3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin: A Groundup Review
Historical Development
If we track the long road that coumarin derivatives have traveled, stories reach back over a century. Early pharmacologists got hooked on the blood-thinning effects after noticing how spoiled sweet clover led to unexplained bleeding in cattle. Once Karl Paul Link and his team at the University of Wisconsin started unraveling the mystery in the 1940s, a wave of coumarin modifications hit the lab bench. 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin grew out of this fever for new anticoagulants, as researchers sought improved activity and different side effect profiles by tweaking the aromatic rings. Even before the warfarin boom, these naphthyl derivatives promised longer therapeutic windows and practical dosing for doctors trying to save lives at a time when hemorrhagic disease often had no answer.
Product Overview
This compound has become a staple in the chemist’s repertoire and the pharmacist’s shelf. It arrives as a white to pale yellow crystalline powder, stable at room temperature with reasonable shelf life when kept in proper containers. Unlike the basic coumarin, the bulky tetrahydronaphthyl moiety transforms it into a more intriguing molecule: lipophilicity rises, and so does the ability to bind proteins and slip through membranes with less resistance. Over the past five decades, this ingredient has featured in the synthesis of next-generation anticoagulants and served as a scaffold for drugs targeting clotting disorders, as well as experimental models for researchers screening new molecules against vitamin K epoxide reductase.
Physical & Chemical Properties
The powder’s melting point usually hovers between 175 and 179 °C. Chemists measure its solubility in polar aprotic solvents like DMSO and DMF, as water barely dissolves it thanks to the nonpolar regions of the core. Its molecular weight sits at about 294.36 g/mol. Compounds like this one pull in attention for their UV absorbance: the coumarin ring imparts a sharp peak around 310 nm, which analytical chemists use for quantification or tracking reaction progress. While the molecule stands up to air, handling near strong acids, oxidizers, or sources of ignition asks for the usual caution that synthetic labs demand.
Technical Specifications & Labeling
Bottles or drums arrive labeled as either 3-(1,2,3,4-Tetrahydro-1-naphthalenyl)-4-hydroxy-2H-chromen-2-one or simply as “THNHC.” Purity grades often list 98% or higher, with minimal moisture and verified by HPLC and NMR. Labels must highlight storage instructions—keep dry and tightly sealed, ideally between 15–25 °C. The product’s MSDS stresses its particular class as an anticoagulant, outlining risks for skin or mucous membrane contact and emphasizing proper PPE. Supply chains now often demand full traceability down to the reactor batch and include QR code linking to digital compliance records.
Preparation Method
Lab-scale synthesis leans into Pechmann condensation as a backbone, then plugs in the cyclic naphthyl moiety via Friedel–Crafts acylation, followed by catalytic hydrogenation of the aromatic ring. Workers add phenol and ethyl acetoacetate under acid catalysis, and after careful drying and purification, the naphthyl group inserts with aluminum chloride guiding the process. Once the structure is locked, a quick column on silica provides the targeted product, but industrial routes scale this up using continuous flow reactors, in-line drying stations, and closed systems that reclaim solvents efficiently.
Chemical Reactions & Modifications
Chemists love a scaffold that takes well to modification. THNHC's coumarin system handles halogenation and nitration to yield new candidates for biological testing. Sulfonation improves water solubility for injectable forms, while the naphthyl ring acts as a sweet spot for adding alkyl groups that can stretch half-lives or change metabolic pathways. Researchers have chased these modifications, sometimes with one eye on improved anticoagulant activity and another on reducing toxicity. The key transformations involve routine organic reactions: reductive amination, esterification, and cross-coupling, giving medicinal chemists a broad playground for exploration.
Synonyms & Product Names
In different corners of the globe or across suppliers, the same compound might carry several names. “THNHC” acts as the lab shorthand, but literature might mention its full IUPAC name: 3-(1,2,3,4-tetrahydro-1-naphthalenyl)-4-hydroxycoumarin. Other identifiers in catalogs include “1-Tetrahydronaphthyl-4-hydroxycoumarin,” or its systematized registry numbers for chemical buyers. Pharmaceutical contexts reference batch codes, and references in anticoagulant research sometimes just call it “the naphthyl-coumarin analog.”
Safety & Operational Standards
Direct exposure to this compound brings the same risks as other potent anticoagulants: easy skin or eye irritation and potential for toxicity if inhaled. Labs handling gram or kilogram quantities operate under fume hoods, with gloves and goggles as standard. Emergency showers and eye stations sit close by, and spill kits for organic solvents line the walls. Waste gets collected as non-chlorinated organic, with regular training on spill response and first aid for exposure. Newer standards push for closed-system transfer, especially in drug synthesis settings, where questions about operator safety drive process redesigns. Most critically, users track inventories to prevent accidental mixing with other potent biologics, as cross-contamination can pose real health hazards.
Application Area
Medical chemistry took early interest in this molecule, especially for its anticoagulant profile. The naphthyl group plays a critical role in mimicking vitamin K, making the molecule ideal for comparing clotting pathway inhibitors. Drug developers borrow its scaffold for binding studies looking at liver enzymes or serine protease receptors. Outside medicine, materials scientists once ran screens using derivatives as fluorescent dyes, but its mainstay rests solidly in hematological research—charting territory between academic curiosity and real-world therapy. Clinical interest also persists in exploring new oral agents for stroke and pulmonary embolism prevention, and each fresh derivative offers a shot at improving outcomes.
Research & Development
R&D in medicinal chemistry never sits still, and this compound keeps making its way into research papers and patent filings. Teams at public universities and drug companies keep testing new modifications for enhanced bioavailability, better metabolic profiles, and reduced risk of adverse reactions. Recent research lasers in on targeting resistance in warfarin-tolerant strains by exploiting slight tweaks in the hydrophobic pocket of vitamin K epoxide reductase. AI-guided modeling now slashes the time between design and bench testing, letting chemists rotate through variant after variant. At the same time, preclinical studies benchmark toxicity in animal models to make sure innovation does not come at a price to safety.
Toxicity Research
Toxicologists bring a hard-nosed approach, running rigorous screens of this coumarin derivative in rats and rabbits. Results track bleeding thresholds and liver enzyme disruption, as well as the compound’s fate when swallowed, inhaled, or absorbed through the skin. Data point to LD50 values that resemble other strong vitamin K antagonists, so overdosing poses real risk of hemorrhage. Long-term studies check for any carcinogenic effects and signal that most problems tie back to blood loss, not direct tissue damage. Regulatory agencies expect these toxicity profiles to accompany every new drug application built around this molecule. Proper animal handling and reporting standards matter, as do honest assessments of any negative findings.
Future Prospects
Looking ahead, this coumarin derivative stands in line for more rounds of molecular tinkering. Pressure from both clinics and health regulators spurs chemists to push for lower toxicity and faster onset drugs. The rise of personalized medicine hints that future patients may one day receive custom-tailored coumarin analogs based on their own DNA, with dosing and monitoring cut to fit their actual needs. Green chemistry draws attention too: the drive for solvent-free reactions and recyclable catalysts crops up in patent applications, as big chemical manufacturers answer the call for less waste and safer workspaces. At the same time, AI-driven compound screening promises to uncover fresh ways to tweak the naphthyl or coumarin core, aiming at not just better drugs, but smarter, leaner paths from idea to pharmacy shelf. Even in the face of new oral anticoagulants from beyond the coumarin class, researchers keep circling back to this molecule, convinced there’s more to wring from its famous scaffold.
What is 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin used for?
Understanding 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin
This compound, with a name that’s a mouthful, has earned a place in the pharmaceutical world for good reason. Most folks wouldn’t recognize it if they saw it on a label, but chemists and doctors have a different perspective. This molecule comes from the coumarin family. People have long looked to coumarins as a base for medicines, and this particular version is no exception.
A closer look at its structure reveals a connection to warfarin, a blood-thinning medicine many people know as an anticoagulant. Warfarin and its cousins, including 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin, come from efforts to manage clotting problems. Blood clots are dangerous—just ask anyone with a family history of stroke or heart attack. Because of this, society depends on medicines like this to keep blood flowing well, especially in people at risk.
Why Anticoagulant Medicines Matter
I’ve seen what unmanaged clotting can do—just one trip to an emergency room paints a clear picture. Without medicine to thin the blood, a clot can cut off oxygen to the brain or block a vessel in the heart. Doctors turn to chemicals like this one because of their proven track record. According to the American Heart Association, nearly 900,000 Americans experience blood clots each year, and more than one-third of them can die if they don’t get timely treatment. That’s not a number you can shrug off.
Doctors often prescribe these medicines to folks with atrial fibrillation, those who have artificial heart valves, or people recovering from certain surgeries. The idea is pretty simple: lower the risk of a clot and you cut the chances of a major stroke or heart attack.
Challenges & Side Effects
Using medicine like this involves trade-offs. Thinning the blood can save your life, but it isn’t always smooth sailing. People on these medicines need to visit the clinic for regular check-ins. Their blood must strike a careful balance—not too thin, not too thick. Even a small scratch can turn into a big problem, and internal bleeding isn’t always easy to spot. WebMD points out that patients on such drugs sometimes deal with bruising, blood in the urine or stool, and nosebleeds.
Doctors and patients manage this dance with habit and routine. Regular testing, sticking to medication schedules, and avoiding certain foods and supplements makes a big difference.
Innovations and Patient Safety
Chemists keep working to make these compounds safer and easier to use. Researchers watch for new formulations that last longer or cause less trouble. Pharmacy teams support patients by explaining side effects and watching for dangerous interactions. Online health records and smartphone apps now help people track their doses.
The World Health Organization adds that broad access remains a concern. Pricing, distribution, and public awareness shape outcomes, especially for low-income families. Some organizations push for generic options to lower costs and expand coverage. This holds promise so that more people can benefit from advances in anticoagulation medicine.
A Community Responsibility
Patients, doctors, researchers, and advocates must all push forward. Regular blood tests, communication with healthcare teams, and community education go a long way. Family conversations about health history sometimes lead to life-saving choices. As research progresses, the hope is that safer and more affordable treatments will reach those who need them most.
Is 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin toxic or hazardous?
Understanding the Chemical
3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin might have a long name, but under the surface, it’s one of many synthetic coumarin derivatives. Across the globe, chemists have developed several types, mostly to target either blood clotting or pests. The backbone structure hints where concern sits. This compound sits in the same chemical family as warfarin, an anticoagulant used in medicine and rodent control. That connection immediately raises eyebrows about potential for toxicity, since coumarin derivatives have a well-known and sometimes troubling track record.
Toxicity: What the Chemistry Suggests
Any time a molecule has a coumarin core paired with naphthyl substituents, questions about safety for people and animals feel justified. Most data on these molecules come from their effect on blood. Warfarin, the best-known compound in this family, disrupts vitamin K recycling in the liver. Without vitamin K, the blood loses its ability to clot properly, so a small dose thins blood, but a large or repeated exposure can cause dangerous internal bleeding.
Scientists designed most coumarin derivatives, including 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin, to act as rodenticides. They target the biology of rodents, but humans share the same clotting proteins. Many people don’t realize how potent anticoagulant rodenticides can be for pets and children—a tiny mistake in handling or storage leads to emergency situations. The leanest data hint that this compound acts comparably to other “first-generation” anticoagulant rodenticides like warfarin and coumatetralyl. Symptoms tend to appear days after exposure: fatigue, unexplained bruising, nosebleeds, gum bleeding, and sometimes life-threatening internal hemorrhages.
Why Experience Matters
I’ve talked to veterinary staff who’ve rushed dogs to the clinic after accidental rodenticide ingestion—the antidote, vitamin K, works, but only if the pet gets help early. Some people brush off rodenticide safety because modern products don’t look frightening. Yet anything engineered to kill small mammals by disrupting vital chemistry doesn’t exactly inspire comfort.
Years watching occupational chemical safety also taught me: reading the label means little without understanding the risks. Most people trust the appearance and packaging, and few read hazard sheets at all. In labs, staff must wear gloves, eye protection, and sometimes full face shields to move rodenticides, no matter the coumarin derivative. Spills end up on work logs, and anything that touches the skin, even unknowingly, goes in the chemical waste bin.
Reducing Hazard Exposure
The easiest way to avoid harm starts with never treating any rodenticide as safe for untrained hands. If a label says “coumarin derivative,” or if it lists ingredients like 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin, assume the risk level matches what veterinarians and pesticide experts expect—significant hazard, especially if inhaled, ingested, or handled frequently. For home and workplace protection, keep storage locked and secure, far from food, pets, or children, and always clean up spills using gloves and proper disposal.
On the regulatory side, agencies like EPA in the US and EC in Europe review anticoagulant rodenticides regularly. Some have faced stricter restrictions or outright bans due to the risk of accidental poisoning and environmental persistence. Safer alternatives for rodent control do exist—traps, physical barriers, or newer poisons with reversal agents already available. Shifting toward prevention and less hazardous control keeps both people and pets safer, and still gets the job done.
What are the storage requirements for 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin?
The Real-World Side of Chemical Storage
Anyone storing chemicals knows that every compound brings its unique quirks. 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin belongs to the family of coumarin derivatives, which tend to be sensitive to air, light, and temperature. It’s tempting to treat each powder or crystalline form as just another batch on the shelf. I learned early in my lab days that taking shortcuts, like popping open a lid for a quick scoop or sticking a bottle near a window, can cause slow disasters. Over months, small mistakes build up: the chemical degrades, research results drift, and, soon enough, there’s no telling what’s in the jar.
What Goes Wrong Without Good Storage?
Light, air, and humidity often team up to ruin sensitive organics. In college, I kept a hydroxycoumarin sample on an open bench because the storeroom was locked. That clear yellow powder turned brownish and clumpy within a week. Exposure to air caused oxidation, and bits of moisture from summer’s humidity probably triggered chemical changes I couldn’t see. Testing compromised material leads to unreliable outcomes, messes with reproducibility, and even puts safety on the back burner.
Key Practices That Actually Work
The first order of business: store this compound in a tightly sealed amber glass bottle. Amber glass shields against the blue and ultraviolet part of the light spectrum, which makes a real difference for compounds prone to photodegradation. Plastics sometimes breathe more than we expect, so glass closes that loophole.
Humidity sneaks through improper seals and brings trouble. I once found a desiccator invaluable in our chemistry storeroom—especially during muggy months. Tossing a few silica gel packs inside isn’t just a minor detail; they do a solid job sucking up ambient moisture. Anybody leaving coumarins in a damp lab finds out soon enough that the stuff sticks together and forms sticky lumps.
Let’s talk temperature. While this coumarin doesn’t demand freezing cold, a cool, steady environment—like a basic storage refrigerator—extends shelf life by slowing chemical breakdown. My habit was to avoid the door shelves, which fluctuate more, and use the middle or lower racks. Avoiding temperature swings is just as important as keeping things cool.
Some chemicals, including this naphthyl-coumarin, can interact with oxygen, kicking off slow, unwanted reactions. Filling headspace with an inert gas like nitrogen definitely isn’t overkill in settings where purity matters. Working in small aliquots rather than dipping into one big container means the main batch isn’t seeing air every week.
Responsibility and Going the Extra Mile
Labels matter, not just for tracking expiration but for giving quick reminders: “Protect from light,” “Keep dry,” or “Refrigerate.” I’ve watched accidents unfold when one person stored an open vial in the wrong spot, and another, in a rush, grabbed what looked like a fresh sample. Consistent training and clear signage help lessen the risk of mix-ups.
As more of us focus on greener, safer chemistry, responsible storage cuts down on waste and lost money. Contamination leads to disposal headaches and avoidable purchases. Proper handling isn’t just bureaucracy—it’s a real investment in safety, data quality, and budget.
For every lab tech, scientist, or even hobbyist who takes on 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin, the day-to-day details keep headaches away. Precision here pays off in cleaner data, longer shelf life, and a safer workplace.
What is the chemical structure of 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin?
The Backbone: Coumarins and Naphthalene Rings
It takes just a glance at the long name to recognize this compound carries pieces from different chemical families. On one side sits coumarin, which is a benzopyrone structure with a lactone ring fused to a benzene ring. Chemists and pharmacists see coumarins as the root of many anticoagulant drugs. On the other, there’s a naphthalene derivative: 1,2,3,4-tetrahydronaphthalene, sometimes called tetralin. Tetralin has a partially saturated ring, which brings in flexibility and new biochemical interactions.
Piecing the Structure Together
The molecule forms by sticking a 1,2,3,4-tetrahydro-1-naphthyl group to the coumarin’s third carbon. Move to the coumarin’s fourth position and you’ll spot a hydroxy group. So, the core features look something like this: a fused benzene and alpha-pyrone ring (the coumarin), a naphthyl moiety bridged by four hydrogen atoms (reducing aromatic character there), and an OH group offering potential for hydrogen bonding or further reactions.
Graphically, you’d find a three-dimensional tangle—aromatic stacks from the coumarin’s benzene ring, the part-saturated naphthalene, and the polar handle from the hydroxy group. This configuration tells a lot about how it fits into receptors in the body: shape, charge, and pocket-filling all decide how a molecule behaves as a drug.
Why This Mix Matters: Real-World Context
Chemists didn’t throw these groups together for fun. History shapes this sort of compound. Warfarin, for instance, stems from the coumarin family and revolutionized blood-thinning treatments. Pharmaceutical innovators often tweak chemical rings, seeking subtle changes—less toxicity, better absorption, improved selectivity for enzymes. Attaching a tetrahydronaphthyl group to coumarin creates a molecule that may dodge certain enzymes while grabbing onto others with greater strength, often changing its path through the body.
Veterinarians and doctors watched for coumarin derivatives that offered high potency with less risk of causing accidental bleeding. Much of the research rested not just on how well it worked, but how predictable and controllable its actions proved to be. The structure’s tweak can mean fewer interactions with food and other medications, or a steadier effect for patients whose liver enzymes vary widely.
Safety and Environmental Footprint
Synthesizing compounds like 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin has consequences outside the lab. Factories vent solvents, researchers handle reagents that may trigger allergies, and leftover material can seep into water or soil. Anyone who stepped into an organic chemistry lab knows the smell and the stains. Regulatory agencies urge strict tracking and waste management, not just to safeguard workers but to protect downstream communities and wildlife.
Lab stories tend to feature unpredictable results—side reactions, nasty byproducts, unexpected toxicities. A compound might sail through cell testing, but show up as problematic in mice. Science doesn’t always hand over clean answers or perfect safety margins.
Charting a More Responsible Path
Today's chemists lean on green chemistry principles, searching for methods that slash waste and scraps while producing high-quality molecules. Shorter synthesis routes, safer reagents, and better recycling help shrink the environmental footprint. Hospitals and clinics depend on reliable supplies and steady quality, so companies must balance high yields with environmental responsibility.
Opening dialogue among researchers, regulators, and the public sparks better understanding. Safety education, investment in greener processes, and clear communication go far in building trust around any molecule’s journey from beaker to bedside. Chemical structures shape more than just their own reactions—they reflect our promises and responsibilities to society and the planet.
Where can I buy 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin?
Understanding the Substance
3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin isn’t something you usually find on a hardware shelf or in a regular pharmacy. This chemical plays a role in medical and research settings, mostly associated with compounds used as anticoagulants or in studies related to blood-clotting disorders. Getting it, though, isn’t as simple as sending an online order.
Real-World Challenges in Sourcing
Back in my lab days, sourcing niche chemicals meant lots of paperwork and even more patience. There’s red tape for a reason: substances with specific biological or toxicological properties get tightly watched by regulators. Suppliers can’t just drop off a package unless they know it’s going into the right hands—the law makes sure of that. Even when I needed small quantities for a controlled experiment, the process took weeks and involved multiple approvals.
Why Quality and Legitimacy Go Hand-in-Hand
I’ve seen the fallout from using questionable sources firsthand. There’s a trust that builds over years with established chemical suppliers. Buying specialty compounds through established distributors means they provide certificates of analysis, batch documentation, and shipment tracking. Sigma-Aldrich, Fisher Scientific, and TCI are just a few companies regularly working with universities and accredited laboratories. They won’t process orders without proper credentials, but there’s peace of mind in knowing the chemical in the bottle is the real deal.
Beware of the Black Market and Shady Sources
The internet buzzes with sellers promising exotic chemicals, no questions asked. Some of these sites pop up overnight, ready to cash in on folks who might not realize the dangers or legal risks. Several high-profile incidents have shown just how badly things can go. Authorities have cracked down on these back-alley suppliers for selling fake or even hazardous substitutes. Worse still, getting caught purchasing controlled substances without official clearance leads to steep penalties.
Regulations That Matter
Chemicals like 3-(1,2,3,4-Tetrahydro-1-Naphthyl)-4-Hydroxycoumarin land under multiple regulations, especially if there’s any perceived drug or toxicology function. In the U.S., DEA and EPA keep close tabs on distribution and usage. Outside the States, the European Chemicals Agency does similar enforcement. During my years behind the bench, it never paid off to cut corners.
Smarter Ways to Move Forward
If you have a legitimate need—whether it’s for university research, industrial quality control, or another professional pursuit—start by getting your credentials in order. Most top-tier suppliers make the process clear. They’ll require proof of business, research intent, and compliance with federal and local laws. Some even help navigate the permitting system, having helped hundreds of labs move forward safely and ethically.
Another Angle: Collaborating
One fix for individual scientists coming up short on credentials is to collaborate with institutions already vetted by suppliers. Universities and research hospitals routinely broker these purchases for affiliated researchers. Joint projects often provide an easier path for approvals, oversight, and funding, too.
The Bottom Line: Smart, Legal Choices
While hunting down this particular compound might sound like a simple procurement task, the risks and responsibilities run deep. The best outcomes come to those who make choices rooted in safety, compliance, and clear purpose. Sticking with reputable channels protects everyone involved and keeps scientific progress above board.
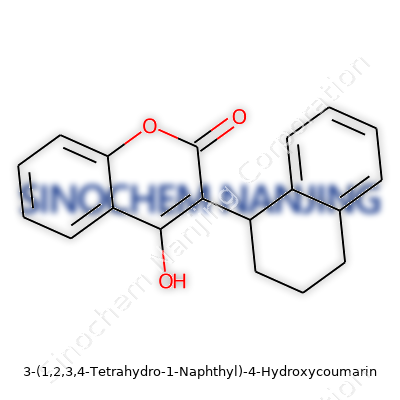
| Names | |
| Preferred IUPAC name | 3-(3,4-Dihydro-1H-naphthalen-1-yl)-4-hydroxy-2H-chromen-2-one |
| Other names |
Brodifacoum Talon Volid Ratak |
| Pronunciation | /θriː baɪ ˈwʌn tuː θriː fɔː ˈtɛtrəˌhaɪdrə wʌn næfˌθɪl fɔː ˈhaɪdrɒksi kuːˈmɑːrɪn/ |
| Identifiers | |
| CAS Number | 2605-59-6 |
| Beilstein Reference | 1445451 |
| ChEBI | CHEBI:9648 |
| ChEMBL | CHEMBL1208392 |
| ChemSpider | 11689744 |
| DrugBank | DB00682 |
| ECHA InfoCard | 03b9ecd5-6770-425c-9509-1ee6ec077905 |
| EC Number | 606-075-00-9 |
| Gmelin Reference | 78635 |
| KEGG | C18606 |
| MeSH | D014781 |
| PubChem CID | 10315804 |
| RTECS number | GN5950000 |
| UNII | E1J8W38T2N |
| UN number | UN3276 |
| Properties | |
| Chemical formula | C19H16O3 |
| Molar mass | 324.37 g/mol |
| Appearance | white to off-white solid |
| Odor | Odorless |
| Density | 1.26 g/cm³ |
| Solubility in water | Insoluble in water |
| log P | 4.6 |
| Vapor pressure | 3.39E-09 mmHg at 25°C |
| Acidity (pKa) | 10.2 |
| Basicity (pKb) | 11.8 |
| Magnetic susceptibility (χ) | -89.27×10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.649 |
| Viscosity | Viscosity: 2.11 cP (23°C) |
| Dipole moment | 3.7594 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 523.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -75.68 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7154 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | B01AA04 |
| Hazards | |
| Main hazards | Harmful if swallowed. Irritating to eyes, respiratory system and skin. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P261, P264, P270, P271, P272, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 132°C |
| Lethal dose or concentration | LD50 oral rat 56 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 276 mg/kg |
| NIOSH | GZ2625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) not established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
Warfarin Acenocoumarol Phenprocoumon Dicoumarol |

