Understanding 21-Hydroxy-20-Methylpregn-4-En-3-One: Behind the Science and Prospects
Historical Development
Once chemists pinpointed the complex architecture of steroids, a race began to develop methods for synthesizing molecules that could unlock new medical advances. 21-Hydroxy-20-Methylpregn-4-en-3-one—known among researchers as a key intermediate in corticosteroid synthesis—has a history rooted in decades of scientific grind. In the 1950s and 60s, researchers sought routes to create corticosteroids with precise functional side groups, and this compound became a reliable stepping stone. Its structure set the stage for breakthroughs in treating inflammatory disorders and autoimmune diseases. Scientists in those early years used painstaking classic organic methods, refining the process step by step: isolation, oxidation, protection and deprotection of functional groups. It wasn’t just about making something new; it was about unlocking ways to precisely control the fate of hormone analogues that changed real lives.
Product Overview
21-Hydroxy-20-Methylpregn-4-en-3-one stands as a synthetic steroid molecule that carries both a 21-hydroxyl and a 20-methyl group. It belongs to a family of progestins and corticosteroid intermediates, and plays a central role in pharmaceutical manufacturing. Research labs and industry rely on this molecule to bridge the gap between raw plant sterols and active medicines, especially those used to treat adrenal dysfunction and for anti-inflammatory drugs. No one seeking reliable, pure starting substances for steroidal research can ignore its importance, and its impact spreads across pharmacology, medicinal chemistry, and hormone replacement fields.
Physical & Chemical Properties
The structure of 21-Hydroxy-20-Methylpregn-4-en-3-one reveals a classic pregnane skeleton, modified at the C20 and C21 positions, which affects both reactivity and solubility. Typically, it forms off-white crystalline solids, bearing limited solubility in water but greater affinity for organic solvents like chloroform and methanol. Its melting point hovers in the range familiar to most corticosteroid intermediates—usually around 180°C. The presence of the 21-hydroxyl group lends itself to targeted derivatization, while the 20-methyl makes this compound stand apart from its peers on the pharmacological landscape. Chemists track purity and structural confirmation through NMR, IR, and MS spectra, all of which reflect the subtle presence of its unique functional groups.
Technical Specifications & Labeling
Precision in labeling and technical documentation fosters trust in research and industry supply. Leading suppliers report purity levels exceeding 98%, backed by detailed chromatographic analyses. Labels display chemical identifiers, batch numbers, expiration dates, and specific storage requirements—cool, dry, and shielded from direct light. Labs rely on stability studies and documentation to ensure consistent results, a big deal when a single lot feeds into thousands of high-value syntheses. Whether packaged as a powder or suspended in ethanol, every batch faces rigorous scrutiny to confirm it aligns with internationally accepted standards set by regulatory agencies and pharmacopeias.
Preparation Method
Preparation kicks off from plant-derived steroids like diosgenin or commercially available progesterone derivatives. Chemists wield a mix of oxidation (often chromium-based) and selective hydrogenation to introduce and set the necessary functional groups. A sharply-tuned protection-deprotection dance secures the hydroxyl group at C21 without unwanted side reactions. Experienced practitioners know the value of gentle purification—think silica gel chromatography and recrystallization—to get rid of side products. High-yield routes may involve phase-transfer catalysis or modern, greener oxidants, cutting down waste and improving safety, all while keeping the sensitive steroid skeleton intact. Hands-on work in the lab teaches you just how challenging, and rewarding, these syntheses turn out.
Chemical Reactions & Modifications
This molecule’s reactivity spins out a range of follow-up reactions—acetylation at C21, oxidation at C3, transformation to keto or hydroxyl derivatives at multiple rings. Each step opens the door to new biological activities, serving as the launchpad for therapeutic agents like prednisolone, dexamethasone, or other designer steroids. Smart modifications of the 21-hydroxyl have produced drug candidates with altered anti-inflammatory properties. In many labs, teams explore selective fluorination or methylation to tweak metabolic stability and receptor affinity. That margin for modification defines how medicinal chemistry evolves—one inspired synthetic route at a time.
Synonyms & Product Names
Those in the know quickly recognize how many names a compound like this can pick up over decades. Besides 21-Hydroxy-20-Methylpregn-4-en-3-one, you’ll come across terms like 21-Hydroxy-20-methylprogesterone, or reference numbers from suppliers. Researchers sometimes slip into shorthand—21-OH-20-MeP or similar—in papers and protocols, a reminder that standardization in nomenclature feels elusive in complex fields like steroid chemistry. Catalogs might list slight variations, but careful reading of the structure and analytical data keeps everyone on the same page.
Safety & Operational Standards
Handling steroid intermediates brings responsibility. Researchers wear gloves, lab coats, and goggles as routine. Material safety data sheets warn of possible skin and eye irritation, suggesting well-ventilated areas and prompt cleaning of any spills. Experienced chemists track exposure not just in the moment, but across careers—recording dates, tracking usage, and working under hoods to limit inhalation. Waste management falls under local and international hazardous substance protocols, as these compounds don’t just vanish after rinsing. Training in emergency procedures, safe storage, and waste labeling forms the backbone of lab work, and anyone who’s faced an accident knows these habits matter most just when the routine falters.
Application Area
The pharmaceutical industry leans heavily on this molecule for corticosteroid synthesis and as a probe for metabolic pathway studies. Research teams create analogues to study receptor binding, metabolic breakdown, and unique target profiles for anti-inflammatory drugs. In hospitals, final derivatives trace back to intermediates like this in medications for asthma, autoimmune diseases, and transplant rejection. Beyond the obvious, agricultural scientists investigate similar steroids for veterinary drugs and even crop-growing aids. The utility of this single intermediate blends into so many research fronts that anyone working at the interface of organic chemistry and medicine views it as a familiar face in the lab notebook.
Research & Development
Innovation in steroid chemistry never seems to slow down. Research teams harness new asymmetric catalysts for greener, higher-yielding routes, and automation streamlines multi-step purification. Studies published in top journals in the last five years show big strides in C–H activation and selective functionalization, pushing the limits of what synthetic methods can do. Universities partner with industry to test new derivatives for improved bioavailability, longer half-lives, or new mechanisms of action. These R&D efforts don’t just offer incremental change; they promise blockbuster therapies and new tools for tackling both rare and common illnesses.
Toxicity Research
Safety can’t take a back seat, and that’s why teams continue to probe the acute and chronic toxicity of parent compounds and their modifications. Animal studies track liver, kidney, and reproductive outcomes, while long-term cell-line research addresses possible mutagenicity. Industry regulators require comprehensive profiling—dosing studies, environmental toxicity evaluations, routes of breakdown. Clinical trial pipelines demand every scrap of toxicity evidence before a derivative can move into human testing. Anyone who’s seen what an overlooked metabolite can cause—unexpected side effects, failed clinical trials, environmental contamination—knows the importance of honest, thorough toxicity work on every intermediate.
Future Prospects
Emerging science in synthetic biology hints at fresh ways to produce steroid intermediates through engineered yeast or bacteria. Advances in CRISPR and metabolic engineering paint a future where old chemical syntheses step back for bio-based processes, bringing both scale and sustainability. Demand for personalized steroids that target specific genetic backgrounds pushes research into new frontiers, while green chemistry stands as a loud call for safer, more efficient manufacturing methods. Teams around the world keep sharing their findings, driven by the impact these compounds bring to health, science, and beyond. Comparing snapshots of the field from even ten years ago to now, the leaps in efficiency, selectivity, and understanding tell us 21-Hydroxy-20-Methylpregn-4-en-3-one isn’t just another compound on the shelf—it’s both a milestone and a launchpad for the future.
What is 21-Hydroxy-20-Methylpregn-4-En-3-One used for?
A Glimpse into the World of Steroid Chemistry
Stepping into a pharmacy or flipping through a biology textbook, you’ll notice complex names like 21-Hydroxy-20-Methylpregn-4-En-3-One. In reality, names like this link back to critical molecules found at the core of hormone biology. This one stands as an intermediate compound—a kind of chemical bridge—sitting along the path to making something we all know: corticosteroids.
How This Compound Connects to Medicine
Pharmaceutical labs use this molecule largely for one purpose: to synthesize corticosteroids. These steroids anchor modern medicine’s response to things like allergies, autoimmune disorders, and addisonian crises—those times when the adrenal glands just don’t work as they should.
People rely on corticosteroids to stop inflammation, control immune flares, and prevent organ rejection after transplantation. The problem: you can’t find most corticosteroids in a river or in a field. Researchers use building blocks like 21-Hydroxy-20-Methylpregn-4-En-3-One because they allow chemical tweaks, paving the way for prednisone or hydrocortisone, meds you see on thousands of prescriptions each day.
Why the Source and Process Matter
Years ago, lab workers made steroid precursors from animal glands, but demand, cost, and ethical questions forced researchers down a different path. Today, many steroid precursors come from plant matter, such as diosgenin isolated from yams. Then, in a chemical plant, this precursor undergoes changes—adding or swapping out chemical groups—eventually landing on this compound and, one or two steps later, arriving at useful corticosteroids.
Lab synthesis isn’t just a technical footnote. Consistency, purity, and cost depend on it. Contaminated batches pose health risks. Poorly regulated sourcing can create shortages or price surges for critical medicines. So the path from this compound to your pharmacy shelf deserves some real attention.
Clinical Connections and Limitations
Doctors don’t hand out pure 21-Hydroxy-20-Methylpregn-4-En-3-One at clinics. Still, the ease and reliability of making this compound influences the medical supply chain. More efficient routes help with supply stability and make essential medicines cheaper. Patients facing asthma, lupus, or severe allergies might never hear the compound’s name, but better production practices could save them hundreds on yearly medical bills.
Pharmaceutical supply chains have faced disruption more than once in my own work. Each time, the backup often traced to shortages with base chemicals used much earlier in synthesis. Streamlining the manufacturing process by improving yields of this precursor can help blunt the impact of shocks—like sudden spikes in demand or breakdowns in international shipping.
What Needs Fixing
Transparency matters here. Companies often don’t disclose the exact origins of their active ingredients. Advocates have pushed for laws that would require more detail. Detailed sourcing helps spot supply chain weaknesses before they lead to missing or unaffordable medicines.
Another challenge comes from environmental costs linked to chemical manufacturing. Many pathways use solvents or generate waste that must be managed carefully. Investments into ‘green chemistry’, like using water-based solvents or bioengineered yeast, could shrink the ecological footprint.
Building for a Resilient Future
Clear labeling, investment in efficient chemistry, and sharing best practices across borders could shore up the supplies of medicines that rely on precursors like 21-Hydroxy-20-Methylpregn-4-En-3-One. Governments, industry, and the public all play a part in making sure corticosteroids continue to reach people across the globe, without the risk of fluctuating prices or dangerous shortages.
Is 21-Hydroxy-20-Methylpregn-4-En-3-One safe to use?
Background on This Compound
Think of 21-Hydroxy-20-Methylpregn-4-En-3-One as a mouthful, yet it fits into a bigger story in medicine. Synthetic steroids and their relatives keep showing up in research, sometimes as potential treatments, sometimes as cautionary tales. This chemical lands in the steroid family tree, sharing some relatives with medicines that have shaped entire fields, like hydrocortisone for inflammation or progesterone for reproductive health.
What Current Science Tells Us
Safety starts with solid evidence. Researchers who study new steroid derivatives often look at effects on hormones, possible toxicity, and how these molecules work in the body. Most of the scientific record on 21-Hydroxy-20-Methylpregn-4-En-3-One comes from animal models or early pharmacology labs. Data points show up in hormone assays and tissue culture findings, which matter for the first impressions but cannot replace real-world, long-term patient outcomes.
One thing stands out: just because a compound looks like an approved drug does not mean it will behave the same. These small changes can flip safe into risky, or vice versa. Early-stage studies sometimes reveal benefits, such as anti-inflammatory action or potential to modulate hormone pathways. That can spark real hope in researchers, especially when looking at unmet needs in medicine.
Safety Profile: Known Risks and Unknowns
People who use new steroidal compounds often track two big concerns—side effects and unexpected long-term consequences. Classical issues pop up with steroids: immune suppression, effects on metabolism, impacts on mood. Animal studies on 21-Hydroxy-20-Methylpregn-4-En-3-One hint at some of these, but real outcomes in people remain mostly a blank page. The chemical does not have a broad track record in clinical use. There are no large-scale safety trials, no population data, and no regulatory green lights for medical treatment. Those gaps leave patients and doctors without much to lean on.
Transparency and Responsibility
Experts—including endocrinologists and pharmacologists—emphasize the need for testing at every step. Without toxicity tests, reproductive system studies, allergy screening, and drug interaction data, risk grows with every unsupervised experiment. Regulatory agencies like the FDA require years of vetting for good reason. No one wants another thalidomide case or a repeat of early hormone drugs that caused more harm than good.
Researchers and users should be clear-eyed: using experimental compounds outside controlled trials is rolling the dice. What helped in a rat or a petri dish does not translate to humans, especially over months or years. This means anyone selling, recommending, or experimenting with 21-Hydroxy-20-Methylpregn-4-En-3-One should share what evidence there is, plain and simple.
Paths Forward
Medical progress always demands balance. Push for innovation, but do not overlook caution. Institutions with experience—academic labs, hospitals, public health agencies—can drive this work with oversight and published data. People interested in using this compound, whether out of hope or desperation, deserve transparency, honest risk assessment, and real support.
Personal experience here counts for a lot. Clinicians and pharmacists have seen compounds with big early promise fall short because incomplete safety data missed a bad side effect. Better to ask hard questions, look for published research, and put safety before speed.
No shortcut replaces peer review and careful study. Trust builds up over years, batch by batch, trial by trial. Until those results come in, caution remains the closest thing to wisdom.
What are the possible side effects of 21-Hydroxy-20-Methylpregn-4-En-3-One?
What Happens Inside Your Body
Prescription steroids like 21-Hydroxy-20-Methylpregn-4-En-3-One don’t usually grab headlines, but their impact goes deeper than you’d guess. Steroid hormones play a huge role in everyday health, controlling stress responses, immune reactions, and metabolism. With the right prescription, these drugs can save lives. My background in pharmacy has shown how sensitive patients can get to even small doses of synthetic hormones. It’s not just about hitting a target number on a blood test—our hormones touch mood, weight, blood pressure, bones, and much more.
Short-Term Effects People Notice Quickly
Anyone starting a new steroid should watch for changes they feel right away. Fluid retention tops the list. Many patients mention that their rings feel tight or they see some new puffiness around their ankles. Appetite can crank up, leading to unexpected weight gain. Sleep troubles often crop up, too. I’ve had patients toss and turn, only to realize their new medicine nudged their body’s timing off track.
People sometimes feel wired, restless, even irritable or down. Some also notice higher blood pressure or faster pulse. Acne can pop up, or old skin issues flare. Each case is different, but these responses often tie back to how steroids affect salt balance, sugar levels, and brain chemistry.
Long-Term Risks No One Wants to Ignore
Doctors usually aim for the smallest dose for the shortest time. Staying on steroids for many months may invite bigger problems. Blood sugar can creep up, with some developing full-blown diabetes. Bones thin out as calcium drains away, leading to osteoporosis. Older adults become more prone to fractures. Muscle weakness can slowly appear in both young and old.
I’ve seen cholesterol numbers shift in long-term users, raising cardiac risks down the line. Kids on these medicines may face slower growth. The body’s own hormone factories take a backseat while synthetic steroids do all the work, making sudden stops risky. It leaves patients vulnerable to stress or infection. Even the immune system calms down under steroids, which matches their use in autoimmune diseases—but it’s a double-edged sword. Infections can get worse, or normal vaccines may not trigger a strong response. In rare cases, psychiatric symptoms like mood swings or even psychosis have been recorded.
What Can People Do?
Facing all these potential side effects feels overwhelming, but taking a hands-on approach helps. Patients need close follow-up. Experienced medical teams set up regular labs—checking blood pressure, sugar, cholesterol, and bone health. Nutritionists in the clinic can help folks adjust diets to cut out extra salt and sugar, easing some early trouble spots. Keeping bones strong with exercise and calcium-rich food makes a real difference for those on long-term steroids.
Education is a tool too often left in the drawer. Pharmacists and nurses can walk patients through what to watch for and when to ask for help, from unusual bruising to high fevers. Tapering a dose slowly—never stopping cold turkey—lets the body’s natural hormones ease back in.
Pharmaceutical science offers newer steroid alternatives with fewer side effects, and research pushes on toward tailored dosing. That said, the reality stays the same: every drug choice means trade-offs. Conversations between patients and doctors do more than any package insert. Vigilance and teamwork make a difference every day for patients relying on steroids like 21-Hydroxy-20-Methylpregn-4-En-3-One.
How should 21-Hydroxy-20-Methylpregn-4-En-3-One be stored?
Finding the Right Spot for This Chemical
In any lab, 21-Hydroxy-20-Methylpregn-4-En-3-One lands in that class of compounds you don’t want getting spilled, exposed, or broken down by sun or heat. This synthetic steroid is an important intermediate in steroid research. Storage sounds simple, but ignoring basics can mean wasted money or safety risks.
Light and Temperature Aren’t Friends Here
Chemicals break down in the wrong conditions. I once watched a colleague lose six months of work because a cleaner left the window shades up, letting sunlight beat down on a bottle. Ultraviolet rays will accelerate decomposition. This happens with many steroid derivatives, and this one is no different. Shelving in a well-shaded cabinet with a lock on it pulls double duty: you keep the chemical from light exposure, and you limit who can access it.
Refrigerators in labs serve more than just lunch. Store 21-Hydroxy-20-Methylpregn-4-En-3-One at low temperatures – usually 2-8°C. This holds back unwanted reactions and preserves the molecule’s structure. In my experience, going below freezing isn't needed. If crystals form, you may not get the same properties in your experiment next time. Regular monitoring with a digital thermometer helps keep things confident. Don’t slip into the habit of keeping chemicals at room temp just for convenience – long-term, it risks more than it saves.
Humidity and Air: The Silent Breakdown Artists
Popping the cap off the vial for ‘just a minute’ can spoil the whole lot. Water vapor finds a way in, and the chemical may take it up, clumping or breaking apart. I store most sensitive steroids with desiccant packs in the primary container and in the outer storage area. That little bag of silica gel is cheap insurance. Always reseal the bottle tightly after use. Vacuum-sealed ampoules offer even better protection, but single-use vials aren’t always budget friendly. Oxygen may also play a role in breakdown, especially over a few months. In one poorly sealed bottle I saw, the powder turned a weird shade and batch results turned unpredictable.
Don’t Skip the Label Details
Sharpie notes blur. Invest in chemical-resistant labels, clearly mark opening dates, and track refrigerated chemicals’ shelf lives. Since regulations ask for full traceability (GMP or GLP labs in particular), an illegible bottle becomes more than an organizational slip – it may trigger audit headaches or worse. I’ve seen research derailed by unclear notes or forgotten bottles.
People Make or Break Chemical Safety
Nothing replaces hands-on reminders. Staff turnover, late nights, or just plain carelessness mean protocols get skipped. Routine walk-throughs catch problems early. Training refreshers help newer students or techs internalize why these chemicals aren’t like storing acetaminophen at home. A decent Standard Operating Procedure, kept visible near storage areas, pays off down the road.
Proper Disposal Closes the Loop
Unused or expired 21-Hydroxy-20-Methylpregn-4-En-3-One shouldn’t end up in regular trash or down the sink. Coordinate with your facility’s hazardous waste team. One summer, a lab fire investigation found several flammable solvents stored close to old steroids – a totally preventable risk. Getting this right matters for health, environment, and compliance.
Reliable Storage Keeps Labs Productive and Safe
Solid science begins long before the first reaction. Protecting a sensitive intermediate like this means thinking about sunlight, temperature, moisture, and access. Care up front lets the science shine, instead of loss creeping in quietly through neglect or short cuts.
Does 21-Hydroxy-20-Methylpregn-4-En-3-One interact with other medications?
The Challenge of Medication Interactions
Walking into a pharmacy with a bag full of prescriptions, most of us forget our medicines might not always “play nice” together. Even in research-heavy environments like teaching hospitals, staff run into complex cases where a new drug enters the mix and throws off the balance. We see this often with hormones and steroid derivatives, including compounds like 21-Hydroxy-20-Methylpregn-4-En-3-One.
Why Doctors Watch Hormone-Like Compounds Closely
Steroids and their relatives—natural or synthetic—serve as chemical messengers. They stir up metabolic changes, kick off immune responses, affect how much salt our kidneys keep, or even alter how we handle stress. It wouldn’t surprise any seasoned clinician to see one of these compounds mess with another drug’s effectiveness or risk profile. Sometimes the interaction feels subtle at first, but complications can build.
This particular compound, structurally related to progesterone, leans on enzymes in the liver for processing. Cytochrome P450 enzymes break down not only most steroid medications, but also antidepressants, blood thinners, anti-seizure drugs, and even common painkillers. Introducing a new hormone analog can tilt the seesaw; it may cause blood levels of well-known medications to rise or fall, leading to less effect or more side effects.
Examples From Everyday Practice
Classic scenarios emerge with drugs that ride the same metabolic “highway.” Oral contraceptives, for example, can lose their punch in the presence of certain steroids—opening the door to unplanned pregnancies. People with epilepsy, already dealing with a fine balance of anti-seizure medication, might suddenly face more seizures or side effects if metabolic rates shift. Folks on anticoagulants—think warfarin—could bleed more easily if a new drug blocks breakdown in the liver.
Striking the Right Balance in Care
A smart approach hinges on a few habits. Pharmacists and doctors rely on solid databases, updated at lightning speed, to flag potential trouble before the patient leaves the pharmacy. Electronic records trigger warnings, but an honest, patient-doctor conversation makes a bigger difference. Listing every pill, supplement, and even herbal tea on a medication list covers blind spots.
Researchers still climb the learning curve with newer or less common steroids. Large clinical trials don’t always catch rare but real problems that turn up only once a drug leaves the research lab and starts landing in daily prescriptions. Independent studies, real-world case reports, and patient registries help fill in missing information for the medical community.
Practical Steps and Solutions
Direct communication from patient to provider remains the bedrock—you talk about new symptoms, track unexpected side effects, and share any recent changes in what you take, from vitamins to over-the-counter allergy pills. Doctors should review the full list at every visit. Pharmacies can run drug interaction checks, but face-to-face advice often trumps an automated alert.
If a new or unusual medication gets added to a routine, smart teams often set up closer lab monitoring. Blood tests check drug levels or key markers, especially with compounds filtered by the liver or kidney. For families and caregivers, a simple chart or smartphone app to log changes and symptoms helps track trends that might signal a brewing problem.
Medicines like 21-Hydroxy-20-Methylpregn-4-En-3-One deserve respect for both their benefits and complexities. Learning about interactions costs a little time but often saves health, money, and peace of mind down the line.
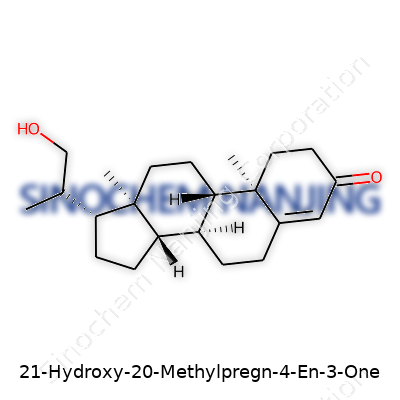
| Names | |
| Preferred IUPAC name | (20S)-21-hydroxy-20-methylpregn-4-en-3-one |
| Other names |
11-Deoxycorticosterone Desoxycorticosterone DOC Cortexone |
| Pronunciation | /ˌtwɛnti wʌn haɪˈdrɒksi ˌtwɛnti ˈmɛθɪlˈprɛgn fɔr ˈiːn θri oʊn/ |
| Identifiers | |
| CAS Number | 3890-23-3 |
| Beilstein Reference | 1856751 |
| ChEBI | CHEBI:34735 |
| ChEMBL | CHEMBL3914109 |
| ChemSpider | 21476557 |
| DrugBank | DB01481 |
| ECHA InfoCard | ECHA InfoCard: 100.207.430 |
| EC Number | 1.1.1.146 |
| Gmelin Reference | Gmelin 409382 |
| KEGG | C16583 |
| MeSH | D009373 |
| PubChem CID | 12412744 |
| RTECS number | KY8170000 |
| UNII | 47D78G1X6H |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID7032517 |
| Properties | |
| Chemical formula | C22H32O3 |
| Molar mass | 332.478 g/mol |
| Appearance | White to Off-White Solid |
| Odor | Odorless |
| Density | 1.09 g/cm³ |
| Solubility in water | practically insoluble |
| log P | 2.17 |
| Vapor pressure | 2.71 x 10^-9 mm Hg at 25 °C |
| Acidity (pKa) | 15.5 |
| Basicity (pKb) | 2.94 |
| Magnetic susceptibility (χ) | -11.28 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.5700 |
| Dipole moment | 3.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 298.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -558.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -8546.7 kJ/mol |
| Pharmacology | |
| ATC code | H02AB02 |
| Hazards | |
| Main hazards | H315, H319, H335 |
| GHS labelling | GHS07, GHS08 |
| Pictograms | C[C@]12CC[C@H]3[C@H]([C@@H]1CC[C@@H]2O)CCC4=CC(=O)CC[C@]34C |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H361 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P305+P351+P338, P362+P364 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | >110°C |
| LD50 (median dose) | LD50: 500mg/kg (rat, oral) |
| PEL (Permissible) | PEL (Permissible) for 21-Hydroxy-20-Methylpregn-4-En-3-One: Not established |
| REL (Recommended) | 0.003 mg/kg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Progesterone 21-Hydroxyprogesterone 20-Methylpregn-4-en-3-one Corticosterone Desoxycorticosterone |

