Names: Synonyms and Brands in the Field
Chemicals gather a crowded set of names—this one’s no exception. Alongside systematic naming conventions, supply catalogs might use shorthand like hydroxyethoxy-pyrrolidinyl diazonium zinc salt. Some journals still prefer the full mouthful. Fine, it’s confusing, but using CAS numbers can often cut through the jargon during procurement or literature review. Anyone who’s ordered the wrong chemical because of an ambiguous synonym knows the value of tying every inquiry to a verified CAS identifier to keep miscommunications in check.
Safety and Smarts: Practical Standards
People in the lab quickly learn that “benzenediazonium” signals extra vigilance. Proper ventilation, gloves, and goggles aren’t optional here, even with its improved stability. Zinc chloride, while less dramatic than some alternatives, still asks for respect—skin contact can cause irritation, so observers enforcing PPE protocols stand on firm ground. I’ve had colleagues recount times where simple procedural lapses—like skipping splash protection during weighing—led to minor but memorable incidents. Companies promoting clear training programs, consistent signage, and up-to-date safety data keep accidents at bay and insurance premiums reasonable. Safe disposal practices cut down on hazardous waste and keep regulators content—a practical win for any institution.
Everyday Impact: Where It Gets Used
This chemistry isn’t just an academic exercise; it pops up in industrial dye manufacture, bioconjugation routines, and as an intermediate in pharmaceuticals. In my own experience consulting for a custom synthesis group, I’ve seen it bridge small-scale R&D and scaled-up production. Its adjustable solubility makes it a favorite among formulators working with complex mixtures, whether laying down precision coatings on electronics or extending the life of printing plates. The hydroxyethoxy-pyrrolidinyl core nudges reactivity toward selective modifications, so custom dye designers rely on it when colorfastness and unique hues are needed under harsh process conditions.
Piloting New Science: Research Paths
Interest has grown in harnessing diazonium salts for cross-coupling and surface modification, and this one attracts attention for exactly those reasons. Integrated into new catalyst systems, it supports greener synthetic pathways—something I’ve seen kick off collaborative projects between academic labs and the specialty chemicals sector. As researchers turn to computational modeling, this molecule’s unique electronic profile provides plenty of material for machine learning studies, helping to speed up reaction design or predict toxicity outcomes in silico. Future breakthroughs often depend on tenacity and creative partnerships between universities and companies, looking to turn small molecular changes into big real-world effects.
Understanding Hazards: Toxicity Investigations
Benzenediazonium derivatives never fly under the radar for toxicologists. Studies routinely track cellular toxicity, environmental impacts, and metabolic fate. The hydroxyethoxy and pyrrolidinyl groups modify absorption rates and distribution in biological systems—a daily point of discussion in regulatory circles. Zinc chloride influences solubility but also raises potential aquatic toxicity during disposal, demanding responsible waste treatment, a topic that rarely lets up in technical forums. As industry pays closer attention to sustainable chemistry, updated toxicological data shapes exposure limits, with ongoing research adjusting operational best practices. Good intentions only get you so far; without robust evidence and accountability, even moderately hazardous chemicals can sideline entire production lines.
On the Horizon: What Tomorrow May Bring
The story never stops at current applications. Researchers see promise in leveraging this compound for next-generation dye-sensitized solar cells, smart materials, and biomedical tagging. Modifying the ligand shell or developing new counterion partners could nudge its reactivity even further, offering new synthetic strategies and safer, more sustainable reagents. As labs develop adroit protocols tailored around its chemistry, manufacturing, and disposal, the potential for innovation grows. The way forward means balancing innovation, safety, and regulatory compliance—and from what I’ve seen, today’s best practices often come out of cross-disciplinary teamwork, continuous learning, and the willingness to revisit yesterday’s solutions with fresh eyes.
What is the primary use of 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride?
A Closer Look at This Specialty Compound
Scientific names can sound intimidating, but here we’re looking at a diazonium salt that plays a focused, important part in the world of imaging and printing technologies. 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride might sound like something to leave to a chemist, but its impact lands much closer to home for anyone using printed circuit boards, architectural blueprints, or specialty graphic arts media.
Setting the Scene: Blueprinting and Imaging
Long before digital plans ruled the table, architects and engineers relied on a lower-tech hero: diazo paper. This compound is at the heart of that process, helping create images by reacting to light. Its most common destination sits in the diazo printing process—think of those deep blue background plans, often called blueprints, with thin white lines showing the technical details of a building or machine. These images don’t come from ink or toner; they form where light activates a chemical reaction in the diazonium salt, transforming treated paper wherever a draftsperson’s pen has left its mark.
The Science Behind the Impression
Diazonium salts play a key part here by breaking down in the presence of ultraviolet light. After exposure, ammonia vapor triggers the rest of the process, helping turn invisible impressions into sharp, visible lines. Without these specialized compounds, entire industries would struggle to produce quick, accurate reproductions of technical documents. Over time, this compound’s stability and responsiveness pushed it to the front of the pack among similar chemicals.
From Engineering Benches to Electronics Labs
Modern electronics depend on intricate copper patterns layered onto circuit boards. Photolithography, the process behind this, asks for pinpoint precision—one error and the board is junk. Here, a diazonium salt acts as a key ingredient in light-sensitive resists. Once again, the compound’s ability to react with light gives printed circuit designers a way to draw razor-sharp lines at a tiny scale. This step, though easy to overlook, underpins many types of electronic devices in daily life, from phones to cars.
Why Standards, Safety, and Reliability Matter
I once worked with a group installing large-format blueprint machines in a busy engineering firm. Cabinets full of diazo paper lined the back wall. Technicians always checked lot numbers and chemical freshness. If the chemical mixture veered off spec, everyone saw blurry or washed-out plans, wasting time and creating costly delays. The people making these chemicals follow strict purity standards and protocols for handling, so workers, users, and the public aren’t put at risk.
Regulation supports safety. The Environmental Protection Agency tracks and sets guidelines for handling these types of specialty chemicals, from storage temperatures to ventilation during printing. Proper labeling, training, and ventilation protect everyone down the chain—from factory workers to building inspectors.
What Lies Ahead?
Everything moves digital, but specialty chemical compounds like this diazonium salt stick around for good reason. While 3D design software floods the market, there’s still no quicker way to produce high-contrast, durable technical documents in some settings. The compound’s story reflects the broader arc of technology: old tools don’t fade overnight, and new ones usually have to prove themselves against what came before. Research into safer, more sustainable alternatives is ongoing, but for now, this specialty salt holds a unique place in industries where speed and accuracy matter.
What are the storage requirements for this chemical?
Why Storage Rules Matter
Ask any lab worker or warehouse manager about their toughest daily challenge, and the topic of handling chemicals rarely gets skipped. Safety protocols for chemical storage have been written in blood—literal and legal. One mistake can prompt everything from contamination scares to serious health emergencies, especially with substances that act violently with air, water, or each other. My time working in a school science department showed that even the usual suspects—like bleach or alcohol—can turn dangerous in the blink of an eye through simple carelessness. Mixing strong cleaning agents seems innocent at first until fumes start rising and eyes begin to water. Stories like that underline why regulations look so strict.
Factors Shaping Storage Demand
Every chemical brings its own set of quirks. Flammable liquids like acetone or ether call for rooms without spark sources and strong venting. Acids don’t play well with bases, so shelving takes on a chessboard pattern, dividing rivals with physical barriers. Humidity poses problems for powders that clump or react to water. Then there’s temperature—exposure to extremes can mean ruined products or dangerous leaks. In my experience, cold storage sometimes blocks access, pushing people to cut corners just to get a job done faster. That’s when bad things happen.
Forget the myth that sturdy shelves solve everything. Many chemicals corrode steel, eat their way through plastic, or shatter glass containers by building up pressure. Certain solvents creep out even through tightly sealed caps, gumming up labels and confusing the system at audit time. Choosing the right vessel for each chemical and knowing when to swap out an old drum can save both product and lives. I’ve watched a supervisor show how to “burp” a solvent container—a skill that rarely shows up in training manuals but definitely belongs there.
Common Sense Beats Convenience
Sometimes it feels like seasoned workers get so comfortable with routine they start dismissing the paperwork—labels, logs, or the monthly inventory check. But the real protection comes from these details. Unlabeled bottles or faded writing give no clues about hazard; more than once, I’ve had to trade guesswork for a cautious wait until an expert arrived. Inventory management also keeps expired or spoiled chemicals from sticking around, where unexpected reactions brew quietly out of sight.
Paths to Safer Practices
Staying safe means more than following codes written in some distant office. Staff should grow comfortable refusing shortcuts, even if it slows down the process. Refresher training does help, especially with real-life photos and stories—people remember lessons better with a context they recognize. Secondary containment, like spill trays and bins, catches leaks before they become problems. Regular cleaning keeps dust, residue, and trash from layering risk on top of risk.
Technology helps, too. Simple things like digital logs and QR-coded labels bring accountability. Automated alerts for approaching expiry dates remove the habit of “out of sight, out of mind.” I’ve seen companies set rewards for spotless inspections, boosting attention and pride in a safe workspace. Culture matters: having everyone buy in, from the boss to the intern, makes safety procedures less about rules and more about respect for each other’s well-being.
Conclusion
Storing chemicals the right way takes commitment, attention to detail, and the willingness to learn from both science and personal stories. Each guideline protects not just property or product, but real people with families and futures. That’s not just policy; it’s common sense, earned the hard way and worth repeating every single day.
Is this product hazardous or toxic to handle?
Sorting Fact from Fiction
Let’s face it—anyone who’s ever read a warning label knows one thing: it’s easy to get nervous about what’s inside the products we use every day. The truth is, some common items in our kitchens, garages, or workspaces will do more harm than good if you handle them carelessly. There’s no need to panic, but brushing off basic safety advice only leads to trouble. I’ve spent years around solvents, cleaners, and chemical products, and honest mistakes taught me why labels exist in the first place.
Why Labels Really Matter
That skull-and-crossbones logo isn’t just for decoration. Hazardous or toxic products can cause burns, breathing problems, or worse if you don’t treat them with respect. The immediate dangers—burns, rashes, or dizziness—give you signals that something isn’t right, but some chemicals do their worst damage slowly, without obvious warning signs. Research from the CDC shows that household exposures to toxins send thousands to emergency rooms every year. What’s striking: most injuries come from skipping basic steps like wearing gloves, working in fresh air, or storing products right.
Facts Over Fear
I remember the first time I spilled a strong cleaner on my skin. The sting made me pay attention, but the real lesson came from reading what I should’ve done in the first place. Government agencies—like OSHA and the EPA—have easy guides that break down which products pose the most risk. For instance, ammonia and bleach are cheap and widely used, but mix them and you create a poisonous gas. Even routine products, like certain adhesives, carry hidden risks for breathing or long-term health. The Environmental Working Group and similar organizations publish lists that flag the highest-risk substances and give clear advice you can trust.
What Keeps You Safe
Washing up after use, wearing proper gloves, or opening a window are the sort of tips you hear all the time for a reason: they work. For jobs that go beyond everyday chores, a bit more caution goes a long way. Safety goggles, masks, or even chemical-resistant aprons cut down on accidents. It pays to store products where curious kids or pets can’t get into them, and not everyone remembers to keep cleaners or solvents in original containers, a habit that cuts down on deadly mistakes. Data from the American Association of Poison Control Centers points to accidental ingestion from improper storage as a leading cause of injury, especially in households with children.
Finding Real Solutions
Safer alternatives exist for nearly every risky product out there. Homeowners can swap in plant-based cleaners for harsh solvents. Look for the “Safer Choice” label from the EPA, which means fewer toxic chemicals and an easier time breathing easy—literally and figuratively. For tough jobs where harsh ingredients can’t be avoided, pushing for better training and more straightforward labeling helps workers and regular folks alike. Companies have a duty to spell out the real risks, not just bury them in fine print. I’ve worked with organizations that switched over to less hazardous supplies, and over time, saw fewer health complaints and much less waste, which benefits everyone.
Every Choice Counts
No one keeps a lab at home, but understanding what’s really on your hands—or what’s swirling in the air you breathe—makes a difference you can feel. Hazards aren’t always obvious, but the tools to keep safe are right in front of us. Read labels, make swaps when you can, and don’t be shy about asking for training or information. Safety isn’t just extra work—it’s peace of mind.
What is the shelf life of 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride?
Why Shelf Life in Specialty Chemicals Cannot Be Ignored
Most people never worry about how long a diazonium salt remains stable. In the world of specialty chemicals, a product’s shelf life connects directly to both safety and economic waste. Relying on expired or degraded chemicals can give botched results, create dangerous situations, and drain budgets. This isn’t just cautious talk—there’s a massive body of research about diazonium compounds losing potency or even breaking apart unpredictably after a certain point.
What Science Says About This Compound
If you look at 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride, it behaves like other similar diazonium salts. Room temperature, light, and air all speed up its decay. Published studies and manufacturers’ tech sheets outline a typical shelf life of 6 to 12 months for related compounds, assuming sealed, dark-storage, and dry conditions. Zinc salts may offer extra stability compared to their nitrate or chloride cousins, but that only stretches the window so far.
Facts on Real-World Handling
From years of working in lab environments, the warning signs start to appear before the “expiration” clock runs out. That lemony-green powder might turn brown, clump, or start smelling off. Wasting a costly reagent because it sat in a humid storeroom happens more often than many will admit. Once I watched a team lose a whole day recalibrating their test after discovering a crucial reagent had decomposed ahead of schedule, even though the label claimed another three months. Quality control labs tend to respect these deadlines the most. They discard anything at the first hint of weirdness. Small operations sometimes try to stretch things, but the risks usually outweigh the savings.
Safety Isn’t Optional
This compound, like all diazoniums, isn’t content to sit quietly if ignored. Breakdown can mean pressure build-up and, in rare cases, release of gases, heat, or even explosions. Every chemical engineer or technician I’ve met treats old diazonium compounds with a healthy respect—often double-checking dates and handling old lots like gentle hand grenades.
Fine-Tuning Storage and Track-Records
Better storage really does stretch shelf life. Chemical refrigerators with desiccators help, but nothing beats a good inventory system. Barcode labeling and computerized inventory have become more standard, making it easier to rotate lots. For many companies, tighter controls mean less lost product and fewer safety incidents. It also means less scrambling during audits. In my own experience, teams using strict first-in, first-out methods almost never run into surprise failures, which keeps workflows smoother.
The Big Picture: Investing in Quality and Efficiency
Reliable shelf life info saves costs and prevents accidents. With diazonium salts, including 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride, the best approach leans toward the cautious side. Regular checks, clear labeling, and honest discussions with suppliers about real-world stability outlast empty claims or hope. Research continues to improve stabilizers, so newer lots sometimes last longer if stored right. For now, nobody wins by pushing luck on old, unstable compounds.
What is the recommended method of disposal for this compound?
Disposal of Chemical Compounds: More Than Just Throwing Away
Plenty of folks assume it’s harmless to pour leftover lab chemicals down the drain or toss them in the trash. Over time, some have learned the hard way that it’s not smart or safe. A little sodium cyanide at the wrong place, and the story goes from a minor mistake to a big disaster. The goal is clear: keep chemicals out of public water, away from landfill leachate, and miles from the food chain.
The Bigger Picture: Health and Environment on the Line
Hospital records and local news coverage show how poor handling leads to unpredictable injuries, contaminated soil, water issues, and even long-term cancer risks. A solvent container tossed into a dumpster in a school neighborhood once ended with an evacuation and a costly clean-up. Real stories up the stakes. Ignoring best practices turns every minor shortcut into a gamble with public safety.
Ignoring an MSDS, skimming past the instructions, or using the kitchen sink as a disposal site all play into the same risky logic. Mercury, old pesticides, or acids work their way into storm drains, catch attention from regulators, and harm more than just the user. Those living near disposal sites often shoulder the cost decades later as contamination persists in groundwater and crops.
The Recommended Path: Practical Steps in Action
Effective disposal starts by checking the compound’s specific Material Safety Data Sheet for instructions. Waste handling rules are stricter than many realize. City governments and state departments across the US run household hazardous waste collection, creating secure drop-off spots for everything from old paint thinner to expired pool chemicals.
Once there, trained staff sort materials by “compatibility” to prevent chemical reactions. Organic solvents go in one drum, acids in another. Authorized handlers ship these batches to treatment facilities instead of letting them linger in garages or under sinks. For new chemicals, labs schedule regular pickups through approved vendors. Medical facilities contract specialized companies, never asking their custodial teams to handle unknown liquids or powders.
Personal Responsibility in Labs and at Home
My high school chemistry teacher kept every chemical bottle in a locked cabinet, never letting us pour leftovers anywhere except the labeled “hazardous waste” jug. Now, in my own garage, I keep used motor oil and weed-killer bottles away from the trash can, waiting for the city’s monthly take-back event. It’s embarrassing to remember the days I flushed paint thinner, thinking water would neutralize anything. Later, after listening to a local water technician describe what trace amounts of solvents actually do to aquatic life and water treatment costs, I changed my habits for good.
Stronger Systems and Fixing Mainstream Gaps
There’s room to improve public awareness and convenience. Many communities lack frequent hazardous waste collection. Funding for education about proper disposal rarely stretches beyond emergency alerts after a spill. Giving clear, easy-to-understand instructions on household chemical labels helps those who want to do the right thing but lack confidence. Local governments can provide more accessible drop-off points and regular reminders, bridging the gap where personal discipline leaves off.
Individual efforts work best when local systems make safe disposal easy. By following guidelines and making careful choices, people keep dangerous substances off kitchen counters and out of rivers—protecting families today and generations tomorrow.
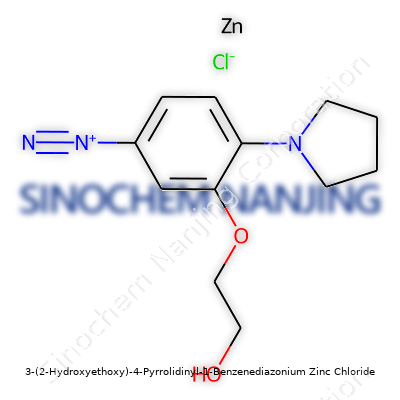
| Names | |
| Preferred IUPAC name | Zinc;4-(pyrrolidin-1-yl)-3-(2-hydroxyethoxy)benzenediazonium;chloride |
| Other names |
Fast Red TR Salt 4-(Pyrrolidin-1-yl)-2-(2-hydroxyethoxy)benzenediazonium zinc chloride FR TR C.I. 37175 |
| Pronunciation | /θriː-tuːˈhaɪdrɒksiˌɛθɒksi-fɔːr-pɪˈrɒlɪdɪnɪl-wʌn-bɛnˈziːndiaːzəˌniːəm-zɪŋk-ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 7149-10-2 |
| 3D model (JSmol) | `3D model (JSmol)` string for **3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride**: ``` [Zn+2].[Cl-].[Cl-].C1=CC(=C(C=C1N=[N+]=N-)OCCO)N2CCOCC2 ``` |
| Beilstein Reference | 3921816 |
| ChEBI | CHEBI:53251 |
| ChEMBL | CHEMBL4065157 |
| ChemSpider | 21517410 |
| DrugBank | DB11638 |
| ECHA InfoCard | 03b9007b-dbc5-4190-9e6e-e1ce2a08763d |
| EC Number | EC 603-725-7 |
| Gmelin Reference | Gmelin Reference: 1082156 |
| KEGG | C18610 |
| MeSH | D013602 |
| PubChem CID | 164095160 |
| RTECS number | GQ6475000 |
| UNII | XM1N0A6K2A |
| UN number | UN3276 |
| Properties | |
| Chemical formula | C12H19Cl2N4O2Zn |
| Molar mass | 379.56 g/mol |
| Appearance | Light yellow to yellow solid |
| Odor | Odorless |
| Density | 1.32 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -2.7 |
| Acidity (pKa) | 11.2 |
| Basicity (pKb) | pKb: 6.29 |
| Refractive index (nD) | 1.596 |
| Dipole moment | 3.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 370.3 J·mol⁻¹·K⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS06, GHS09 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 1-3-0-W |
| Flash point | >100°C |
| Lethal dose or concentration | LD₅₀ (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 15 mg/kg |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 3-(2-Hydroxyethoxy)-4-Pyrrolidinyl-1-Benzenediazonium Zinc Chloride: Not established |
| REL (Recommended) | REL (Recommended): "10 mg/m3 |
| Related compounds | |
| Related compounds |
3-(2-Hydroxyethoxy)-4-pyrrolidinyl-1-benzenediazonium chloride 3-(2-Hydroxyethoxy)-4-pyrrolidinylaniline 4-Pyrrolidinylbenzenediazonium zinc chloride 3-(2-Hydroxyethoxy)aniline |

