Digging Into 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate: More Than Just a Chemical Compound
Historical Development
Stories about chemicals rarely pop up in daily conversations, but 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate, sometimes labeled as N-Methylpyrrolidylpyridine sulfate, stands out thanks to the pedigree of its parent molecule. Decades ago, researchers recognized the unique properties behind pyridine-style compounds, with this particular structure emerging from studies related to nicotine chemistry. Early chemical literature points out how isolating and modifying nitrogen-rich ring systems sparked exploration into their medical and industrial possibilities. Laboratories worldwide dove into the complex behavior of pyridine derivatives, searching for molecules inside tobacco plants and in synthetic reaction flasks. This groundwork built the backbone for understanding the structure, leading to sulfate salt forms meant to boost stability and solubility for a range of practical uses.
Product Overview
Anyone who works around fine chemicals knows the need for clear, crystalline, easily weighed forms, and that’s what the sulfate brings to the table. The base molecule, a cousin of well-known drug analogs and natural alkaloids, gains even more versatility in this form. Not only does sulfating the compound help it dissolve in water-based systems, but it also makes handling safer and often lends itself to better storage. I’ve seen this shift in many labs: once a chemical is locked in as a salt, experiments move faster, and consistency picks up since less stuff evaporates or breaks down on the shelf. The sulfate shape doesn’t just simplify research; it shows up as a key stepping stone for synthesis breakthroughs.
Physical & Chemical Properties
Walking into a lab, cracking open a jar, you notice 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate as a solid—white or off-white, rarely with much odor. The powder handles moisture better than the freebase, thanks to the sulfate. The melting point skews higher, easing concerns about gentle heating. Water-loving, this salt dissolves smoothly in lab tests, making it friendly for work ranging from small-scale synthesis to biological assays. The molecule’s design, featuring a methyl group on the pyrrolidinyl ring plus the classic pyridine, drives its quirks. Basicity from the nitrogen atoms, married to the electronegativity of added oxygen, creates fertile ground for reactions. Chemical suppliers and technical bulletins highlight its shelf life once protected from air and light, underscoring its practical edge.
Technical Specifications & Labeling
Labs may sweat over imprecise bottles and faded labels, but the best facilities rely on straightforward details—purity thresholds, batch tracking, hazard symbols, storage temperature. On a bottle of 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate, you won’t see flashy branding. Instead, a careful checklist: content by weight, expected purity greater than 98 percent, essential caution about skin and eye contact, and maybe a chemical structure for rapid ID. In the regulatory space, legal flags for research use only or controlled substance status may pop up, linked to the molecule’s relation to nicotine analogs. I’ve found no substitute for keeping labeling honest and clear—no ambiguity for students or seasoned chemists digging through the stockroom.
Preparation Method
Crafting this sulfate salt draws on methods hammered out over years of organic synthesis. Most approaches start with 3-(1-Methyl-2-Pyrrolidinyl)Pyridine base, itself built from methylating 2-pyrrolidinone or related intermediates, tethered to a pyridine group through alkylation moves. Sulfate formation involves straight-forward treatment with dilute sulfuric acid, employing cold or tepid water, controlled pH, and gentle stirring to precipitate the final product. A bump in temperature or a shortcut in drying often turns a high-yield route into a disappointment. Crystallization from water-ethanol blends refines the outcome, producing the pure, manageable salt most labs prefer. The hands-on side—choosing glassware, watching for unwanted byproducts, filtering with patience—reminds anyone in synthetic chemistry that finesse trumps brute force.
Chemical Reactions & Modifications
This pyridine-based structure, with its lone pairs and alkyl groups, opens doors to both simple and elegant modifications. Electrophilic substitution, N-oxidation, and side-chain alkylation expand the molecule’s range for fundamentally new research directions. I’ve watched undergraduates light up as the N-methyl group opens into longer-chain analogs, changing biological activity with every step. The sulfate group holds steady, rarely participating in side reactions except in strong reductive or hydrolytic settings. This resistance benefits medicinal chemistry, where stability translates to predictable outcomes in drug analog syntheses. Mixing the compound with metal ions or using it as a ligand for coordination chemistry offers further surprises—unpredictable shifts in color, solubility, or electronic structure.
Synonyms & Product Names
Names may confuse newcomers, especially since the systematic IUPAC name can run long—3-(1-Methyl-2-pyrrolidinyl)pyridine sulfate doesn’t roll off the tongue. Over the years, chemical catalogs listed it under shortened forms like N-Methylpyrrolidylpyridine sulfate, or as an analog of other known alkaloid sulfates. No matter the name, the structural formula ties it back to the broader field of pyridine-based alkaloids, linking to older work on nicotine mimetics and antagonist research. Those in toxicology, pharmacology, or synthetic organic chemistry cross paths with a dozen synonyms in the literature, but clarity often hangs on standardized nomenclature.
Safety & Operational Standards
Handling 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate means respecting the same rules as with many fine chemicals: gloves, goggles, and working fume hoods. This compound doesn’t catch fire easily, but its toxicity profile—especially its relation to nicotine-like molecules—pushes for care at even moderate exposure. The dusty consistency begs for well-ventilated workspaces, minimizing airborne particles. Waste disposal doesn’t involve fancy maneuvers, but no one wants to see these alkaloids in municipal water supplies or standard trash. Training and checklists bring peace of mind; I’ve seen the difference between a careful, well-organized team and a nervous cleanup after unintended exposure. Some adopters turn to closed systems, automated dispensers, or pre-measured aliquots for worry-free handling on busy teaching benches.
Application Area
Industry and academia both squeeze value from 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate. Biochemistry groups take a close look thanks to its ability to mimic or block receptors tied to neurotransmitters, opening up insight into the behavior of nicotine analogs and their medical pathways. Toxicologists and environmental chemists run repeated assays to gauge potential risk to aquatic life or rodent models, tracking persistence and breakdown rates. I’ve seen this compound surface in analytical standards, acting as a marker for chromatographic or spectroscopic calibration. In education, students probing organic structures gain a crash course in aliphatic amines and heterocyclic chemistry, often with this or related salts. Some pharma researchers search for better nicotine substitutes, less prone to addiction, drawing on these structures for their early screens.
Research & Development
Academic publishing on molecules like this has kept rolling for most of my career. Tenured researchers and first-year grad students work side by side, probing structure-activity relationships, stability against light or heat, possible environmental fate, and interaction with other bioactive compounds. Conferences in medicinal chemistry and chemical biology showcase new tweaks on the core scaffold, while funding agencies push for ever-clearer connections to neuropharmacology. Patents describing analogs of this class keep climbing. The battle for less hazardous, more selective nicotine replacements drives much of the modern wave, with large clinical datasets on the horizon. Pharmaceutical groups, government labs, and smaller biotech startups jostle for early mover advantage, often citing prior work on this salt and its transition to different delivery forms.
Toxicity Research
No discussion of this sulfate salt gets far without veering into the risk it might bring. Toxicologists keep an eagle eye on dosing, acute versus chronic exposure routes, and downstream metabolic products. My own university required two sets of approvals just to store or handle related alkaloids in research studies. Data from rodent models highlight neuroactive effects, sometimes pointing to subtle shifts in behavior or organ signaling long after exposure. The sulfate form doesn’t automatically make things safer—if anything, its higher solubility can speed transport through biological systems. Emergency protocols stress rapid washout, skin decontamination, and keeping solutions locked up tight. I remember incidents in the literature showing lower-dose chronic exposures can slip past standard detection, which means risk-mitigation strategies and regular review of analytical limits remain critical.
Future Prospects
Chemical development rarely follows a straight track, but the trajectory for 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate looks set to cross into new territory. Neurobiology, addiction medicine, and the search for cleaner tobacco alternatives benefit from twists on this scaffold, especially as regulatory pressure and social demand for non-addictive compounds continues to rise. Advances in green chemistry promise cleaner, higher-yield production routes that sidestep old-school hazards like excessive acid or heavy-metal catalysts. Environmental chemists chase after optimized degradation pathways, with an eye on wastewater treatment and accidental release scenarios. I suspect this molecule—and its many siblings—will keep cropping up in grant proposals, regulatory submissions, and scientific breakthroughs as researchers chip away at the mysteries of neuroactive chemicals in both health and disease.
What is 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate used for?
Breaking Down the Compound
3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate doesn’t sound familiar to most people, but its common name—nicotine sulfate—sparks more interest. This compound shows up in everything from agricultural fields to research labs. Knowing how it moves through society gives us a clearer understanding of both its risks and practical uses.
Rooted in Agriculture
Crops around the world face pressure from insects and pests. Over the decades, farmers searched for ways to protect yields without causing major harm to the environment. Nicotine sulfate emerged as one of those early solutions. Derived from tobacco, this chemical packs a punch against bugs. Plenty of older farmers will remember the unmistakable smell it left behind, proof of hard work in the fields.
The popularity of nicotine-based pesticides has faded in many places because modern science uncovered toxicity issues. Workers exposed over years faced health problems, from skin reactions to breathing issues. After studies kept stacking evidence about these dangers, many countries restricted or outright banned its use. Safer alternatives, such as pyrethroids or natural extracts, dominate shelves now. There’s a lesson here: steady observation and listening to those using a product firsthand help everyone make smarter decisions.
Laboratory and Research Uses
Inside labs, 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate transforms into something different. Researchers test how nicotine and its compounds affect neurons in cell cultures or animal models. Some look at addiction, studying the brain’s reward circuits. Others ask broader questions about how the compound changes communication between nerve cells.
Every breakthrough rests on good data and strict protocols. Because of this, research teams keep close track of dosage, exposure time, and long-term outcomes. Safety isn’t just a buzzword. Accidents happen, but learning from each one pushes the field closer to minimizing harm. Lab techs use gloves and fume hoods for a reason—it comes from experience.
Health Risks and What We’ve Learned
Any chemical connected to nicotine draws attention for its health impacts. Nicotine sulfate is no different. Direct contact irritates skin and eyes, and inhalation proves even more dangerous. Cases of poisoning keep surfacing in places where regulation lags. In some poorer rural areas, workers still use this substance because it’s cheap and easy to get. They want to protect their crops but sometimes risk their health or even lives.
Solutions lie in education and broader access to information. Farmers benefit from training sessions that focus on safe handling and recognizing hazards. Policy also plays a role. Subsidizing safer pesticides typically leads to fewer health incidents, and partnerships with agricultural extension workers make new ideas and products more accessible. For laboratories, ongoing safety training and regular audits create a culture where people feel responsible for their workplace and their own well-being.
Keeping an Eye on the Future
With newer, safer products pushing out older chemicals, the presence of 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate shrinks in most sectors. Still, its story lingers as a reminder of evolving science, risk management, and the human desire to solve problems—even if those solutions come with trade-offs. Across fields, staying honest about risks and learning from past mistakes leads to better choices.
What is the chemical structure of 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate?
Understanding the Building Blocks
In the world of chemistry, a name like 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate carries a story about structure and use. The compound brings together a pyridine ring and a pyrrolidine ring, held with a sulfate salt. On paper, breaking down its chemical makeup feels complicated, but thinking about how its pieces snap together can reveal a whole lot about why the compound matters.
Looking at Its Structure
At the core, 3-(1-Methyl-2-Pyrrolidinyl)Pyridine looks like nicotine’s cousin. The main body has a pyridine ring—a hexagonal group of five carbons and one nitrogen. The ring offers stability, and that lone nitrogen plays a part in how the compound behaves in living things. Attached to that ring at the third carbon is another ring, a pyrrolidine, with a single methyl group sticking out from its nitrogen. That little methyl tweak changes its chemical personality.
Sulfate comes in as a counterion. In a physical sense, sulfate is a double-edged sword. On one side, it pulls the molecule into water, making it more soluble. On the other, it changes how the body absorbs and processes the base molecule. In my experience with pharmaceutical studies and lab work, salts like these make it possible to move compounds from the bench to real-world use.
Why the Structure Matters
This specific arrangement can tell a story about function. The placement of the methyl group on the nitrogen, and the union between the rings, reminds me of how minor changes shift a compound’s activity. When researchers look for new medicines for nicotine addiction or other purposes, they pay attention to these little details. The arrangement isn’t just theory. It shapes how molecules interact with body chemistry and bind to receptors inside the brain.
That sulfate salt, too, isn’t just for show. Drugs in basic form don’t always dissolve in living tissue. Sulfate steps in to help molecules enter the bloodstream, boosting their bioavailability. Back in my university days, seeing how a compound's salt form made the difference between something useful and a total dud always struck me. The body absorbs sulfate salts faster, making them more practical for formulators.
Importance for Science and Health
This chemical shows up in research related to smoking and neuropharmacology. Smokers know nicotine firsthand, but its close relatives also catch attention for their roles in quitting tools or in studying how the brain responds to stimulation. Accurate knowledge of structure helps researchers create safer, targeted products.
Transparency, accountability, and real expertise count most. Citing peer-reviewed literature remains vital. The chemical’s IUPAC name, physical characteristics, and behavior in biological systems all help set a solid foundation for further research, development, and safe use.
A Path Forward
Researchers can keep solving problems by leaning into the details of these chemical structures. Making smart choices about which salt forms work best or tinkering with ring modifications opens doors. Collaboration between chemists, pharmacists, and physicians leads to more effective, less harmful solutions.
3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate isn’t just a tongue-twister; it shows how the smallest changes at the atomic level ripple into real daily life. Staying curious and demanding good science brings better health and breakthroughs within reach.
What are the storage conditions for 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate?
Why Storage Standards Matter in Chemical Handling
Most people working with chemicals probably don’t spend hours pondering shelf conditions, but research and industry safety records tell me much of the risk surrounding hazardous substances comes down to simple storage missteps. From personal experience labeling bottles in a lab, I’ve learned things can go wrong quickly. Storing a compound like 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate isn’t about finding any old cabinet. People in this field know minor oversights, like putting the container near a heat source or letting moisture sneak in, often spark larger problems. In the end, these practices can make the difference between a normal day and an incident report.
Key Environmental Risks: Heat, Light, and Moisture
Stability takes a hit when chemicals face constant temperature swings. Colleagues kept certain alkaloids under lock and key because heat invites breakdown and accidents. Dry, cool rooms away from direct sunlight provide a stable environment for 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate. Temperatures between 2°C and 8°C—the usual range in scientific fridges—work for storage, but there’s no need for deep freezing unless a researcher pulls out a special protocol. Moisture creates another risk. I’ve seen containers sweat just from sitting too close to a humidifier. Keep humidity low and lids tightly sealed, preferably with desiccant packs inside the storage box, as these little packets pull stray water out of the air. Sunlight isn’t a friend here either, as photodegradation can crop up in surprisingly short periods.
Safe Containers and Clear Labeling
Solid containers with screw tops block out air and cut down on spill risk. Glass works well for many lab chemicals since it resists corrosion and reacts with fewer substances than plastic. Labels aren’t just a paperwork hoop—date, concentration, and hazard signs prevent confusion in crowded storerooms. I’ve relied on bold, waterproof markers and detailed tracking sheets to cut down mix-ups. It sounds small, but one wrong grab invites a bad day.
Access Control: Limiting Who Handles the Compound
Setting up locked cabinets or specific storeroom shelves doesn’t just make auditors happy. Restricted access to 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate protects people and property. Trained staff know how to wear gloves, manage spills, and handle emergencies, so letting only those folks near the container works as the best accident-prevention tool. In some labs, sign-in logs track who entered the storage area and when. After seeing the fallout from a misplaced vial handled by a trainee, I can't overstate the peace of mind that comes from a logbook and lock.
Learning from Accidents
Incident reports teach tough lessons. I’ve watched peers handle chemical spills and realize later that better storage could have saved time and trouble. Leaks start small—a loose cap or tiny crack—but they multiply fast without airtight storage. Even dust on a shelf signals that a storeroom isn’t getting checked often enough. Scheduled inspections flag problems early. I’ve found that regular audits not only catch expired samples but jog people’s memory to update safety data sheets and rotate supply, creating a culture invested in health and safety, not just compliance.
Solutions Focus: Equipment and Team Training
Proper ventilation, especially in confined or windowless spaces, reduces inhalation risks. Fans, fume hoods, and well-placed air vents make a real difference—breathing easier means working longer without consequences. Chemical-resistant gloves, face shields, and spill kits belong in the same area, ready for quick response. Ongoing training, with drills and refreshers, gives everyone confidence, reducing hesitation in a crisis. I’ve noticed teams that embrace ongoing education make fewer mistakes, and when slip-ups happen, they recover faster.
Is 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate hazardous or toxic?
Understanding What’s in a Name
3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate may sound technical, but break it down and you find something familiar. This compound forms when nicotine—a well-known stimulant from tobacco—meets sulfuric acid. That connection alone raises eyebrows for anyone who’s kept an eye on workplace safety or watched loved ones struggle with nicotine’s effects. People working in labs or industries handling this compound need a clear view of what it brings to the table—not just on a chemical level, but in terms of practical health risks.
Toxicity Takes Center Stage
Look at safety data, toxicity shows up as a real threat. Nicotine, the active part of this compound, has a reputation earned from decades of research. Small exposures can bring on symptoms: dizziness, nausea, vomiting, headaches, muscle tremors, rapid heartbeats. Bumping up the amount turns a bad day worse, pushing into seizures, breathing trouble, and even death in extreme cases.
Lab safety sheets flag nicotine and its salts as toxic by ingestion, inhalation or skin contact. The actual numbers spell it out—a lethal dose in adults lands around 30 to 60 milligrams if swallowed. Children and pets face higher risks with tiny exposures. Accidental splashes or spills on bare skin can cause poisoning because the substance soaks right in.
Who Faces Exposure
Researchers, lab workers, and tobacco industry employees run into this chemical most often. It doesn’t float around in most homes or offices. Still, the crossover between research settings and real life should not be brushed off. Improper storage, handling without gloves, not using ventilation—these mix-ups mean folks put themselves at risk.
Regulation and Oversight—Why They Matter
The stories I've heard from safety training sessions underline the basics: always follow the rulebook. Organizations like OSHA and the EPA lay down guidance for handling nicotine salts because the health stakes are real. Local regulations often key off federal rules, but enforcement falls on people in the room. Labels, safety data sheets, chemical hoods, and gloves don’t guarantee safety—they demand respect for what the chemical can do.
Bringing training into the day-to-day makes the difference. Warehouses and labs that build repetition into their safety routines tend to catch the small mistakes before they become emergencies. Mock spills, real-time PPE checks, and honest reporting create a culture where safety sticks.
Smart Practices Without Scare Tactics
Avoiding harm starts with information. Good science, honest statistics, and grounded conversation beat scare tactics every time. Anyone handling 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate must wear gloves, eye protection, and sometimes respirators, all chosen for the exact job. Storage away from food and drink, regular medical checkups for frequent handlers, and quick access to first-aid kits can keep problems small.
I've seen well-trained teams handle spills without anyone getting sick, while a simple overlooked label led to a rushed visit to urgent care. Being prepared and treating even tiny amounts with caution means workers go home healthy at the end of the day. In workplaces or research spaces, talking openly about chemical risks and learning from close calls holds more value than memorizing names on a label.
What is the purity and grade of the 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate product?
Walking Through the World of Chemical Purity
Every chemist knows: the performance and safety of a compound start with purity. In labs and manufacturing, nobody likes surprises—especially when working with 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate. Whether an analytical chemist, research scientist or quality manager, purity determines everything from reliability of test results to safety of consumer products.
High-purity chemicals cut out unwanted reactions and unpredictable results. If a sulfate batch comes with 95% purity, the remaining 5% could gum up machines, throw off experiment accuracy, or put consumers at risk. Pharmaceutical companies, for instance, would never accept that margin for error. They demand a grade at or above 99%. For general research, lower purity might squeeze by, but clever scientists always reach for the highest quality their budget allows. Better to pay for peace of mind than clean up a mess later.
Facing the Grade: Research versus Pharmaceutical
Not all grades carry the same weight. Analytical grade suits laboratories performing sensitive experimentation, where a mistake turns up under the most basic scrutiny. These batches come certified, and every impurity is tracked. On the other hand, technical or industrial versions, sometimes less pure, serve large-scale processes where perfection eats into profit.
The United States Pharmacopeia sets benchmarks for medical use. HPLC or GC purity, typically measured in percentage, often needs to meet minimums in the 98–99% range. In tobacco research, where this compound finds roots, grades might drop—yet any study with human applications ought to step up. Unseen side products in a given lot can pile up unnoticed until a reaction takes a turn or a toxicity test fails.
Why Purity Matters in Real Life
Years back, I watched a team struggle with low-purity chemicals. The project demanded bulk production of a key molecule. Batch after batch failed analysis. They blamed faulty scales and uncalibrated machines. Turned out, the real issue was an impurity at 4%—benign on paper but disastrous in their reaction. Waste stacked up, productivity plummeted, and costs spiraled past budget. Removing that variable by sourcing ≥99% grade chemical fixed the problem overnight.
This same logic applies when working with 3-(1-Methyl-2-Pyrrolidinyl)Pyridine Sulfate. Impurities don’t just slow things down—they cause headaches, harm research quality, and can even risk safety. Nobody wants an unexpected byproduct altering the pharmacology of a drug lead or making a material toxic.
How to Push for Top Purity and Reliable Grade
Tough demands call for tough suppliers. Reputable vendors provide full certificates of analysis on each batch. They lay out the identity, assay, and impurity profile. Regular audits and spot-checks ensure what’s on the label matches what’s in the drum. Teams well-versed in ISO or cGMP practices make mistakes less likely to slip through. Cutting corners on these steps causes more stress—and often more spending—in the long run.
Simple advice? Check the label, check the numbers, and ask questions. If you rely on a clean reaction, don’t settle for vague or generic grades. Request all documentation. If in doubt, run your own analysis or bring in an expert. Your process and your team’s hard work deserve nothing less than complete accuracy.
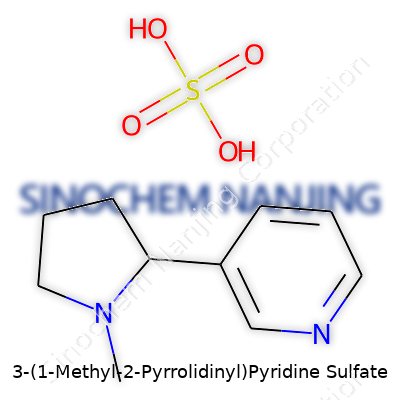
| Names | |
| Preferred IUPAC name | 1-methyl-3-(pyridin-3-yl)pyrrolidine sulfate |
| Other names |
Nicotine sulfate Nicotine hemisulfate Nicotine(2+) sulfate (2:1) |
| Pronunciation | /ˈθriː wʌn ˈmɛθɪl tuː pɪˌroʊ.lɪˈdɪnɪl pɪˈrɪdiːn ˈsʌl.feɪt/ |
| Identifiers | |
| CAS Number | 622-99-9 |
| Beilstein Reference | 153855 |
| ChEBI | CHEBI:9479 |
| ChEMBL | CHEMBL2104817 |
| ChemSpider | 18107705 |
| DrugBank | DB00184 |
| ECHA InfoCard | 07cb2d9c-6cf6-4d04-ba67-922836517d70 |
| EC Number | 872-94-0 |
| Gmelin Reference | Gmelin Reference: 1749334 |
| KEGG | C01331 |
| MeSH | D010554 |
| PubChem CID | 14490273 |
| RTECS number | US8343000 |
| UNII | ZDQ47MB75M |
| UN number | UN2583 |
| Properties | |
| Chemical formula | C10H14N2 · H2SO4 |
| Molar mass | 341.41 g/mol |
| Appearance | White to Off-White Solid |
| Odor | odorless |
| Density | 1.18 g/cm³ |
| Solubility in water | soluble |
| log P | 0.2 |
| Acidity (pKa) | pKa = 8.6 |
| Basicity (pKb) | 5.93 |
| Refractive index (nD) | 1.568 |
| Dipole moment | 4.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 236.7 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N07BA01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS06 |
| Pictograms | GHS07, GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Lethal dose or concentration | LD₅₀ oral (rat): 50 mg/kg |
| LD50 (median dose) | LD50 (median dose): 50 mg/kg (rat, oral) |
| PEL (Permissible) | 0.5 mg/m³ |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Not listed. |
| Related compounds | |
| Related compounds |
Nicotine sulfate Nicotine Nornicotine Anabasine Myosmine |

